Impact of Pretreatment on Hydrothermally Carbonized Spruce
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
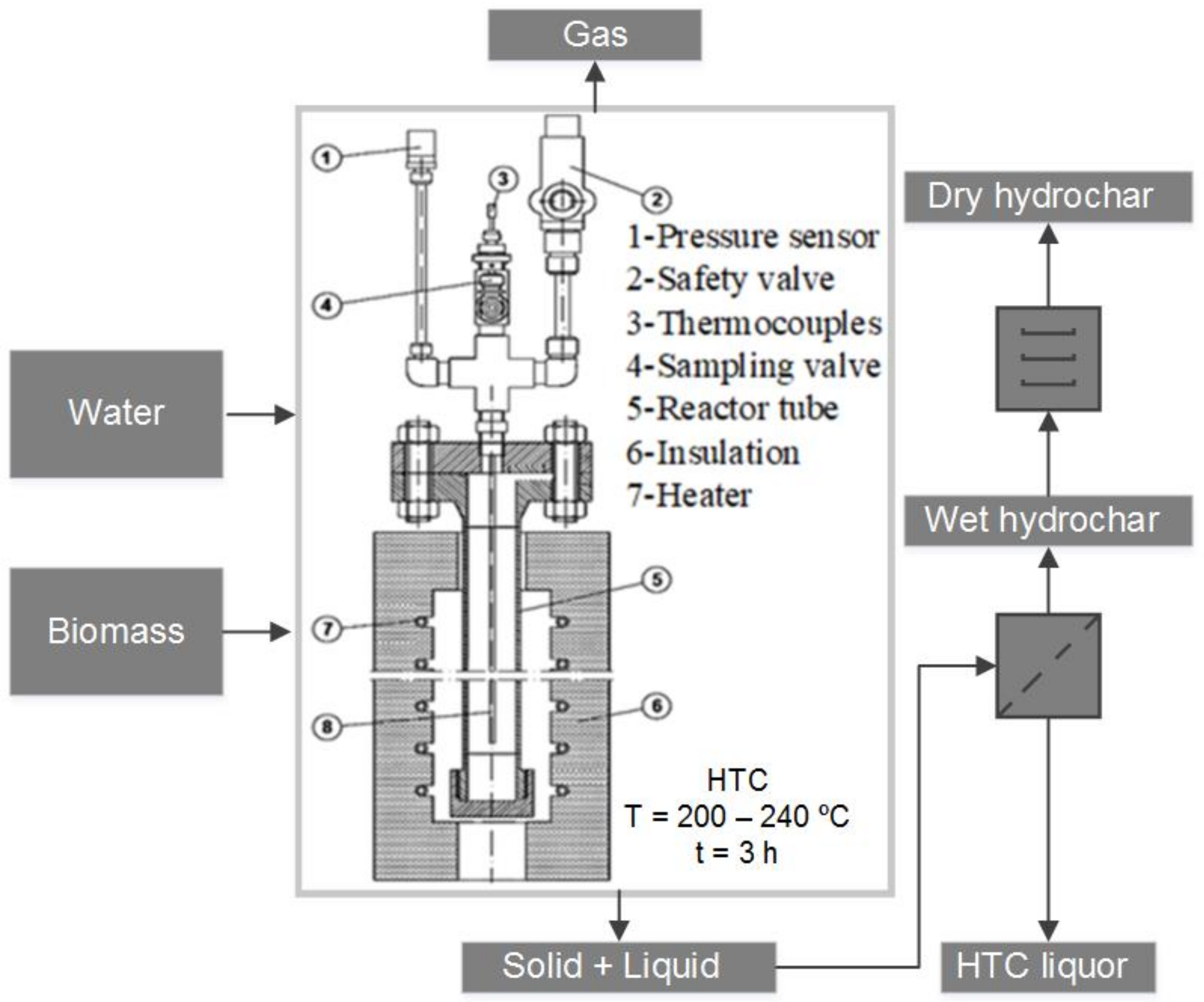
2.2. Instrumentation
2.3. Methods
3. Results and Discussion
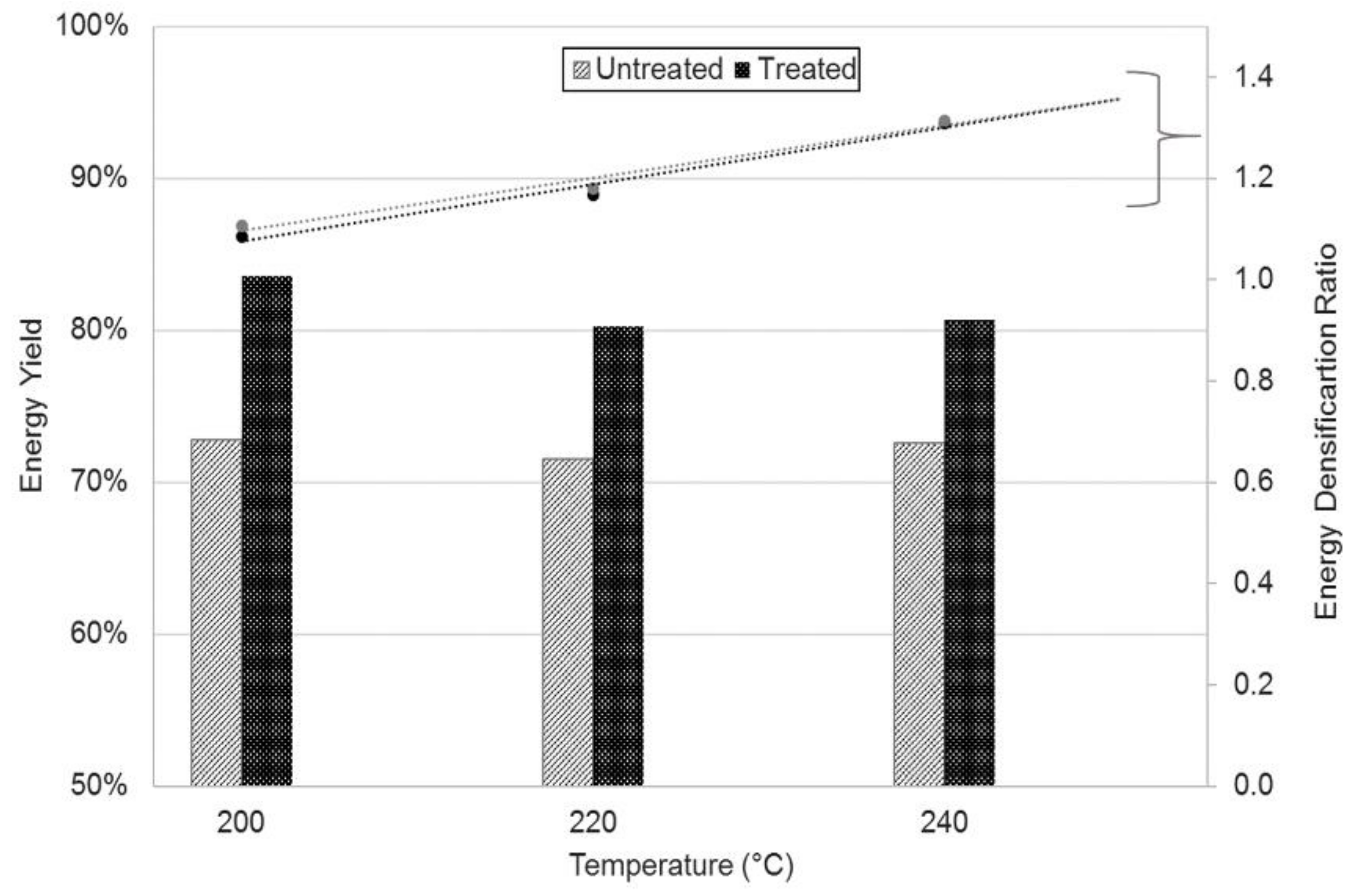
3.1. Mass and Energy Yields
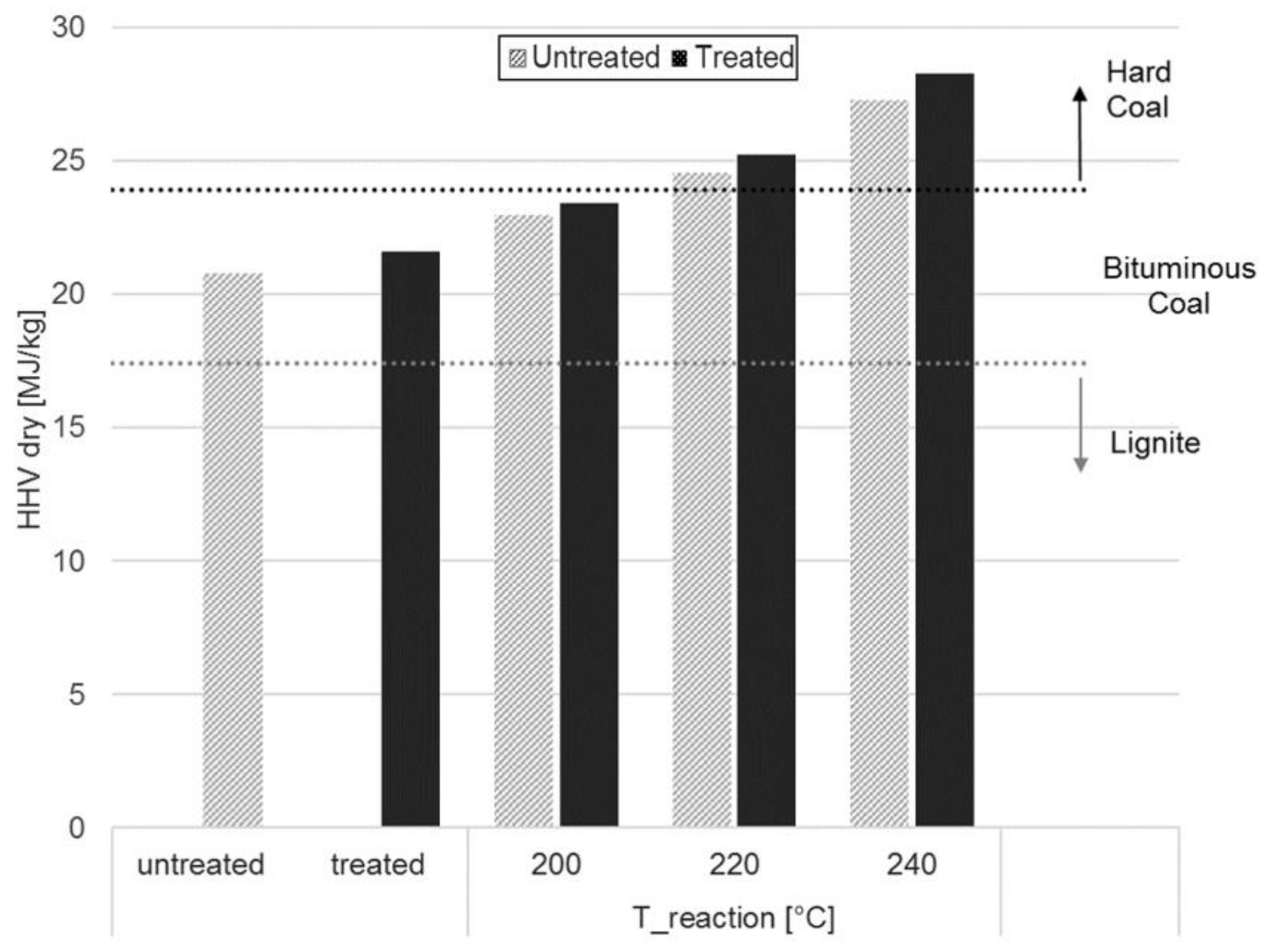
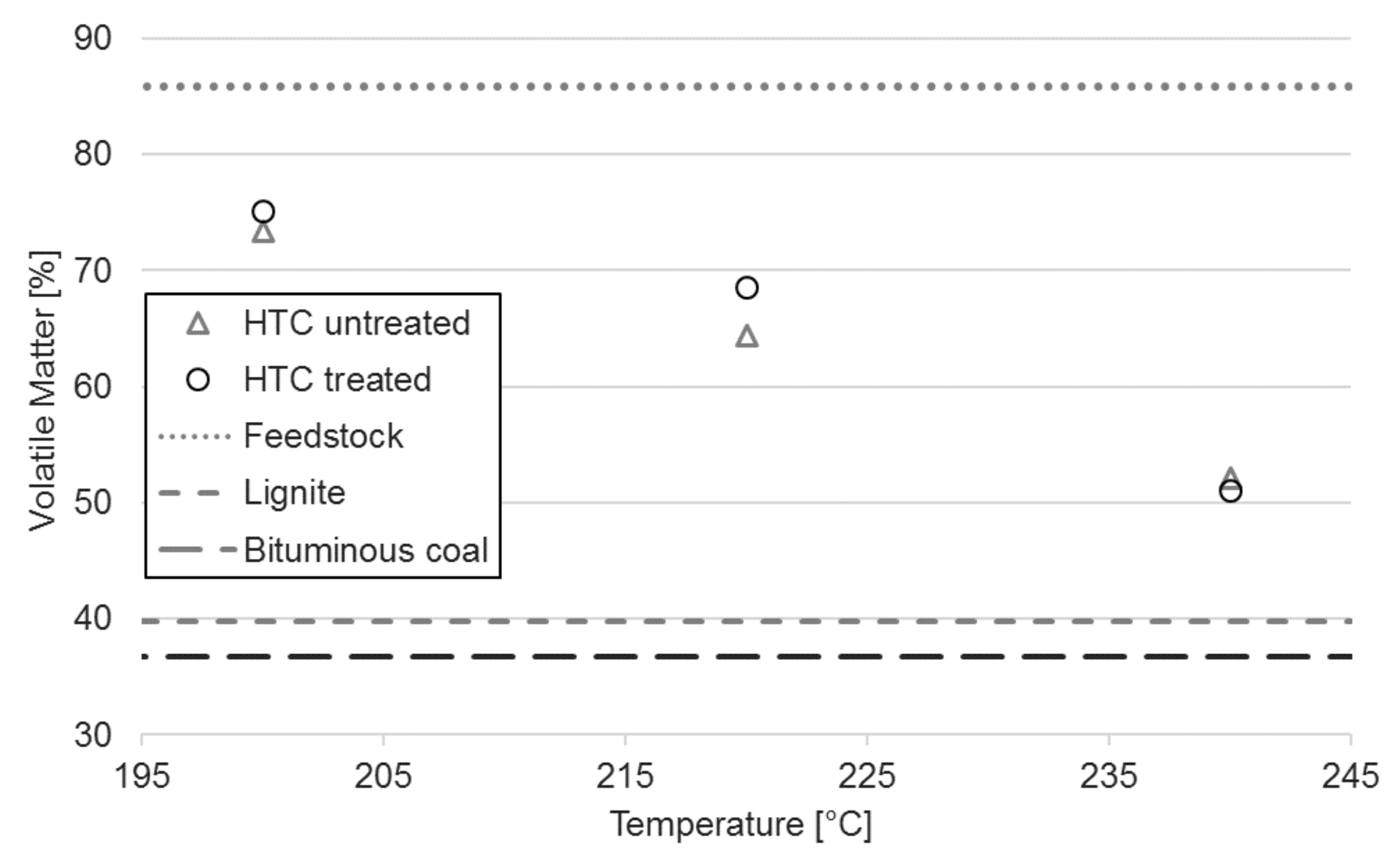
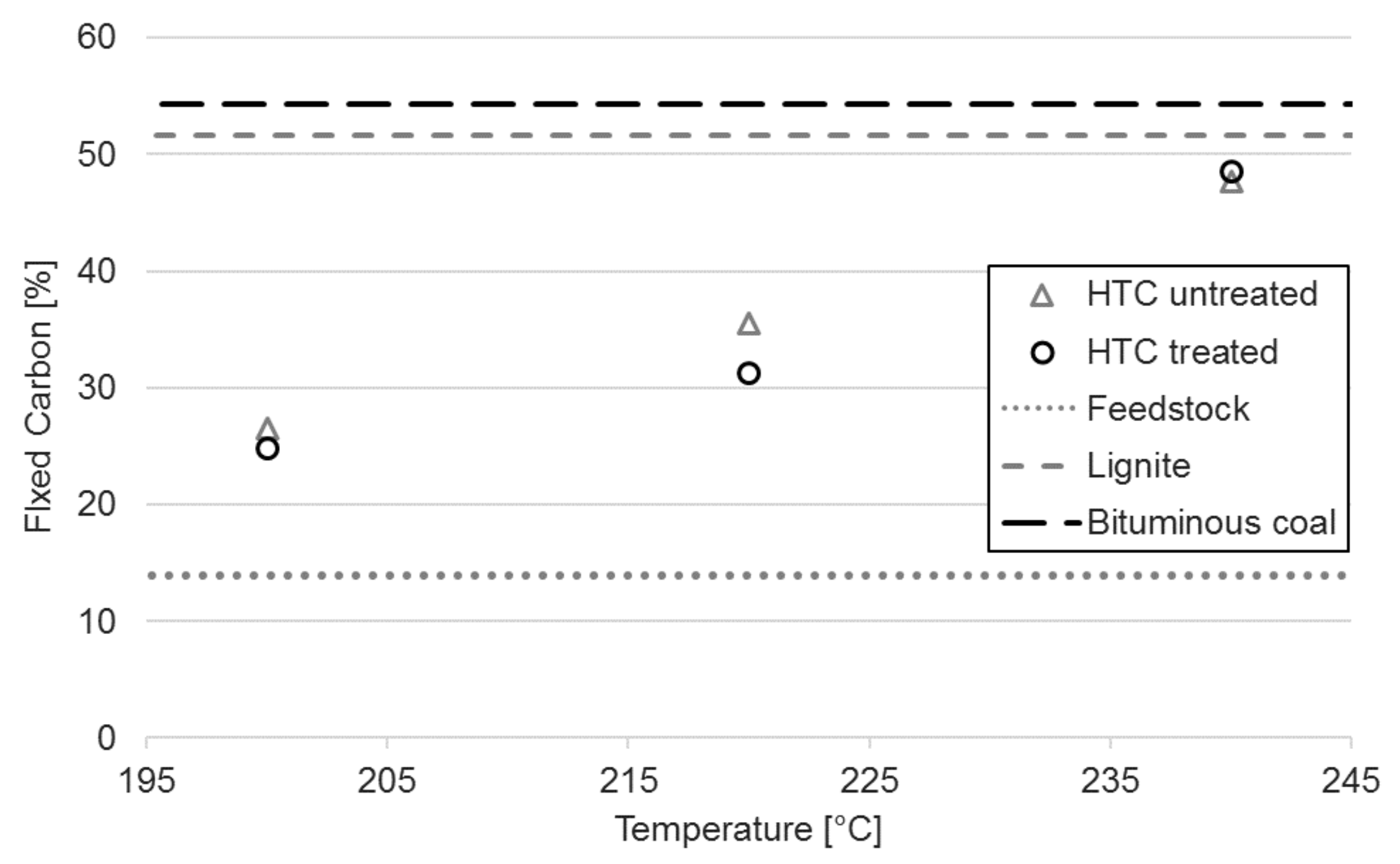
3.2. Proximate Analysis
3.3. Lignocellulosic Composition
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hasan, M.; Haseli, Y.; Karadogan, E. Correlations to predict elemental compositions and heating value of torrefied biomass. Energies 2018, 11, 2443. [Google Scholar] [CrossRef]
- Lucian, M.; Volpe, M.; Fiori, L. Hydrothermal carbonization kinetics of lignocellulosic agro-wastes: Experimental data and modeling. Energies 2019, 12, 516. [Google Scholar] [CrossRef]
- Azadi, P.; Inderwildi, O.R.; Farnood, R.; King, D.A. Liquid fuels, hydrogen and chemicals from lignin: A critical review. Renew. Sustain. Energy Rev. 2013, 21, 506–523. [Google Scholar] [CrossRef]
- Mainka, H.; Hilfert, L.; Busse, S.; Edelmann, F.; Haak, E.; Herrmann, A.S. Characterization of the major reactions during conversion of lignin to carbon fiber. J. Mater. Res. Technol. 2015, 4, 377–391. [Google Scholar] [CrossRef]
- Davis, K.M.; Rover, M.; Brown, R.C.; Bai, X.; Wen, Z.; Jarboe, L.R. Recovery and utilization of lignin monomers as part of the biorefinery approach. Energies 2016, 9, 808. [Google Scholar] [CrossRef]
