Value-Added Performance and Thermal Decomposition Characteristics of Dumped Food Waste Compost by Pyrolysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
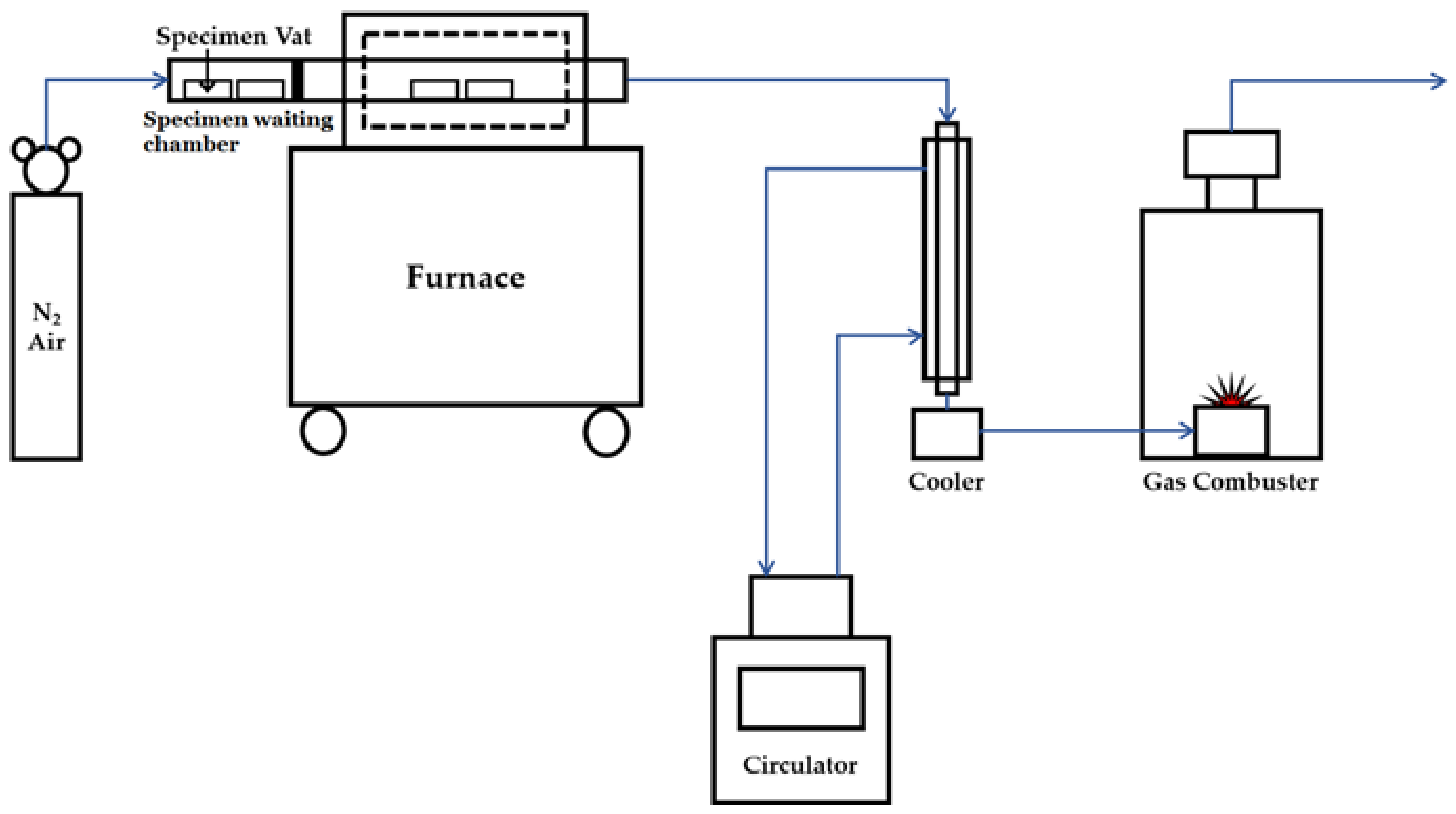
2.2. Experimental Method
2.3. Analysis Method
3. Results and Discussion
3.1. Characterization of Raw Material and Biochar
