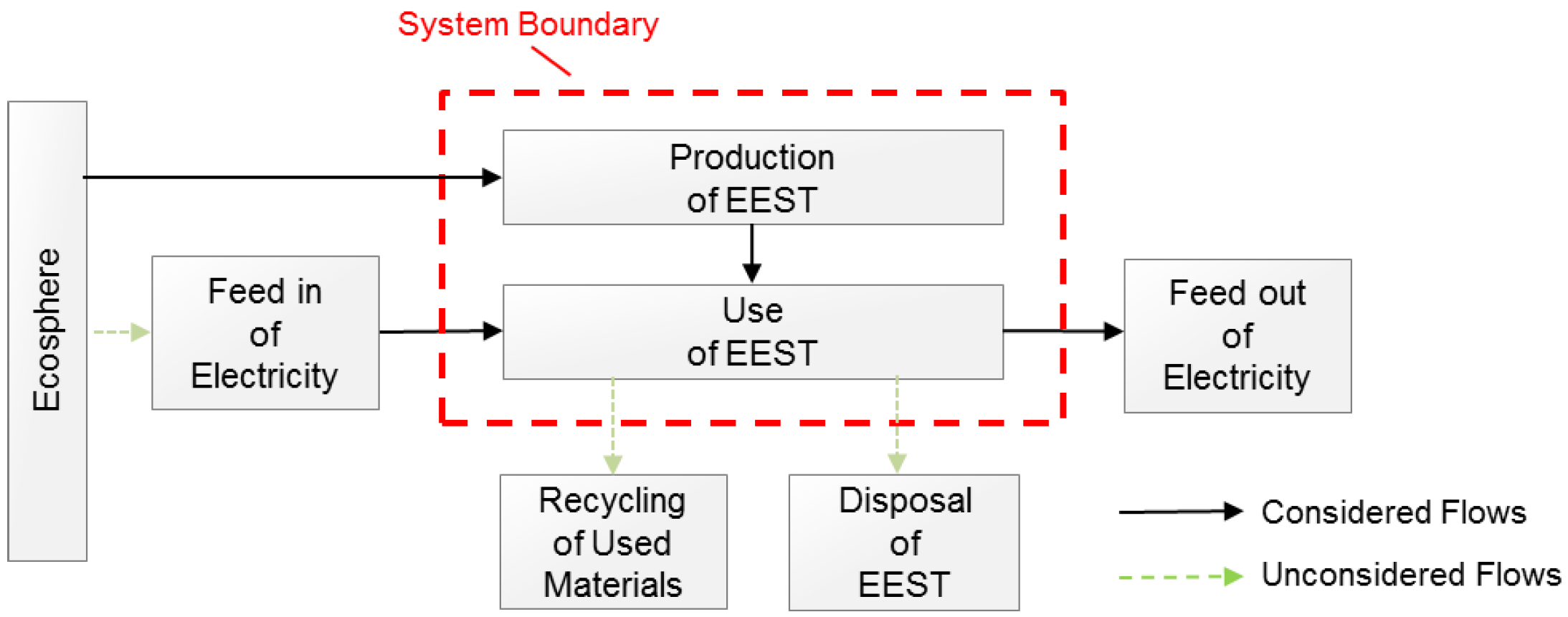
Appendix A
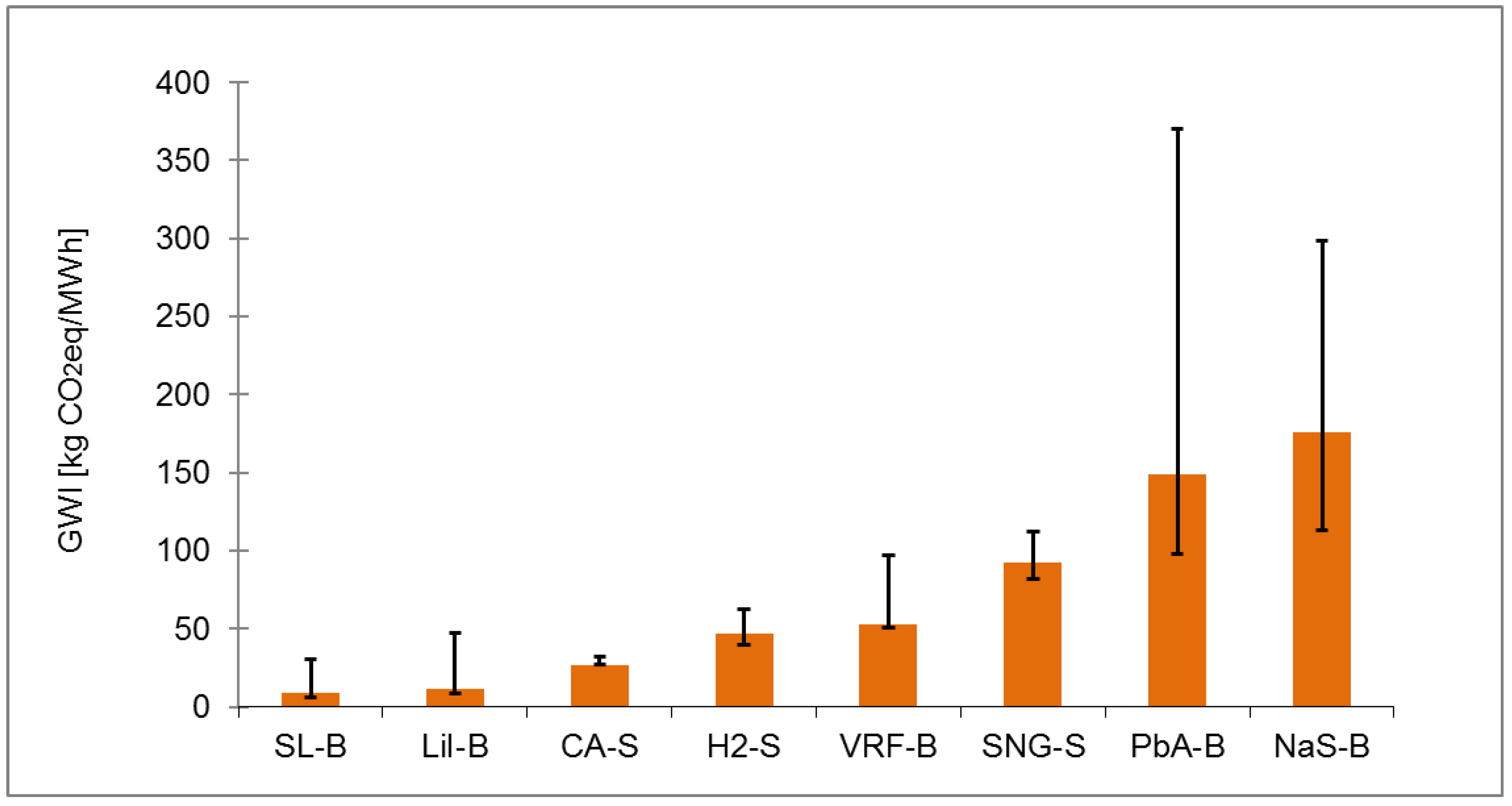
Table A1.