- Buzala, K.P.; Kalinowska, H.; Malachowska, E.; Boruszewski, P.; Krajewski, K.; Przybysz, P. The effect of lignin content in birch and beech kraft cellulosic pulps on simple sugar yields from the enzymatic hydrolysis of cellulose. Energies 2019, 12, 2952. [Google Scholar] [CrossRef]
- Chen, Z.; Wan, C. Biological valorization strategies for converting lignin into fuels and chemicals. Renew. Sustain. Energy Rev. 2017, 73, 610–621. [Google Scholar] [CrossRef]
- Watkins, D.; Nuruddin, M.; Hosur, M.; Tcherbi-Narteh, A.; Jeelani, S. Extraction and characterization of lignin from different biomass resources. J. Mater. Res. Technol. 2015, 4, 26–32. [Google Scholar] [CrossRef]
- Hansen, N.M.; Plackett, D. Sustainable films and coatings from hemicelluloses: A review. Biomacromolecules 2008, 9, 1493–1505. [Google Scholar] [CrossRef] [PubMed]
- Söderqvist-Lindblad, M.; Ranucci, E.; Albertsson, A.-C. Biodegradable Polymers from Renewable Sources. New Hemicellulose-Based Hydrogels. Macromol. Rapid Commun. 2001, 22, 962–967. [Google Scholar]
- Valoppi, F.; Maina, N.; Allén, M.; Miglioli, R.; Kilpeläinen, P.O.; Mikkonen, K.S. Spruce galactoglucomannan-stabilized emulsions as essential fatty acid delivery systems for functionalized drinkable yogurt and oat-based beverage. Eur. Food Res. Technol. 2019, 245, 1387–1398. [Google Scholar] [CrossRef]
