Prospecting for Oleaginous and Robust Chlorella spp. for Coal-Fired Flue-Gas-Mediated Biodiesel Production
Abstract
1. Introduction
2. Results and Discussion
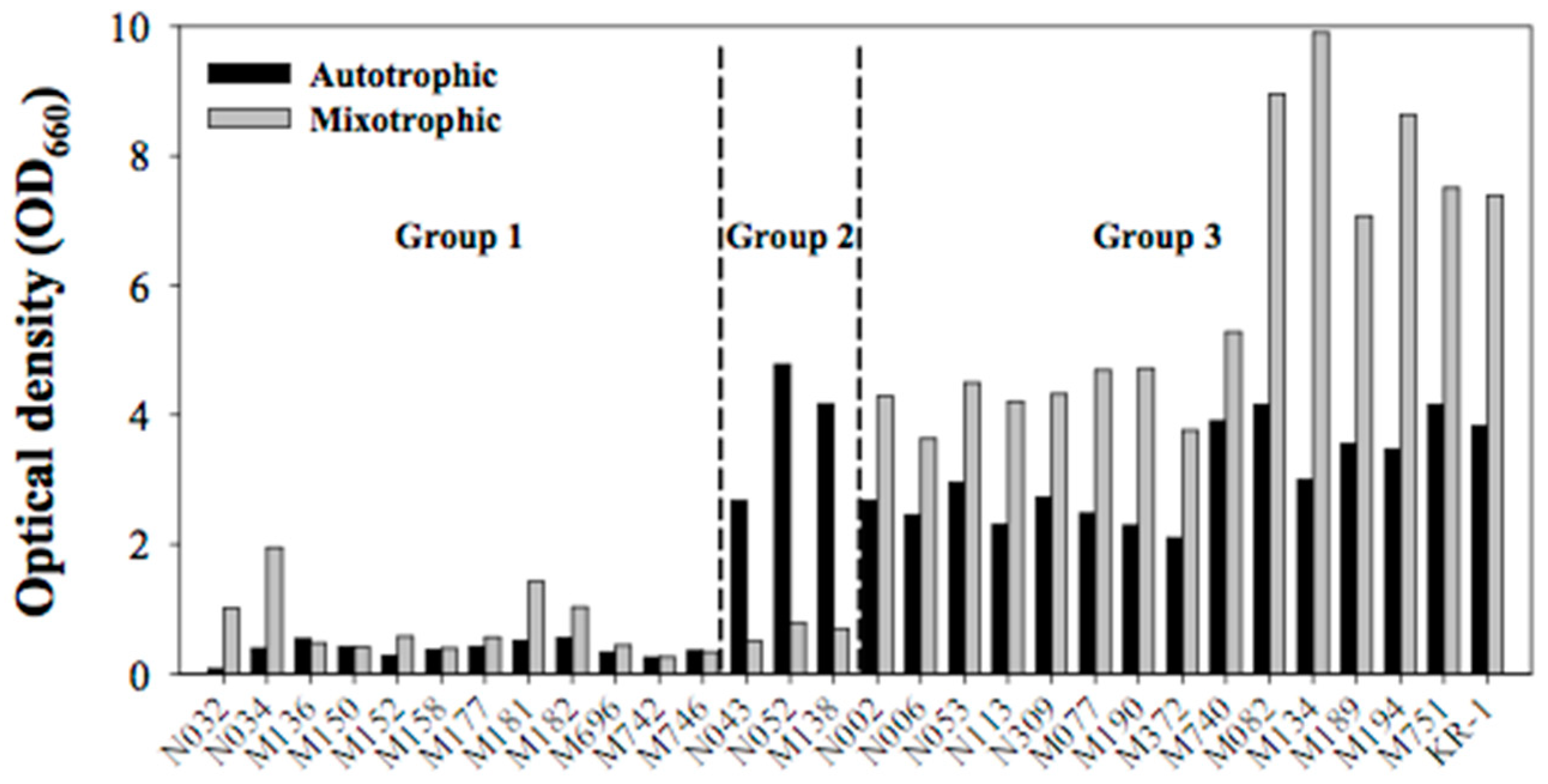
2.1. Preliminary Screening Based on Growth Rate
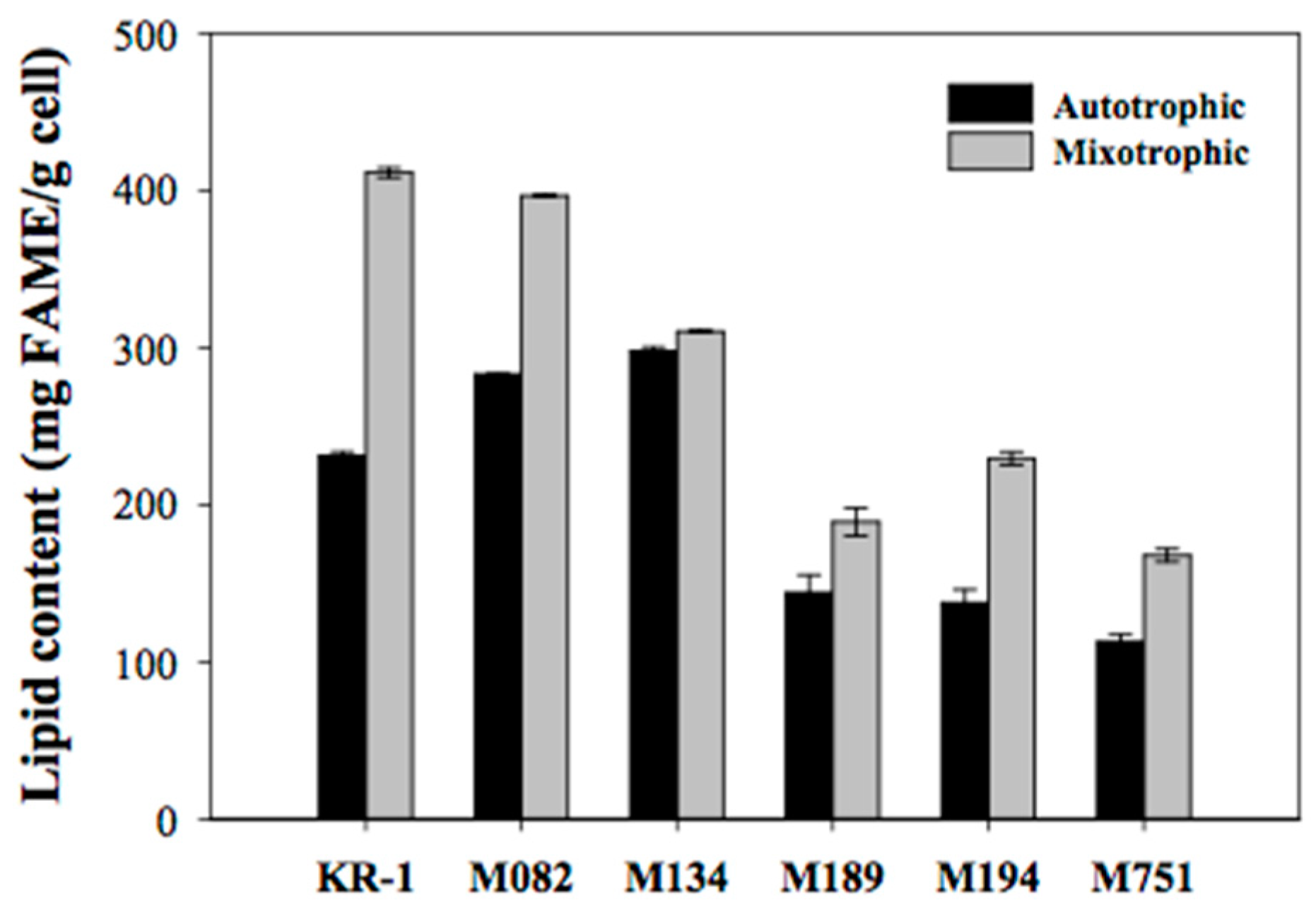
2.2. Secondary Screening Based on Lipid Content and Composition
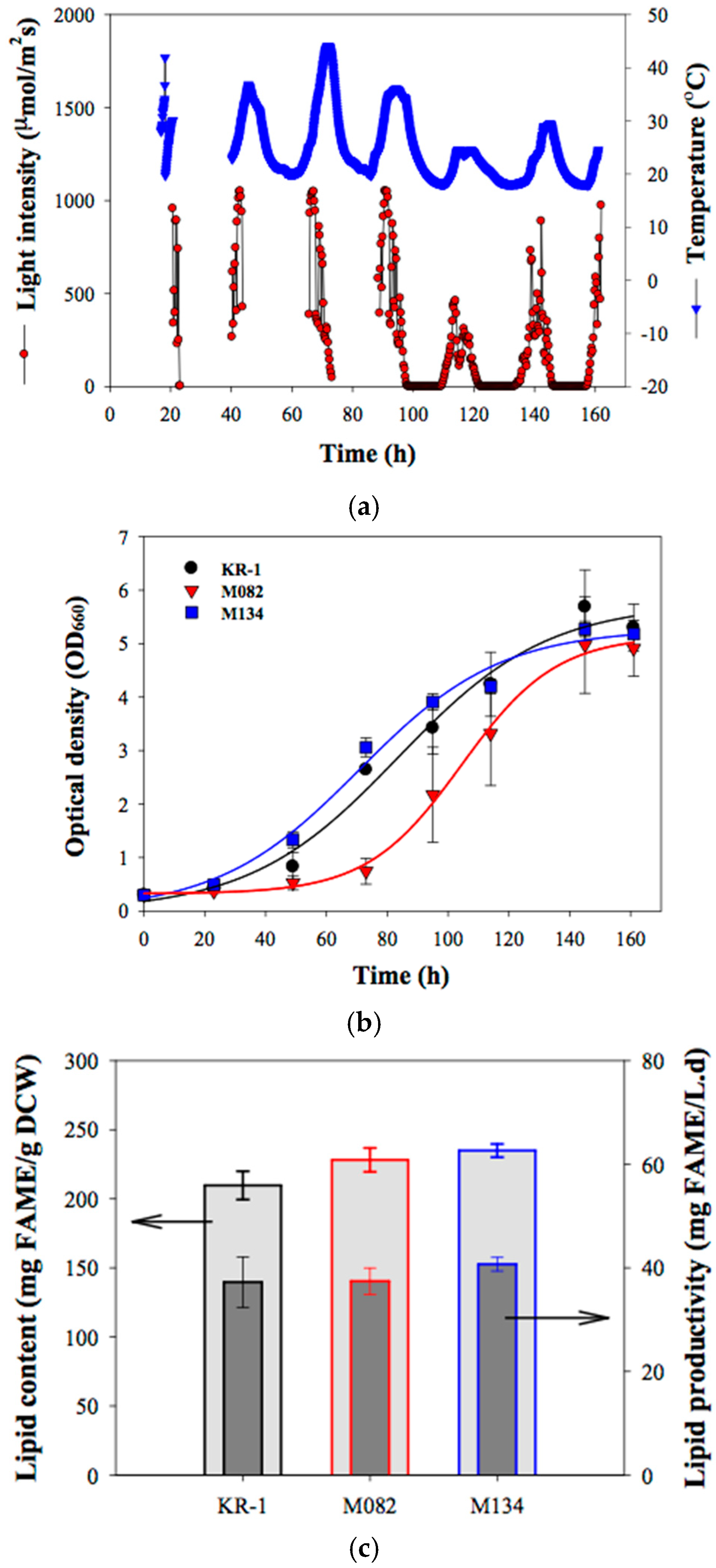
2.3. Outdoor Flue-Gas Cultivation and High-Temperature Tolerance Experimentation
2.4. Theoretical Estimation of Biodiesel Properties from Fatty Acid Profiles
3. Materials and Methods
3.1. Microalgal Strains and Culture Medium
3.2. Seed Flask Culture
3.3. Indoor Culture Using Photobioreactor with 10% (v/v) CO2
3.4. Outdoor Culture Using Photobioreactor with Actual Flue-gas
3.5. Analytical Methods
3.6. Estimation of Biodiesel Properties
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mata, T.M.; Martins, A.A.; Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energ. Rev. 2010, 14, 217–232. [Google Scholar] [CrossRef]
- Praveenkumar, R.; Kim, B.; Lee, J.; Vijayan, D.; Lee, K.; Nam, B.; Jeon, S.G.; Kim, D.M.; Oh, Y.K. Mild pressure induces rapid accumulation of neutral lipid (triacylglycerol) in Chlorella spp. Bioresour. Technol. 2016, 220, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.; Luong, T.T.; Lee, D.; Oh, Y.K.; Lee, T. Reuse of effluent water from a municipal wastewater treatment plant in microalgae cultivation for biofuel production. Bioresour. Technol. 2011, 102, 8639–8645. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Lee, S.Y.; Na, J.G.; Jeon, S.G.; Praveenkumar, R.; Kim, D.M.; Chang, W.S.; Oh, Y.K. Magnetophoretic harvesting of oleaginous Chlorella sp. by using biocompatible chitosan/magnetic nanoparticle composites. Bioresour. Technol. 2013, 149, 575–578. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Lee, S.Y.; Praveenkumar, R.; Kim, B.; Seo, J.Y.; Jeon, S.G.; Na, J.G.; Park, J.Y.; Kim, D.M.; Oh, Y.K. Repeated use of stable magnetic flocculant for efficient harvest of oleaginous Chlorella sp. Bioresour. Technol. 2014, 167, 284–290. [Google Scholar] [CrossRef] [PubMed]
