The Effect of Biochar Addition on the Biogas Production Kinetics from the Anaerobic Digestion of Brewers’ Spent Grain
Abstract
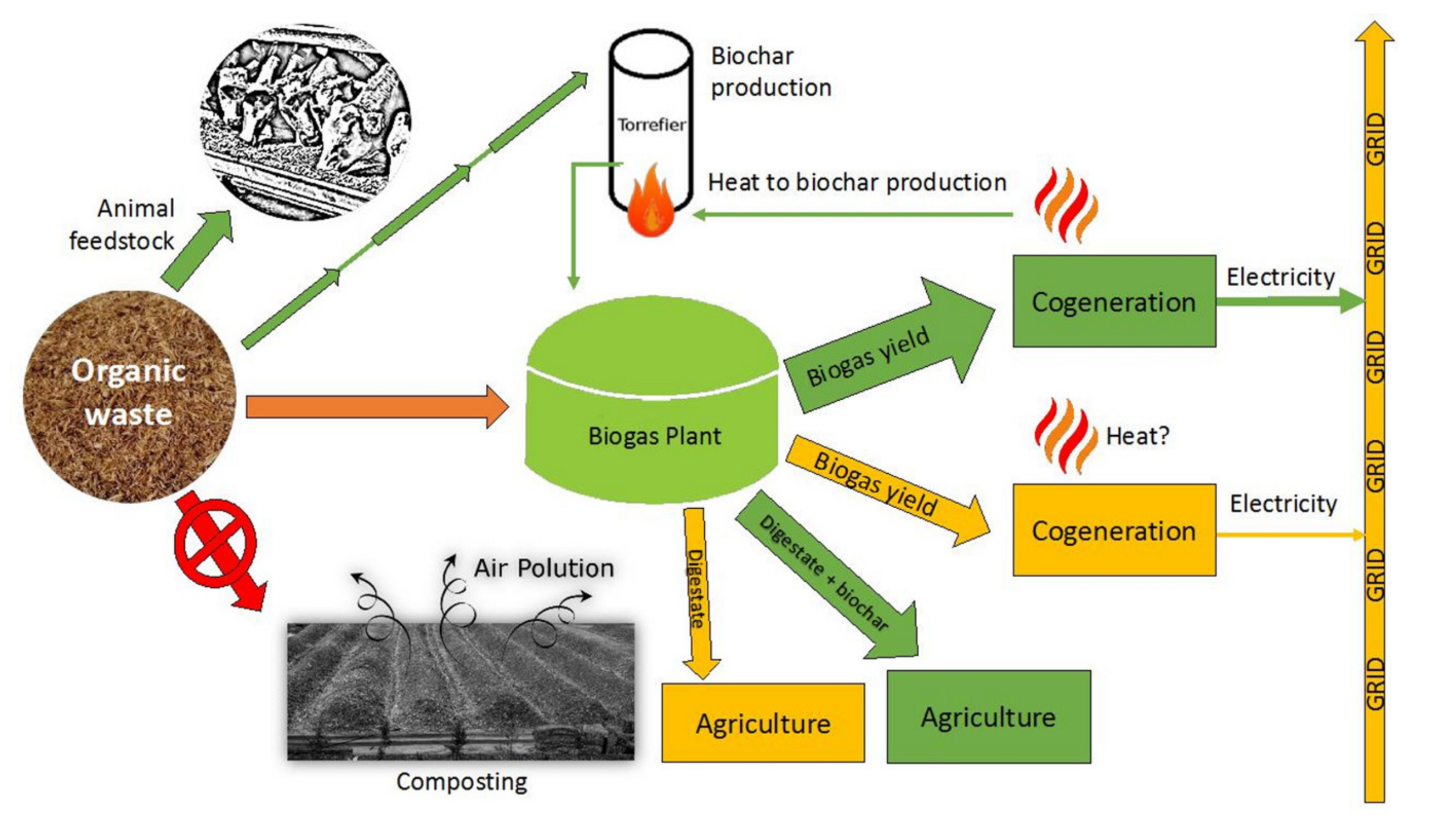
:1. Introduction
2. Materials and Methods
2.1. Used Materials
2.2. Experimental Setup
- incubation time = 21 d,
- incubation temperature = 37 °C,
- the total mass of substrates = 35 g.
2.3. Experimental Design
2.4. Standard Analytical Methods
- Organic matter was determined in accordance with PN-EN 15169:2011 using the muffle furnace (SNOL, model 8.1/1100, Utena, Lithuania),
- Ash content in accordance with the PN-G-04516:1998 standard using the same muffle furnace and Radwag PS 3500.R2 analytical balance,