- Mänttäri, M.; Manasrah MAl Strand, E.; Laasonen, H.; Preis, S.; Puro, L.; Xu, C.; Kisonen, V.; Korpinen, R.; Kallioinen, M. Improvement of ultrafiltration performance by oxidation treatment in the recovery of galactoglucomannan from wood autohydrolyzate. Sep. Purif. Technol. 2015, 149, 428–436. [Google Scholar] [CrossRef]
- Kambo, H.; Dutta, A. Strength, storage, and combustion characteristics of densified lignocellulosic biomass produced via torrefaction and hydrothermal carbonization. Appl. Energy 2014, 135, 182–191. [Google Scholar]
- Sermyagina, E. Modelling of Torrefaction and Hydrothermal Carbonization and Heat Integration of Torrefaction with a CHP Plant. Ph.D. Thesis, Lappeenranta University of Technology, Lappeenranta, Finland, 2016. [Google Scholar]
- Mesfun, S.; Matsakas, L.; Rova, U.; Christakopoulos, P. Technoeconomic Assessment of Hybrid Organosolv–Steam Explosion Pretreatment of Woody Biomass. Energies 2019, 12, 4206. [Google Scholar] [CrossRef]
- Libra, J.A.; Ro, K.S.; Kammann, C.; Funke, A.; Berge, N.D.; Neubauer, Y.; Titirici, M.-M.; Fuhner, K.; Bens, O.; Kern, J.; et al. Hydrothermal carbonization of biomass residuals: A comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels 2011, 2, 89–124. [Google Scholar] [CrossRef]
- Bhatt, D.; Shrestha, A.; Dahal, R.; Acharya, B.; Basu, P.; MacEwen, R. Hydrothermal Carbonization of Biosolids from Waste Water Treatment Plant. Energies 2018, 11, 2286. [Google Scholar] [CrossRef]
- Li, Z.; Yi, W.; Li, Z.; Tian, C.; Fu, P.; Zhang, Y.; Teng, J. Preparation of Solid Fuel Hydrochar over Hydrothermal Carbonization of Red Jujube Branch. Energies 2020, 13, 480. [Google Scholar] [CrossRef]