- Praveenkumar, R.; Kim, B.; Choi, E.; Lee, K.; Park, J.Y.; Lee, J.S.; Lee, Y.C.; Oh, Y.K. Improved biomass and lipid production in a mixotrophic culture of Chlorella sp. KR-1 with addition of coal-fired flue-gas. Bioresour. Technol. 2014, 171, 500–505. [Google Scholar] [CrossRef] [PubMed]
- Praveenkumar, R.; Shameera, K.; Mahalakshmi, G.; Akbarsha, M.A.; Thajuddin, N. Influence of nutrient deprivations on lipid accumulation in a dominant indigenous microalga Chlorella sp., BUM11008: Evaluation for biodiesel production. Biomass Bioenergy 2012, 37, 60–66. [Google Scholar] [CrossRef]
- Remmers, I.M.; Wijffels, R.N.; Barbosa, M.; Lamers, P.P. Can We Approach Theoretical Lipid Yields in Microalgae? Trend Biotechnol. 2018, 36, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Chiu, S.Y.; Kao, C.Y.; Huang, T.-T.; Lin, C.J.; Ong, S.C.; Chen, C.D.; Chang, J.S.; Lin, C.S. Microalgal biomass production and on-site bioremediation of carbon dioxide, nitrogen oxide and sulfur dioxide from flue gas using Chlorella sp. cultures. Bioresour. Technol. 2011, 102, 9135–9142. [Google Scholar] [CrossRef] [PubMed]
- Hende, S.V.D.; Vervaeren, H.; Boon, N. Flue gas compounds and microalgae:(Bio-) chemical interactions leading to biotechnological opportunities. Biotechnol. Adv. 2012, 30, 1405–1424. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Sarkany, N.; Cui, Y. Biomass and lipid productivities of Chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotechnol. Lett. 2009, 31, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Guccione, A.; Biondi, N.; Sampietro, G.; Rodolfi, L.; Bassi, N.; Tredici, M.R. Chlorella for protein and biofuels: From strain selection to outdoor cultivation in a green wall panel photobioreactor. Biotechnol. Biofuel 2014, 7, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Muthuraj, M.; Kumar, V.; Palabhanvi, B.; Das, D. Evaluation of indigenous microalgal isolate Chlorella sp. FC2 IITG as a cell factory for biodiesel production and scale up in outdoor conditions. J. Ind. Microbiol. Biotechnol. 