- Elementary composition of C, H, N, O, and S, using Perkin Elmer 2400 Series CHNS/O with Radwag, MYA 2.4 Y analyzer in accordance with PN-EN 15104: 2011
- Lower heating value and higher heating value by means of the IKA C2000 Basic calorimeter in accordance with the PN-Z-15008-04:1993 standard.
2.5. Data analysis—Assessment of Biogas Production Potential
- ng—the number of moles of gas in the vessel, mol,
- p—accumulated gas pressures in the vessel, Pa,
- Vv—the volume of the vessel, m3
- R—universal gas constant (R = 8.314 J∙mol−1∙K−1), J∙mol−1∙K−1
- T—incubation temperature, K.
- Vg—the volume of biogas produced, m3,
- Vmol—the molar volume of gas under normal conditions (Vmol = 0.0224 m3), m3,
- GB21—the amount of gas produced from 1 kg of dry organic matter (BSG), dm3∙kg−1d.o.m.,
- md.o.m.—dry organic mass in substrates, kg.
- Bt—the amount of biogas obtained from the substrate (BSG) after process time t, dm3∙kg−1d.o.m,
- B0—maximum production of biogas from the substrate, dm3∙kg−1d.o.m,
- k—reaction constant rate, d−1,
- t—time, d.
- r—biogas production rate, dm3∙kg−1d.o.m. d−1,
3. Results
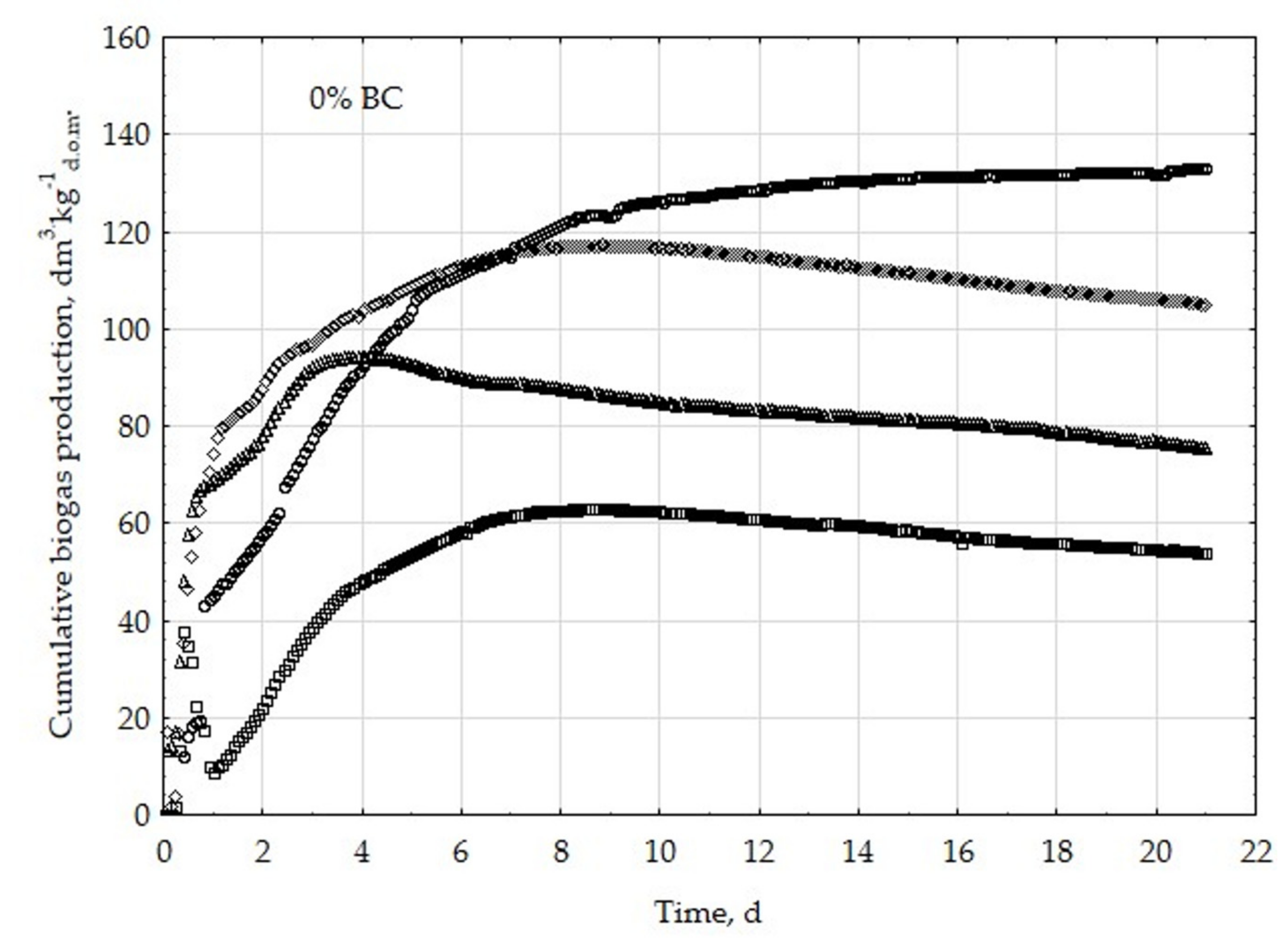
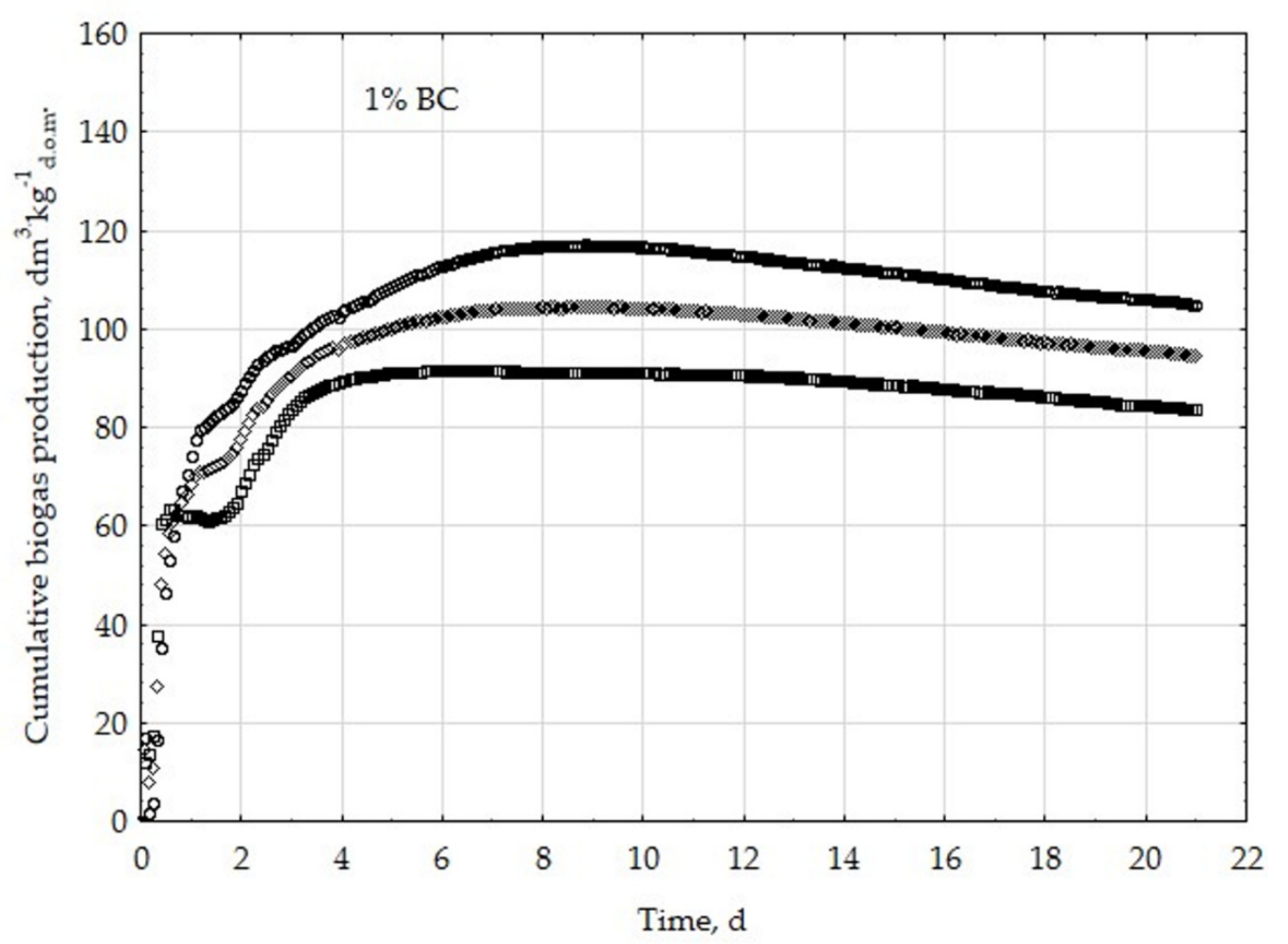
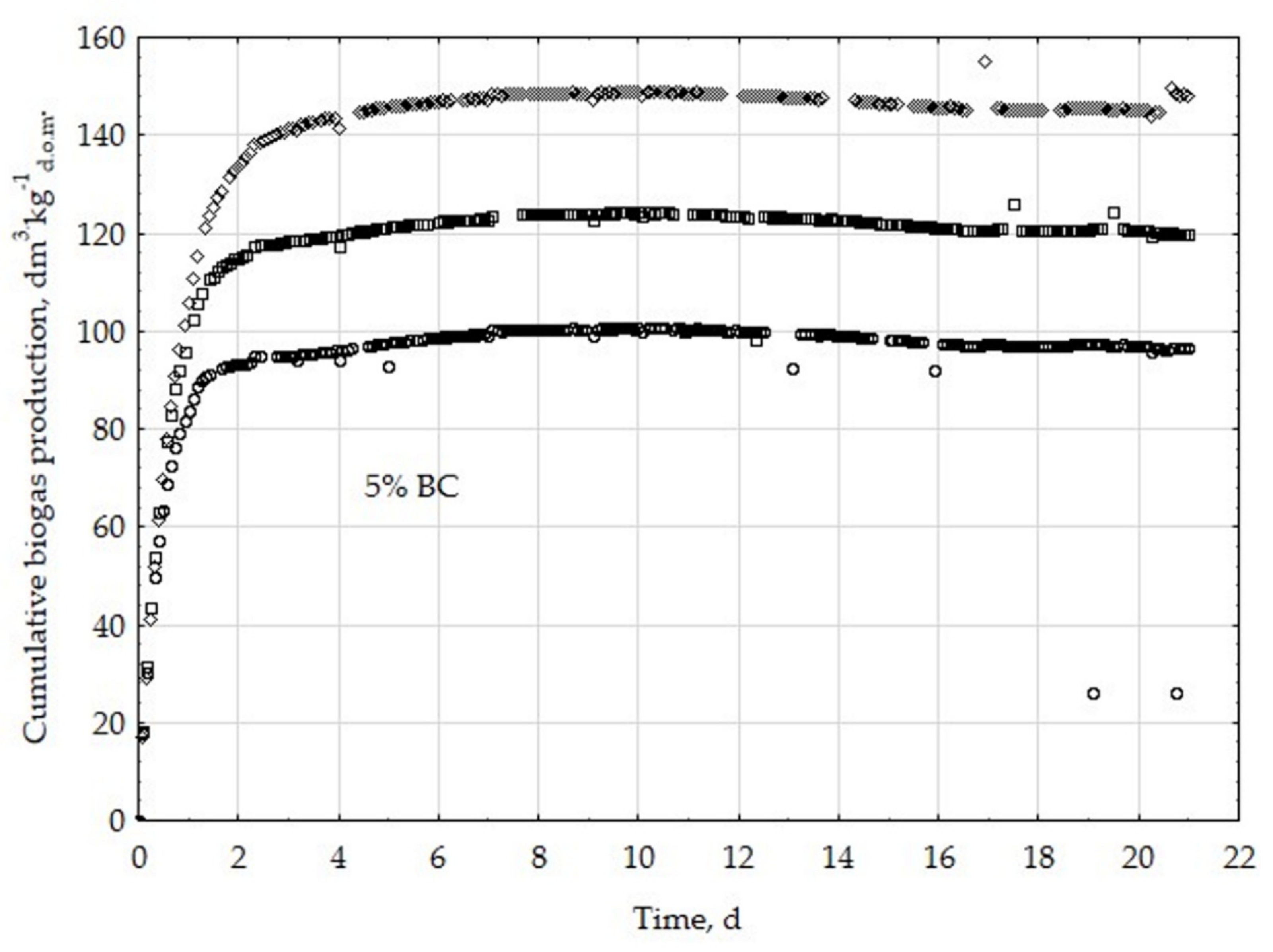
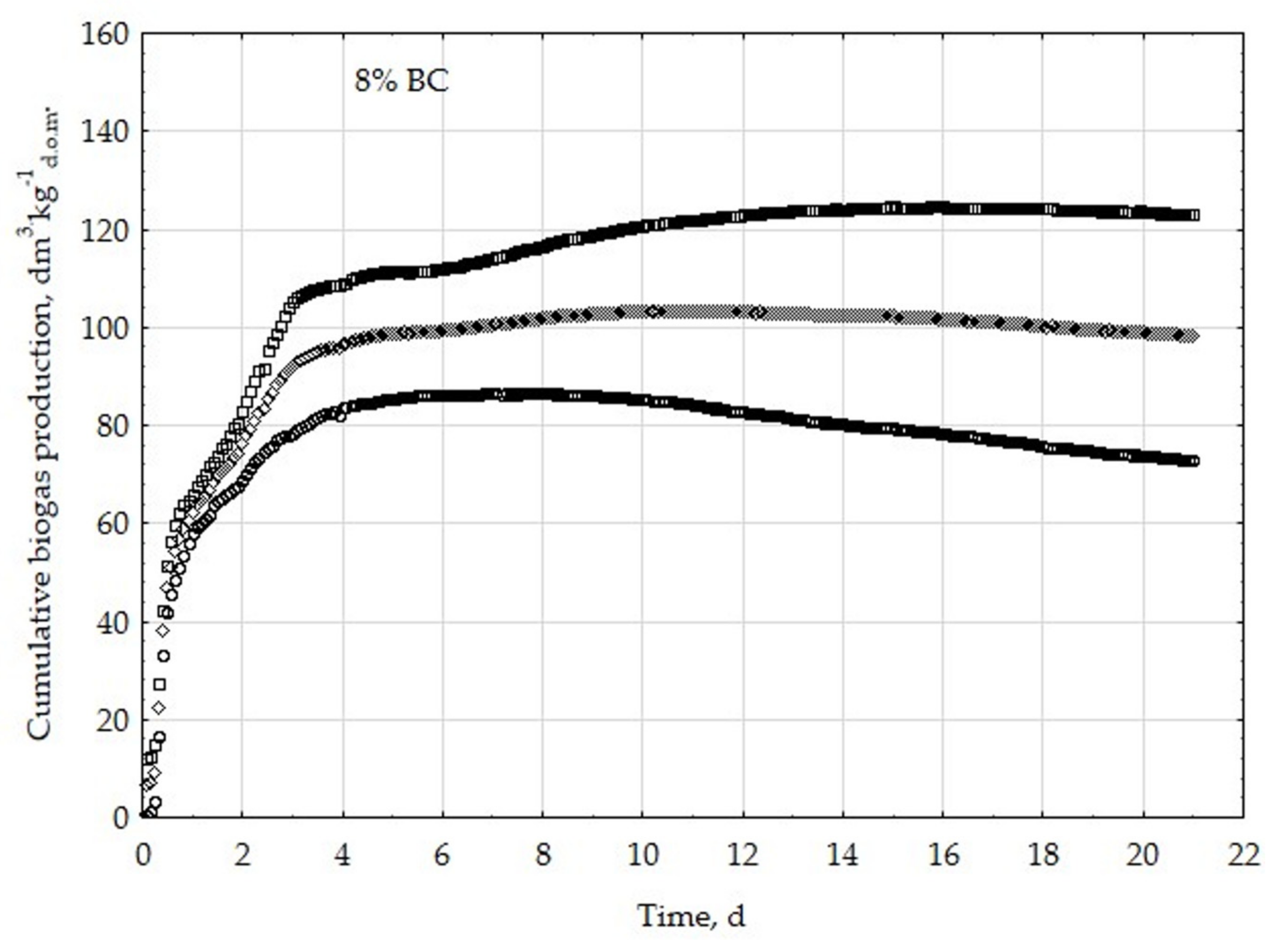
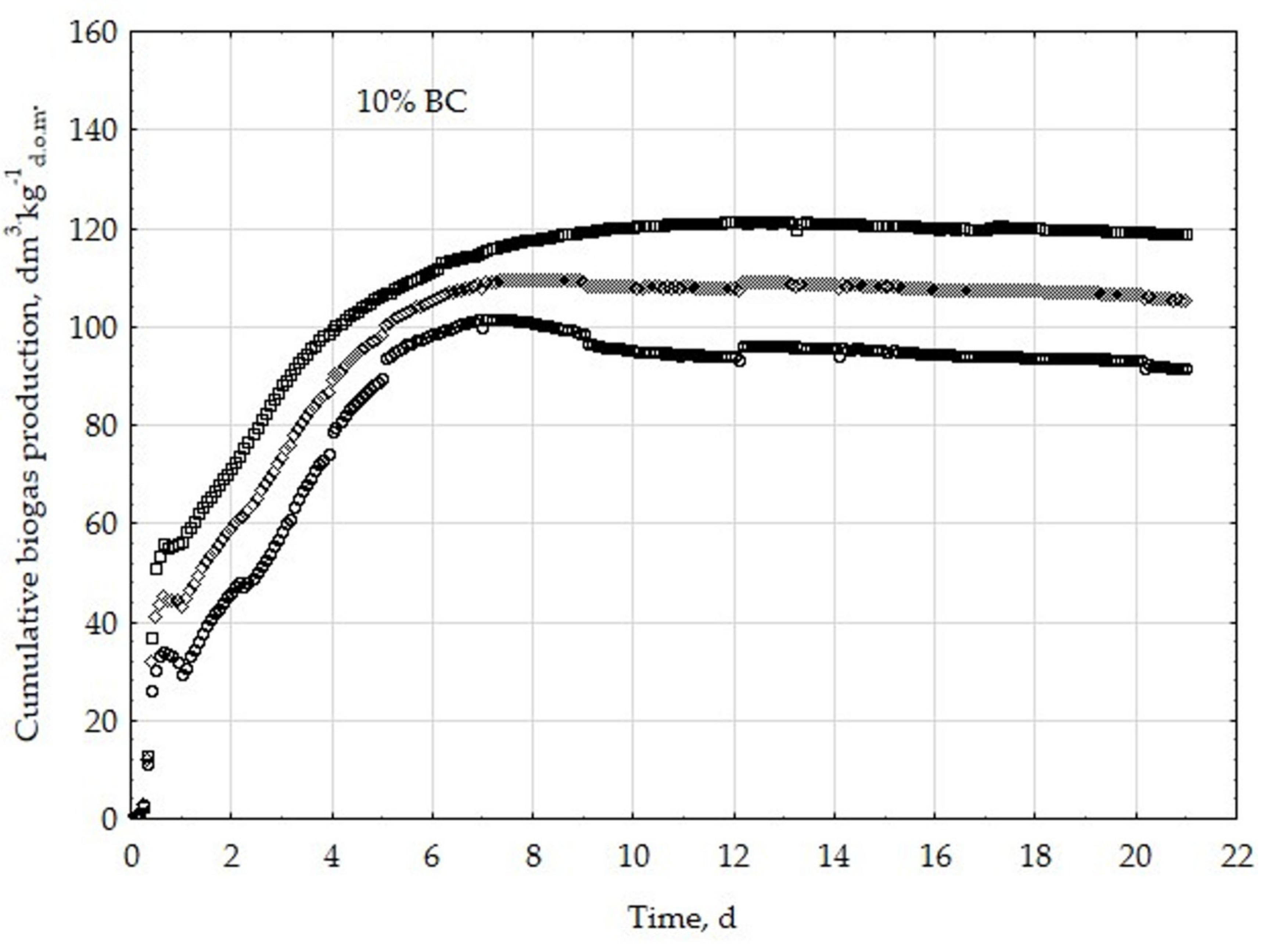
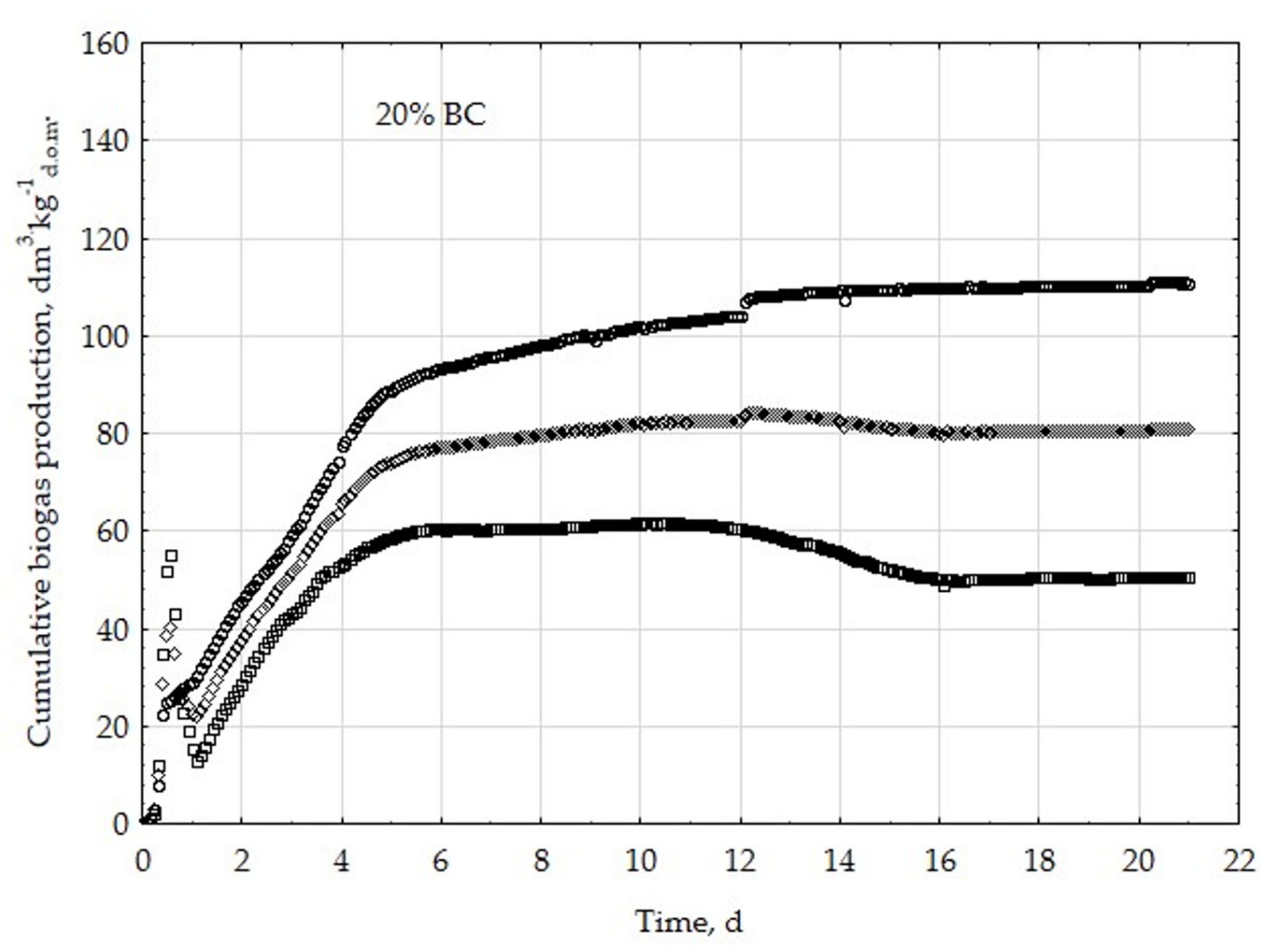