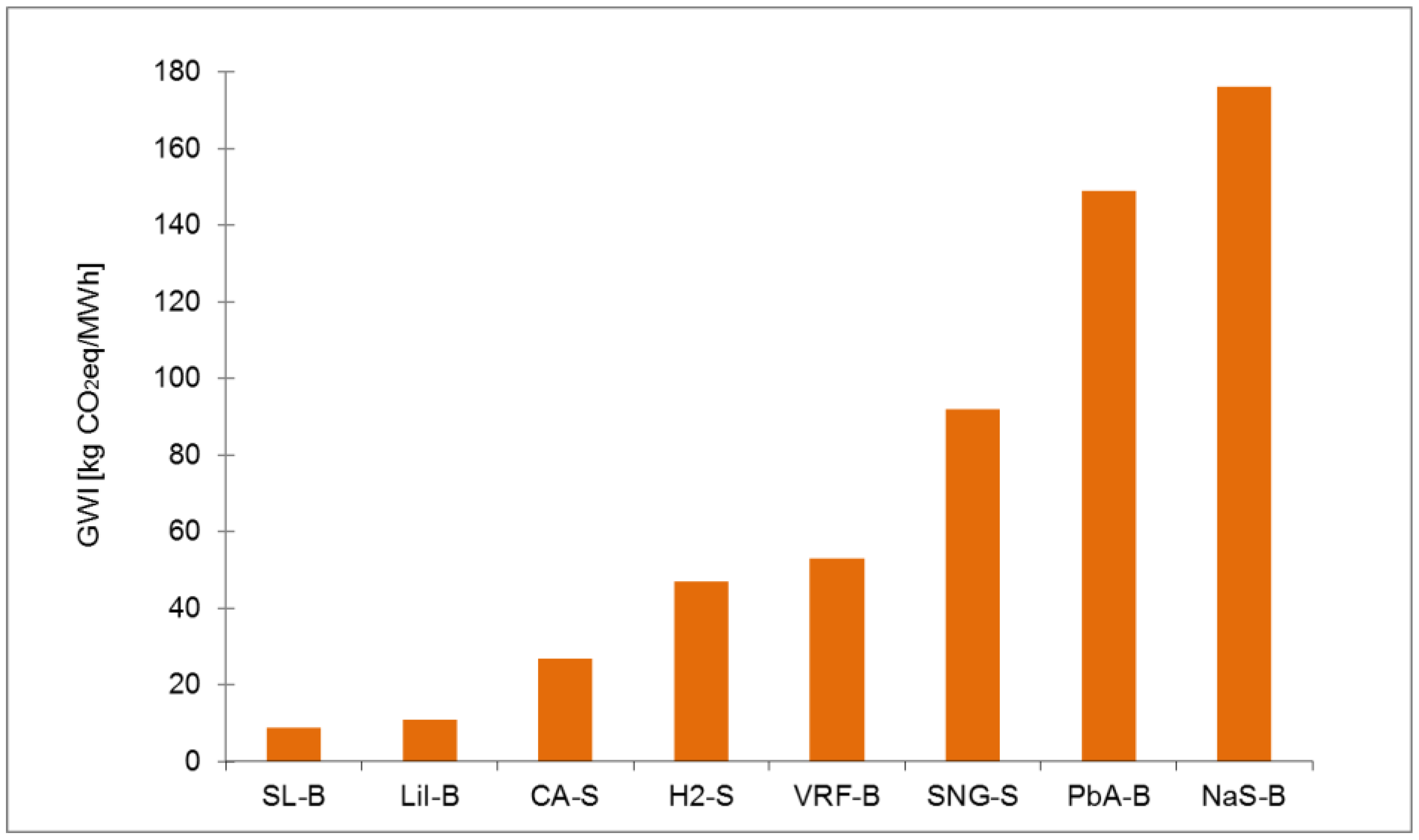
Ranking of electrical energy storage technologies for GWI with normal, shorter and longer lifetime.
Table A1.
Ranking of electrical energy storage technologies for GWI with normal, shorter and longer lifetime.
| GWI (kg CO2eq·MWh−1) | SL-B | LiI-B | CA-S | H2-S | VRF-B | SNG-S | PbA-B | NaS-B |
|---|
| Normal lifetime Value | 8.88 | 11.41 | 27.33 | 46.56 | 53.08 | 92.09 | 148.77 | 175.84 |
| Rank | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| Shorter lifetime | 30.37 | 47.42 | 32.37 | 62.08 | 97.29 | 112.16 | 370.08 | 298.48 |
| Change [%] | +241.9 | +315.5 | +18.4 | +33.3 | +83.3 | +21.8 | +148.8 | +69.7 |
| Rank | 1 | 3 | 2 | 4 | 5 | 6 | 8 | 7 |
| Longer lifetime | 6.15 | 8.66 | 27.33 | 38.92 | 50.51 | 82.20 | 94.70 | 112.67 |
| Change [%] | −30.8 | −24.1 | 0.0 | −16.4 | −4.9 | −10.7 | −36.3 | −35.9 |