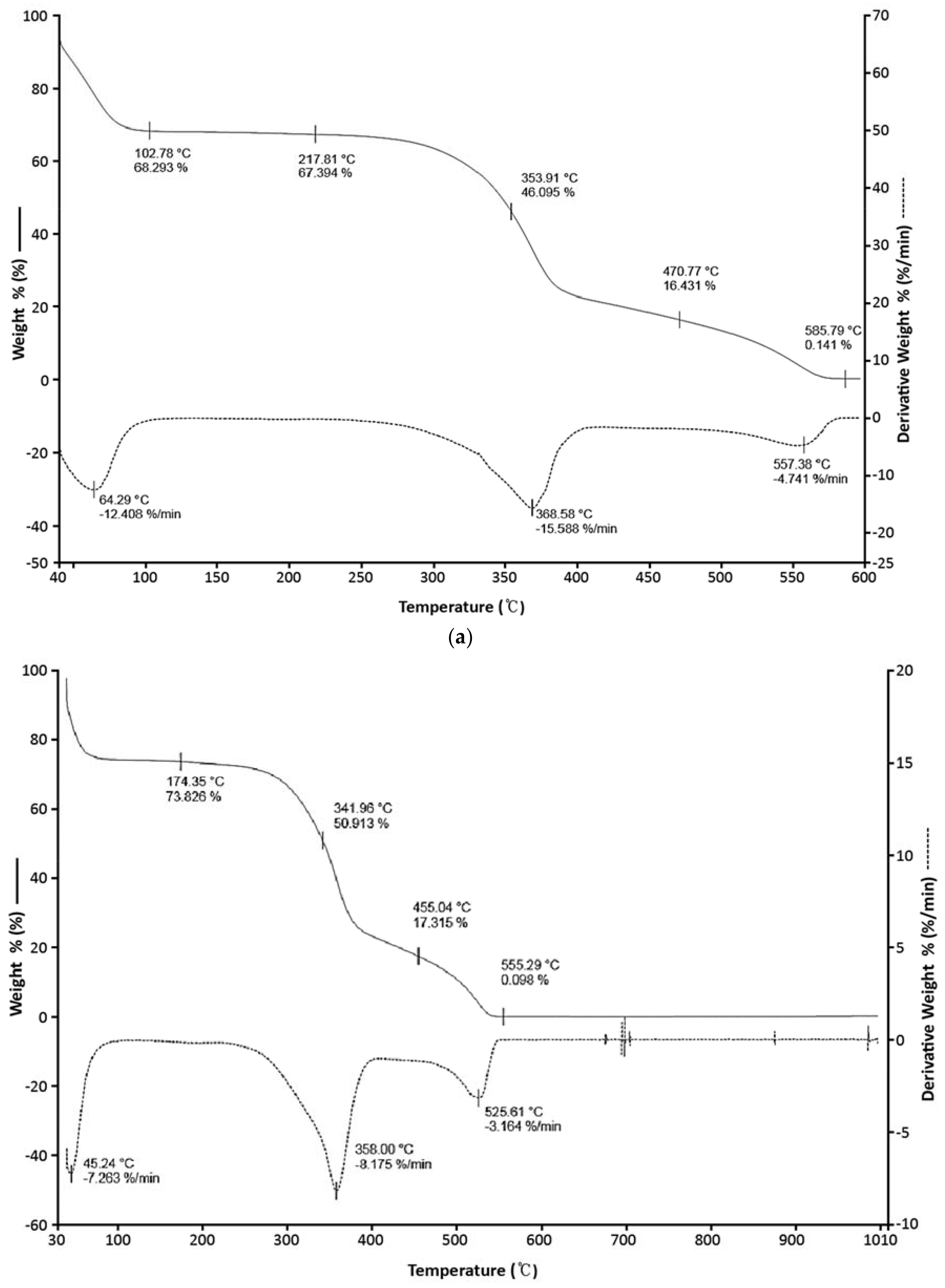
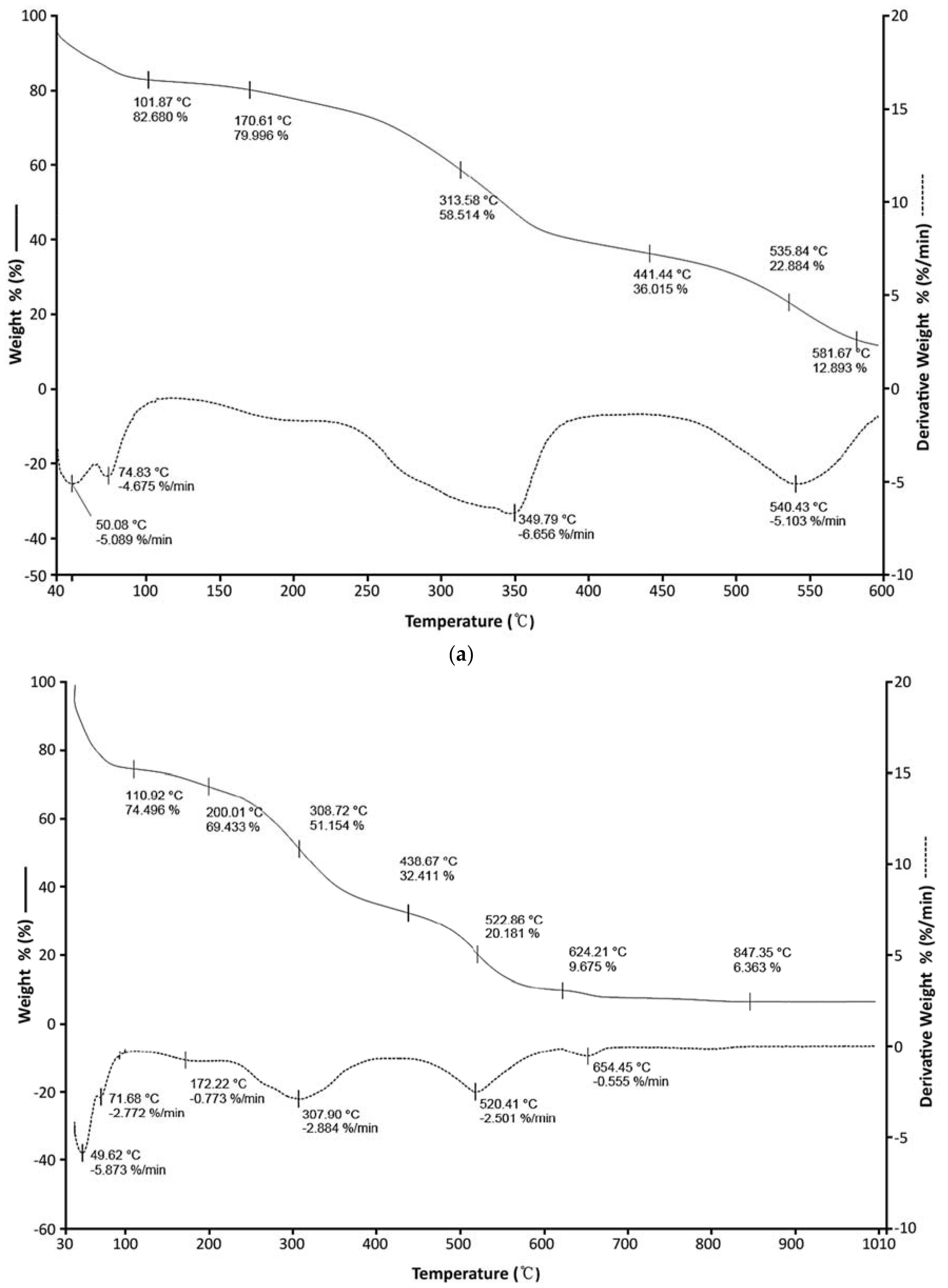
3.2. Thermogravimetric Analysis
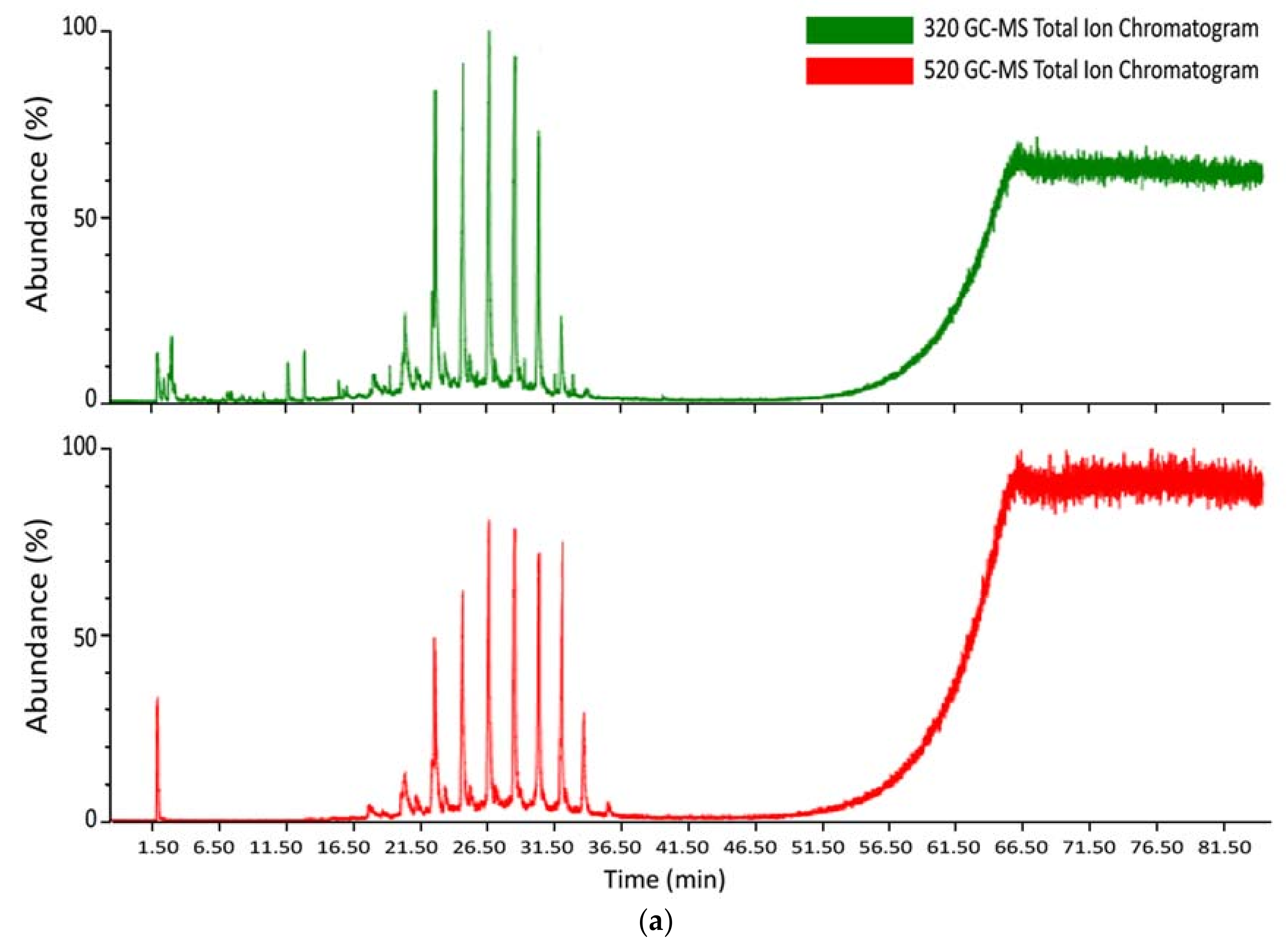
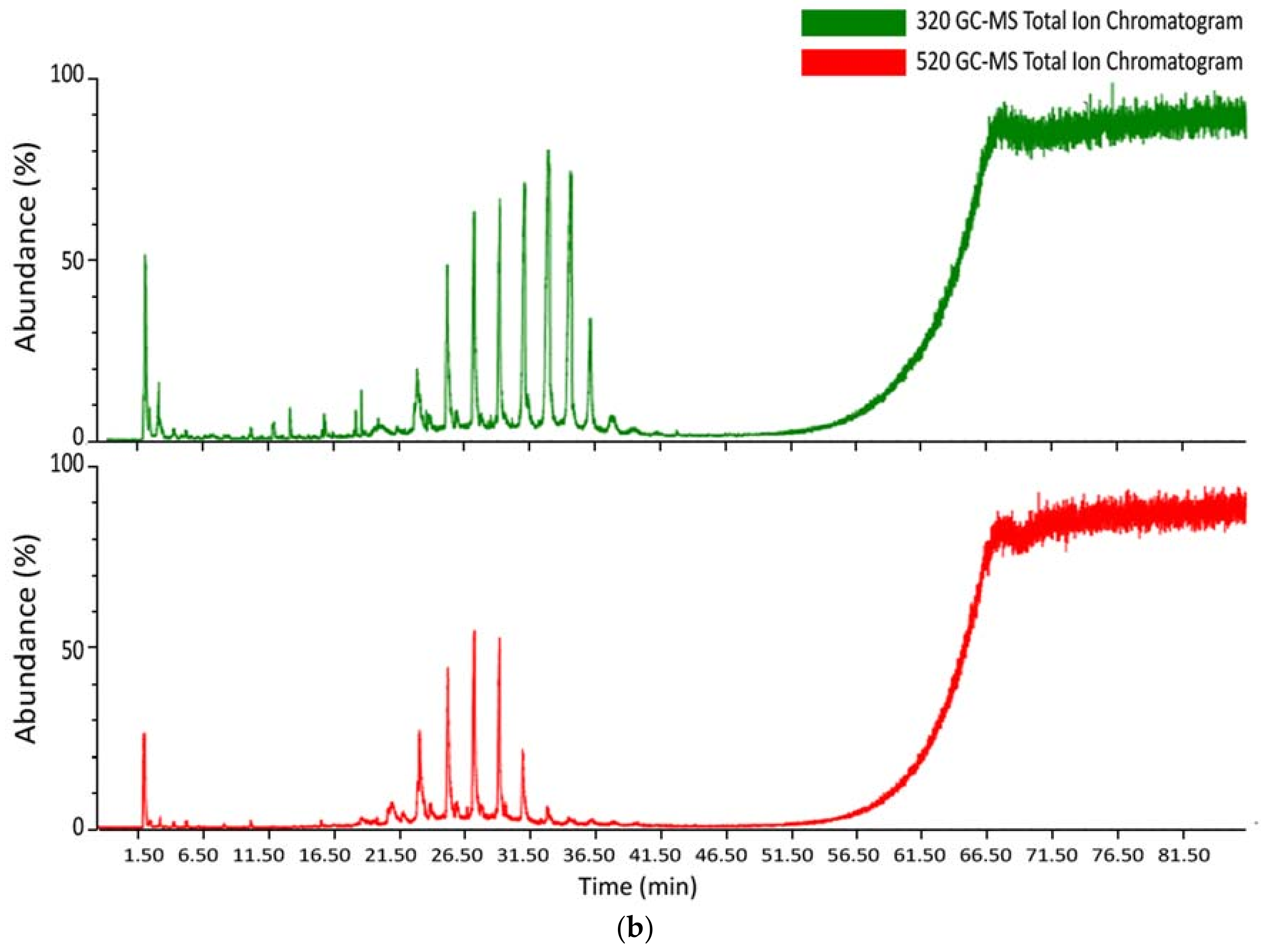
3.3. Mass Spectrometry Analysis
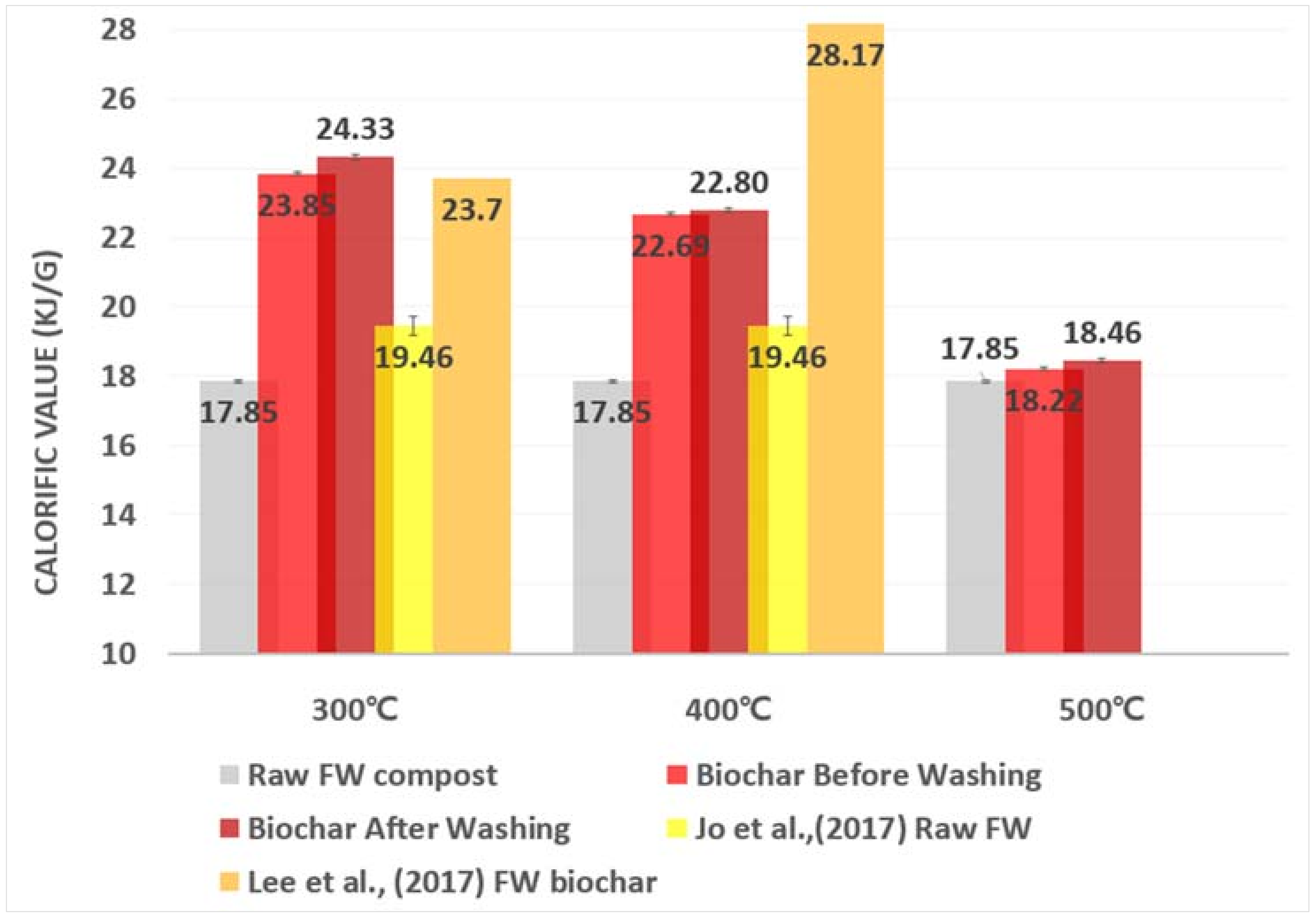
3.4. Biochar Production
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lee, S.E.; Ahn, H.J.; Youn, S.K.; Kim, S.M.; Jung, K.W. Application effect of food waste compost abundant in NaCl on the growth and cationic balance of rice plant in paddy soil. Kor. J. Soil Sci. Fertil. 2000, 33, 100–108. [Google Scholar]
- Zhao, S.-X.; Ta, N.; Wang, X.-D. Effect of Temperature on the Structural and Physicochemical Properties of Biochar with Apple Tree Branches as Feedstock Material. Energies 2017, 10, 1293. [Google Scholar]
- Lee, J.J.; Park, R.D.; Kim, Y.W.; Shim, J.H.; Chae, D.H.; Rim, Y.S.; Sohn, B.K.; Kim, T.H; Kim, K.Y. Effect of food waste compost on microbial population, soil enzyme activity and lettuce growth. Bioresour. Technol. 2004, 93, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Environment. 2015 Food Waste Treatment Facility Installation and Operation Status (Nationwide); Ministry of Environment: Sejong City, Korea, 2015.
