The Use of Acidic Hydrolysates after Furfural Production from Sugar Waste Biomass as a Fermentation Medium in the Biotechnological Production of Hydrogen
Abstract
1. Introduction
2. Materials and Methods
2.1. Biological Material
2.1.1. Sugar Beet Pulp
2.1.2. Inoculum
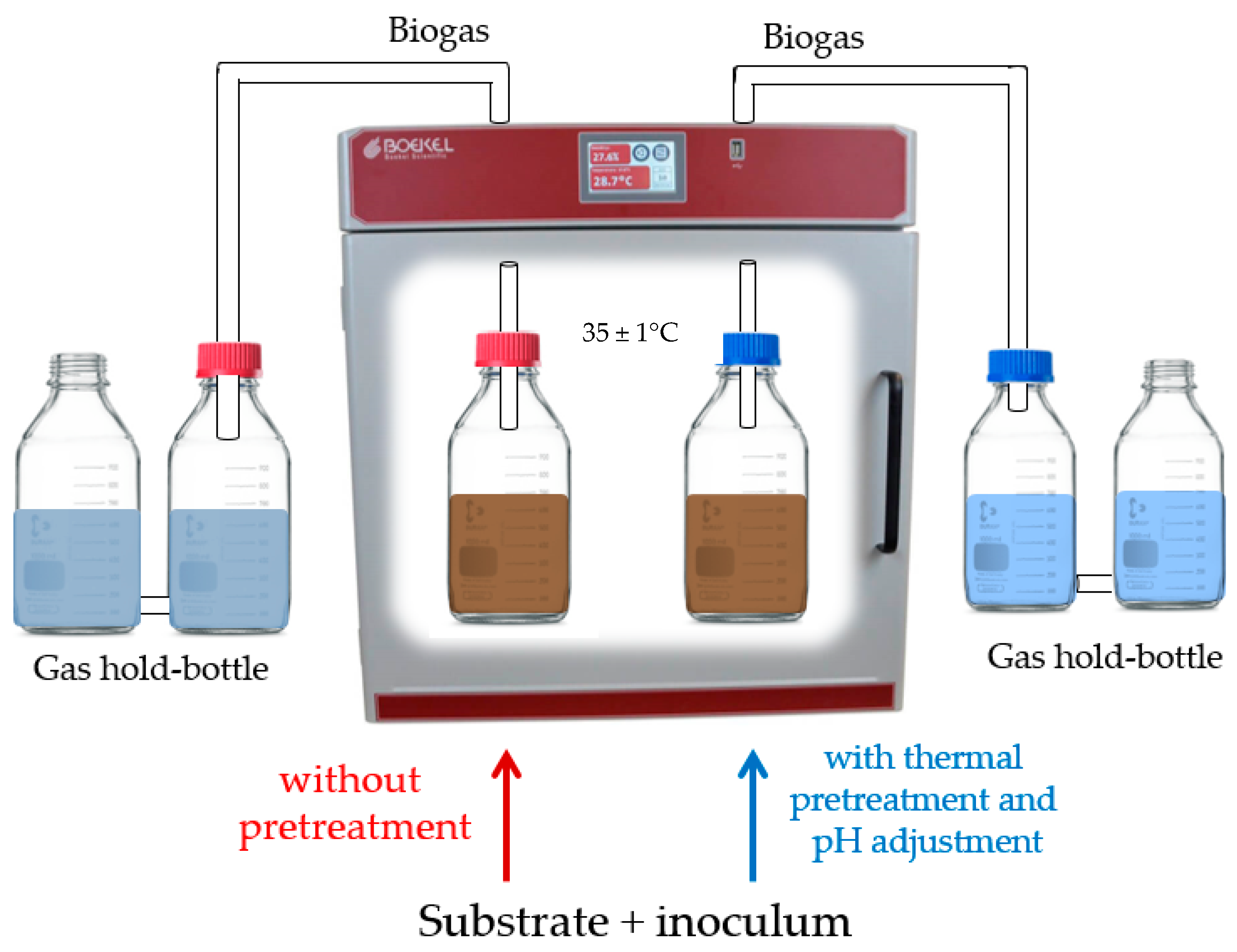
2.2. Experimental Setup and Operational Conditions
2.2.1. Sugar Beet Pulp Hydrolysis
2.2.2. Dark Fermentation
2.3. Analytical Methods
2.3.1. Substrate Characterization
2.3.2. Monosaccharide Content
2.3.3. Methane and Hydrogen Potential Tests
2.3.4. Furfural
2.4. Statistical Analysis
3. Results and Discussion
3.1. Composition of Sugar Beet Pulp Hydrolysates
3.2. Batch Tests
3.3. Volatile Fatty Acid Productions
3.4. Mass and Energy Balance
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 5-HMF | 5-hydroxymethylfurfural |
| AD | anaerobic digestion |
| COD | chemical oxygen demand |
| DF | dark fermentation |
| FID | flame ionization detector |
| HSBP | sugar beet pulp enzymatic hydrolysate |
| LA | levulinic acid |
| OLR | organic loading rate |
| PEG | nitroterephthalic-acid-modified polyethylene glycol |
| SBP | sugar beet pulp |
| SGP | specific gas production |
| SHP | specific hydrogen production |
| SMP | specific methane production |
| SRT | solids retention time |
| TSBP | sugar beet pulp after thermal treatment |
| TASBP | sugar beet pulp after thermal and acidic treatment |
| TAN | total ammonium nitrogen |
| TS | total solids |
| VFA | volatile fatty acids |
| VS | volatile solids |
Appendix A
References
- Judzińska, A. Przemiany w polskim premyśle cukrowniczym w latach 2010–2016. Roczniki Naukowe Stowarzyszenia Ekonomistów Rolnictwa i Agrobiznesu; Stowarzyszenie Ekonomistów Rolnictwa i Agrobiznesu: Poznań, Poland, 2017; pp. 104–109. [Google Scholar]
- Chudoba, L. Sugar market in Poland-currently and after the integration with the EU. In Rynek Cukru w Polsce Obecnie i po Integr. z Unia Eur; IERiGZ-PIB: Warszawa, Poland, 2004. [Google Scholar]
- Modelska, M.; Berlowska, J.; Kregiel, D.; Cieciura, W.; Antolak, H.; Tomaszewska, J.; Binczarski, M.; Szubiakiewicz, E.; Witonska, I. Concept for Recycling Waste Biomass from the Sugar Industry for Chemical and Biotechnological Purposes. Molecules 2017, 22, 1544. [Google Scholar] [CrossRef] [PubMed]
- Tomaszewska, J.; Bieliński, D.; Binczarski, M.; Berlowska, J.; Dziugan, P.; Piotrowski, J.; Stanishevsky, A.; Witońska, I.A. Products of sugar beet processing as raw materials for chemicals and biodegradable polymers. RSC Adv. 2018, 8, 3161–3177. [Google Scholar] [CrossRef]
- Güell, E.J.; Maru, B.T.; Chimentão, R.J.; Gispert-Guirado, F.; Constantí, M.; Medina, F. Combined heterogeneous catalysis and dark fermentation systems for the conversion of cellulose into biohydrogen. Biochem. Eng. J. 2015, 101, 209–219. [Google Scholar] [CrossRef]