- Atallah, E.; Kwapinski, W.; Ahmad, M.N.; Leahy, J.J.; Al-Muhtaseb, A.H.; Zeaiter, J. Hydrothermal carbonization of olive mill wastewater: Liquid phase product analysis. J. Environ. Chem. Eng. 2019, 7, 102833. [Google Scholar] [CrossRef]
- Fuente-Hernández, A.; Lee, R.; Béland, N.; Zamboni, I.; Lavoie, J.M. Reduction of furfural to furfuryl alcohol in liquid phase over a biochar-supported platinum catalyst. Energies 2017, 10, 286. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, Z.; Liu, Y.; Meng, X.; Qu, J.; Liu, C.; Qu, B. A Review on the Transformation of Furfural Residue for Value-Added Products. Energies 2019, 13, 21. [Google Scholar] [CrossRef]
- Khan, T.A.; Saud, A.S.; Jamari, S.S.; Rahim MH, A.; Park, J.W.; Kim, H.J. Hydrothermal carbonization of lignocellulosic biomass for carbon rich material preparation: A review. Biomass Bioenergy 2019, 130, 105384. [Google Scholar] [CrossRef]
- Zhuang, X.; Zhan, H.; Song, Y.; He, C.; Huang, Y.; Yin, X.; Wu, C. Insights into the evolution of chemical structures in lignocellulose and non-lignocellulose biowastes during hydrothermal carbonization (HTC). Fuel 2019, 236, 960–974. [Google Scholar] [CrossRef]
- Hoekman, S.K.; Broch, A.; Robbins, C.; Zielinska, B.; Felix, L. Hydrothermal carbonization (HTC) of selected woody and herbaceous biomass feedstocks. Biomass Conv. Biorefin. 2013, 3, 113–126. [Google Scholar] [CrossRef]
- Bach, Q.-V.; Skreiberg, O. Upgrading biomass fuels via wet torrefaction: A review and comparison with dry torrefaction. Renew. Sustain. Energy Rev. 2016, 54, 665–677. [Google Scholar] [CrossRef]