- Ghorbel, L.; Rouissi, T.; Brar, S.K.; López-González, D.; Ramirez, A.A.; Godbout, S. Value-added performance of processed cardboard and farm breeding compost by pyrolysis. Waste Manag. 2015, 38, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.T. Waste Treatment and Disposal; John Wiley & Sons: New York, NY, USA, 2005. [Google Scholar]
- Brassard, P.; Godbout, S.; Raghavan, V.; Palacios, J.H.; Grenier, M.; Zegan, D. The Production of Engineered Biochars in a Vertical Auger Pyrolysis Reactor for Carbon Sequestration. Energies 2017, 10, 288. [Google Scholar] [CrossRef]
- Imam, T.; Capareda, S. Characterization of bio-oil, syn-gas and bio-char from switchgrass pyrolysis at various temperatures. J. Anal. Appl. Pyrolysis 2012, 93, 170–177. [Google Scholar] [CrossRef]
- Jahirul, M.I.; Rasul, M.G.; Chowdhury, A.A.; Ashwath, N. Biofuels production through biomass pyrolysis—A technological review. Energies 2012, 5, 4952–5001. [Google Scholar] [CrossRef]
- Barneto, A.G.; Carmona, J.A.; Conesa Ferrer, J.A.; Díaz Blanco, M.G. Kinetic study on the thermal degradation of a biomass and its compost: Composting effect on hydrogen production. Fuel 2010, 89, 462–473. [Google Scholar] [CrossRef]
- Ryu, C.; Finney, K.; Sharifi, V.N.; Swithenbank, J. Pelletised fuel production from coal tailings and spent mushroom compost—Part I: Identification of pelletisation parameters. Fuel Process. Technol. 2008, 89, 269–275. [Google Scholar] [CrossRef]
- Lee, Y.-E.; Jo, J.-H.; Kim, S.-M.; Yoo, Y.-S. Recycling Possibility of the Salty Food Waste by Pyrolysis and Water Scrubbing. Energies 2017, 10, 210. [Google Scholar] [CrossRef]
- Phan, A.N.; Ryu, C.; Sharifi, V.N.; Swithenbank, J. Characterisation of slow pyrolysis products from segregated wastes for energy production. J. Anal. Appl. Pyrolysis 2008, 81, 65–71. [Google Scholar] [CrossRef]
- Ryu, C.; Sharifi, V.N.; Swithenbank, J. Waste pyrolysis and generation of storable char. Int. J. Energy Res. 2007, 31, 177–191. [Google Scholar] [CrossRef]
- Jo, J.-H.; Kim, S.-S.; Shim, J.-W.; Lee, Y.-E.; Yoo, Y.-S. Pyrolysis Characteristics and Kinetics of Food Wastes. Energies 2017, 10, 1191. [Google Scholar] [CrossRef]
- Zhang, R.; El-Mashad, H.M.; Hartman, K.; Wang, F.; Liu, G.; Choate, C.; Gamble, P. Characterization of food waste as feedstock for anaerobic digestion. Bioresour. Technol. 2007, 98, 929–935. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.-H.; Lee, D.-H. Evaluation of Korean food waste composting with fed-batch operations I: Using water extractable total organic carbon contents (TOCw). Process Biochem. 2004, 39, 1183–1194. [Google Scholar] [CrossRef]
- Chang, I.J.; Hsu, T. Effects of compositions on food waste composting. Bioresour. Technol. 2008, 99, 8068–8074. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-E.; Jo, J.-H.; Kim, I.-T.; Yoo, Y.-S. Chemical Characteristics and NaCl Component Behavior of Biochar Derived from the Salty Food Waste by Water Flushing. Energies 2017, 10, 1555. [Google Scholar] [CrossRef]
- EN, BS. 15359: 2011 Solid Recovered Fuels. Specifications and Classes; BSI: London, UK, 2011; p. 30.
