Biomass Chars: The Effects of Pyrolysis Conditions on Their Morphology, Structure, Chemical Properties and Reactivity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Biomass
2.2. Experimental EFR Set Up
2.3. Pyrolysis Conditions
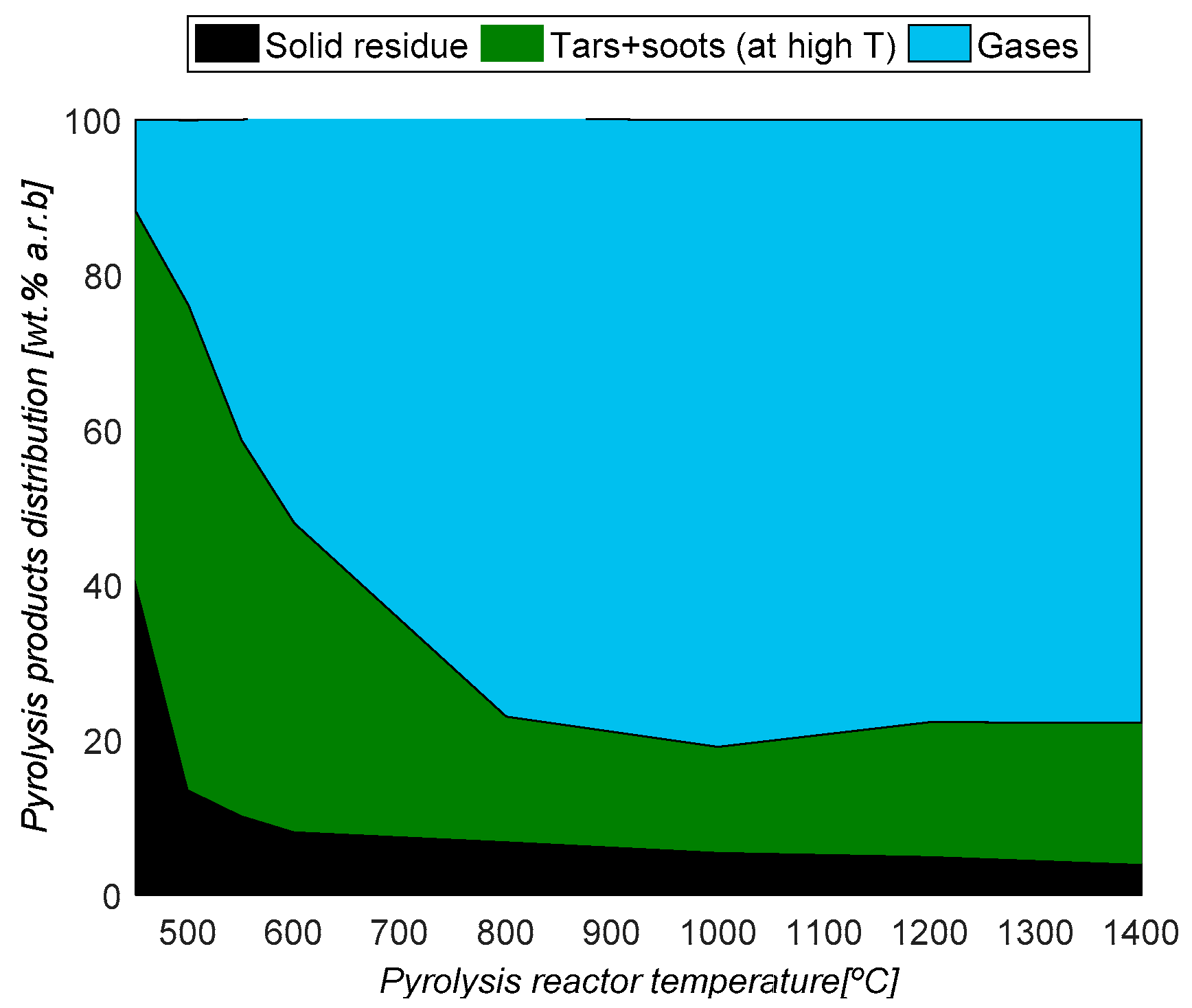
- The first is related to medium-temperature pyrolysis (500–600 °C), aiming at maximizing bio-oil production.