- Leijdekkers, A.G.M.; Bink, J.P.M.; Geutjes, S.; Schols, H.A.; Gruppen, H. Enzymatic saccharification of sugar beet pulp for the production of galacturonic acid and arabinose; a study on the impact of the formation of recalcitrant oligosaccharides. Bioresour. Technol. 2013, 128, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Berlowska, J.; Cieciura, W.; Borowski, S.; Dudkiewicz, M.; Binczarski, M.; Witonska, I.; Otlewska, A.; Kregiel, D. Simultaneous Saccharification and Fermentation of Sugar Beet Pulp with Mixed Bacterial Cultures for Lactic Acid and Propylene Glycol Production. Molecules 2016, 21, 1380. [Google Scholar] [CrossRef] [PubMed]
- Concha, J.; Weinstein, C.; Zúñiga, M.E. Production of pectic extracts from sugar beet pulp with antiproliferative activity on a breast cancer cell line. Front. Chem. Sci. Eng. 2013, 7, 482–489. [Google Scholar] [CrossRef]
- Zheng, Y.; Lee, C.; Yu, C.; Cheng, Y.-S.; Zhang, R.; Jenkins, B.M.; VanderGheynst, J.S. Dilute acid pretreatment and fermentation of sugar beet pulp to ethanol. Appl. Energy 2013, 105, 1–7. [Google Scholar] [CrossRef]
- Berlowska, J.; Cieciura-Wloch, W.; Kalinowska, H.; Kregiel, D.; Borowski, S.; Pawlikowska, E.; Binczarski, M.; Witonska, I. Enzymatic Conversion of Sugar Beet Pulp: A Comparison of Simultaneous Saccharification and Fermentation and Separate Hydrolysis and Fermentation for Lactic Acid Production. Food Technol. Biotechnol. 2018, 56, 188. [Google Scholar] [CrossRef] [PubMed]
- Mardones, W.; Callegari, E.; Eyzaguirre, J. Corncob and sugar beet pulp induce specific sets of lignocellulolytic enzymes in Penicillium purpurogenum. Mycology 2019, 10, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Fishman, M.L.; Hicks, K.B.; Liu, C.-K. Biodegradable Composites from Sugar Beet Pulp and Poly (lactic acid). J. Agric. Food Chem. 2005, 53, 9017–9022. [Google Scholar] [CrossRef] [PubMed]
- Cárdenas-Fernández, M.; Bawn, M.; Hamley-Bennett, C.; Bharat, P.K.V.; Subrizi, F.; Suhaili, N.; Ward, D.P.; Bourdin, S.; Dalby, P.A.; Hailes, H.C.; et al. An integrated biorefinery concept for conversion of sugar beet pulp into value-added chemicals and pharmaceutical intermediates. Faraday Discuss. 2017, 202, 415–431. [Google Scholar] [CrossRef]
- Kucherov, F.A.; Romashov, L.V.; Galkin, K.I.; Ananikov, V.P. Chemical Transformations of Biomass-Derived C6-Furanic Platform Chemicals for Sustainable Energy Research, Materials Science, and Synthetic Building Blocks. ACS Sustain. Chem. Eng. 2018, 6, 8064–8092. [Google Scholar] [CrossRef]
- Kougioumtzis, M.A.; Marianou, A.; Atsonios, K.; Michailof, C.; Nikolopoulos, N.; Koukouzas, N.; Triantafyllidis, K.; Lappas, A.; Kakaras, E. Production of 5-HMF from Cellulosic Biomass: Experimental Results and Integrated Process Simulation. Waste Biomass Valorization 2018, 9, 2433–2445. [Google Scholar] [CrossRef]