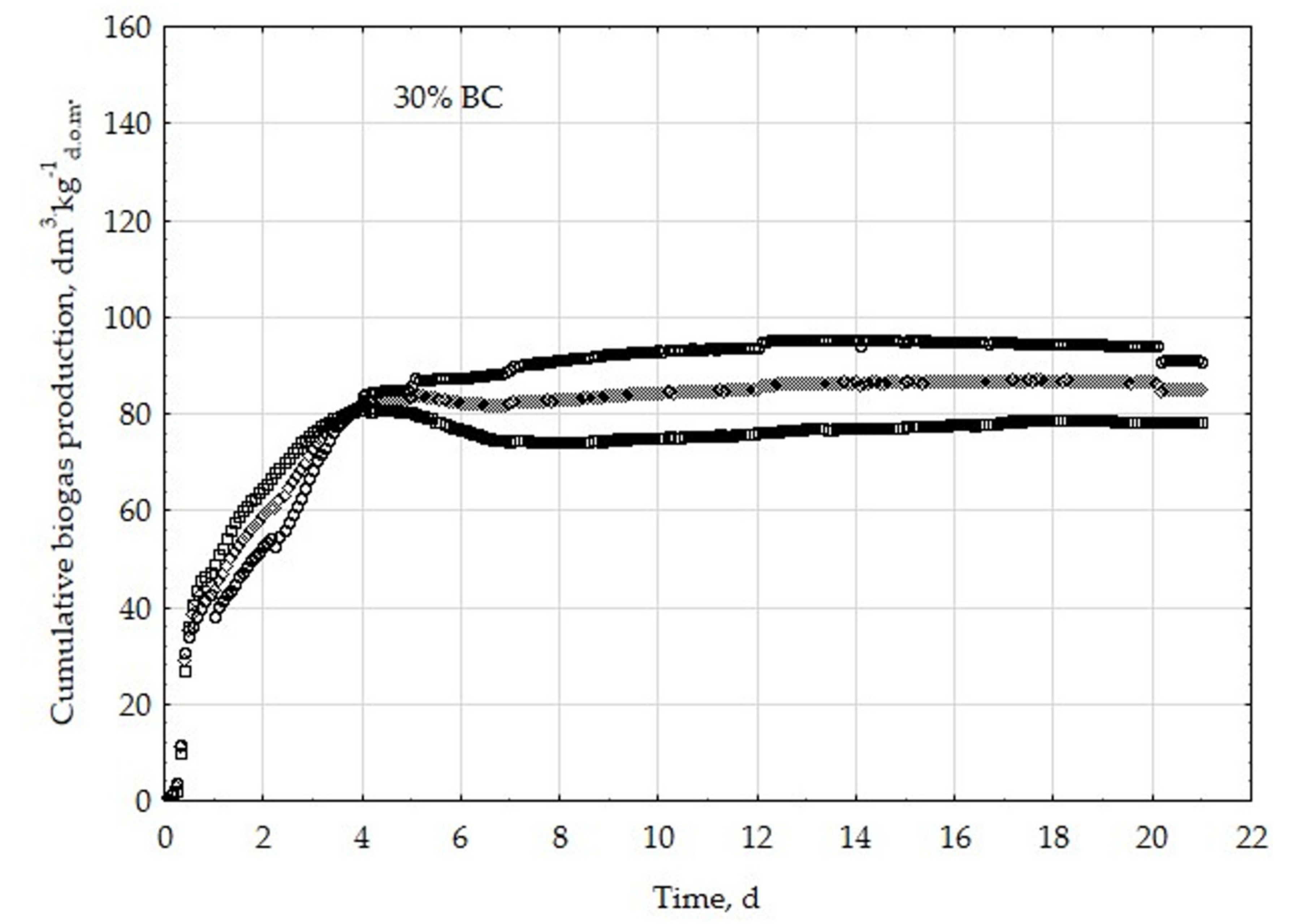
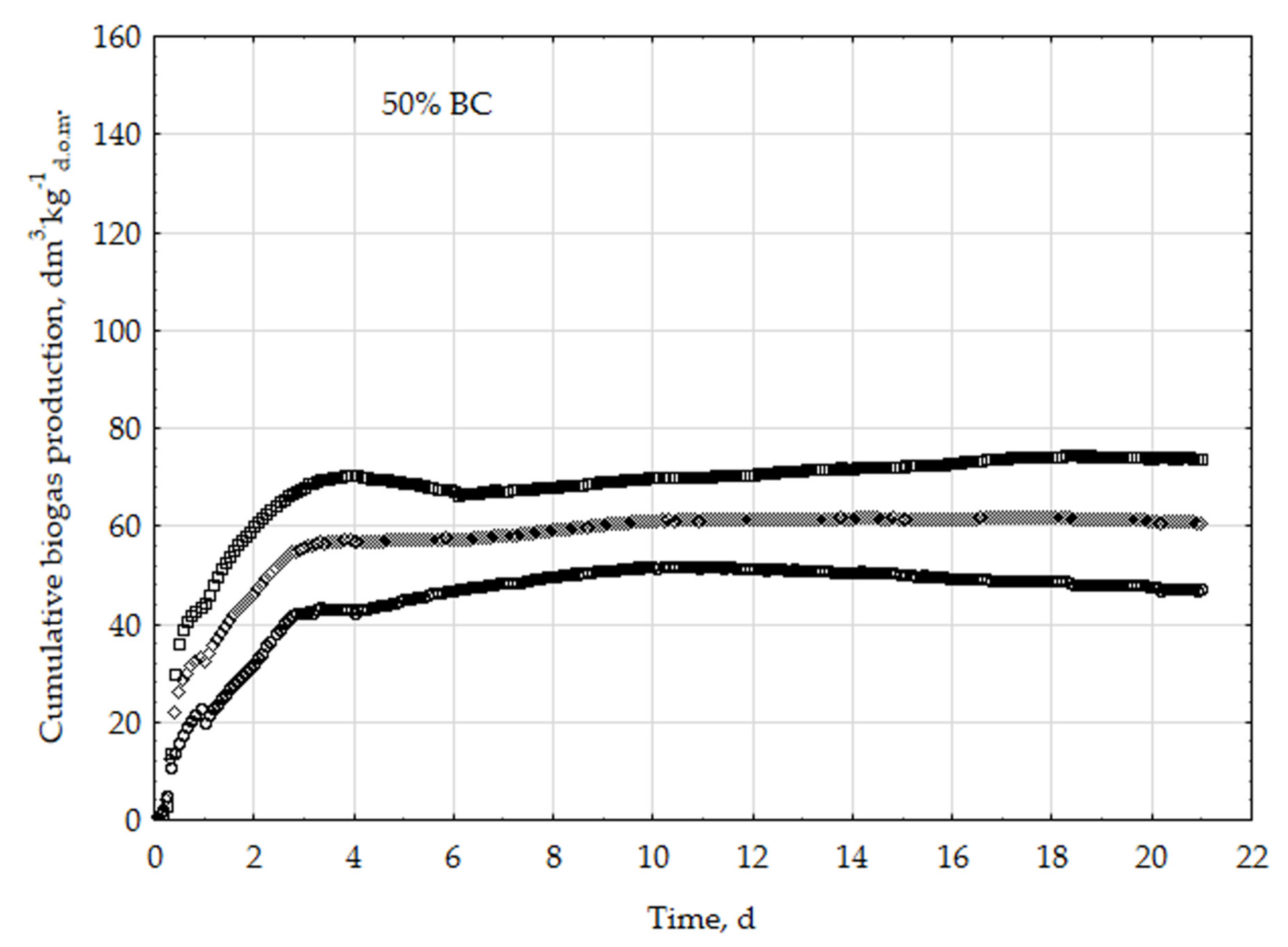
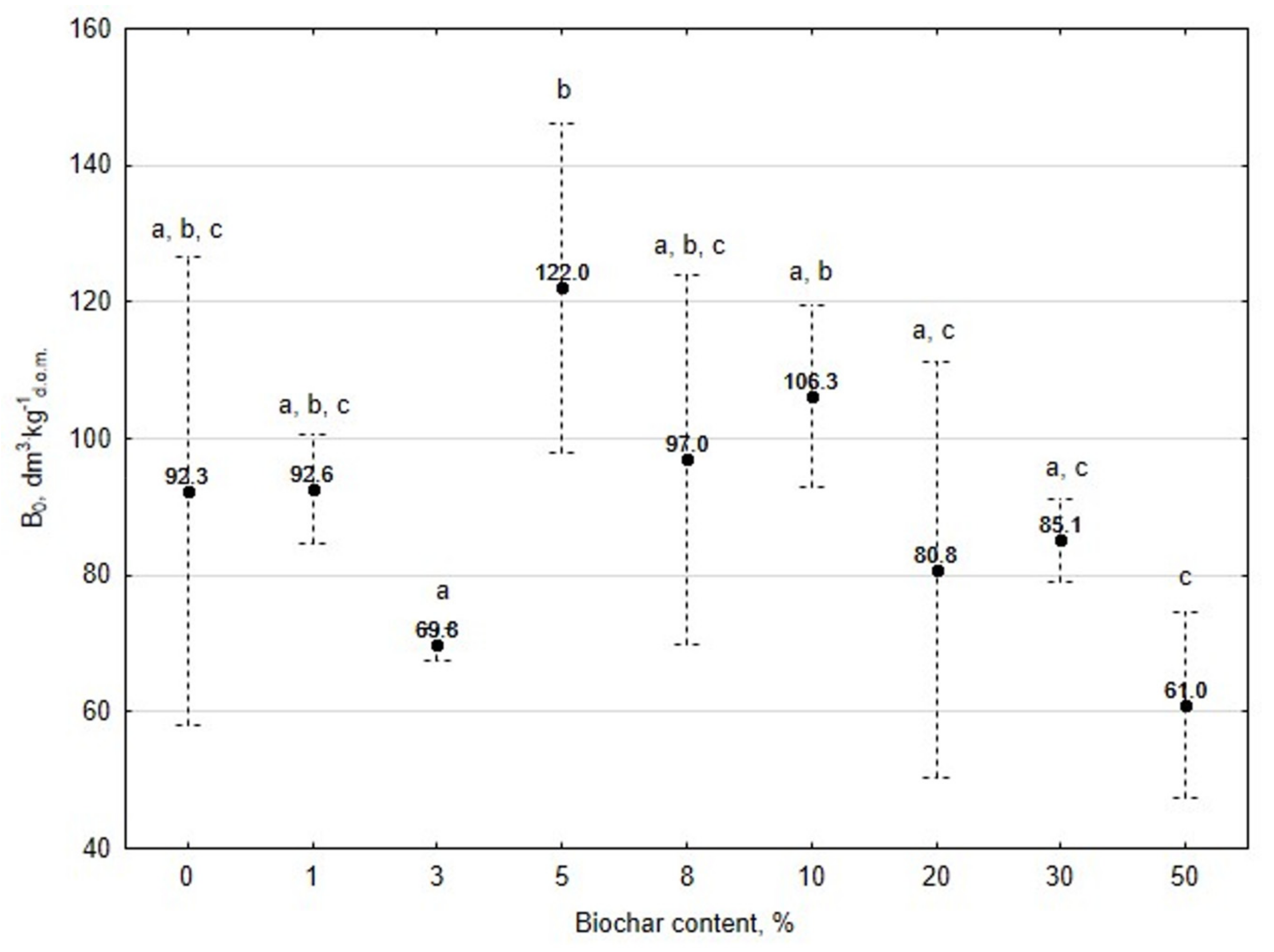
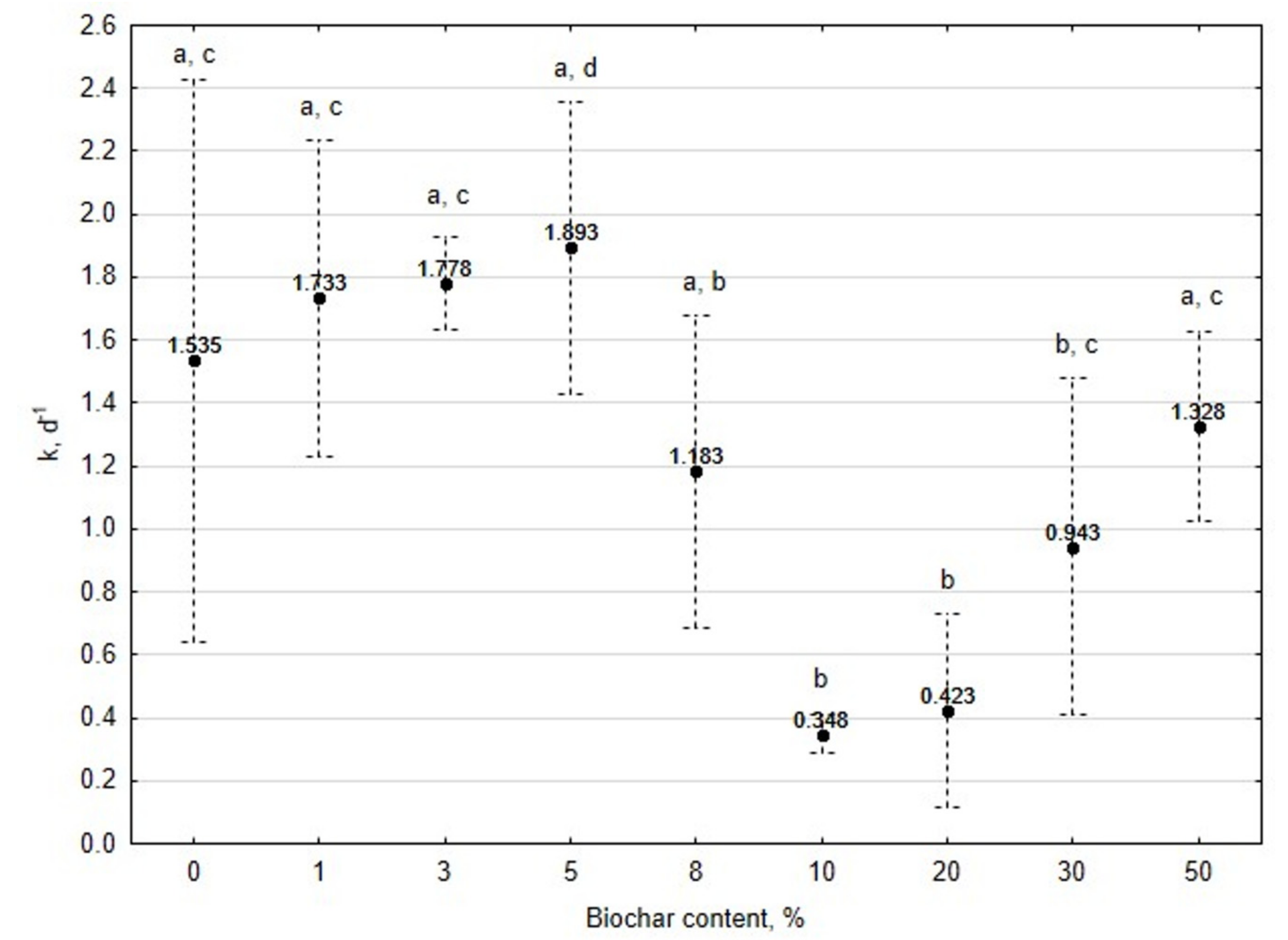
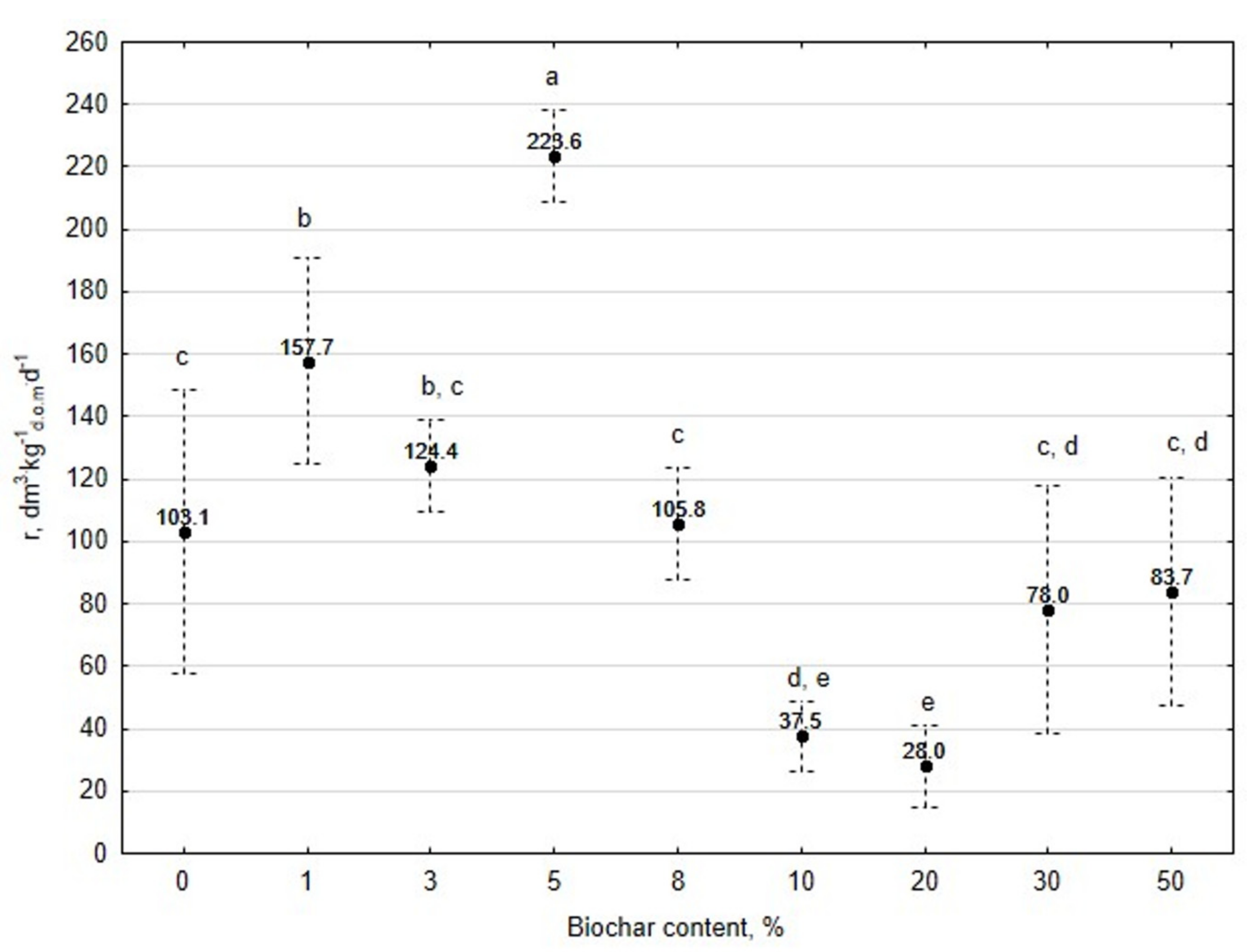
3.1. Assessment of Biogas Production Potential
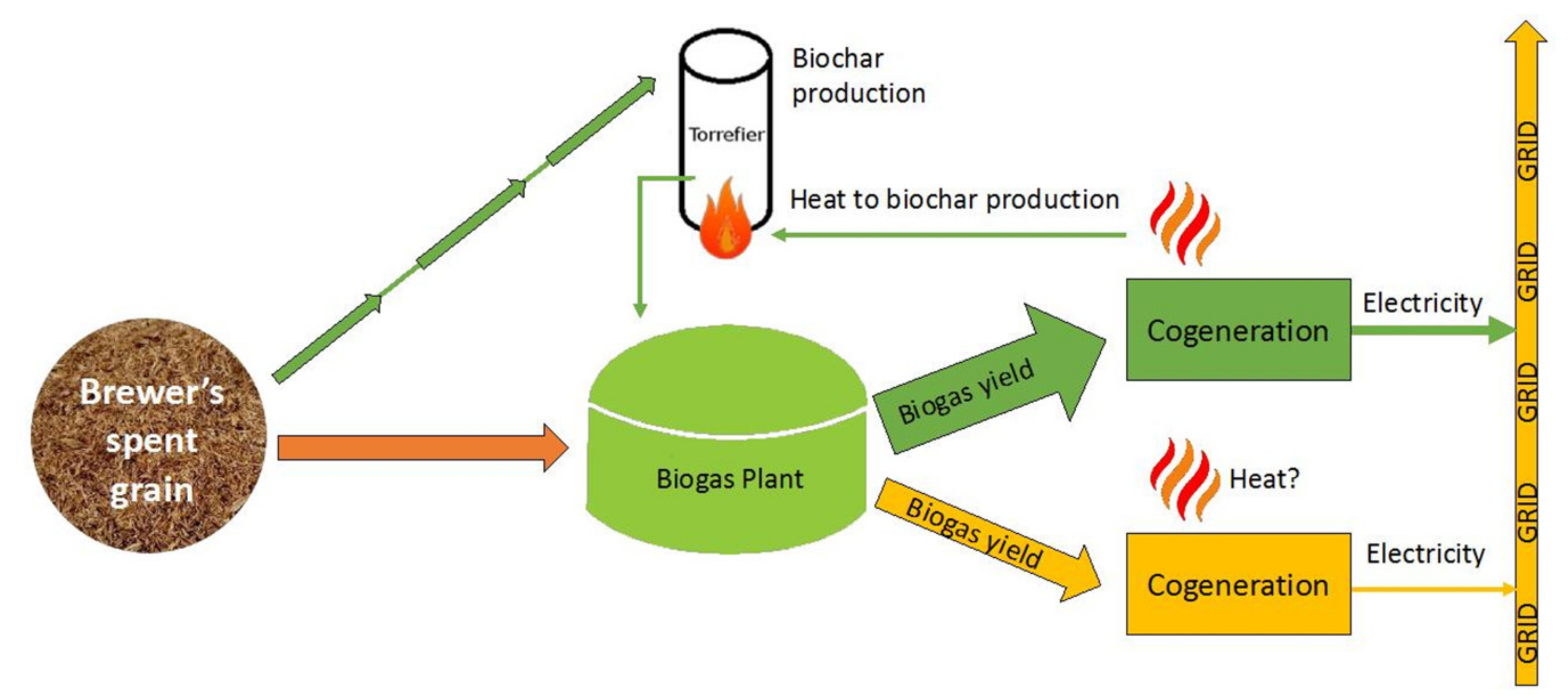
3.2. Simplified Energy Balance Comparing Digestion with and without Biochar from BSG
- Scenario (1): anaerobic digestion of BSG in the presence of BC (obtained with torrefaction of BSG);
- Scenario (2): anaerobic digestion of BSG.
- -
- BSG moisture content = 78.7% (Table 1); 1 ton of BSG contains 787 kg of water, which must be removed.
- -
- The specific heat capacity of water = 4.2 kJ (kg °C)−1;
- -