- The second concerns high-temperature pyrolysis aiming at maximizing the gas fraction [23].
2.4. Characterization Techniques of the Biomass Chars
2.4.1. Proximate and Ultimate Analysis
2.4.2. Morphology
2.4.3. Surface Chemistry
2.4.4. Structure
- ID: D band intensity height.
- Dpos: D band position.
- IG: G band intensity height.
- Gpos: G band position.
- IV: Valley region intensity height.
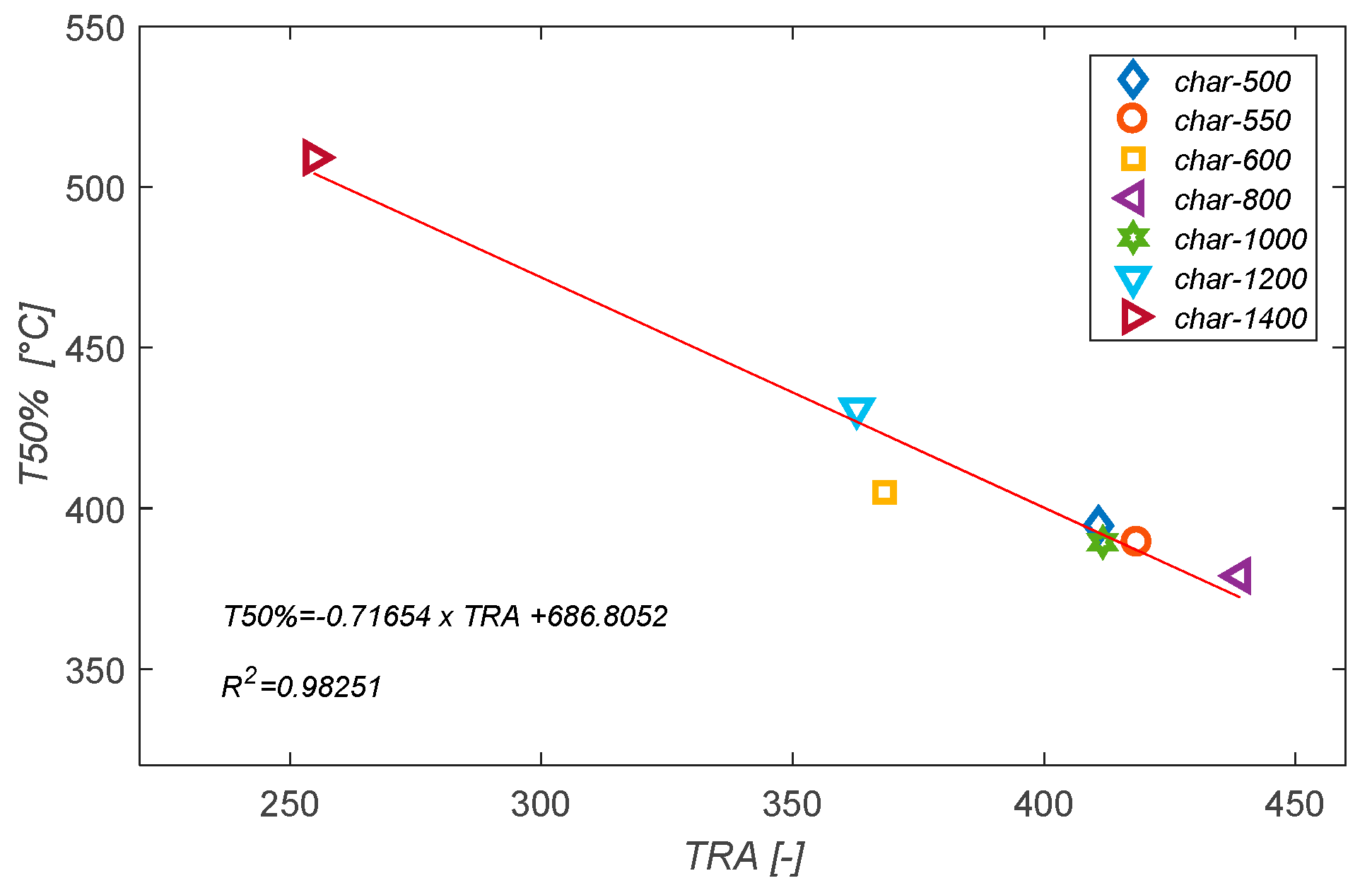
- The Total Raman Area (TRA):
2.4.5. Reactivity towards Oxygen
- is the conversion level.