2014, 41, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, I.A.; Marques, S.S.I.; Cabanelas, I.T.D.; Pereira, S.A.; Druzian, J.I.; de Souza, C.O.; Vich, D.V.; de Carvalho, G.C.; Nascimento, M.A. Screening microalgae strains for biodiesel production: Lipid productivity and estimation of fuel quality based on fatty acids profiles as selective criteria. Bioenergy Res. 2013, 6, 1–13. [Google Scholar] [CrossRef]
- Sun, Z.; Zhou, Z.G.; Gerken, H.; Chen, F.; Liu, J. Screening and characterization of oleaginous Chlorella strains and exploration of photoautotrophic Chlorella protothecoides for oil production. Bioresour. Technol. 2015, 184, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Cui, J.; Liu, Q.; Ding, Y.; Liu, J. Screening and phylogenetic analysis of lipid-rich microalgae. Algal Res. 2015, 11, 381–386. [Google Scholar] [CrossRef]
- Xia, L.; Song, S.; He, Q.; Yang, H.; Hu, C. Selection of microalgae for biodiesel production in a scalable outdoor photobioreactor in north China. Bioresour. Technol. 2014, 174, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Cheirsilp, B.; Torpee, S. Enhanced growth and lipid production of microalgae under mixotrophic culture condition: Effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour. Technol. 2012, 110, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Praveenkumar, R.; Lee, J.; Nam, B.; Kim, D.M.; Lee, K.; Lee, Y.C.; Oh, Y.K. Magnesium aminoclay enhances lipid production of mixotrophic Chlorella sp. KR-1 while reducing bacterial populations. Bioresour. Technol. 2016, 219, 608–613. [Google Scholar] [CrossRef] [PubMed]
- Abomohra, A.E.-F.; El-Sheekh, M.; Hanelt, D. Screening of marine microalgae isolated from the hypersaline Bardawil lagoon for biodiesel feedstock. Renew. Energy 2017, 101, 1266–1272. [Google Scholar] [CrossRef]