- Yu, I.K.M.; Tsang, D.C.W. Conversion of biomass to hydroxymethylfurfural: A review of catalytic systems and underlying mechanisms. Bioresour. Technol. 2017, 238, 716–732. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Qi, B.; Wan, Y. Inhibitory effect of vanillin on cellulase activity in hydrolysis of cellulosic biomass. Bioresour. Technol. 2014, 167, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.; Fu, J.; Zhang, G. From lignocellulosic biomass to levulinic acid: A review on acid-catalyzed hydrolysis. Renew. Sustain. Energy Rev. 2018, 94, 340–362. [Google Scholar] [CrossRef]
- Pileidis, F.D.; Titirici, M.-M. Levulinic Acid Biorefineries: New Challenges for Efficient Utilization of Biomass. ChemSusChem 2016, 9, 562–582. [Google Scholar] [CrossRef] [PubMed]
- Cai, C.M.; Nagane, N.; Kumar, R.; Wyman, C.E. Coupling metal halides with a co-solvent to produce furfural and 5-HMF at high yields directly from lignocellulosic biomass as an integrated biofuels strategy. Green Chem. 2014, 16, 3819–3829. [Google Scholar] [CrossRef]
- Rasmussen, H.; Sørensen, H.R.; Meyer, A.S. Formation of degradation compounds from lignocellulosic biomass in the biorefinery: Sugar reaction mechanisms. Carbohydr. Res. 2014, 385, 45–57. [Google Scholar] [CrossRef]
- Mittal, A.; Black, S.K.; Vinzant, T.B.; O’Brien, M.; Tucker, M.P.; Johnson, D.K. Production of Furfural from Process-Relevant Biomass-Derived Pentoses in a Biphasic Reaction System. ACS Sustain. Chem. Eng. 2017, 5, 5694–5701. [Google Scholar] [CrossRef]
- Luo, Y.; Li, Z.; Li, X.; Liu, X.; Fan, J.; Clark, J.H.; Hu, C. The production of furfural directly from hemicellulose in lignocellulosic biomass: A review. Catal. Today 2019, 319, 14–24. [Google Scholar] [CrossRef]
- Li, X.; Jia, P.; Wang, T. Furfural: A Promising Platform Compound for Sustainable Production of C 4 and C 5 Chemicals. ACS Catal. 2016, 6, 7621–7640. [Google Scholar] [CrossRef]
- Chu, C.-F.; Li, Y.-Y.; Xu, K.-Q.; Ebie, Y.; Inamori, Y.; Kong, H.-N. A pH- and temperature-phased two-stage process for hydrogen and methane production from food waste. Int. J. Hydrogen Energy 2008, 33, 4739–4746. [Google Scholar] [CrossRef]
- Lee, D.-Y.; Ebie, Y.; Xu, K.-Q.; Li, Y.-Y.; Inamori, Y. Continuous H2 and CH4 production from high-solid food waste in the two-stage thermophilic fermentation process with the recirculation of digester sludge. Bioresour. Technol. 2010, 101, S42–S47. [Google Scholar] [CrossRef] [PubMed]
- Nathao, C.; Sirisukpoka, U.; Pisutpaisal, N. Production of hydrogen and methane by one and two stage fermentation of food waste. Int. J. Hydrogen Energy 2013, 38, 15764–15769. [Google Scholar] [CrossRef]
- Xiao, L.; Deng, Z.; Fung, K.Y.; Ng, K.M. Biohydrogen generation from anaerobic digestion of food waste. Int. J. Hydrogen Energy 2013, 38, 13907–13913. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, Y. A bench scale study of fermentative hydrogen and methane production from food waste in integrated two-stage process. Int. J. Hydrogen Energy 2009, 34, 245–254. [Google Scholar] [CrossRef]