- is the temperature at which .
- is the temperature at which .
- is the temperature at which .
- is the mean reaction rate is the maximum observed reaction rate.
- is the temperature at the maximum reaction rate.
- is the mean reactivity.
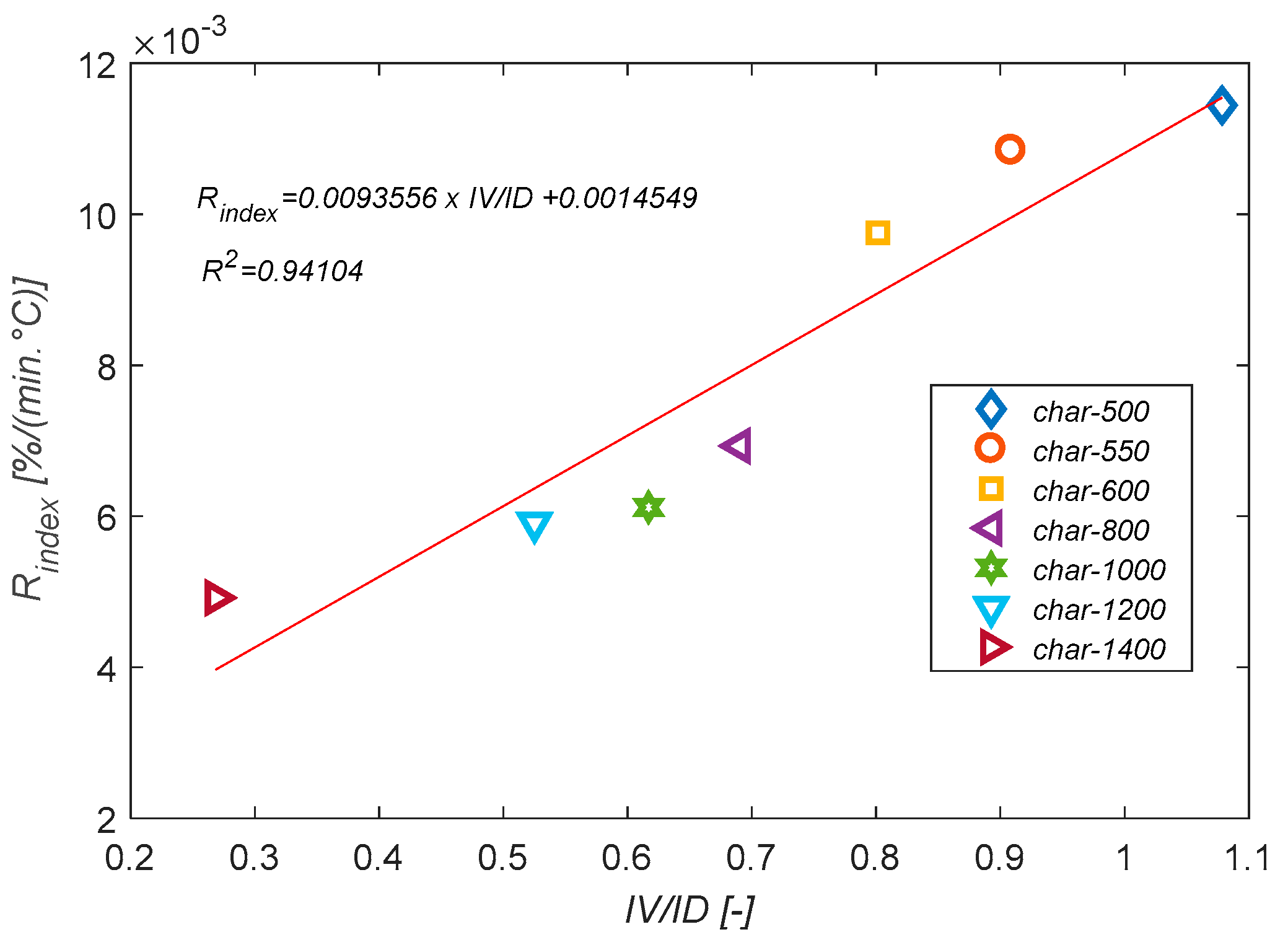
- is the reactivity index.