- Tanimura, A.; Takashima, M.; Sugita, T.; Endoh, R.; Kikukawa, M.; Yamaguchi, S.; Sakuradani, E.; Ogawa, J.; Shima, J. Selection of oleaginous yeasts with high lipid productivity for practical biodiesel production. Bioresour. Technol. 2014, 153, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Vello, V.; Phang, S.M.; Chu, W.L.; Majid, N.A.; Lim, P.E.; Loh, S.K. Lipid productivity and fatty acid composition-guided selection of Chlorella strains isolated from Malaysia for biodiesel production. J. Appl. Phycol. 2014, 26, 1399–1413. [Google Scholar] [CrossRef]
- Praveenkumar, R.; Kim, B.; Choi, E.; Lee, K.; Cho, S.; Hyun, J.; Park, J.; Lee, Y.; Lee, H.; Lee, J. Mixotrophic cultivation of oleaginous Chlorella sp. KR-1 mediated by actual coal-fired flue gas for biodiesel production. Bioproc. Biosyst. Eng. 2014, 37, 2083–2094. [Google Scholar] [CrossRef] [PubMed]
- Hoekman, S.K.; Broch, A.; Robbins, C.; Ceniceros, E.; Natarajan, M. Review of biodiesel composition, properties, and specifications. Renew. Sustain. Energy Rev. 2012, 16, 143–169. [Google Scholar] [CrossRef]
- Valdez-Ojeda, R.; González-Muñoz, M.; Us-Vázquez, R.; Narváez-Zapata, J.; Chavarria-Hernandez, J.C.; López-Adrián, S.; Barahona-Pérez, F.; Toledano-Thompson, T.; Garduño-Solórzano, G.; Medrano, R.M.E.-G. Characterization of five fresh water microalgae with potential for biodiesel production. Algal Res. 2015, 7, 33–44. [Google Scholar] [CrossRef]
- Sung, K.D.; Lee, J.S.; Shin, C.S.; Park, S.C.; Choi, M.J. CO2 fixation by Chlorella sp. KR-1 and its cultural characteristics. Bioresour. Technol. 1999, 68, 269–273. [Google Scholar] [CrossRef]
- Yun, M.; Oh, Y.K.; Praveenkumar, R.; Seo, Y.S.; Cho, S. Contaminated bacterial effects and qPCR application to monitor a specific bacterium in Chlorella sp. KR-1 culture. Biotechnol. Bioprocess. Eng. 2017, 22, 150–160. [Google Scholar] [CrossRef]
| Fatty Acid Methyl Ester | Ratio (%) of Each FAME to Total FAME (Mean ± Standard Deviation) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| M082 | M134 | M189 | M194 | M751 | KR-1 | ||||||||
| Auto | Mixo | Auto | Mixo | Auto | Mixo | Auto | Mixo | Auto | Mixo | Auto | Mixo | ||
| Caprate | C10:0 | 0.05 ± 0.07 | 0.08 ± 0.00 | nd | 0.1 ± 0.01 | nd | nd | nd | nd | nd | nd | nd | 0.19 ± 0.01 |