- Angeriz-Campoy, R.; Álvarez-Gallego, C.J.; Romero-García, L.I. Thermophilic anaerobic co-digestion of organic fraction of municipal solid waste (OFMSW) with food waste (FW): Enhancement of bio-hydrogen production. Bioresour. Technol. 2015, 194, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Elsamadony, M.; Tawfik, A. Potential of biohydrogen production from organic fraction of municipal solid waste (OFMSW) using pilot-scale dry anaerobic reactor. Bioresour. Technol. 2015, 196, 9–16. [Google Scholar] [CrossRef]
- De Gioannis, G.; Friargiu, M.; Massi, E.; Muntoni, A.; Polettini, A.; Pomi, R.; Spiga, D. Biohydrogen production from dark fermentation of cheese whey: Influence of pH. Int. J. Hydrogen Energy 2014, 39, 20930–20941. [Google Scholar] [CrossRef]
- Castelló, E.; Santos, C.G.Y.; Iglesias, T.; Paolino, G.; Wenzel, J.; Borzacconi, L.; Etchebehere, C. Feasibility of biohydrogen production from cheese whey using a UASB reactor: Links between microbial community and reactor performance. Int. J. Hydrogen Energy 2009, 34, 5674–5682. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Z.; Zhu, S.; Zhang, H.; Zhang, Y.; Zhang, T.; Zhang, Q. Comparison of bio-hydrogen production yield capacity between asynchronous and simultaneous saccharification and fermentation processes from agricultural residue by mixed anaerobic cultures. Bioresour. Technol. 2018, 247, 1210–1214. [Google Scholar] [CrossRef] [PubMed]
- Júnior, A.D.N.F.; Etchebehere, C.; Zaiat, M. High organic loading rate on thermophilic hydrogen production and metagenomic study at an anaerobic packed-bed reactor treating a residual liquid stream of a Brazilian biorefinery. Bioresour. Technol. 2015, 186, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Wicher, E.; Seifert, K.; Zagrodnik, R.; Pietrzyk, B.; Laniecki, M. Hydrogen gas production from distillery wastewater by dark fermentation. Int. J. Hydrogen Energy 2013, 38, 7767–7773. [Google Scholar] [CrossRef]
- Wang, W.; Xie, L.; Luo, G.; Zhou, Q. Enhanced fermentative hydrogen production from cassava stillage by co-digestion: The effects of different co-substrates. Int. J. Hydrogen Energy 2013, 38, 6980–6988. [Google Scholar] [CrossRef]
- Dhar, B.R.; Elbeshbishy, E.; Hafez, H.; Lee, H.S. Hydrogen production from sugar beet juice using an integrated biohydrogen process of dark fermentation and microbial electrolysis cell. Bioresour. Technol. 2015, 198, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.C.; Rosa, P.R.F.; Sakamoto, I.K.; Varesche, M.B.A.; Silva, E.L. Continuous thermophilic hydrogen production and microbial community analysis from anaerobic digestion of diluted sugar cane stillage. Int. J. Hydrogen Energy 2014, 39, 9000–9011. [Google Scholar] [CrossRef]
- Arreola-Vargas, J.; Flores-Larios, A.; González-Álvarez, V.; Corona-González, R.I.; Méndez-Acosta, H.O. Single and two-stage anaerobic digestion for hydrogen and methane production from acid and enzymatic hydrolysates of Agave tequilana bagasse. Int. J. Hydrogen Energy 2016, 41, 897–904. [Google Scholar] [CrossRef]
- Lee, H.-S.; Vermaas, W.F.J.; Rittmann, B.E. Biological hydrogen production: Prospects and challenges. Trends Biotechnol. 2010, 28, 262–271. [Google Scholar] [CrossRef]