- Zhuang, X.; Zhan, H.; Huang, Y.; Song, Y.; Yin, X.; Wu, C. Conversion of industrial biowastes to clean solid fuels via hydrothermal carbonization (HTC): Upgrading mechanism in relation to coalification process and combustion behavior. Bioresour. Technol. 2018, 267, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Sermyagina, E.; Saari, J.; Kaikko, J.; Vakkilainen, E. Hydrothermal carbonization of coniferous biomass: Effect of process parameters on mass and energy yields. J. Anal. Appl. Pyrolysis 2015, 113, 551–556. [Google Scholar] [CrossRef]
- Saha, N.; Saba, A.; Reza, M.T. Effect of hydrothermal carbonization temperature on pH, dissociation constants, and acidic functional groups on hydrochar from cellulose and wood. J. Anal. Appl. Pyrolysis 2019, 137, 138–145. [Google Scholar] [CrossRef]
- Wilk, M.; Magdziarz, A.; Jayaraman, K.; Szymańska-Chargot, M.; Gökalp, I. Hydrothermal carbonization characteristics of sewage sludge and lignocellulosic biomass. A comparative study. Biomass Bioenergy 2019, 120, 166–175. [Google Scholar] [CrossRef]
- Titirici, M.-M.; Funke, A.; Kruse, A. Recent Advances in Thermo-Chemical Conversion of Biomass; Chapter 12—Hydrothermal Carbonization of Biomass; Elsevier B.V.: Waltham, MA, USA, 2015; pp. 325–351. [Google Scholar]
- Kim, D.; Lee, K.; Park, K. Upgrading the characteristics of biochar from cellulose, lignin, and xylan for solid biofuel production from biomass by hydrothermal carbonization. J. Ind. Eng. Chem. 2016, 42, 95–100. [Google Scholar] [CrossRef]