- Vamvuka, D.; Kakaras, E.; Kastanaki, E.; Grammelis, P. Pyrolysis characteristics and kinetics of biomass residuals mixtures with lignite☆. Fuel 2003, 82, 1949–1960. [Google Scholar] [CrossRef]
- Yang, H.; Yan, R.; Chen, H.; Lee, D.H.; Zheng, C. Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel 2007, 86, 1781–1788. [Google Scholar] [CrossRef]
- Gao, N.; Li, A.; Quan, C.; Du, L.; Duan, Y. TG–FTIR and Py–GC/MS analysis on pyrolysis and combustion of pine sawdust. J. Anal. Appl. Pyrolysis 2013, 100, 26–32. [Google Scholar] [CrossRef]
- Ulloa, C.A.; Gordon, A.L.; García, X.A. Thermogravimetric study of interactions in the pyrolysis of blends of coal with radiata pine sawdust. Fuel Process. Technol. 2009, 90, 583–590. [Google Scholar] [CrossRef]
- De Souza, A.G.; Santos, J.C.O.; Conceição, M.M.; Silva1, M.C.D.; Prasad, S. A thermoanalytic and kinetic study of sunflower oil. Braz. J. Chem. Eng. 2004, 21, 265–273. [Google Scholar] [CrossRef]
- Bihari-Varga, M.; Sepulchre, C.; Moczár, E. Thermoanalytical studies on protein-polysaccharide complexes of connective tissues. J. Therm. Anal. Calorim. 1975, 7, 675–683. [Google Scholar] [CrossRef]
- Gu, X.; Ma, X.; Li, L.; Liu, C.; Cheng, K.; Li, Z. Pyrolysis of poplar wood sawdust by TG-FTIR and Py–GC/MS. J. Anal. Appl. Pyrolysis 2013, 102, 16–23. [Google Scholar] [CrossRef]
- Som, M.-P.; Lemée, L.; Amblès, A. Stability and maturity of a green waste and biowaste compost assessed on the basis of a molecular study using spectroscopy, thermal analysis, thermodesorption and thermochemolysis. Bioresour. Technol. 2009, 100, 4404–4416. [Google Scholar] [CrossRef] [PubMed]
- Smidt, E.; Eckhardt, K.U.; Lechner, P.; Schulten, H.R.; Leinweber, P. Characterization of different decomposition stages of biowaste using FT-IR spectroscopy and pyrolysis-field ionization mass spectrometry. Biodegradation 2005, 16, 67–79. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, P.G. Whole microorganisms studied by pyrolysis-gas chromatography-mass spectrometry: Significance for extraterrestrial life detection experiments. Appl. Microbiol. 1970, 20, 567–572. [Google Scholar] [PubMed]
- Chen, G.; Leung, D.Y.C. Experimental investigation of biomass waste, (rice straw, cotton stalk, and pine sawdust), pyrolysis characteristics. Energy Sources 2003, 25, 331–337. [Google Scholar] [CrossRef]
- Kim, K.-S.; Choi, E.-A.; Ryu, J.-S.; Lee, Y.P.; Park, J.-Y.; Choi, S.-H.; Park, S.-J. A study on pyrolysis characterization and heating value of semi-carbonized wood chip. Appl. Chem. Eng. 2012, 23, 440–444. [Google Scholar]
- Demirbas, A. Effects of temperature and particle size on bio-char yield from pyrolysis of agricultural residues. J. Anal. Appl. Pyrolysis 2004, 72, 243–248. [Google Scholar] [CrossRef]
| Sample | C | H | N | Moisture | Dry Basis | Reference | ||
|---|---|---|---|---|---|---|---|---|
| Ash | Volatile | Fixed Carbon | ||||||
| Food waste compost | 29.7 | 4.3 | 2.9 | 21.6 4.4 | 9.8 | 78.3 | 11.9 | This study |
| Sawdust | 24.4 | 2.9 | - | 28.6 3.1 | 0.6 | 83.3 | 16.14 | |
| Mixed food | 47.5 | 12.2 | 2.9 | 85.7 2.9 | 3.8 0.1 | 79.0 2.7 | 17.25 | Jo et al. [15] |