- Zagrodnik, R.; Laniecki, M. The role of pH control on biohydrogen production by single stage hybrid dark- and photo-fermentation. Bioresour. Technol. 2015, 194, 187–195. [Google Scholar] [CrossRef]
- Pachapur, V.L.; Kutty, P.; Pachapur, P.; Brar, S.K.; Le Bihan, Y.; Galvez-Cloutier, R.; Buelna, G. Seed Pretreatment for Increased Hydrogen Production Using Mixed-Culture Systems with Advantages over Pure-Culture Systems. Energies 2019, 12, 530. [Google Scholar] [CrossRef]
- Ghimire, A.; Frunzo, L.; Pirozzi, F.; Trably, E.; Escudie, R.; Lens, P.N.L.; Esposito, G. A review on dark fermentative biohydrogen production from organic biomass: Process parameters and use of by-products. Appl. Energy 2015, 144, 73–95. [Google Scholar] [CrossRef]
- Cieciura-Włoch, W.; Borowski, S. Biohydrogen production from waste of plant and animal origin via dark fermentation. J. Environ. Eng. Landsc. Manag. 2019, 27, 101–113. [Google Scholar] [CrossRef]
- Tsapekos, P.; Kougias, P.G.; Treu, L.; Campanaro, S.; Angelidaki, I. Process performance and comparative metagenomic analysis during co-digestion of manure and lignocellulosic biomass for biogas production. Appl. Energy 2017, 185, 126–135. [Google Scholar] [CrossRef]
- Rice, E.W.; Baird, R.B.; Eaton, A.D.; Clesceri, L.S. APHA Standard Methods for the Examination of Water and Waste Water, 22nd ed.; American Public Health Association; American Water Works Association; Water Environment Federation: Washington, DC, USA, 2012. [Google Scholar]
- Zhong, W.; Zhang, Z.; Luo, Y.; Sun, S.; Qiao, W.; Xiao, M. Effect of biological pretreatments in enhancing corn straw biogas production. Bioresour. Technol. 2011, 102, 11177–11182. [Google Scholar] [CrossRef] [PubMed]
- Chheda, J.N.; Román-Leshkov, Y.; Dumesic, J.A. Production of 5-hydroxymethylfurfural and furfural by dehydration of biomass-derived mono- and poly-saccharides. Green Chem. 2007, 9, 342–350. [Google Scholar] [CrossRef]
- Montané, D. High-temperature dilute-acid hydrolysis of olive stones for furfural production. Biomass Bioenergy 2002, 22, 295–304. [Google Scholar] [CrossRef]
- Marzialetti, T.; Olarte, M.B.V.; Sievers, C.; Hoskins, T.J.C.; Agrawal, P.K.; Jones, C.W. Dilute Acid Hydrolysis of Loblolly Pine: A Comprehensive Approach. Ind. Eng. Chem. Res. 2008, 47, 7131–7140. [Google Scholar] [CrossRef]
- Sun, J.-B.; Severson, R.F.; Kays, S.J. Effect of heating temperature and microwave pretreatment on the formation of sugars and volatiles in jewel sweetpotato. J. Food Qual. 1994, 17, 447–456. [Google Scholar] [CrossRef]
- Ariunbaatar, J.; Panico, A.; Frunzo, L.; Esposito, G.; Lens, P.N.L.; Pirozzi, F. Enhanced anaerobic digestion of food waste by thermal and ozonation pretreatment methods. J. Environ. Manag. 2014, 146, 142–149. [Google Scholar] [CrossRef]
- Luo, J.; Xu, Y.; Zhao, L.; Dong, L.; Tong, D.; Zhu, L.; Hu, C. Two-step hydrothermal conversion of Pubescens to obtain furans and phenol compounds separately. Bioresour. Technol. 2010, 101, 8873–8880. [Google Scholar] [CrossRef] [PubMed]