- Borrero-López, A.M.; Masson, E.; Celzard, A.; Fierro, V. Modelling the reactions of cellulose, hemicellulose and lignin submitted to hydrothermal treatment. Ind. Crop. Prod. 2018, 124, 919–930. [Google Scholar] [CrossRef]
- Sheng, K.; Zhang, S.; Liu, J.; Shuang, E.; Jin, C.; Xu, Z.; Zhang, X. Hydrothermal carbonization of cellulose and xylan into hydrochars and application on glucose isomerization. J. Clean. Prod. 2019, 237, 117831. [Google Scholar] [CrossRef]
- Kilpeläinen, P.O.; Hautala, S.S.; Byman, O.O.; Tanner, L.J.; Korpinen, R.I.; Lillandt, M.K.-J.; Pranovich, A.V.; Kitunen, V.H.; Willför, S.M.; Ilvesniemi, H.S. Pressurized hot water flow-through extraction system scale up from the laboratory to the pilot scale. Green Chem. 2014, 16, 3186–3194. [Google Scholar] [CrossRef]
- SFS EN 14775 Solid Biofuels. Determination of Ash Content; Standard; Finnish Standards Association SFS: Helsinki, Finland, 2010. [Google Scholar]
- SFS EN 15148 Solid Biofuels. Determination of the Content of Volatile Matter; Standard; Finnish Standards Association SFS: Helsinki, Finland, 2010. [Google Scholar]
- SFS-EN 14918 Solid Biofuels. Determination of Calorific Value; Standard; Finnish Standards Association SFS: Helsinki, Finland, 2010. [Google Scholar]
- TAPPI Method T264 cm-97; Standard; TAPPI Press: Atlanta, GA, USA, 1997.
- TAPPI T 222 om-11; Standard; TAPPI Press: Atlanta, GA, USA, 2011.