| Rank | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
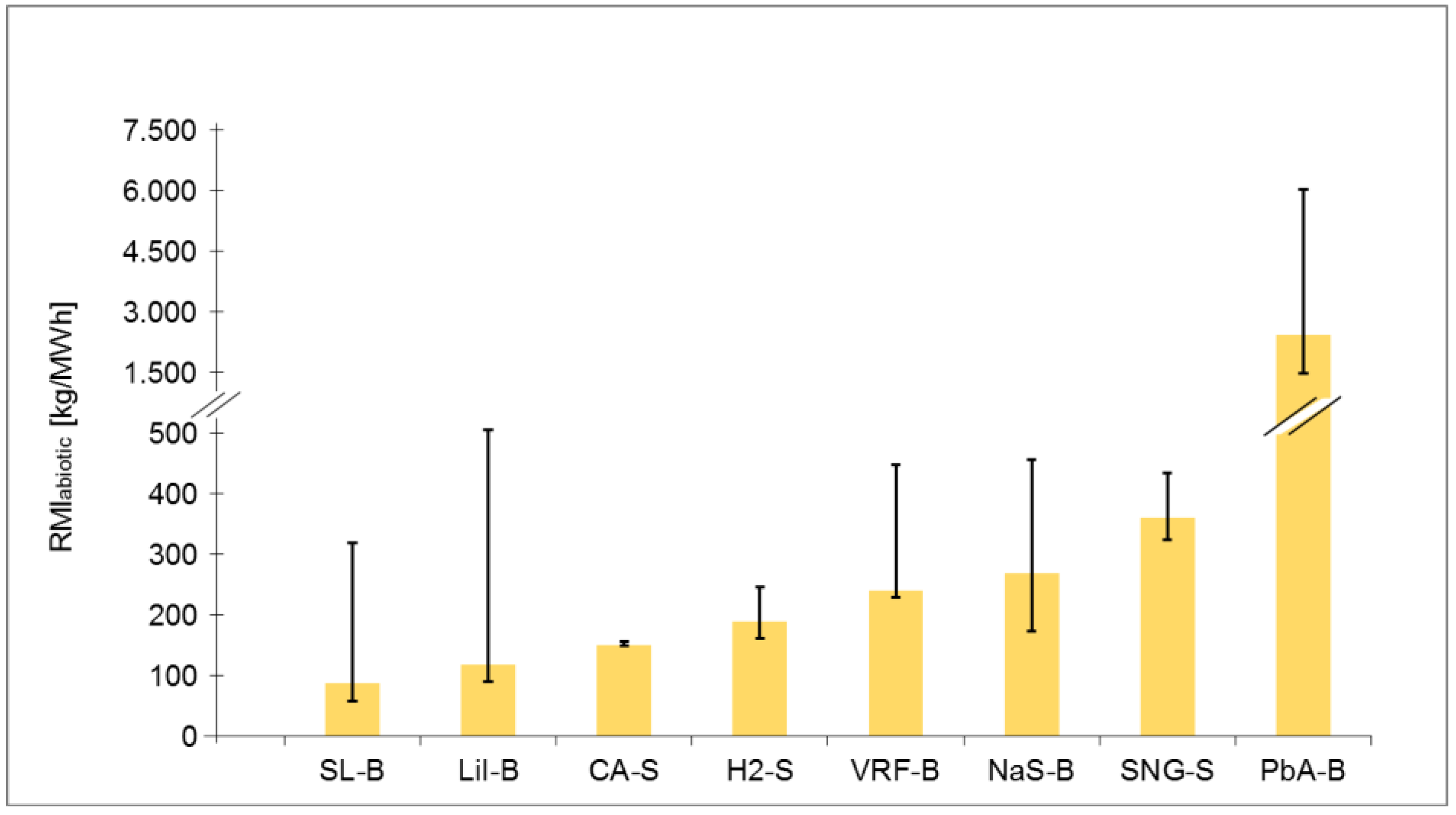
Table A2.
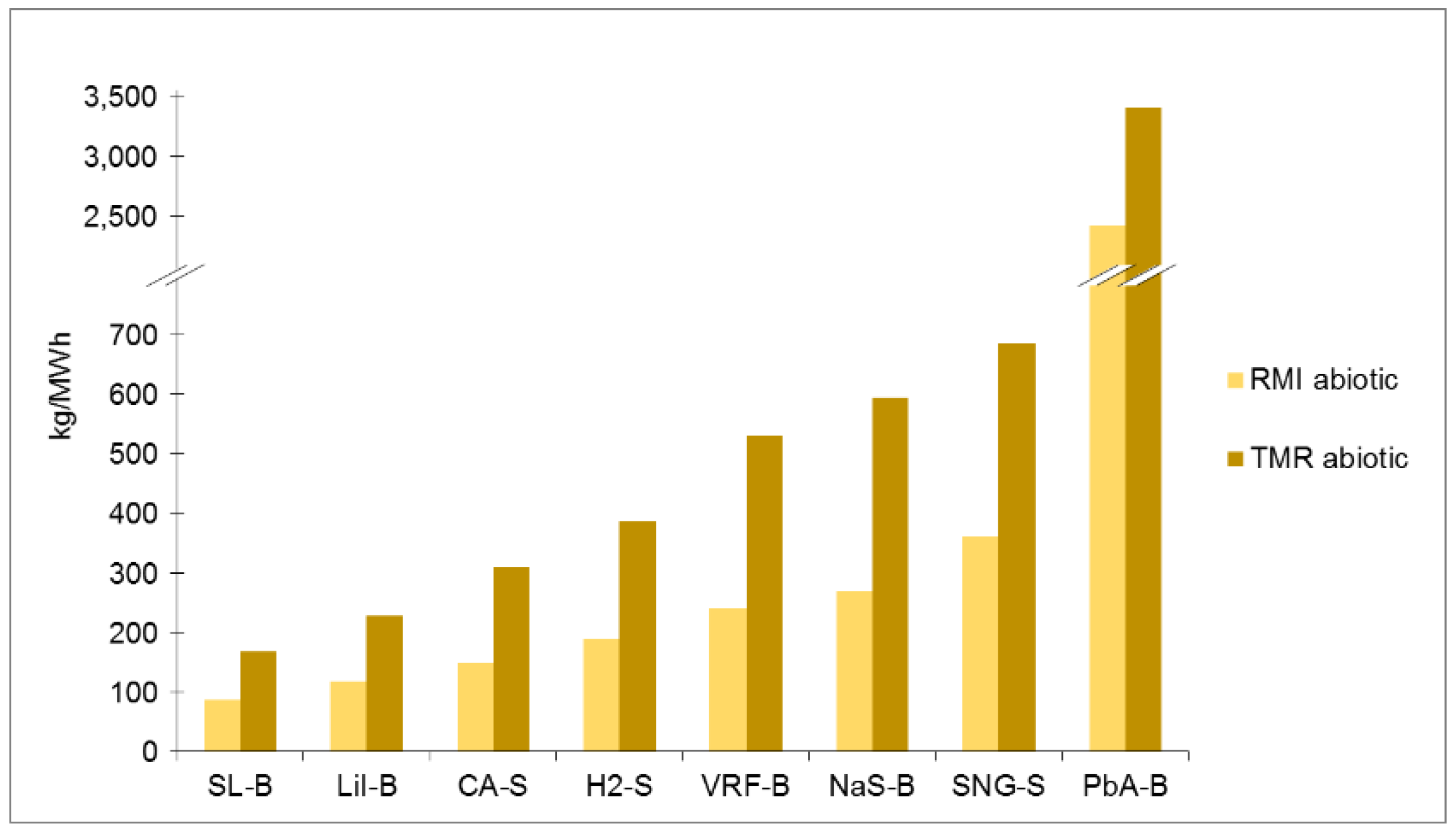
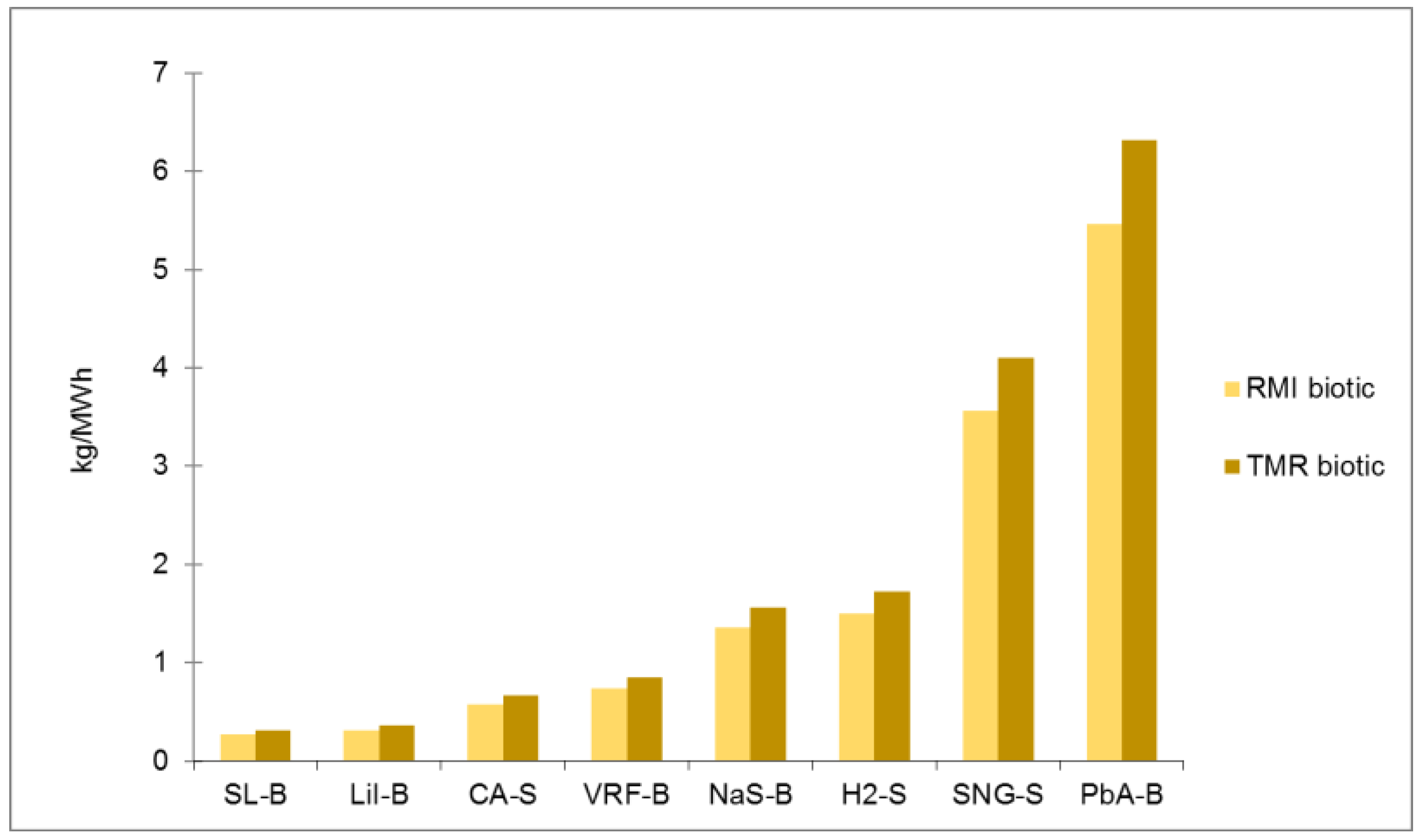
Ranking of electrical energy storage technologies for RMI abiotic with normal, shorter and longer lifetime.
Table A2.
Ranking of electrical energy storage technologies for RMI abiotic with normal, shorter and longer lifetime.
| RMIabiotic (kg·MWh−1) | SL-B | LiI-B | CA-S | H2-S | VRF-B | SNG-S | PbA | NaS-B |
|---|
| Normal lifetime Value | 87 | 118 | 149 | 189 | 240 | 360 | 2414 | 269 |
| Rank | 1 | 2 | 3 | 4 | 5 | 7 | 8 | 6 |
| Shorter lifetime | 318 | 505 | 155 | 246 | 447 | 433 | 6032 | 455 |
| Change [%] | +266.5 | +327.3 | +3.8 | +30.1 | +86.0 | +20.5 | +149.8 | +69.3 |
| Rank | 3 | 7 | 1 | 2 | 5 | 4 | 8 | 6 |