- Ziemiński, K.; Kowalska-Wentel, M. Effect of Different Sugar Beet Pulp Pretreatments on Biogas Production Efficiency. Appl. Biochem. Biotechnol. 2017, 181, 1211–1227. [Google Scholar] [CrossRef] [PubMed]
- Brazdausks, P.; Vedernikovs, N.; Puke, M.; Kruma, I. Effect of the Acid Hydrolysis Temperature on the Conversion of Birch Wood Hemicelluloses into Furfural. Key Eng. Mater. 2014, 604, 245–248. [Google Scholar] [CrossRef]
- Berlowska, J.; Pielech-Przybylska, K.; Balcerek, M.; Cieciura, W.; Borowski, S.; Kregiel, D. Integrated Bioethanol Fermentation/Anaerobic Digestion for Valorization of Sugar Beet Pulp. Energies 2017, 10, 1255. [Google Scholar] [CrossRef]
- Singh, R.; White, D.; Demirel, Y.; Kelly, R.; Noll, K.; Blum, P. Uncoupling fermentative synthesis of molecular hydrogen from biomass formation in Thermotoga maritima. Appl. Environ. Microbiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Tevatia, R.; White, D.; Demirel, Y.; Blum, P. Comparative kinetic modeling of growth and molecular hydrogen overproduction by engineered strains of Thermotoga maritima. Int. J. Hydrogen Energy 2019, 44, 7125–7136. [Google Scholar] [CrossRef]
- Ren, N. Mechanism and controlling strategy of the production and accumulation of propionic acid for anaerobic wastewater treatment. Sci. China Ser. B Chem. 2002, 45, 319. [Google Scholar] [CrossRef]
- Wang, B.; Wan, W.; Wang, J. Inhibitory effect of ethanol, acetic acid, propionic acid and butyric acid on fermentative hydrogen production. Int. J. Hydrogen Energy 2008, 33, 7013–7019. [Google Scholar] [CrossRef]
- Fang, H.H.; Yu, H. Mesophilic acidification of gelatinaceous wastewater. J. Biotechnol. 2002, 93, 99–108. [Google Scholar] [CrossRef]
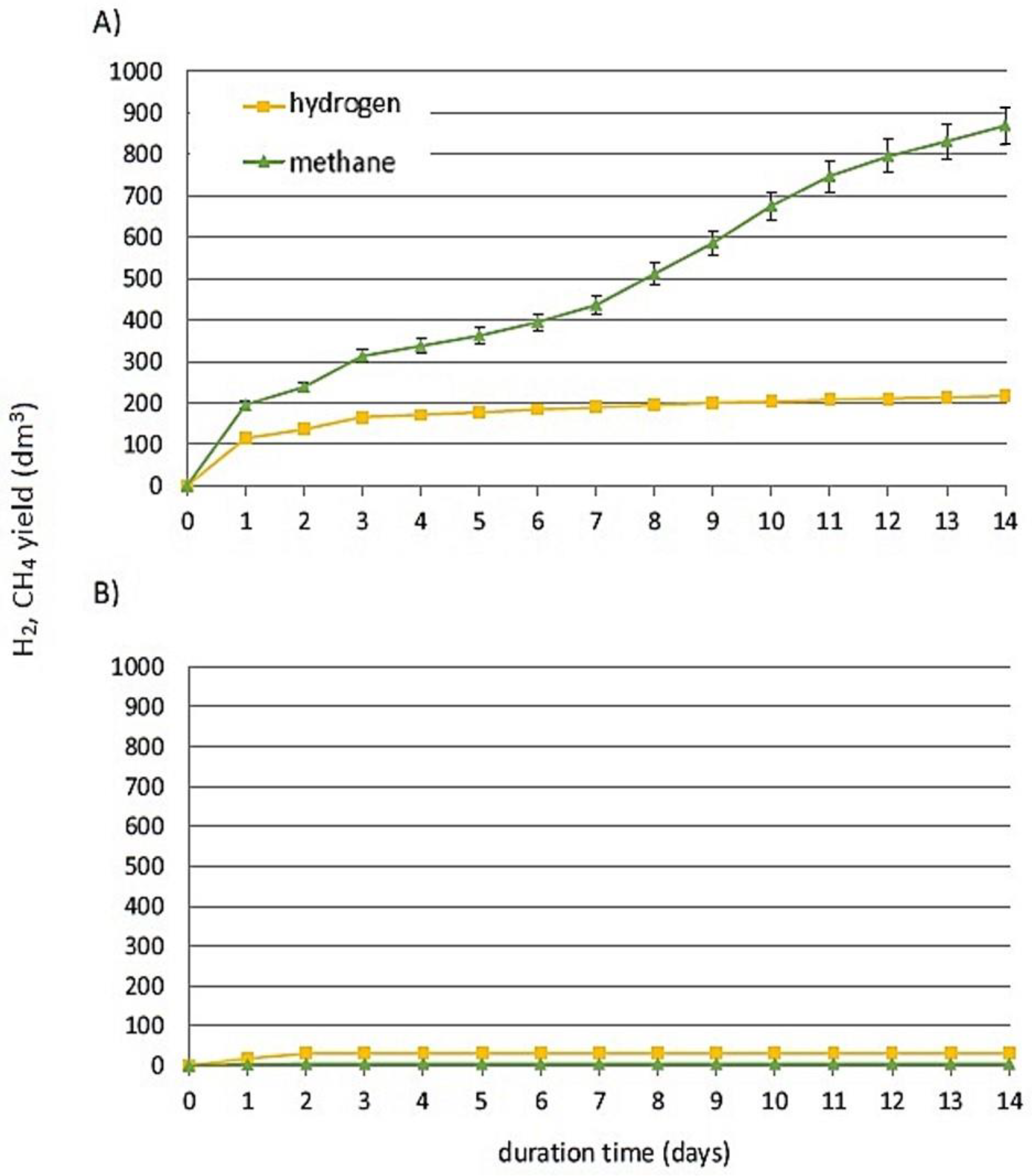
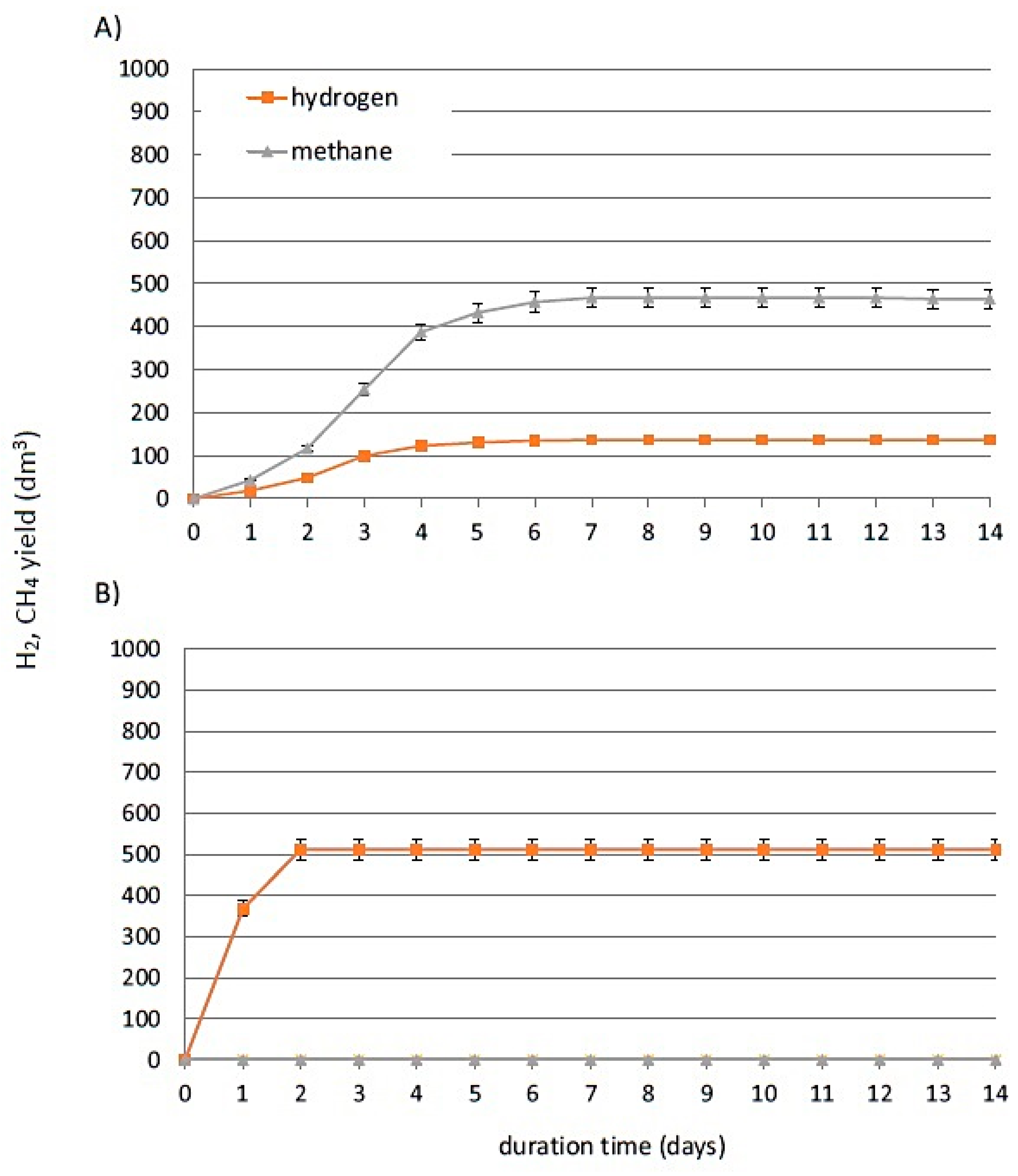
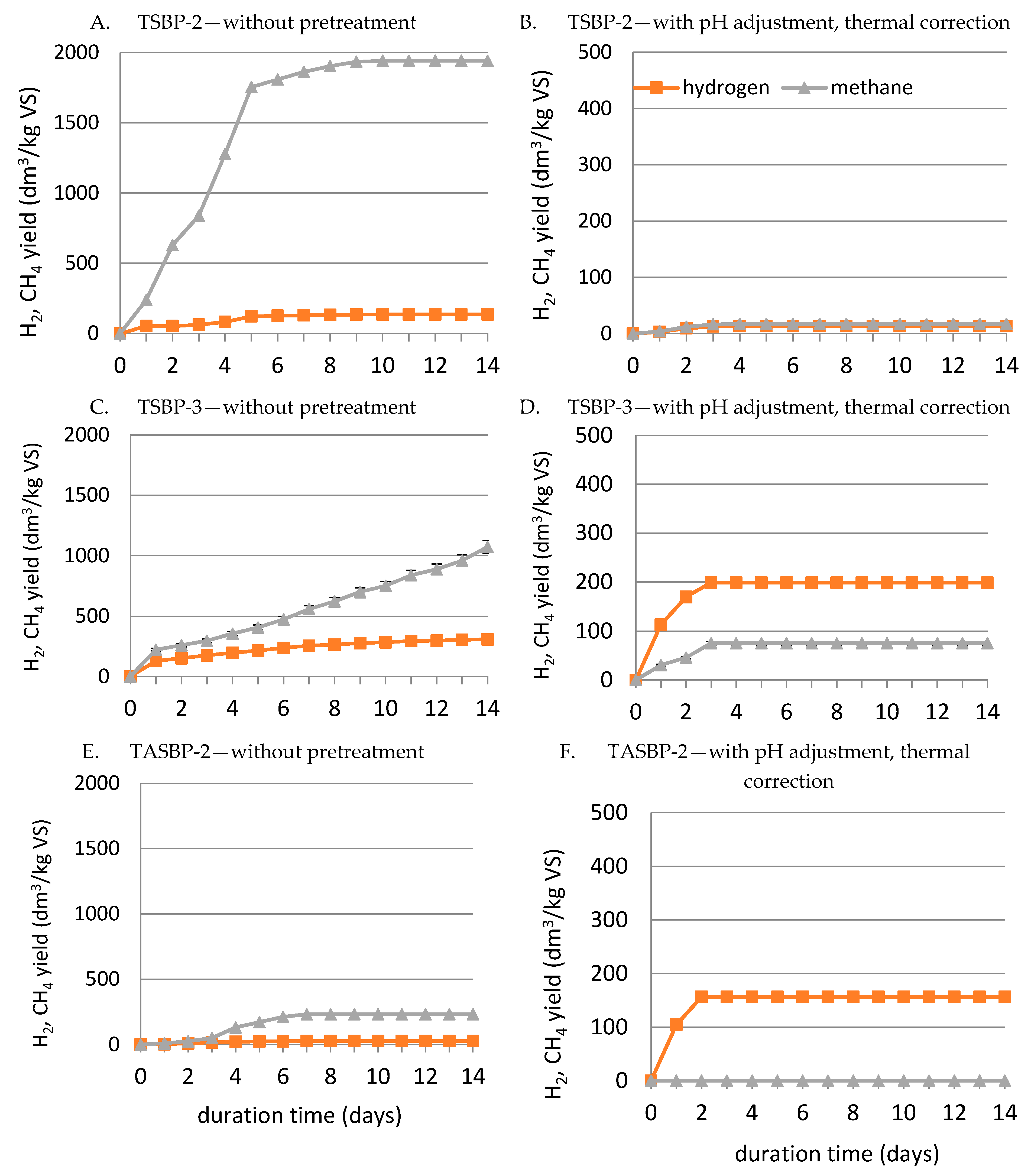
| Experimental Run | Inoculum | HSBP | TSBP-1 | TSBP-2 | TSBP-3 | TASBP-1 | TASBP-2 |
|---|---|---|---|---|---|---|---|
| Dilution (SBP: water) | - | 2:1 | 2:5 | 1:5 | 1:5 | 1:5 | 1:5 |
| Pretreatment time (h) | - | 10 | 6 | 0 | 6 | 0 | 1 |
| Pretreatment temp (°C) | - | 45 | 140 | 140 | 140 | 140 | 140 |
| H2SO4 dose | - | - | - | - | - | 2% | 5% |
| Total solids (g/kg) | 24.59 ± 0.18 | 161.32 ± 1.98 | 50.24 ± 0.25 | 29.48 ± 0.89 | 27.56 ± 0.94 | 35.58 ± 0.52 | 27.21 ± 0.33 |
| Volatile solids (g/kg) | 15.74 ± 0.12 | 149.65 ± 1.87 | 47.41 ± 0.34 | 25.88 ± 0.56 | 24.21 ± 0.53 | 32.94 ± 0.37 | 23.66 ± 0.63 |
| Furfural (g/L) | - | 0.00 ± 0.00 | 1.63 ± 0.12 | 1.19 ± 0.15 | 1.16 ± 0.11 | 4.05 ± 0.39 | 12.12 ± 1.36 |
| Yield (%) a | - | 0.00 ± 0.00 | 0.14 ± 0.02 | 0.10 ± 0.01 | 0.10 ± 0.01 | 0.33 ± 0.02 | 1.00 ± 0.05 |
| Glucose (g/L) | - | 18.42 ± 0.98 | 3.14 ± 0.25 | 2.52 ± 0.34 | 3.24 ± 0.38 | 1.74 ± 0.18 | 1.71 ± 0.25 |
| Fructose (g/L) | - | 7.89 ± 0.42 | 1.48 ± 0.05 | 1.72 ± 0.28 | 3.12 ± 0.46 | 0.63 ± 0.02 | 0.28 ± 0.01 |
| Mannose (g/L) | - | 4.25 ± 0.12 | 6.24 ± 0.48 | 7.30 ± 0.55 | 10.75 ± 1.18 | 0.28 ± 0.01 | 0.11 ± 0.01 |