3. Results and Discussion
3.1. Pyrolysis Product Yields
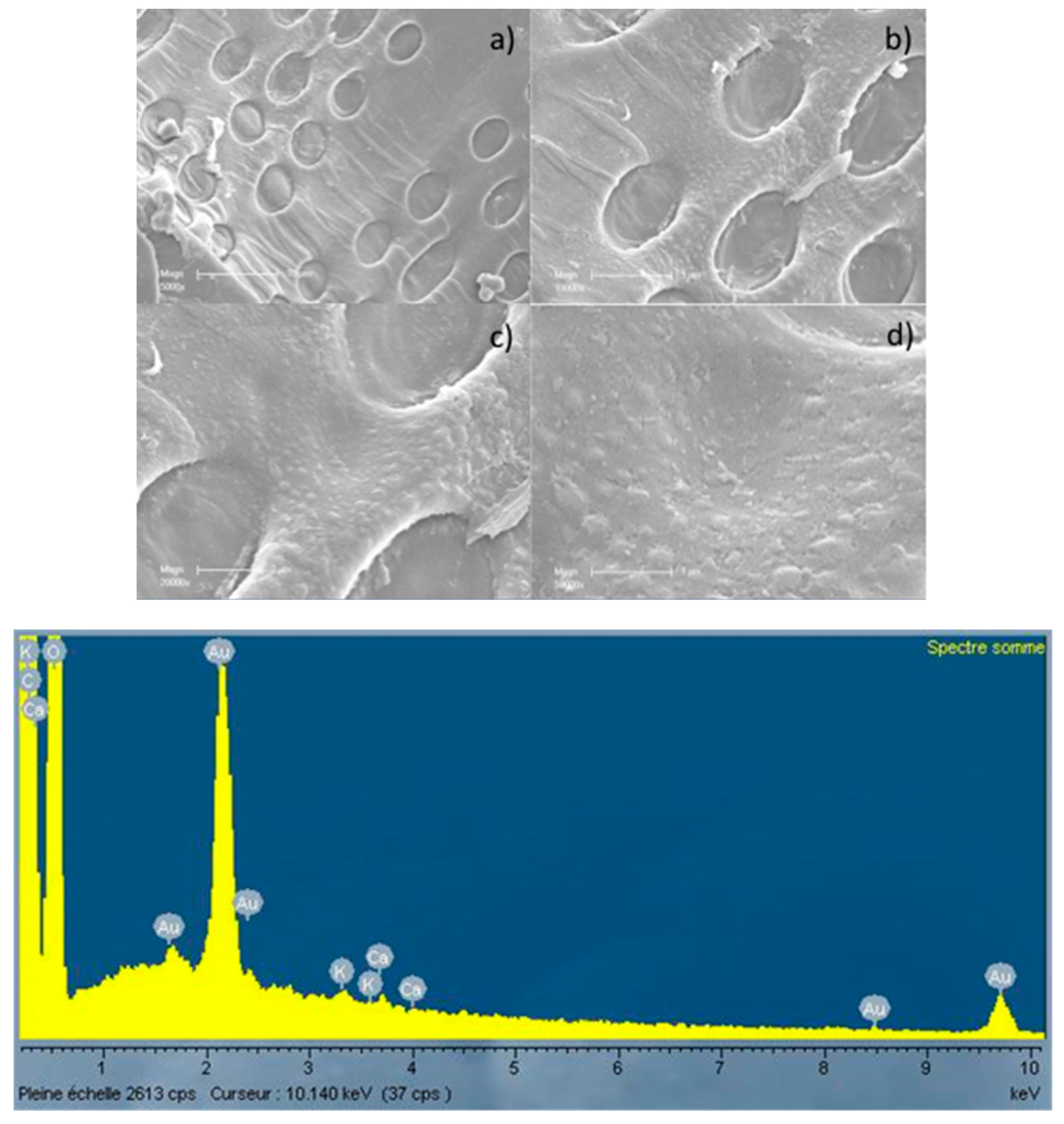