| Longer lifetime | 57 | 89 | 149 | 161 | 228 | 323 | 1531 | 173 |
| Change [%] | −33.9 | −25.0 | 0.0 | −14.8 | −5.0 | −10.1 | −36.6 | −35.7 |
| Rank | 1 | 2 | 3 | 4 | 6 | 7 | 8 | 5 |
Table A3.
Ranking of electrical energy storage technologies for TMR abiotic with normal, shorter and longer lifetime.
Table A3.
Ranking of electrical energy storage technologies for TMR abiotic with normal, shorter and longer lifetime.
| TMRabiotic (kg·MWh−1) | SL-B | LiI-B | CA-S | H2-S | VRF-B | SNG-S | PbA | NaS-B |
|---|
| Normal lifetime | 168 | 229 | 309 | 387 | 531 | 686 | 3411 | 595 |
| Rank | 1 | 2 | 3 | 4 | 5 | 7 | 8 | 6 |
| Shorter lifetime | 614 | 976 | 320 | 499 | 988 | 830 | 8517 | 1008 |
| Change [%] | +265.5 | +326.8 | +3.6 | +28.8 | +86.0 | +21.0 | +149.7 | +69.3 |
| Rank | 3 | 5 | 1 | 2 | 6 | 4 | 8 | 7 |
| Longer lifetime | 111 | 172 | 309 | 332 | 504 | 615 | 2163 | 383 |