- Biller, P.; Ross, A.B. Handbook of Biofuels Production: Processing and Technologies, 2nd ed.; no. 98; Woodhead Publishing Series in Energy17–Production of biofuels via hydrothermal conversion; Elsevier Ltd., Ch.: Amsterdam, The Netherlands, 2016; pp. 509–547. [Google Scholar]
- Funke, A.; Ziegler, F. Hydrothermal carbonization of biomass: A summary and discussion of chemical mechanisms for process engineering. Biofuels Bioprod. Biorefining 2010, 4, 160–177. [Google Scholar] [CrossRef]
- Donahue, C.J.; Rais, E.A. Proximate analysis of coal. J. Chem. Educ. 2009, 86, 222–224. [Google Scholar] [CrossRef]
- Gordobil, O.; Moriana, R.; Zhang, L.; Labidi, J.; Sevastyanova, O. Assessment of technical lignins for uses in biofuels and biomaterials: Structure-related properties, proximate analysis and chemical modification. Ind. Crop. Prod. 2016, 83, 155–165. [Google Scholar] [CrossRef]
- Broch, A.; Jena, U.; Hoekman, S.K.; Langford, J. Analysis of solid and aqueous phase products from hydrothermal carbonization of whole and lipid-extracted algae. Energies 2014, 7, 62–79. [Google Scholar] [CrossRef]
- Hoekman, S.K.; Broch, A.; Felix, L.; Farthing, W. Hydrothermal carbonization (htc) of loblolly pine using a continuous, reactive twin-screw extruder. Energy Convers. Manag. 2017, 134, 247–259. [Google Scholar] [CrossRef]
- Yan, W.; Acharjee, T.C.; Coronella, C.J.; Vasquez, V.R. Thermal Pretreatment of Lignocellulosic Biomass. Environ. Prog. Sustain. Energy 2009, 28, 435–440. [Google Scholar] [CrossRef]
- Mäkelä, M.; Volpe, M.; Volpe, R.; Fiori, L.; Dahl, O. Spatially resolved spectral determination of polysaccharides in hydrothermally carbonized biomass. Green Chem. 2018, 20, 1114–1120. [Google Scholar] [CrossRef]
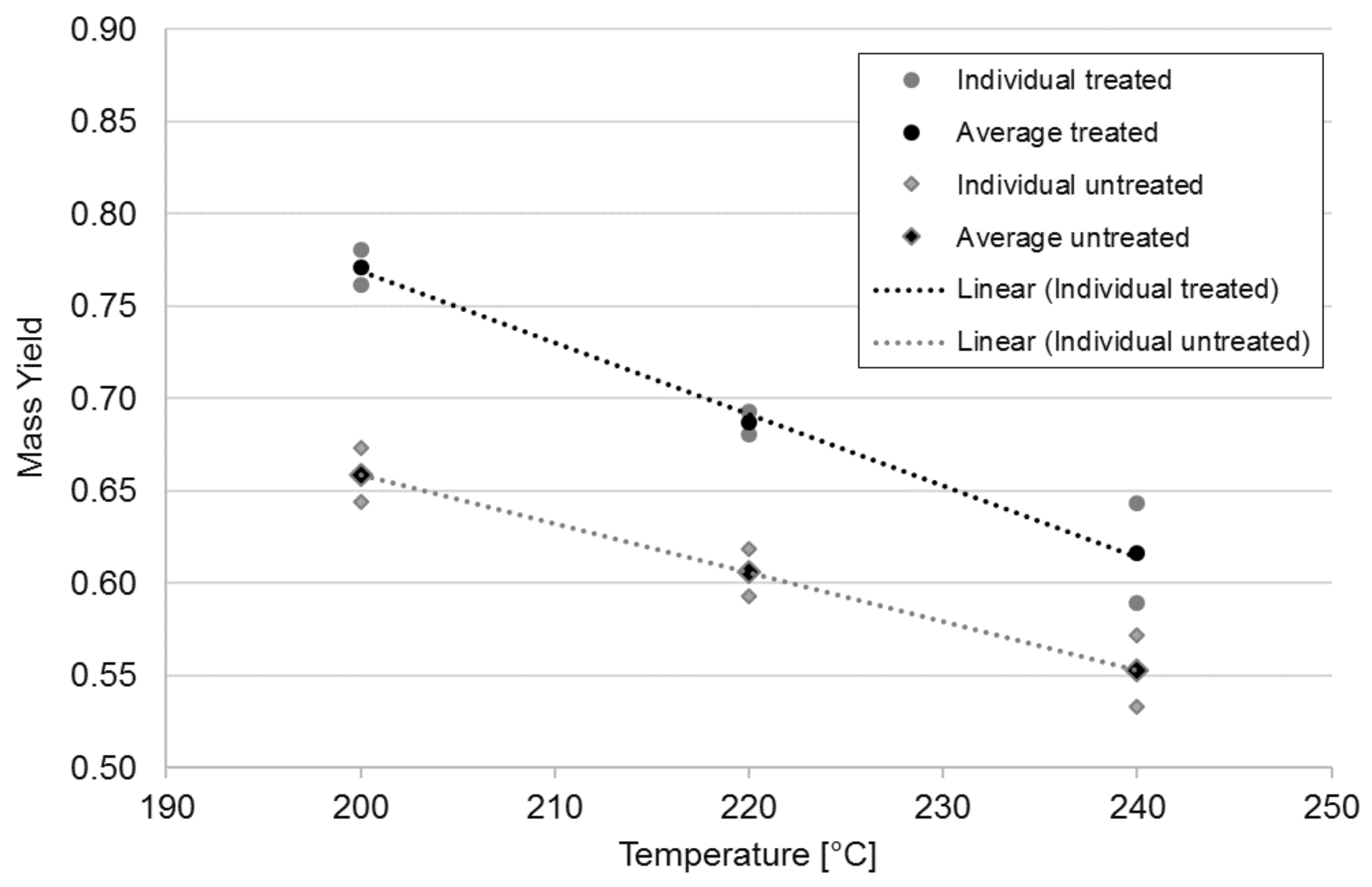
| Samples | Slope | Lower Bound | Upper Bound |
|---|---|---|---|
| treated | −0.00387 | −0.00534 | −0.00239 |
| untreated | −0.00265 | −0.00399 | −0.00131 |
| Sample | Proximate Composition ± Standard Deviations 1 | - | HTC Temperature | ||
|---|---|---|---|---|---|
| Raw | 200 °C | 220 °C | 240 °C | ||
| Untreated | VM [%] | 85.7 ± 1.2 | 73.4 ± 0.9 | 64.4 ± 0.6 | 52.2 ± 0.6 |
| AC [%] | 0.3 ± 0.07 | 0.2 ± 0.01 | 0.1 ± 0.10 | 0.2 ± 0.01 | |