- The specific heat of BSG = 2.0 kJ (kg °C)−1; the specific heat capacity of BSG has not been reported yet. Thus, we used the specific heat capacity of barley ~1.2 kJ (kg °C)−1 [56] and increased it to be conservative;
- -
- The temperature gradient between room temperature of 20 °C and the water boiling point 100 °C is 80 °C;
- -
- The heat of water evaporation is 2257 kJ kg−1.
3.2.1. Scenario 1: Anaerobic Digestion of BSG in the Presence of BC (Obtained with Torrefaction of BSG)
3.2.2. Scenario 2: Only BSG is Anaerobically Digested
3.3. Changes of Mixtures Properties Due to Anaerobic Digestion
4. Conclusions
- The highest biogas production rate (r) resulted due to the 5% BC addition and it was 227 dm3∙kg−1d.o.m.∙d−1. This production rate was significantly higher (p < 0.05) compared with all other treatments (0, 1, 3, 8, 10, 20, 30, and 50% BC).
- The 5% BC dose resulted in the maximum production of biogas from the substrate (B0) of 122 dm3∙kg−1d.o.m. but it was not significantly different from 0% BC variant. The significant (p < 0.05) inhibition of B0 in comparison to 5% BC was found for variants with 20–50% BC.
- The 5% BC dose resulted in the highest reaction rate constant (k) 1.89 d−1, but it was not significantly different from 0% BC variant. The significant (p < 0.05) decrease of the k was found for variants 10%, 20%, and 30% BC in relation to 5% BC.