| Change [%] | −33.8 | −25.0 | 0.00 | −14.2 | −5.0 | −10.3 | −36.6 | −35.7 |
| Rank | 1 | 2 | 3 | 4 | 6 | 7 | 8 | 5 |
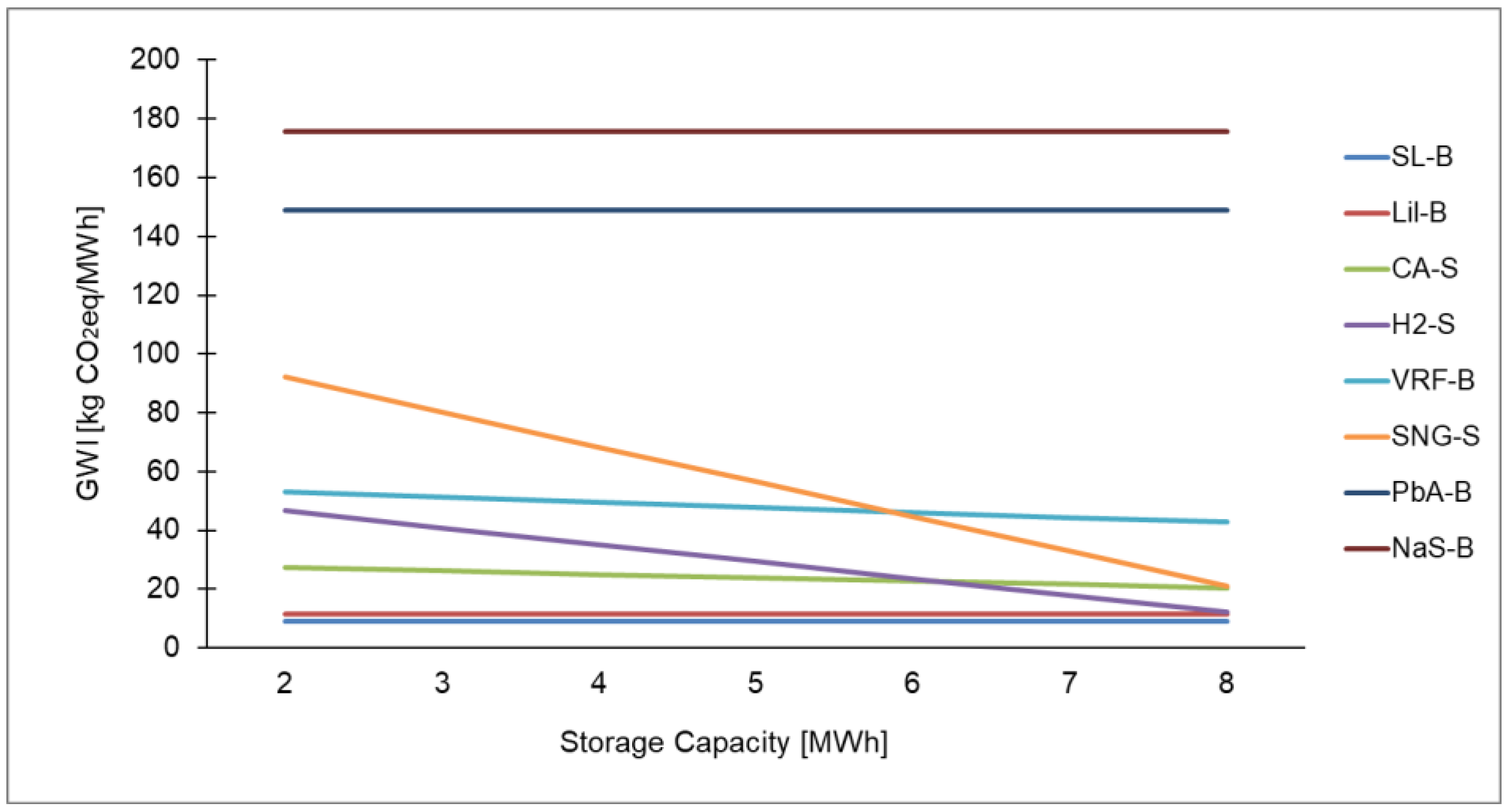
Table A4.
Ranking of electrical energy storage technologies for GWI with 2 and 8 MWh storage capacity.
Table A4.
Ranking of electrical energy storage technologies for GWI with 2 and 8 MWh storage capacity.
| GWI (kg CO2eq·MWh−1) | SL-B | LiI-B | CA-S | H2-S | VRF-B | SNG-S | PbA-B | NaS-B |
|---|
| 2 MWh | 8.88 | 11.41 | 27.33 | 46.56 | 53.08 | 92.09 | 148.77 | 175.84 |
| Rank | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
| 8 MWh | 8.88 | 11.41 | 20.45 | 12.07 | 42.70 | 20.96 | 148.77 | 175.84 |
| Change (%) | 0.0 | 0.0 | −25.2 | −74.1 | −19.6 | −77.2 | 0.0 | 0.0 |
| Rank | 1 | 2 | 4 | 3 | 6 | 5 | 7 | 8 |
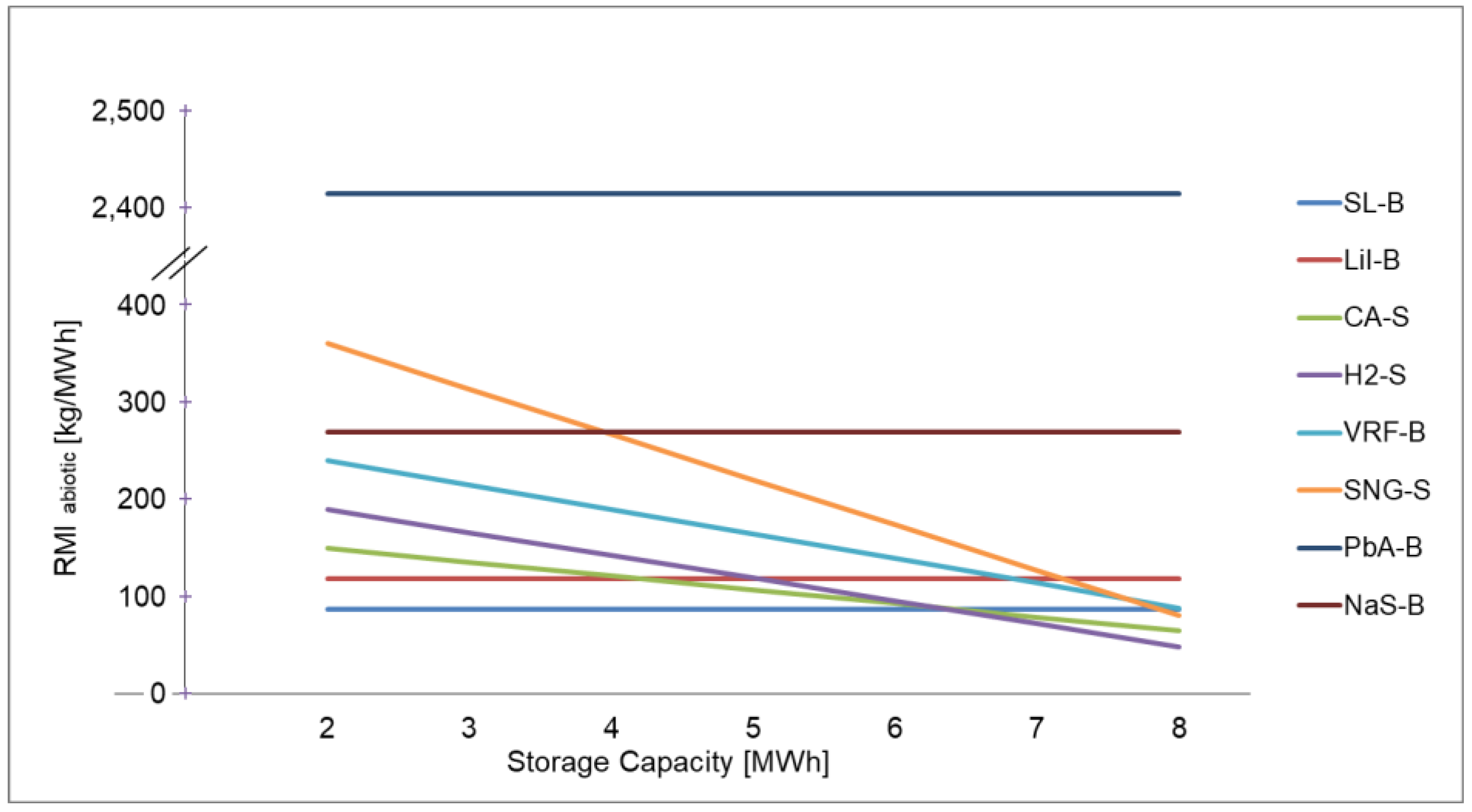
Table A5.