| Arabinose (g/L) | - | 6.07 ± 0.18 | 2.22 ± 0.29 | 0.27 ± 0.01 | 4.04 ± 0.62 | 13.28 ± 0.18 | 4.32 ± 0.28 |
| Galactose (g/L) | - | 8.25 ± 0.25 | 1.79 ± 0.16 | 0.24 ± 0.02 | 3.65 ± 0.35 | 8.94 ± 0.85 | 4.29 ± 0.32 |
| Rhamnose (g/L) | - | 7.04 ± 0.15 | 0.02 ± 0.00 | 0.02 ± 0.00 | 0.05 ± 0.00 | 4.87 ± 0.53 | 4.27 ± 0.49 |
| Raffinose (g/L) | - | 11.84 ± 0.28 | 1.76 ± 0.22 | 1.05 ± 0.10 | 1.41 ± 0.32 | 11.66 ± 1.45 | 6.87 ± 0.72 |
| Xylose (g/L) | - | 3.98 ± 0.15 | 0.86 ± 0.19 | 0.75 ± 0.01 | 0.68 ± 0.02 | 1.10 ± 0.12 | 0.45 ± 0.02 |
| Galacturonic acid (g/L) | – | 4.11 ± 0.12 | 10.87 ± 0.85 | 0.55 ± 0.01 | 10.07 ± 0.68 | 2.70 ± 0.02 | 0.28 ± 0.01 |
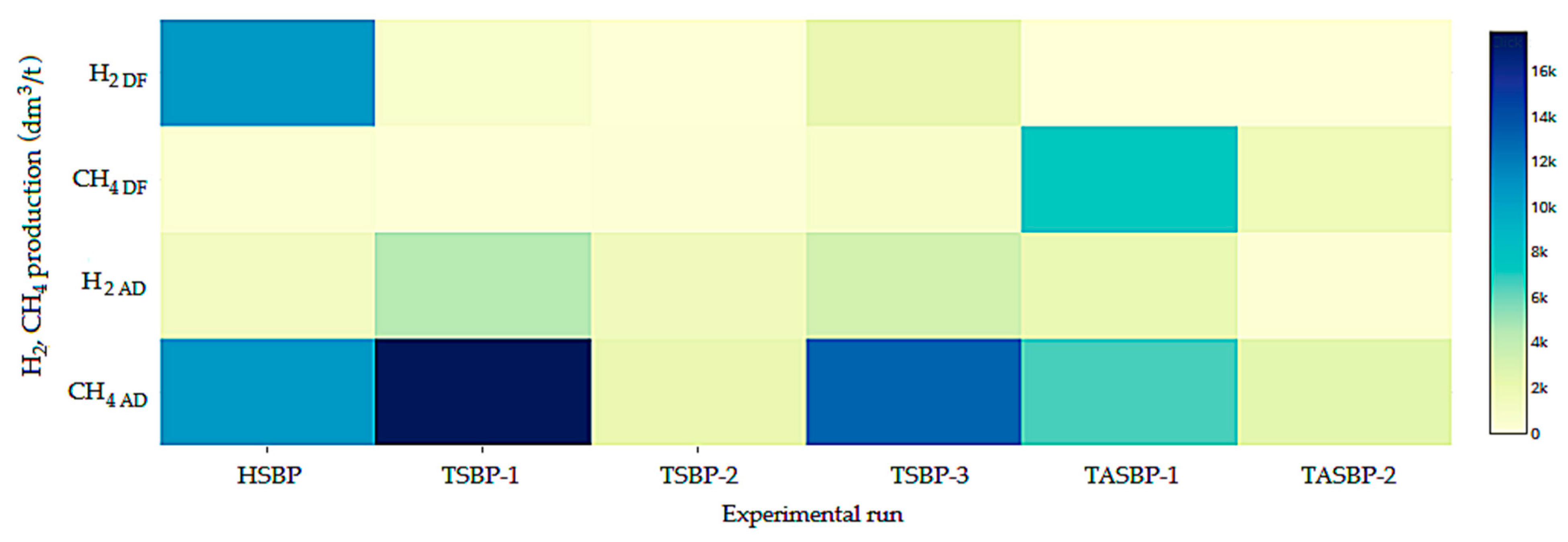
| Substrate | Specific Gas Production (SGP) dm3/kg VS | Specific Methane Production (SMP) dm3 CH4/kg VS | Specific Hydrogen Production (SHP) dm3 H2/kg VS | pHinitial | pHfinal |
|---|---|---|---|---|---|
| HSBP | 913.22 ± 28.54 | 402.26 ± 21.51 | 112.59 ± 8.94 | 7.25 ± 0.02 | 7.09 ± 0.05 |
| TSBP-1 | 945.22 ± 130.93 | 374.24 ± 45.09 | 93.42 ± 15.61 | 7.33 ± 0.08 | 7.14 ± 0.01 |
| TSBP-2 | 170.30 ± 0.36 | 83.34 ± 0.52 | 5.84 ± 0.10 | 7.08 ± 0.05 | 6.29 ± 0.08 |
| TSBP-3 | 894.50 ± 15.78 | 391.16 ± 12.08 | 80.68 ± 3.60 | 7.13 ± 0.05 | 6.76 ± 0.02 |
| TASBP-1 | 61.60 ± 3.49 | 40.16 ± 4.85 | 3.92 ± 0.22 | 7.05 ± 0.01 | 6.51 ± 0.01 |
| TASBP-2 | 36.95 ± 4.43 | 11.82 ± 1.35 | 0.77 ± 0.11 | 7.11 ± 0.05 | 6.34 ± 0.01 |
| Substrate | Specific Gas Production (SGP) dm3/kg VS | Specific Methane Production (SMP) dm3 CH4/kg VS | Specific Hydrogen Production (SHP) dm3 H2/kg VS | pHinitial | pHfinal |
|---|---|---|---|---|---|
| HSBP | 197.39 ± 31.21 | 7.23 ± 1.79 | 109.89 ± 13.58 | 5.54 ± 0.04 | 5.32 ± 0.01 |
| TSBP-1 | 22.60 ± 3.23 | 1.95 ± 0.28 | 13.01 ± 1.86 | 5.61 ± 0.02 | 5.36 ± 0.01 |
| TSBP-2 | 82.29 ± 3.76 | 4.46 ± 0.17 | 15.67 ± 0.62 | 5.49 ± 0.01 | 5.18 ± 0.02 |
| TSBP-3 | 200.87 ± 32.28 | 32.43 ± 3.80 | 85.44 ± 36.08 | 5.48 ± 0.04 | 5.37 ± 0.05 |
| TASBP-1 | 447.22 ± 5.17 | 0.00 ± 0.00 | 219.67 ± 2.54 | 5.54 ± 0.05 | 5.40 ± 0.04 |
| TASBP-2 | 214.67 ± 4.92 | 0.00 ± 0.00 | 67.08 ± 1.54 | 5.61 ± 0.02 | 5.22 ± 0.01 |
| Substrate | N-NH4 (mg/L) | P-PO43 (mg/L) | N-NH4 (mg/L) | P-PO43 (mg/L) |
|---|---|---|---|---|
| Without Pretreatment | With pH Adjustment and Thermal Pretreatment | |||
| HSBP | 825 ± 0.02 | 1665 ± 0.05 | 797 ± 0.02 | 1580 ± 0.02 |
| TSBP-1 | 751 ± 0.02 | 1552 ± 0.05 | 662 ± 0.02 | 1220 ± 0.02 |
| TSBP-2 | 623 ± 0.05 | 1020 ± 0.02 | 524 ± 0.02 | 1560 ± 0.05 |
| TSBP-3 | 543 ± 0.02 | 1032 ± 0.01 | 790 ± 0.01 | 1460 ± 0.05 |
| TASBP-1 | 489 ± 0.01 | 964 ± 0.02 | 800 ± 0.05 | 1030 ± 0.01 |
| TASBP-2 | 431 ± 0.01 | 904 ± 0.02 | 725 ± 0.05 | 980 ± 0.05 |
| Substrate/Experimental Run | HSBP | TSBP-1 | TSBP-2 | TSBP-3 | TASBP-1 | TASBP-2 |
|---|---|---|---|---|---|---|
| Acetic acid (g/L) | 0.8855 | 1.0775 | 0.9355 | 0.9930 | 0.7685 | 0.6235 |
| Propionic acid (g/L) | 0.1280 | 0.0780 | 0.1445 | 0.2335 | 0.0890 | 0.0535 |