- The high biochar addition (20–50% BC) did significantly decrease biogas production in relation to variant 5% BC.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A

References
- Camia, A.; Robert, N.; Jonsson, R.; Pilli, R.; García-Condado, S.; López-Lozano, R.; van der Velde, M.; Ronzon, T.; Gurría, P.; M’Barek, R.; et al. Biomass Production, Supply, Uses and Flows in the European Union. First Results from an Integrated Assessment; EUR 28993 EN; Publications Office of the European Union: Luxembourg, 2018; ISBN 978-92-79-77237-5. [Google Scholar] [CrossRef]
- Čater, M.; Fanedl, L.; Malovrh, Š.; Marinšek Logar, R. Biogas production from brewery spent grain enhanced by bioaugmentation with hydrolytic anaerobic bacteria. Bioresour. Technol. 2015, 186, 261–269. [Google Scholar] [CrossRef]
- Mancini, E.; Arzoumanidis, I.; Raggi, A. Evaluation of potential environmental impacts related to two organic waste treatment options in Italy. Clean. Prod. 2019. [Google Scholar] [CrossRef]
- Zhang, C.; Su, H.; Tan, T. Bioresource Technology Batch and semi-continuous anaerobic digestion of food waste in a dual solid—Liquid system. Bioresour. Technol. 2013, 145, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Dobre, P.; Nicolae, F.; Matei, F. Main factors affecting biogas production—An overview. Roman. Biotechnol. Lett. 2014, 19, 9283–9296. [Google Scholar]
- Panico, A.; d’Antonio, G.; Esposito, G.; Frunzo, L.; Iodice, P.; Pirozzi, P. The Effect of Substrate-Bulk Interaction on Hydrolysis Modeling in Anaerobic Digestion Process. Sustainability 2014, 6, 8348–8363. [Google Scholar] [CrossRef]
- Lorenz, H.; Fischer, P.; Schumacher, B.; Adler, P. Current EU-27 technical potential of organic waste streams for biogas and energy production. Waste Manag. 2013, 33, 2434–2448. [Google Scholar] [CrossRef]
- Pazera, A.; Ślęzak, R.; Krzystek, L.; Ledakowicz, S. The potential of biogas production from food and beverage industry waste. Inżynieria i Aparatura Chemiczna 2015, 54, 194–195. Available online: http://yadda.icm.edu.pl/yadda/element/bwmeta1.element.baztech-a5f79585-17cf-4ada-818b-ab22372a9ecd (accessed on 11 February 2019).
- Photiaades, P. Beer Statistics, 2018th ed.; The Brewers of Europe: Bruxelles, Belgium, 2018; Available online: https://brewersofeurope.org/uploads/mycms-files/documents/publications/2018/EU-beer-statistics-2018-web.pdf (accessed on 11 February 2019).
- Salihu, A.; Muntari, B. Brewer’s spent grain: A review of its potentials and applications. Afr. J. Biotechnol. 2011, 10, 324–331. [Google Scholar] [CrossRef]
- González-García, S.; Morales, P.C.; Gullón, B. Estimating the environmental impacts of a brewery waste—Based biorefinery: Bio-ethanol and xylooligosaccharides joint production case study. Ind. Crop. Prod. 2018, 123, 331–340. [Google Scholar] [CrossRef]
- Klímek, P.; Wimmer, R.; Kumar Mishra, P.; Kúdela, J. Utilizing brewer’s-spent-grain in wood-based particleboard manufacturing. J. Clean. Prod. 2017, 141, 812–817. [Google Scholar] [CrossRef]
- Stojceskaa, V.; Ainswortha, P.; Plunketta, A.; Ibanoglu, S. The recycling of brewer’s processing by-product into ready-to-eat snacks using extrusion technology. J. Cereal Sci. 2008, 47, 469–479. [Google Scholar] [CrossRef]
- Kerby, C.; Vriesekoop, F. An Overview of the Utilisation of Brewery By-Products as Generated by British Craft Breweries. Energies 2017, 3, 24. [Google Scholar] [CrossRef]
- Panjičko, M.; Zupančič, G.D.; Fanedl, L.; Logar, R.M.; Tišma, M.; Zelić, B. Biogas production from brewery spent grain as a mono-substrate in a two-stage process composed of solid-state anaerobic digestion and granular biomass reactors. J. Clean. Prod. 2017, 166, 519–529. [Google Scholar] [CrossRef]
- Meyer-Kohlstock, D.; Haupt, T.; Heldt, E.; Heldt, N.; Kraft, E. Biochar as additive in biogas-production from bio-waste. Energies 2016, 9, 247. [Google Scholar] [CrossRef]
- Weber, K.; Quicker, P. Properties of biochar. Fuel 2018, 217, 240–261. [Google Scholar] [CrossRef]
- Onay, O. Influence of pyrolysis temperature and heating rate on the production of bio-oil and char from safflower seed by pyrolysis, using a well-swept fixed-bed reactor. Fuel Process. Technol. 2007, 88, 523–531. [Google Scholar] [CrossRef]
- Kongkeaw, N.; Patumsawad, S. Thermal Upgrading of Biomass as a Fuel by Torrefaction. In Proceedings of the 2nd International Conference on Environmental Engineering and Applications, Shanghai, China, 19–21 August 2011; Volume 17, pp. 38–42. [Google Scholar]
- Maurer, D.L.; Koziel, J.A.; Kalus, K.; Andersen, D.S.; Opalinski, S. Pilot-scale testing of non-activated biochar for swine manure treatment and mitigation of ammonia, hydrogen sulphide, odorous VOCs, and greenhouse gas emissions. Sustainability 2017, 9, 929. [Google Scholar] [CrossRef]
- Stępień, P.; Białowiec, A. Kinetic parameters of torrefaction process of alternative fuel produced from municipal solid waste and characteristic of carbonized refuse derived fuel. Detritus 2018, 3, 75–83. [Google Scholar] [CrossRef]
- Cha, J.S.; Park, S.H.; Jung, S.C.; Ryu, C.; Jeon, J.K.; Shin, M.C.; Park, Y.K. Production and utilization of biochar: A review. J. Ind. Eng. Chem. 2016, 40, 1–15. [Google Scholar] [CrossRef]
- Rodger, J.-M.; Ganagin, W.; Krieg, A.; Roth, C.; Loewen, A. Steigerung des Biogasertrages durch die Zugabe von Pflanzenkohle (Stimulation of biogas production by adding biochar). Mull und Abfall 2013, 9, 476–481. [Google Scholar]
- Kumar, S.; Jain, M.C.; Chhonkar, P.K. A note on stimulation of biogas production from cattle dung by addition of charcoal. Biol. Wastes 1987, 20, 209–215. [Google Scholar] [CrossRef]
- Inthapanya, S.; Preston, T.R.; Leng, R.A. Biochar increases biogas production in a batch digester charged with cattle manure. Livest. Res. Rural Dev. 2012, 24, 212. [Google Scholar]
- Inthapanya, S.; Preston, T.R. Biochar marginally increases biogas production but decreases methane content of the gas in continuous-flow biodigesters charged with cattle manure. Livest. Res. Rural Dev. 2013, 25, 3869. [Google Scholar]
- Mumme, J.; Srocke, F.; Heeg, K.; Werner, M. Use of biochars in anaerobic digestion. Bioresour. Technol. 2014, 164, 189–197. [Google Scholar] [CrossRef]
- Libra, J.A.; Ro, K.S.; Kammann, C.; Funke, A.; Berge, N.D.; Neubauer, Y.; Titirici, M.M.; Fühner, C.; Bens, O.; Kern, J.; et al. Hydrothermal carbonization of biomass residuals: A comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels 2011, 2, 89–124. [Google Scholar] [CrossRef]
- Białowiec, A.; Pulka, J.; Gołaszewski, J.; Manczarski, P.; Stępień, P. The RDF torrefaction: An effect of temperature on characterization of the product—Carbonized Derived Fuel. Waste Manag. 2017, 70, 91–100. [Google Scholar] [CrossRef]
- Stępień, P.; Pulka, J.; Serowik, M.; Białowiec, A. Thermogravimetric and calorimetric characteristics of alternative fuel in terms of its use in low-temperature pyrolysis. Waste Biomass Valoriz. 2018. [Google Scholar] [CrossRef]
- Białowiec, A.; Micuda, M.; Koziel, J.A. Waste to carbon: Densification of torrefied refuse-derived fuels. Energies 2018, 11, 3233. [Google Scholar] [CrossRef]
- Pulka, J.; Wiśniewski, D.; Gołaszewski, J.; Białowiec, A. Is the biochar produced from sewage sludge a good quality solid fuel? Arch. Environ. Prot. 2016, 42, 125–134. [Google Scholar] [CrossRef]
- Pulka, J.; Manczarski, P.; Koziel, J.A.; Białowiec, A. Torrefaction of sewage sludge: Kinetics and fuel properties of biochars. Energies 2019, 12, 565. [Google Scholar] [CrossRef]
- Luo, C.; Lü, F.; Shao, L.; He, P. Application of eco-compatible biochar in anaerobic digestion to relieve acid stress and promote the selective colonization of functional microbes. Water Res. 2015, 68, 710–718. [Google Scholar] [CrossRef] [PubMed]