| Food waste | 46.8 | - | 3.2 | - | - | 85.3 0.7 | 4.54 | Zhang, Ruihong, et al. [16] |
| Cafeteria food waste | - | - | - | 80.0 2.3 | 1.3 0.3 | 93.6 1.9 | - | Kwon and Lee et al. [17] |
| (wt %) | C | H | N | S | Cl | Na |
|---|---|---|---|---|---|---|
| Compost | 29.73 | 4.27 | 2.90 | - | 0.66 | 0.77 |
| 300 1 | 51.04 0.11 | 5.53 0.09 | 4.55 0.09 | - | 0.18 0.03 | 0.64 0.02 |
| 300 2 | 51.83 0.16 | 5.39 0.10 | 4.68 0.22 | - | 1.13 0.14 | 1.57 0.12 |
| 400 1 | 48.67 0.46 | 4.28 0.09 | 3.74 0.05 | - | 0.30 0.03 | 1.11 0.08 |
| 400 2 | 47.58 0.45 | 3.75 0.74 | 3.25 0.88 | - | 1.73 0.03 | 1.91 0.05 |
| 500 1 | 51.72 1.31 | 2.24 0.21 | 3.39 0.31 | - | 1.03 0.24 | 1.52 0.01 |
| 500 2 | 48.42 0.55 | 1.94 0.07 | 3.61 0.39 | - | 2.52 0.02 | 2.64 0.00 |
| Temperature | Time (min) | MW | Compound Name | Formula |
|---|---|---|---|---|
| 320 °C | 1.973 | 130 | thiocyanic acid, methylene ester | C3H2N2S2 |
| 2.279 | 122 | Butanoic acid, 4-chloro | C4H7O2Cl | |
| 6.617 | 96 | furfural | C5H4O2 | |
| 7.21 | 98 | furanmethanol | C5H6O2 | |
| 8.503 | 84 | 2(5H)-furanone | C4H4O2 |
| Temperature | Time (min) | MW | Compound Name | Formula |
|---|---|---|---|---|
| 320 °C | 1.962 | 285 | fumaric Acid 2-dimethylaminoethyl-heptyl ester (N-contain) | C15H27O4N |
| 2.296 | 88 | Thiophene, Tetrahydro | C4H8S | |
| 6.634 | 96 | 1H-pyrazole, 3,4-dimethyl(C) | C5H8N2 | |
| 7.213 | 88 | methylene cyclo propane carboxylic acid | C5H6O2 | |
| 8.497 | 84 | 2(5H)-furanone(C,P) | C4H4O2 | |
| 9.694 | 106 | Benzaldehyde | C7H6O | |
| 9.892 | 157 | Glycine, N-Cyclopropylcarbonyl-methyl ester | C7H11O3N | |
| 520 °C | 2.356 | 202 | 1,3-propanediol, 2-methyl-dipropanoate(Fatty acids, aliphatic compounds) | C10H18O4 |
| 2.866 | 78 | Benzene | C6H6 | |
| 3.334 | 258 | oxalic acid butyl 2-Ethylhexyl ester(Fatty acids, aliphatic compounds) | C14H26O4 | |
| 5.764 | 214 | pentanoic acid 2-Ethylhexyl ester | C13H26O2 | |
| 8.384 | 214 | propanoic acid, 2,2-Dimethyl-,2-Ethylhexyl ester | C13H26O2 |
| Temperature (°C) | Before Pyrolysis (g) | After Pyrolysis (g) | Yield (%) |
|---|---|---|---|
| 300 | 200.52 0.38 | 88.52 2.07 | 44.14 0.95 |
| 400 | 200.32 0.16 | 60.57 0.04 | 30.24 0.05 |
| 500 | 200.31 0.12 | 45.09 3.20 | 22.51 1.61 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, Y.-E.; Jo, J.-H.; Kim, I.-T.; Yoo, Y.-S. Value-Added Performance and Thermal Decomposition Characteristics of Dumped Food Waste Compost by Pyrolysis. Energies 2018, 11, 1061. https://doi.org/10.3390/en11051061
Lee Y-E, Jo J-H, Kim I-T, Yoo Y-S. Value-Added Performance and Thermal Decomposition Characteristics of Dumped Food Waste Compost by Pyrolysis. Energies. 2018; 11(5):1061. https://doi.org/10.3390/en11051061
Chicago/Turabian StyleLee, Ye-Eun, Jun-Ho Jo, I-Tae Kim, and Yeong-Seok Yoo. 2018. "Value-Added Performance and Thermal Decomposition Characteristics of Dumped Food Waste Compost by Pyrolysis" Energies 11, no. 5: 1061. https://doi.org/10.3390/en11051061
APA StyleLee, Y.-E., Jo, J.-H., Kim, I.-T., & Yoo, Y.-S. (2018). Value-Added Performance and Thermal Decomposition Characteristics of Dumped Food Waste Compost by Pyrolysis. Energies, 11(5), 1061. https://doi.org/10.3390/en11051061