| Tridecanoate | C13:0 | 0.26 ± 0.01 | nd | 0.26 ± 0.01 | nd | 0.34 ± 0.03 | 0.18 ± 0.00 | 0.37 ± 0.01 | 0.14 ± 0.01 | 0.49 ± 0.06 | 0.32 ± 0.00 | 0.23 ± 0.03 | 0.11 ± 0.01 |
| Myristate | C14:0 | nd | 0.94 ± 0.01 | nd | 0.57 ± 0.10 | 0.61 ± 0.03 | 0.59 ± 0.00 | 0.64 ± 0.03 | 0.48 ± 0.00 | 0.36 ± 0.02 | 0.51 ± 0.51 | 0.23 ± 0.00 | 0.22 ± 0.00 |
| Pentadecanoate | C15:0 | 0.12 ± 0.00 | 0.19 ± 0.01 | 0.1 ± 0.00 | 0.22 ± 0.00 | nd | 0.15 ± 0.00 | 0.21 ± 0.01 | 0.14 ± 0.00 | nd | 0.16 ± 0.00 | nd | 0.13 ± 0.00 |
| Palmitate | C16:0 | 23 ± 0.08 | 26.78 ± 0.06 | 25.99 ± 0.05 | 26.79 ± 0.06 | 22.36 ± 0.99 | 24.55 ± 0.37 | 22.21 ± 1.34 | 24.6 ± 0.08 | 20.96 ± 0.16 | 23.91 ± 0.52 | 24.49 ± 0.30 | 28.55 ± 0.02 |
| Palmitoleate | C16:1 | 0.49 ± 0.01 | 0.29 ± 0.00 | 0.55 ± 0.01 | 0.37 ± 0.00 | 0.35 ± 0.01 | 0.4 ± 0.01 | 0.31 ± 0.00 | 0.36 ± 0.00 | 0.14 ± 0.20 | 0.3 ± 0.02 | 0.36 ± 0.01 | 0.32 ± 0.00 |
| Stearate | C18:0 | 3.72 ± 0.01 | 6.26 ± 0.01 | 4.32 ± 0.01 | 5.71 ± 0.02 | 0.78 ± 0.00 | 1.01 ± 0.00 | 0.72 ± 0.03 | 1.52 ± 0.35 | 0.5 ± 0.04 | 0.97 ± 0.00 | 4.57 ± 0.02 | 4 ± 0.02 |
| Oleate | C18:1n9c | 21.45 ± 0.02 | 21.8 ± 0.05 | 20.76 ± 0.02 | 19.25 ± 0.03 | 11.54 ± 0.03 | 16.06 ± 0.10 | 10.07 ± 0.22 | 22.02 ± 0.03 | 7.28 ± 0.01 | 15.26 ± 0.05 | 19.07 ± 0.10 | 18.7 ± 0.02 |
| Linoleate | C18:2n6c | 24.64 ± 0.04 | 25.42 ± 0.17 | 23.66 ± 0.07 | 27.47 ± 0.14 | 20.61 ± 0.55 | 21.43 ± 0.18 | 20.2 ± 0.69 | 21.6 ± 0.02 | 18.3 ± 0.21 | 19.43 ± 0.25 | 25.6 ± 0.30 | 24.08 ± 0.03 |
| γ-Linoleate | C18:3n6 | 0.34 ± 0.00 | 0.31 ± 0.00 | 0.32 ± 0.00 | 0.35 ± 0.01 | 0.4 ± 0.03 | 0.35 ± 0.03 | 0.43 ± 0.01 | 0.33 ± 0.01 | 0.4 ± 0.02 | 0.38 ± 0.00 | 0.35 ± 0.01 | 0.29 ± 0.01 |
| Linolenate | C18:3n3 | 6.69 ± 0.00 | 4.22 ± 0.06 | 6.22 ± 0.01 | 4.43 ± 0.01 | 12.87 ± 0.38 | 12.12 ± 0.16 | 13.09 ± 0.56 | 9.89 ± 0.01 | 14.52 ± 0.19 | 12.18 ± 0.21 | 6.5 ± 0.09 | 7.49 ± 0.02 |
| Arachidate | C20:0 | 0.14 ± 0.19 | 0.35 ± 0.00 | 0.25 ± 0.01 | 0.15 ± 0.01 | nd | nd | nd | nd | nd | nd | 0.32 ± 0.03 | 0.26 ± 0.01 |
| cis-11-iocosenoate | C20:1 | 0.14 ± 0.20 | 0.05 ± 0.07 | nd | nd | nd | nd | nd | nd | nd | nd | nd | nd |