| FC [%] | 14.0 ± 1.2 | 26.5 ± 0.9 | 35.5 ± 0.6 | 47.6 ± 0.6 | |
| HHV [MJ/kg] | 20.77 ± 0.15 | 22.96 ± 0.08 | 24.38 ±0.26 | 27.28 ± 0.08 | |
| Treated | VM [%] | 85.9 ± 0.2 | 75.1 ± 0.8 | 68.6 ± 0.3 | 51.1 ± 1.0 |
| AC [%] | 0.2 ± 0.05 | 0.2 ± 0.03 | 0.1 ± 0.05 | 0.4 ± 0.06 | |
| FC [%] | 13.9 ± 0.2 | 24.7 ± 0.8 | 31.3 ± 0.3 | 48.5 ± 1.0 | |
| HHV [MJ/kg] | 21.60 ± 0.03 | 23.41 ± 0.02 | 25.23 ± 0.02 | 28.27 ± 0.17 | |
| Sample | Chemical Composition (%) ± Standard Deviations 1 | - | HTC Temperature | ||
|---|---|---|---|---|---|
| Raw | 200 °C | 220 °C | 240 °C | ||
| Untreated | Extractives | 3.65 ± 0.15 | 11.16 ± 0.18 | 11.23 ± 0.15 | 11.35 ± 0.08 |
| Insoluble lignin 2 | 30.10 ± 2.20 | 50.66 ± 2.41 | 67.21 ± 4.99 | 87.85 ± 6.96 | |
| Soluble lignin 2 | 0.032 ± 0.003 | 0.027 ± 0.002 | 0.030 ± 0.002 | 0.039 ± 0.003 | |
| Total lignin 2 | 30.13 ± 2.20 | 50.69 ± 2.41 | 67.24 ± 4.99 | 87.89 ± 6.96 | |
| Holocellulose 2 | 66.22 ± 2.21 | 38.15 ± 2.41 | 21.53 ± 5.00 | 0.76 ± 6.96 | |
| Treated | Extractives | 12.11 ± 0.20 | 10.22 ± 0.33 | 12.58 ± 0.07 | 11.50 ± 0.12 |
| Insoluble lignin 2 | 37.44 ± 0.92 | 50.77 ± 2.51 | 63.90 ± 0.06 | 87.34 ± 2.00 | |
| Soluble lignin 2 | 0.066 ± 0.001 | 0.052 ± 0.002 | 0.016 ± 0.002 | 0.032 ± 0.001 | |
| Total lignin 2 | 37.50 ± 0.92 | 50.82 ± 2.51 | 63.92 ± 0.06 | 87.37 ± 2.00 | |
| Holocellulose 2 | 50.39 ± 0.92 | 38.97 ± 2.53 | 23.50 ± 0.10 | 1.13 ± 2.01 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Partridge, A.; Sermyagina, E.; Vakkilainen, E. Impact of Pretreatment on Hydrothermally Carbonized Spruce. Energies 2020, 13, 2984. https://doi.org/10.3390/en13112984
Partridge A, Sermyagina E, Vakkilainen E. Impact of Pretreatment on Hydrothermally Carbonized Spruce. Energies. 2020; 13(11):2984. https://doi.org/10.3390/en13112984
Chicago/Turabian StylePartridge, Anna, Ekaterina Sermyagina, and Esa Vakkilainen. 2020. "Impact of Pretreatment on Hydrothermally Carbonized Spruce" Energies 13, no. 11: 2984. https://doi.org/10.3390/en13112984
APA StylePartridge, A., Sermyagina, E., & Vakkilainen, E. (2020). Impact of Pretreatment on Hydrothermally Carbonized Spruce. Energies, 13(11), 2984. https://doi.org/10.3390/en13112984