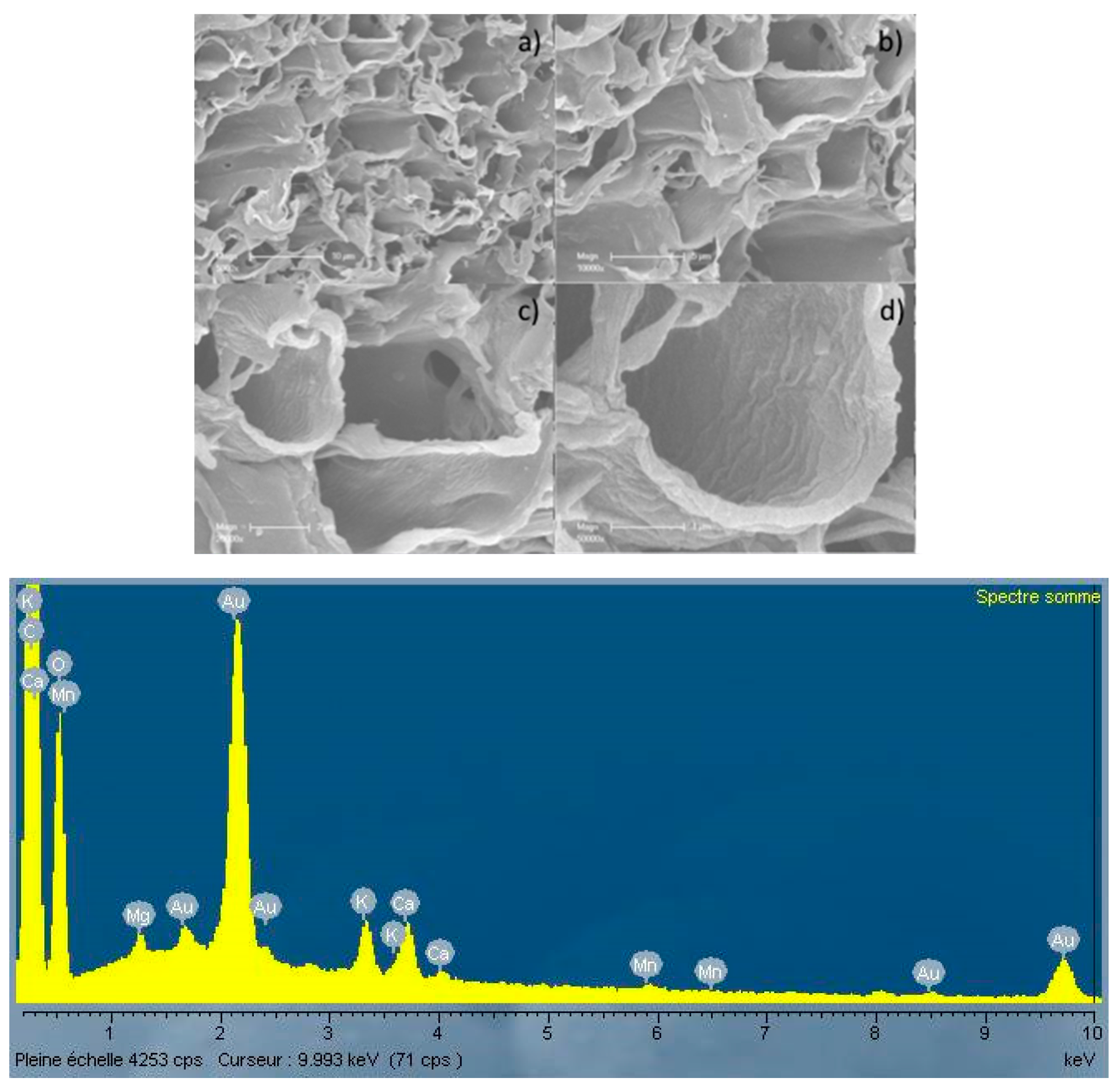
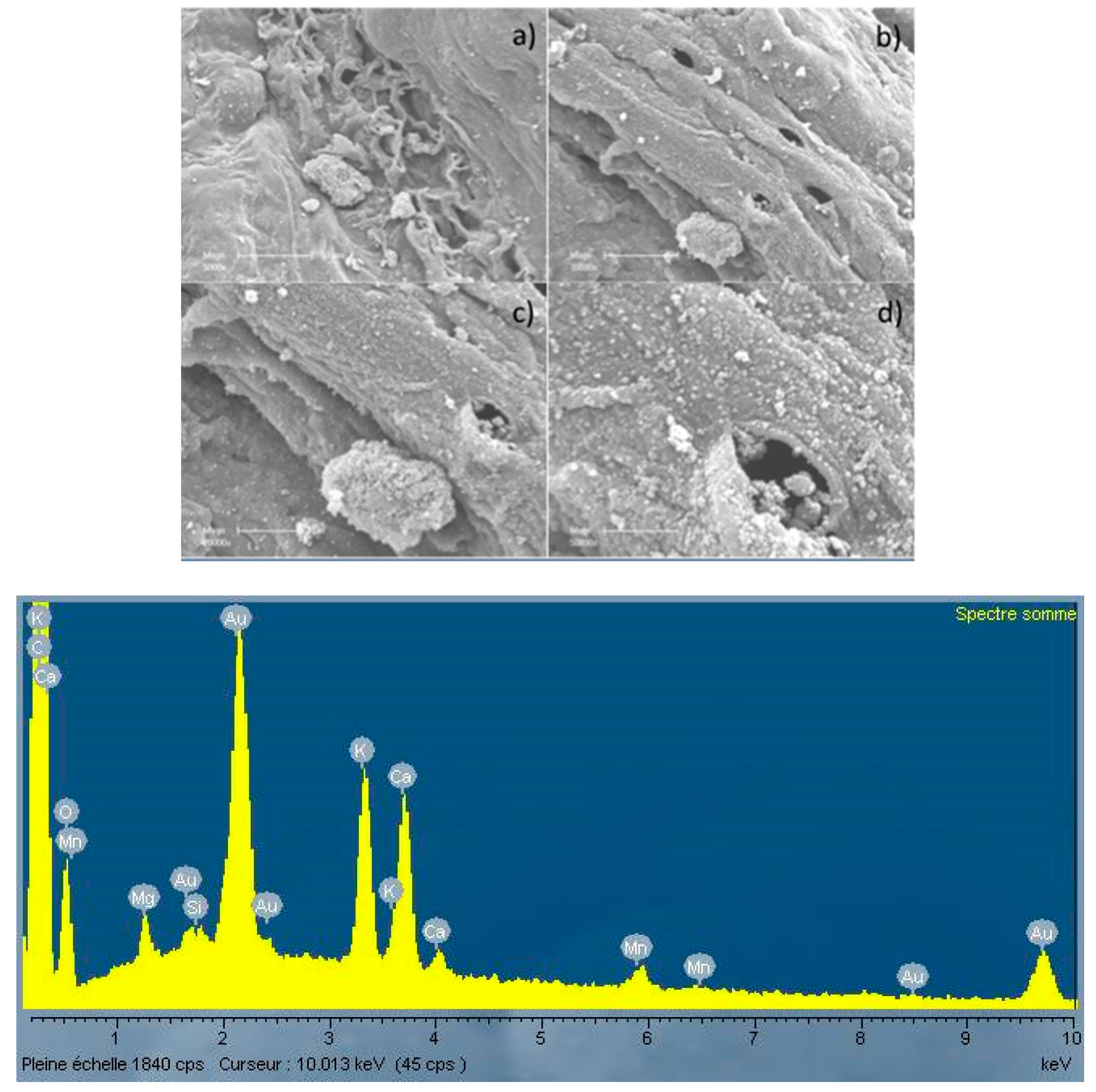