Ranking of electrical energy storage technologies for RMI abiotic with 2 and 8 MWh storage capacity.
Table A5.
Ranking of electrical energy storage technologies for RMI abiotic with 2 and 8 MWh storage capacity.
| RMIabiotic (kg·MWh−1) | SL-B | LiI-B | CA-S | H2-S | VRF-B | SNG-S | PbA | NaS-B |
|---|
| 2 MWh | 87 | 118 | 149 | 189 | 240 | 360 | 2414 | 269 |
| Rank | 1 | 2 | 3 | 4 | 5 | 7 | 8 | 6 |
| 8 MWh capacity | 87 | 118 | 64 | 48 | 88 | 80 | 2414 | 269 |
| Change (%) | 0.0 | 0.0 | −57.0 | −74.6 | −63.3 | −77.8 | 0.0 | 0.0 |
| Rank | 4 | 6 | 2 | 1 | 5 | 3 | 8 | 7 |
Table A6.
Ranking of electrical energy storage technologies for TMR abiotic with 2 and 8 MWh storage capacity.
Table A6.
Ranking of electrical energy storage technologies for TMR abiotic with 2 and 8 MWh storage capacity.
| TMRabiotic (kg·MWh−1) | SL-B | LiI-B | CA-S | H2-S | VRF-B | SNG-S | PbA | NaS-B |
|---|
| 2 MWh | 168 | 229 | 309 | 387 | 531 | 686 | 3411 | 595 |
| Rank | 1 | 2 | 3 | 4 | 5 | 7 | 8 | 6 |
| 8 MWh | 168 | 229 | 123 | 98 | 208 | 158 | 3411 | 595 |
| Change (%) | 0.0 | 0.0 | −60.2 | −75.7 | −60.8 | −77.0 | 0.0 | 0.0 |
| Rank | 4 | 6 | 2 | 1 | 5 | 3 | 8 | 7 |
Appendix B
Table A7.
Manufacturing, Intermodal container 40’ high cube.
Table A7.
Manufacturing, Intermodal container 40’ high cube.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| steel, low-alloyed, hot rolled—GLO | kg | 1896 | market for steel, low-alloyed, hot rolled, cut-off, U-GLO |
| roundwood, meranti from sustainable forest management, under bark—GLO | m3 | 0.4 | market for roundwood, meranti from sustainable forest management, under bark, cut-off, U-GLO |
| Outputs | | | |
| Intermodal container 40’ high-cube | Item (s) | 1 | |
Table A8.
Manufacturing, Intermodal container 20’ high cube.
Table A8.
Manufacturing, Intermodal container 20’ high cube.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| steel, low-alloyed, hot rolled—GLO | kg | 3232 | market for steel, low-alloyed, hot rolled, cut-off, U-GLO |
| roundwood, meranti from sustainable forest management, under bark—GLO | m3 | 0.8 | market for roundwood, meranti from sustainable forest management, under bark, cut-off, U-GLO |
| Outputs | | | |
| Intermodal container 20’ high-cube | Item (s) | 1 | |
Table A9.
Manufacturing, Electrolyseur.
Table A9.
Manufacturing, Electrolyseur.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| transport, freight, sea, transoceanic ship—GLO | kg * km | 1.61 × 108 | market for transport, freight, sea, transoceanic ship, cut-off, U-GLO |
| steel, chromium steel 18/8—GLO | t | 19.38 | market for steel, chromium steel 18/8, cut-off, U-GLO |
| steel, low-alloyed, hot rolled—GLO | t | 16.53 | market for steel. low-alloyed, hot rolled, cut-off, U-GLO |
| Iridium, in ground | g | 15.96 | |
| Platinum—GLO | g | 5.32 | market for platinum, cut-off, U-GLO |
| Intermodal container 40’ high-cube | Item(s) | 2 | Manufacturing, Intermodal container 40’ high cube |
| Outputs | | | |
| Electrolyseur | Item (s) | 1 | |
Table A10.
Production, Hydrogen.
Table A10.
Production, Hydrogen.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| water, ultrapure—DE | kg | 8.92 | water production for electrolysis, ultrapure—DE |
| Electrolyseur | Item (s) | 3.75 × 10−6 | Manufacturing, Electrolyseur |
| electricity, low voltage—DE | kWh | 0 | electricity voltage transformation from high to low voltage, wind power (1–3 MW) |
| Outputs | | | |
| Hydrogen | kg | 1 | |
Table A11.
Transport Hydrogen.
Table A11.
Transport Hydrogen.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| transport, pipeline, long distance, natural gas—DE | t * km | 0.05 | transport, pipeline, long distance, hydrogen, cut-off, U-DE |
| Hydrogen | kg | 1 | Production, Hydrogen |
| Outputs | | | |
| Hydrogen, transported | kg | 1 | |
Table A12.
Reconversion, Hydrogen.
Table A12.
Reconversion, Hydrogen.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| Hydrogen, transported | kg | 266,717 | Transport, Hydrogen |
| heat and power co-generation unit, 1MW electrical, components for electricity only—RER | Item (s) | 1 | heat and power co-generation unit construction, 1MW electrical, components for electricity only, cut-off, U-RER |
| Outputs | | | |
| electricity, high voltage—DE | MWh | 3557 | |
Table A13.
Capturing CO2 from biogas.
Table A13.
Capturing CO2 from biogas.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| electricity, low voltage—DE | kWh | 0.1136 | electricity voltage transformation from high to low voltage, wind power (1–3 MW) |
| Outputs | | | |
| Captured CO2 | kg | 1 | |
Table A14.
Production and Transport, SNG.
Table A14.
Production and Transport, SNG.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| Captured CO2 | kg | 2.68 | Capturing CO2 from biogas |
| Hydrogen | kg | 0.5061 | Production, Hydrogen |
| transport, pipeline, long distance, natural gas—DE | t * km | 0.05 | transport, pipeline, SNG, cut-off, U-DE |
| synthetic gas factory—DE | Item (s) | 3.21 × 10−7 | synthetic gas factory construction, cut-off, U-DE |
| electricity, low voltage—DE | kWh | 0 | electricity voltage transformation from high to low voltage, wind power (1–3 MW) |
| Outputs | | | |
| SNG | kg | 1 | |
Table A15.
Reconversion, SNG.
Table A15.
Reconversion, SNG.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| SNG | t | 513 | Production and Transport, SNG |
| heat and power co-generation unit, 1MW electrical, components for electricity only—RER | Item (s) | 1 | heat and power co-generation unit construction, 1MW electrical, components for electricity only, cut-off, U-RER |
| Outputs | | | |
| electricity, high voltage—DE | MWh | 2676 | |
Table A16.
Manufacturing Electrode, PbA-B.
Table A16.