| Behenate | C22:0 | 0.12 ± 0.01 | 0.19 ± 0.00 | nd | 0.05 ± 0.07 | nd | nd | nd | nd | nd | nd | nd | 0.1 ± 0.00 |
| Lignocerate | C24:0 | nd | 0.15 ± 0.00 | nd | 0.15 ± 0.05 | nd | nd | nd | nd | nd | nd | nd | nd |
| Saturated | 27.41 ± 0.19 | 34.95 ± 0.07 | 30.91 ± 0.06 | 33.75 ± 0.16 | 24.1 ± 1.05 | 26.49 ± 0.36 | 24.14 ± 1.40 | 26.62 ± 0.05 | 22.31 ± 0.09 | 25.87 ± 0.53 | 29.85 ± 0.32 | 33.55 ± 0.04 | |
| Monounsaturated | 22.08 ± 0.21 | 22.14 ± 0.02 | 21.31 ± 0.03 | 19.63 ± 0.03 | 11.89 ± 0.03 | 16.47 ± 0.11 | 10.38 ± 0.22 | 22.38 ± 0.04 | 7.42 ± 0.22 | 15.55 ± 0.07 | 19.43 ± 0.09 | 19.02 ± 0.02 | |
| Polyunsaturated | 31.66 ± 0.05 | 29.95 ± 0.22 | 30.2 ± 0.07 | 32.25 ± 0.14 | 33.89 ± 0.90 | 33.9 ± 0.32 | 33.73 ± 1.26 | 31.82 ± 0.01 | 33.22 ± 0.39 | 31.99 ± 0.46 | 32.45 ± 0.40 | 31.85 ± 0.03 | |
| Others | 18.85 ± 0.04 | 12.96 ± 0.13 | 17.58 ± 0.02 | 14.37 ± 0.01 | 30.12 ± 0.19 | 23.15 ± 0.15 | 31.75 ± 0.36 | 19.18 ± 0.09 | 37.05 ± 0.51 | 26.59 ± 0.13 | 18.26 ± 0.01 | 15.57 ± 0.09 | |
| Total | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | |
| Chlorella Strain | Viscosity [mm/s2] | *Specific Gravity | Cloud Point [°C] | Cetane Number | Iodine Value | HHV [MJ/kg] | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Auto | Mixo | Auto | Mixo | Auto | Mixo | Auto | Mixo | Auto | Mixo | Auto | Mixo | ||
| Indoor culture | KR-1 | 4.63 ± 0.00 | 4.63 ± 0.00 | 0.88 ± 0.00 | 0.88 ± 0.00 | 7.81 ± 0.07 | 7.91 ± 0.01 | 56.79 ± 0.04 | 56.84 ± 0.00 | 80.53 ± 0.42 | 80.02 ± 0.05 | 40.14 ± 0.01 | 40.13 ± 0.00 |
| M082 | 4.62 ± 0.00 | 4.66 ± 0.00 | 0.88 ± 0.00 | 0.88 ± 0.00 | 7.67 ± 0.01 | 8.44 ± 0.05 | 56.72 ± 0.00 | 57.10 ± 0.03 | 81.35 ± 0.05 | 77.05 ± 0.29 | 40.16 ± 0.00 | 40.05 ± 0.01 | |
| M134 | 4.65 ± 0.00 | 4.64 ± 0.01 | 0.88 ± 0.00 | 0.88 ± 0.00 | 8.21 ± 0.01 | 8.12 ± 0.03 | 56.99 ± 0.00 | 56.94 ± 0.02 | 78.34 ± 0.06 | 78.83 ± 0.16 | 40.09 ± 0.00 | 40.09 ± 0.00 | |
| M189 | 4.62 ± 0.01 | 4.59 ± 0.00 | 0.88 ± 0.00 | 0.88 ± 0.00 | 7.58 ± 0.20 | 7.07 ± 0.06 | 56.68 ± 0.10 | 56.42 ± 0.04 | 81.83 ± 1.11 | 84.65 ± 0.35 | 40.17 ± 0.03 | 40.23 ± 0.01 | |