- Ö-NORM S 2027-3. Beurteilung von Abfällen aus der Mechanisch-Biologischen Behandlung. Teil 3: Stabilitätsparameter—Gasbildung im Gärtest (GB21); Austrian Standards Institute: Vienna, Austria, 2012. [Google Scholar]
- Agopsowicz, M.; Białowiec, A.; Radziemska, M. Municipal waste disposal in energetic piles in SWECO technology—Seven years of operation and what now? Arch. Environ. Prot. 2006, 32, 55–66. [Google Scholar]
- Bernat, K.; Białowiec, A.; Wojnowska-Baryła, I. The co-fermentation in sewage sludge digesters. Arch. Environ. Prot. 2008, 34, 103–114. [Google Scholar]
- Luna-del Risco, M.; Normak, A.; Orupoõld, K. Biochemical methane potential of different organic wastes and energy crops from Estonia. Agron. Res. 2011, 9, 331–342. [Google Scholar]
- Lay, J.; Li, Y.; Noike, T. Interaction between homoacetogens and methanogens in lake sediments. J. Ferment. Bioeng. 1998, 86, 467–471. [Google Scholar] [CrossRef]
- Achinas, S.; Euverink, G.J.W. Effect of Combined Inoculation on Biogas Production from Hardly Degradable Material. Energies 2019, 12, 217. [Google Scholar] [CrossRef]
- Ariunbaatar, J.; Panico, A.; Frunzo, L.; Esposito, G.; Lens, P.N.; Pirozzi, F. Enhanced anaerobic digestion of food waste by thermal and ozonation pretreatment methods. J. Environ. Manag. 2014, 146, 142–149. [Google Scholar] [CrossRef]
- Carrere, H.; Dumas, C.; Battimeli, A.; Batstone, D.J.; Delgenes, J.P.; Steyer, J.P.; Ferrer, I. Pretreatment methods to improve sludge anaerobic degradability: A review. Hazard. Mater. 2010, 183, 1–15. [Google Scholar] [CrossRef]
- Vavillin, V.A.; Fernandez, B.; Palatsi, J.; Flotats, X. Hydrolysis kinetics in anaerobic degradation or particulate organic material: An overview. Waste Manag. 2008, 28, 939–951. [Google Scholar] [CrossRef]
- Forssell, P.; Kontkanen, H.; Schols, H.A.; Hinz, S.; Eijsink, V.G.H.; Treimo, J.; Robertson, J.A.; Waldron, K.W.; Faulds, C.B.; Buchert, B. Hydrolysis of Brewers’ Spent Grain by Carbohydrate Degrading Enzymes. J. Inst. Brew. 2008, 114, 306–314. [Google Scholar] [CrossRef]
- Lynch, K.M.; Steffen, E.J.; Arendt, E.K. Brewers’ spent grain: A review with an emphasis on food and health. J. Inst. Brew. 2016, 122, 553–568. [Google Scholar] [CrossRef]
- Bougrier, C.; Delgenes, J.P.; Carrere, H. Effects of thermal treatments on five different waste activated sludge samples solubilisation, physical properties and anaerobic digestion. Chem. Eng. J. 2008, 139, 236–244. [Google Scholar] [CrossRef]
- Protot, A.; Julien, L.; Christophe, D.; Partick, L. Sludge disintegration during heat treatment at low temperature: A better understanding of involved mechanisms with a multi-parametric approach. Biochem. Eng. J. 2011, 54, 178–184. [Google Scholar] [CrossRef]
- Appels, L.; Degreve, J.; van der Bruggen, B.; van Impe, J.; Dewil, R. Influence of low temperature thermal pre-treatment on sludge solubilization, heavy metal release and anaerobic digestion. Bioresour. Technol. 2010, 101, 5743–5748. [Google Scholar] [CrossRef] [PubMed]
- Breure, A.M.; Mooijman, K.A.; van Andel, J.G. Protein degradation in anaerobic digestion: Influence of volatile fatty acids and carbohydrates on hydrolysis and acidogenic fermentation of gelatin. Appl. Microbiol. Biotechnol. 1986, 24, 426–431. [Google Scholar] [CrossRef]
- Weiss, I.M.; Muth, C.; Drumm, R.; Kirchner, H.O.K. Thermal decomposition of the amino acids glycine, cysteine, aspartic acid, asparagine, glutamic acid, glutamine, arginine and histidine. BMC Biophys. 2018, 11. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.H.; Chu, Y.S.; Liu, J.L.; Chang, J.S. Thermal degradation of carbohydrates, proteins and lipids in microalgae analyzed by evolutionary computation. Energy Convers. Manag. 2018, 160, 209–219. [Google Scholar] [CrossRef]
- Lyu, H.; He, Y.; Tang, J.; Hecker, M.; Liu, Q.; Jones, P.D.; Codling, G.; Giesy, J.P. Effect of pyrolysis temperature on potential toxicity of biochar if applied to the environment. Environ. Pollut. 2016, 218, 1–7. [Google Scholar] [CrossRef]
- Stelmach, E.; Stelmach, J.; Krzystek, L.; Ledakowicz, S. Wpływ zawartości węgla i azotu na szybkość produkcji biogazu z organicznej frakcji stałych odpadów komunalnych. Inżynieria i Aparatura Chemiczna 2010, 49, 107–108. [Google Scholar]
- Pan, J.; Liu, J.M.X.; Zhai, L.; Ouyang, X.; Liu, H. Effects of different types of biochar on the anaerobic digestion of chicken manure. Bioresour. Technol. 2019, 275, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Stelt, M.J.C. Chemistry and Reaction Kinetics of BIOWASTE Torrefaction Eindhoven; Technische Universiteit Eindhoven: Eindhoven, The Netherlands, 2011. [Google Scholar]
- Otten, L.; Saman, G. Determination of the specific heat of agricultural materials: Part II. Experimental results. Can. Agric. Eng. 1980, 22, 25–27. [Google Scholar]

| Measured Property | Brewer’s Spent Grain (BSG) | Biochar from BSG (BC) | Digestate (D) (Used for Inoculation) |
|---|---|---|---|
| Moisture content in wet BSG, % | 78.7 ± 3.21 | - | - |
| Dry mass content, % | 100 (dried) | 100 (torrefied) | 3.10 ± 0.27 |
| Organic metter content *, % | 96.90 ± 0.80 | 93.30 ± 0.70 | 70.40 ± 0.60 |
| Ash content *, % | 3.10 ± 0.80 | 6.70 ± 0.70 | 29.60 ± 0.60 |
| C content *, % | 50.47 ± 1.56 | 55.90 ± 0.75 | 35.60 ± 0.82 |
| H content *, % | 7.17 ± 0.06 | 5.78 ± 0.17 | 4.30 ± 0.05 |
| N content *, % | 3.63 ± 0.09 | 4.58 ± 0.23 | 4.04 ± 0.23 |
| S content *, % | 0.24 ± 0.01 | 0.23 ± 0.00 | 0.65 ± 0.02 |
| O content *, % | 34.37 ± 0.71 | 24.58 ± 3.28 | 25.81 ± 0.60 |
| Higher heating value*, MJ·kg−1 | 20.00 ± 0.30 | 24.90 ± 0.30 | 16.00 ± 0.20 |
| Lower heating value, MJ·kg−1 | 17.74 ± 0.25 | 23.6 ± 0.33 | 15.00 ± 0.24 |
| Components | Variant (Mass, g) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| D (Control) | S0 | S1 | S3 | S5 | S8 | S10 | S20 | S30 | S50 | |
| D * | 30.0000 | 29.7500 | 29.7500 | 29.7500 | 29.7500 | 29.7500 | 29.7500 | 29.7500 | 29.7500 | 29.7500 |
| BSG ** | 0.0000 | 5.2500 | 5.1975 | 5.0925 | 4.9875 | 4.8300 | 4.72500 | 4.2000 | 3.6800 | 2.6250 |
| BC ** | 0.0000 | 0.0000 | 0.0525 | 0.1575 | 0.2625 | 0.4200 | 0.52500 | 1.0500 | 1.5700 | 2.6250 |
| Parameter | Variant | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| S0 | S1 | S3 | S5 | S8 | S10 | S20 | S30 | S50 | |
| Initial OM * content, % | 96.2 ± 0.75 | 96.1 ± 0.75 | 96.1 ± 0.75 | 96.0 ± 0.74 | 95.9 ± 0.74 | 95.8 ± 0.74 | 95.5 ± 0.74 | 95.1 ± 0.73 | 94.4 ± 0.72 |
| Final OM * content, % | 91.41 ± 2.78 | 92.21 ± 2.39 | 91.00 ± 1.49 | 90.98 ± 0.27 | 90.86 ± 0.46 | 90.60 ± 0.23 | 90.66 ± 0.51 | 90.78 ± 0.27 | 90.64 ± 0.41 |
| Relative change, % | 5.21 | 4.26 | 5.56 | 5.51 | 5.54 | 5.76 | 5.31 | 4.78 | 4.18 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dudek, M.; Świechowski, K.; Manczarski, P.; Koziel, J.A.; Białowiec, A. The Effect of Biochar Addition on the Biogas Production Kinetics from the Anaerobic Digestion of Brewers’ Spent Grain. Energies 2019, 12, 1518. https://doi.org/10.3390/en12081518
Dudek M, Świechowski K, Manczarski P, Koziel JA, Białowiec A. The Effect of Biochar Addition on the Biogas Production Kinetics from the Anaerobic Digestion of Brewers’ Spent Grain. Energies. 2019; 12(8):1518. https://doi.org/10.3390/en12081518
Chicago/Turabian StyleDudek, Marta, Kacper Świechowski, Piotr Manczarski, Jacek A. Koziel, and Andrzej Białowiec. 2019. "The Effect of Biochar Addition on the Biogas Production Kinetics from the Anaerobic Digestion of Brewers’ Spent Grain" Energies 12, no. 8: 1518. https://doi.org/10.3390/en12081518
APA StyleDudek, M., Świechowski, K., Manczarski, P., Koziel, J. A., & Białowiec, A. (2019). The Effect of Biochar Addition on the Biogas Production Kinetics from the Anaerobic Digestion of Brewers’ Spent Grain. Energies, 12(8), 1518. https://doi.org/10.3390/en12081518