Manufacturing Electrode, PbA-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| lead—RER | kg | 11.73 | treatment of scrap lead acid battery, remelting, cut-off, U-RER |
| graphite, battery grade—GLO | kg | 3.67 | market for graphite, battery grade, cut-off, U-GLO |
| lead—GLO | kg | 2.93 | primary lead production from concentrate, cut-off, U-GLO |
| sulfuric acid—GLO | kg | 2.20 | market for sulfuric acid, cut-off, U-GLO |
| water, ultrapure—GLO | kg | 1.47 | market for water, ultrapure, cut-off, U-GLO |
| heat, district or industrial, natural gas—RoW | kWh | 1.32 | market for heat, district or industrial, natural gas, cut-off, U-RoW |
| Outputs | | | |
| Electrode, PbA-B | kg | 1 | |
Table A17.
Manufacturing Electrolyte, PbA-B.
Table A17.
Manufacturing Electrolyte, PbA-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| silica fume, densified—GLO | kg | 1.07 | market for silica fume, densified, cut-off, U-GLO |
| water, ultrapure—DE | kg | 0.7 | water production for electrolysis, ultrapure-DE |
| sulfuric acid—RER | kg | 0.3 | sulfuric acid production, cut-off, U-RER |
| Outputs | | | |
| Electrolyte, PbA-B | kg | 1 | |
Table A18.
Manufacturing, PbA-B.
Table A18.
Manufacturing, PbA-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| heat, district or industrial, natural gas—RoW | MJ | 167.75 | market for heat, district or industrial, natural gas, cut-off, U-RoW |
| electricity, medium voltage—CN | MJ | 120.12 | market for electricity, medium voltage, cut-off, U-CN |
| Electrode, PbA-B | kg | 22 | Manufacturing Electrode, PbA-B |
| lead—RER | kg | 13.2 | treatment of scrap lead acid battery, remelting, cut-off, U-RER |
| Electrolyte, PbA-B | kg | 11 | Manufacturing Electrolyte, PbA-B |
| polypropylene, granulate—GLO | kg | 4.4 | market for polypropylene, granulate, cut-off, U-GLO |
| lead—GLO | kg | 3.3 | primary lead production from concentrate, cut-off, U-GLO |
| phenolic resin—GLO | kg | 1.1 | market for phenolic resin, cut-off, U-GLO |
| Outputs | | | |
| PbA-B | Item (s) | 1 | |
Table A19.
Manufacturing Storage, PbA-B.
Table A19.
Manufacturing Storage, PbA-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| PbA-B | Item (s) | 6.739 | Manufacturing, PbA-B |
| Intermodal container 40’ high-cube | Item (s) | 2 | Manufacturing, Intermodal container 40’ high cube |
| transport, freight, sea, transoceanic ship—GLO | kg * km | 7.92 × 109 | market for transport, freight, sea, transoceanic ship, cut-off, U-GLO |
| Outputs | | | |
| Storage, PbA-B | Item (s) | 1 | |
Table A20.
Use Storage, PbA-B.
Table A20.
Use Storage, PbA-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| Storage, PbA-B | Item (s) | 1 | Manufacturing Storage, PbA-B |
| electricity, high voltage—DE | MWh | 0 | electricity production, wind, 1–3 MW turbine, onshore, cut-off, U-DE |
| Outputs | | | |
| electricity, high voltage—DE | MWh | 11,899 | |
Table A21.
Manufacturing, VRF-B.
Table A21.
Manufacturing, VRF-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| water, ultrapure—DE | kg | 62,506 | water production for electrolysis, ultrapure—DE |
| sulfuric acid—RER | kg | 33,906 | sulfuric acid production, cut-off, U-RER |
| steel, low-alloyed, hot rolled—GLO | kg | 13,978 | market for steel, low-alloyed, hot rolled, cut-off, U-GLO |
| vanadium pentoxide | kg | 13,161 | vanadium pentoxide, production |
| copper—GLO | kg | 7360 | market for copper, cut-off, U-GLO |
| polypropylene, granulate—GLO | kg | 3333 | market for polypropylene, granulate, cut-off, U-GLO |
| graphite, battery grade—GLO | kg | 2400 | market for graphite, battery grade, cut-off, U-GLO |
| heat, district or industrial, natural gas—RoW | GJ | 1929.58 | market for heat, district or industrial, natural gas, cut-off, U-RoW |
| electricity, medium voltage—CN | GJ | 516.25 | market for electricity, medium voltage, cut-off, U-CN |
| Outputs | | | |
| VRF-B | Item (s) | 1 | |
Table A22.
Manufacturing Storage, VRF-B.
Table A22.
Manufacturing Storage, VRF-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| VRF-B | Item (s) | 2.11 | Manufacturing, VRF-B |
| Intermodal container 20’ high-cube | Item (s) | 10 | Manufacturing, Intermodal container 20’ high cube |
| transport, freight, sea, transoceanic ship—GLO | kg * km | 7.28 × 109 | market for transport, freight, sea, transoceanic ship, cut-off, U-GLO |
| Outputs | | | |
| Storage, VRF-B | Item (s) | 1 | |
Table A23.
Use Storage, VRF-B.
Table A23.
Use Storage, VRF-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| Storage, VRF-B | Item (s) | 1 | Manufacturing Storage, VRF-B |
| electricity, high voltage—DE | MWh | 0 | electricity production, wind, 1–3 MW turbine, onshore, cut-off, U-DE |
| Outputs | | | |
| electricity, high voltage—DE | MWh | 11,240 | |
Table A24.
Manufacturing, LiI-B.
Table A24.
Manufacturing, LiI-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| anode, graphite, for lithium-ion battery—GLO | kg | 0.4011 | market for anode, graphite, for lithium-ion battery, cut-off, U-GLO |
| cathode, LiMn2O4, for lithium-ion battery—GLO | kg | 0.3269 | market for cathode, LiMn2O4, for lithium-ion battery, cut-off, U-GLO |
| ethylene carbonate—GLO | kg | 0.1596 | market for ethylene carbonate, cut-off, U-GLO |
| electricity, medium voltage—CN | kWh | 0.1056 | market for electricity, medium voltage, cut-off, U-CN |
| polyethylene, low density, granulate—GLO | kg | 0.0733 | market for polyethylene, low density, granulate, cut-off, U-GLO |
| extrusion, plastic film—GLO | kg | 0.0733 | market for extrusion, plastic film, cut-off, U-GLO |
| heat, district or industrial, natural gas—RoW | MJ | 0.0652 | market for heat, district or industrial, natural gas, cut-off, U-RoW |
| battery separator—GLO | kg | 0.0537 | market for batt. sep., cut-off, U-GLO |
| lithium hexafluorophosphate—GLO | kg | 0.0190 | market for lithium hexafluorophosphate, cut-off, U-GLO |
| sheet rolling, aluminium—GLO | kg | 0.0165 | market for sheet rolling, aluminium, cut-off, U-GLO |
| aluminium, wrought alloy—GLO | kg | 0.0165 | market for aluminium, wrought alloy, cut-off, U-GLO |
| nitrogen, liquid—RoW | kg | 0.01 | market for nitrogen, liquid, cut-off, U-RoW |
| chemical factory, organics—GLO | Item (s) | 4.00 × 10−10 | market for chemical factory, organics, cut-off, U-GLO |
| Outputs | | | |
| LiI-B | Item (s) | 1 | |
Table A25.