| M194 | 4.63 ± 0.01 | 4.60 ± 0.00 | 0.88 ± 0.00 | 0.88 ± 0.00 | 7.79 ± 0.27 | 7.14 ± 0.00 | 56.78 ± 0.14 | 56.46 ± 0.00 | 80.66 ± 1.52 | 84.28 ± 0.02 | 40.14 ± 0.04 | 40.23 ± 0.00 | |
| M751 | 4.64 ± 0.01 | 4.62 ± 0.00 | 0.88 ± 0.00 | 0.88 ± 0.00 | 8.14 ± 0.11 | 7.69 ± 0.11 | 56.95 ± 0.05 | 56.74 ± 0.05 | 78.74 ± 0.61 | 81.20 ± 0.56 | 40.09 ± 0.02 | 40.15 ± 0.02 | |
| ¶Outdoor culture | KR-1 | 4.52 ± 0.00 | na | 0.88 ± 0.00 | Na | 5.37 ± 0.04 | na | 55.58 ± 0.02 | na | 94.12 ± 0.23 | na | 40.46 ± 0.01 | na |
| M082 | 4.55 ± 0.00 | na | 0.88 ± 0.00 | Na | 6.02 ± 0.00 | na | 55.90 ± 0.00 | na | 90.50 ± 0.01 | na | 40.37 ± 0.00 | na | |
| M134 | 4.57 ± 0.00 | na | 0.88 ± 0.00 | Na | 6.54 ± 0.02 | na | 56.16 ± 0.01 | na | 87.64 ± 0.12 | na | 40.31 ± 0.00 | na | |
| Vegetable oil | †Jatropa | 4.48 | 0.88 | 4.67 | 55.23 | 98.02 | 40.55 | ||||||
| ‡Palm | 4.61 | 0.87 | 14.00 | 61.90 | 54.00 | 40.60 | |||||||
| ‡Rapeseed | 4.50 | 0.88 | -3.00 | 53.70 | 116.10 | 41.10 | |||||||
| ‡Soy bean | 4.26 | 0.88 | 0.00 | 51.30 | 125.50 | 39.70 | |||||||
| ‡Petroleum diesel | 2-3 | 0.85 | Country specific | 40-45 | na | na | |||||||
| International standard | EN14214 | 3.5–5.0 | 0.86–0.9 | na | Minimum 51.0 | Maximum 120 | na | ||||||
| ASTM D6751 | 1.9–6.0 | 0.85–0.9 | na | Minimum 47.0 | na | na | |||||||
| No. | Strain | Species | Origin |
|---|---|---|---|
| 1 | N002 | Chlorella vulgaris | Paddy field/Ssukgol, Gyeonggi |
| 2 | N006 | C. vulgaris | Paddy field/Suwon, Gyeonggi |
| 3 | N032 | C. vulgaris | Reservoir/Andongho, Gyeongbuk |
| 4 | N034 | C. vulgaris | Reservoir/Andongho, Gyeongbuk |
| 5 | N043 | C. vulgaris | Reservoir/Imhaho, Gyeongbuk |
| 6 | N052 | C. vulgaris | Reservoir/Youngsanho, Chungnam |
| 7 | N053 | Chlorella sp. | Reservoir/ Andong, Gyeongbuk |
| 8 | N113 | Chlorella sp. | Youngpyung, Gangwon |
| 9 | N309 | Chlorella sp. | Local fresh water body |
| 10 | M077 | Chlorella sp. | Local fresh water body |
| 11 | M082 | Chlorella sp. | Local fresh water body |
| 12 | M134 | Chlorella sp. | Local fresh water body |