| i-Butyric acid (g/L) | 0.0255 | 0.1205 | 0.1005 | 0.2330 | 0.0210 | 0.0335 |
| n-Butyric acid (g/L) | 0.0315 | 0.0905 | 0.0480 | 0.0780 | 0.0440 | 0.0485 |
| i-Valeric acid (g/L) | 0.0000 | 0.0790 | 0.1025 | 0.0570 | 0.0795 | 0.0600 |
| n-Valeric acid (g/L) | 0.0000 | 0.0000 | 0.0000 | 0.0125 | 0.1265 | 0.0780 |
| i-Caproic acid (g/L) | 0.0000 | 0.0425 | 0.0145 | 0.0165 | 0.0000 | 0.0000 |
| n-Caproic acid (g/L) | 0.0180 | 0.0060 | 0.0055 | 0.0055 | 0.0065 | 0.0075 |
| i-Heptanoic acid (g/L) | 0.0080 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 |
| Total VFA | 1.0965 | 1.4940 | 1.3510 | 1.6285 | 1.1350 | 0.9045 |
| Fuel | Volume (m3/tSBP) | Specific Energy Yield per Volume (kWh/m3) | Theoretical Energy Yield (kWh/tSBP) |
|---|---|---|---|
| TSBP-1 | |||
| Methane | 17.74 | 10.21 | +181.07 |
| Hydrogen | 0.61 | 3.58 | +2.19 |
| Heat treatment | - | - | −41.17 |
| Total energy yield | − | − | ∑142.09 |
| TSBP-2 | |||
| Methane | 3.92 | 10.21 | +40.00 |
| Hydrogen | 0.27 | 3.58 | +0.95 |
| Heat treatment | − | − | −18.67 |
| Total energy yield | − | − | ∑22.28 |
| TSBP-3 | |||
| Methane | 13.09 | 10.21 | +133.69 |
| Hydrogen | 2.07 | 3.58 | +7.42 |
| Heat treatment | − | − | −41.17 |
| Total energy yield | − | − | ∑99.94 |
| TASBP-1 | |||
| Methane | 6.59 | 10.21 | +67.30 |
| Hydrogen | 7.21 | 3.58 | +25.82 |
| Heat treatment | − | − | −18.67 |
| Total energy yield | − | − | ∑74.45 |
| TASBP-2 | |||
| Methane | 2.37 | 10.21 | +24.17 |
| Hydrogen | 1.59 | 3.58 | +5.70 |
| Heat treatment | − | − | −22.42 |
| Total energy yield | − | − | ∑7.45 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cieciura-Włoch, W.; Binczarski, M.; Tomaszewska, J.; Borowski, S.; Domański, J.; Dziugan, P.; Witońska, I. The Use of Acidic Hydrolysates after Furfural Production from Sugar Waste Biomass as a Fermentation Medium in the Biotechnological Production of Hydrogen. Energies 2019, 12, 3222. https://doi.org/10.3390/en12173222
Cieciura-Włoch W, Binczarski M, Tomaszewska J, Borowski S, Domański J, Dziugan P, Witońska I. The Use of Acidic Hydrolysates after Furfural Production from Sugar Waste Biomass as a Fermentation Medium in the Biotechnological Production of Hydrogen. Energies. 2019; 12(17):3222. https://doi.org/10.3390/en12173222
Chicago/Turabian StyleCieciura-Włoch, Weronika, Michał Binczarski, Jolanta Tomaszewska, Sebastian Borowski, Jarosław Domański, Piotr Dziugan, and Izabela Witońska. 2019. "The Use of Acidic Hydrolysates after Furfural Production from Sugar Waste Biomass as a Fermentation Medium in the Biotechnological Production of Hydrogen" Energies 12, no. 17: 3222. https://doi.org/10.3390/en12173222
APA StyleCieciura-Włoch, W., Binczarski, M., Tomaszewska, J., Borowski, S., Domański, J., Dziugan, P., & Witońska, I. (2019). The Use of Acidic Hydrolysates after Furfural Production from Sugar Waste Biomass as a Fermentation Medium in the Biotechnological Production of Hydrogen. Energies, 12(17), 3222. https://doi.org/10.3390/en12173222