Manufacturing Storage, LiI-B.
Table A25.
Manufacturing Storage, LiI-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| LiI-B | kg | 34,800 | Manufacturing, LiI-B |
| Intermodal container 40’ high-cube | Item (s) | 1 | Manufacturing, Intermodal container 40’ high cube |
| transport, freight, sea, transoceanic ship—GLO | kg * km | 8.09 × 108 | market for transport, freight, sea, transoceanic ship, cut-off, U-GLO |
| Outputs | | | |
| Storage, LiI-B | Item (s) | 1 | |
Table A26.
Use Storage, LiI-B.
Table A26.
Use Storage, LiI-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| Storage, LiI-B | Item (s) | 1 | Manufacturing Storage, LiI-B |
| electricity, high voltage—DE | MWh | 0 | electricity production, wind, 1–3 MW turbine, onshore, cut-off, U-DE |
| Outputs | | | |
| electricity, high voltage—DE | MWh | 13,651 | |
Table A27.
Manufacturing, NaS-B.
Table A27.
Manufacturing, NaS-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| heat, district or industrial, natural gas—RoW | MJ | 495 | market for heat, district or industrial, natural gas, cut-off, U-RoW |
| electricity, medium voltage—CN | MJ | 81.41 | market for electricity, medium voltage, cut-off, U-CN |
| steel, chromium steel 18/8—GLO | kg | 1.6324 | market for steel, chromium steel 18/8, cut-off, U-GLO |
| sulfur—GLO | kg | 1.0547 | market for sulfur, cut-off, U-GLO |
| aluminium oxide—GLO | kg | 0.9635 | market for aluminium oxide, cut-off, U-GLO |
| sodium—GLO | kg | 0.6307 | market for sodium, cut-off, U-GLO |
| glass, borosilicate | kg | 0.3392 | glass, borosilicate, cut-off, U-RoW |
| aluminium oxide—GLO | kg | 0.2332 | market for aluminium oxide, cut-off, U-GLO |
| graphite, battery grade—GLO | kg | 0.2067 | market for graphite, battery grade, cut-off, U-GLO |
| copper—GLO | kg | 0.1325 | market for copper, cut-off, U-GLO |
| sodium aluminate, powder—GLO | kg | 0.0857 | market for sodium aluminate, powder, cut-off, U-GLO |
| magnesium oxide—GLO | kg | 0.0214 | market for magnesium oxide, cut-off, U-GLO |
| Outputs | | | |
| NaS-B | Item (s) | 1 | |
Table A28.
Manufacturing Storage, NaS-B.
Table A28.
Manufacturing Storage, NaS-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| steel, low-alloyed, hot rolled—GLO | t | 7.2 | market for steel, low-alloyed, hot rolled, cut-off, U-GLO |
| sand—GLO | kg | 141,000 | market for sand, cut-off, U-GLO |
| steel, chromium steel 18/8—GLO | kg | 4982 | market for steel, chromium steel 18/8, cut-off, U-GLO |
| battery cell, NaS—JP | Item (s) | 36,096 | Battery cell production, NaS-JP |
| transport, freight, sea, transoceanic ship—GLO | kg * km | 4.03 × 109 | market for transport, freight, sea, transoceanic ship, cut-off, U-GLO |
| Outputs | | | |
| Storage, NaS-B | Item (s) | 1 | |
Table A29.
Use Storage, NaS-B.
Table A29.
Use Storage, NaS-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| Storage, NaS-B | Item (s) | 1 | Manufacturing Storage, NaS-B |
| electricity, high voltage—DE | MWh | 0 | electricity production, wind, 1–3 MW turbine, onshore, cut-off, U-DE |
| Outputs | | | |
| electricity, high voltage—DE | MWh | 11,169 | |
Table A30.
Manufacturing, CA-S.
Table A30.
Manufacturing, CA-S.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| transport, freight, sea, transoceanic ship—GLO | kg * km | 1.61 × 108 | market for transport, freight, sea, transoceanic ship, cut-off, U-GLO |
| naphtha—RoW | kg | 44,640 | market for naphtha, cut-off, U-RoW |
| steel, low-alloyed, hot rolled—GLO | kg | 6052 | market for steel, low-alloyed, hot rolled, cut-off, U-GLO |
| polyethylene terephthalate, granulate, amorphous—RER | kg | 3317 | polyethylene terephthalate production, granulate, amorphous, cut-off, U-RER |
| nylon 6-6—RER | kg | 3103 | nylon 6-6 production, cut-off, U-RER |
| steel, chromium steel 18/8—GLO | kg | 2253 | market for steel, chromium steel 18/8, cut-off, U-GLO |
| electric motor, electric passenger car—GLO | kg | 996 | electric motor production, vehicle (electric powertrain), cut-off, U-GLO |
| air compressor, screw-type compressor, 300 kW—RER | Item (s) | 6.27 | air compressor production, screw-type compressor, 300 kW, cut-off, U-RER |
| generator, 200 kW electrical—RER | Item (s) | 5.36 | generator production, 200 kW electrical, cut-off, U-RER |
| air compressor, screw-type compressor, 300 kW—RER | Item (s) | 4.3 | air compressor production, screw-type compressor, 300 kW, cut-off, U-RER |
| Intermodal container 40’ high-cube | Item (s) | 2 | Manufacturing, Intermodal container 40’ high cube |
| Outputs | | | |
| CA-S | Item (s) | 1 | |
Table A31.
Use, CA-S.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| CA-S | Item (s) | 1 | Manufacturing, CA-S |
| electricity, high voltage—DE | kWh | 0 | electricity production, wind, 1–3 MW turbine, onshore, cut-off, U-DE |
| Outputs | | | |
| electricity, high voltage—DE | MWh | 7826 | |
Table A32.
Manufacturing Storage, SL-B.
Table A32.
Manufacturing Storage, SL-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| battery cell, Li-ion—CN | kg | 25,000 | battery cell production, Li-ion, cut-off, U _w/o used battery—CN |
| Intermodal container 40’ high-cube | Item (s) | 2 | Manufacturing, Intermodal container 40’ high cube |
| transport, freight, sea, transoceanic ship—GLO | kg * km | 6.84 × 108 | market for transport, freight, sea, transoceanic ship, cut-off, U-GLO |
| Outputs | | | |
| Storage, SL-B | Item (s) | 1 | |
Table A33.
Use Storage, SL-B.
Table A33.
Use Storage, SL-B.
| | Unit | Amount | Process |
|---|
| Inputs | | | |
| Storage, SL-B | Item (s) | 1 | Manufacturing storage, SL-B |
| electricity, high voltage—DE | MWh | 0 | electricity production, wind, 1–3 MW turbine, onshore, cut-off, U-DE |
| Outputs | | | |
| electricity, high voltage—DE | MWh | 13,651 | |