| 13 | M136 | Chlorella sp. | Local fresh water body |
| 14 | M138 | Chlorella sp. | Local fresh water body |
| 15 | M150 | Chlorella sp. | Local fresh water body |
| 16 | M152 | Chlorella sp. | Local fresh water body |
| 17 | M158 | Chlorella sp. | Local fresh water body |
| 18 | M177 | Chlorella sp. | Local fresh water body |
| 19 | M181 | Chlorella sp. | Local fresh water body |
| 20 | M182 | Chlorella sp. | Local fresh water body |
| 21 | M189 | Chlorella sp. | Local fresh water body |
| 22 | M190 | Chlorella sp. | Local fresh water body |
| 23 | M194 | Chlorella sp. | Local fresh water body |
| 24 | M372 | Chlorella sp. | Local fresh water body |
| 25 | M696 | C. vulgaris | Reservoir/Yongsoho Jeju |
| 26 | M740 | C. sorokiniana | Reservoir/Daedongho, Hampyung |
| 27 | M742 | C. vulgaris | Reservoir/Daedongho, Hampyung |
| 28 | M746 | C. vulgaris | Reservoir/Daedongho, Hampyung |
| 29 | M751 | C. vulgaris | Local fresh water body |
| 30 | KR-1 | Chlorella sp. | Yeongwol, Gangwon |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, B.; Praveenkumar, R.; Choi, E.; Lee, K.; Jeon, S.G.; Oh, Y.-K. Prospecting for Oleaginous and Robust Chlorella spp. for Coal-Fired Flue-Gas-Mediated Biodiesel Production. Energies 2018, 11, 2026. https://doi.org/10.3390/en11082026
Kim B, Praveenkumar R, Choi E, Lee K, Jeon SG, Oh Y-K. Prospecting for Oleaginous and Robust Chlorella spp. for Coal-Fired Flue-Gas-Mediated Biodiesel Production. Energies. 2018; 11(8):2026. https://doi.org/10.3390/en11082026
Chicago/Turabian StyleKim, Bohwa, Ramasamy Praveenkumar, Eunji Choi, Kyubock Lee, Sang Goo Jeon, and You-Kwan Oh. 2018. "Prospecting for Oleaginous and Robust Chlorella spp. for Coal-Fired Flue-Gas-Mediated Biodiesel Production" Energies 11, no. 8: 2026. https://doi.org/10.3390/en11082026
APA StyleKim, B., Praveenkumar, R., Choi, E., Lee, K., Jeon, S. G., & Oh, Y.-K. (2018). Prospecting for Oleaginous and Robust Chlorella spp. for Coal-Fired Flue-Gas-Mediated Biodiesel Production. Energies, 11(8), 2026. https://doi.org/10.3390/en11082026