3.2. Microscopic Analysis of the Biochar Surfaces
3.3. Chemical Composition
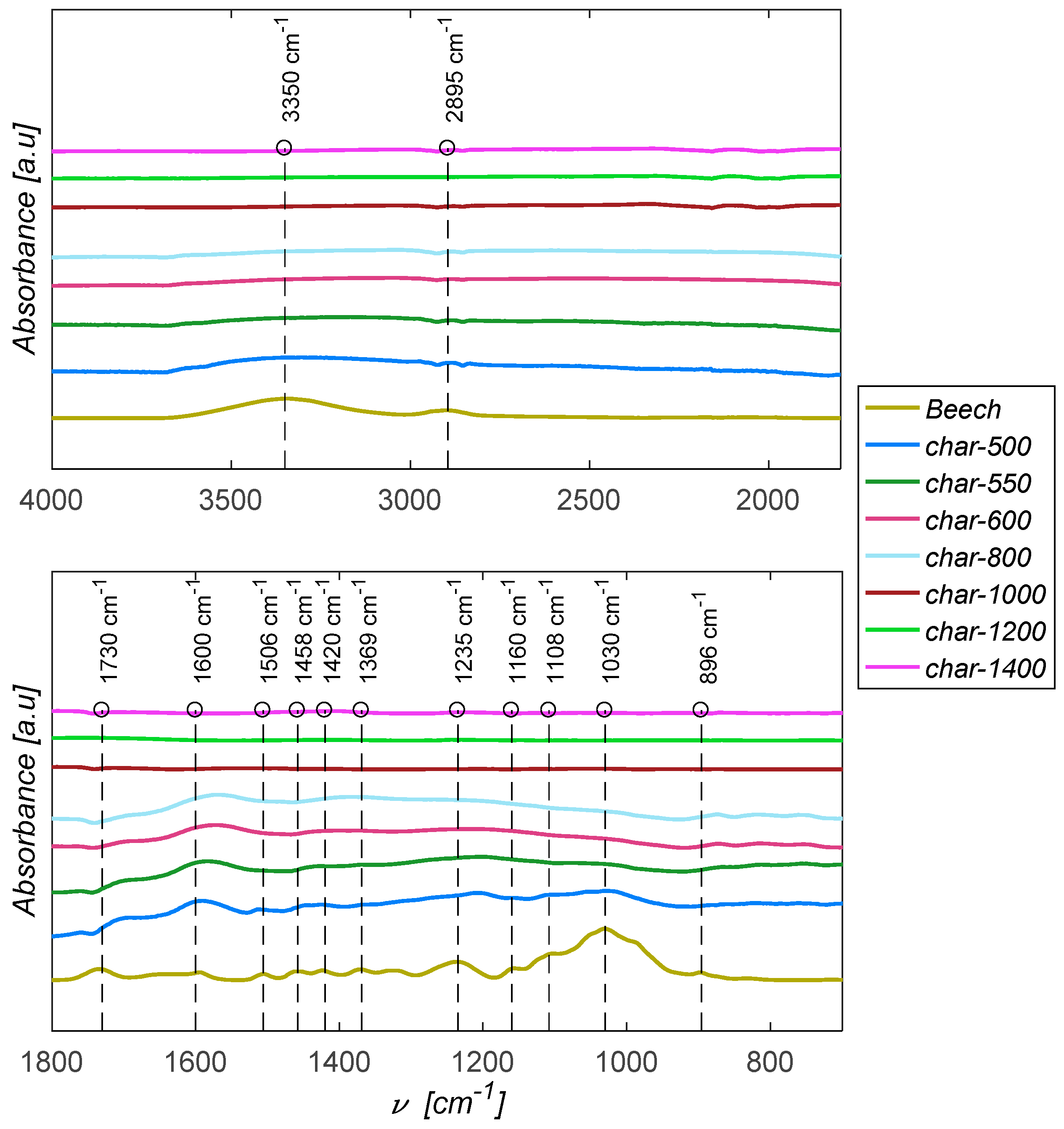
3.4. FTIR Spectroscopy
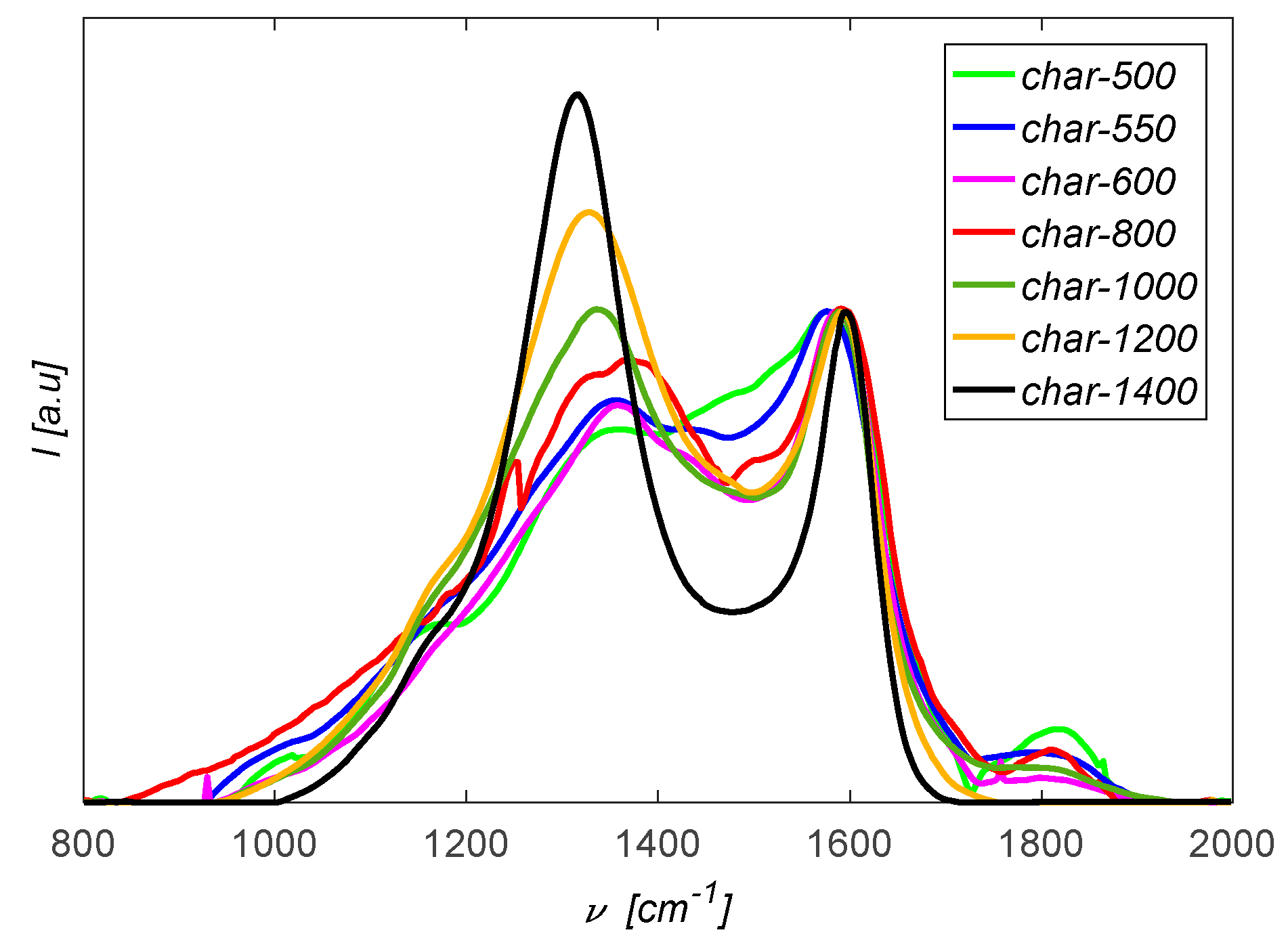
3.5. Structural Changes of the Biomass Particles as Revealed by Raman Spectroscopy
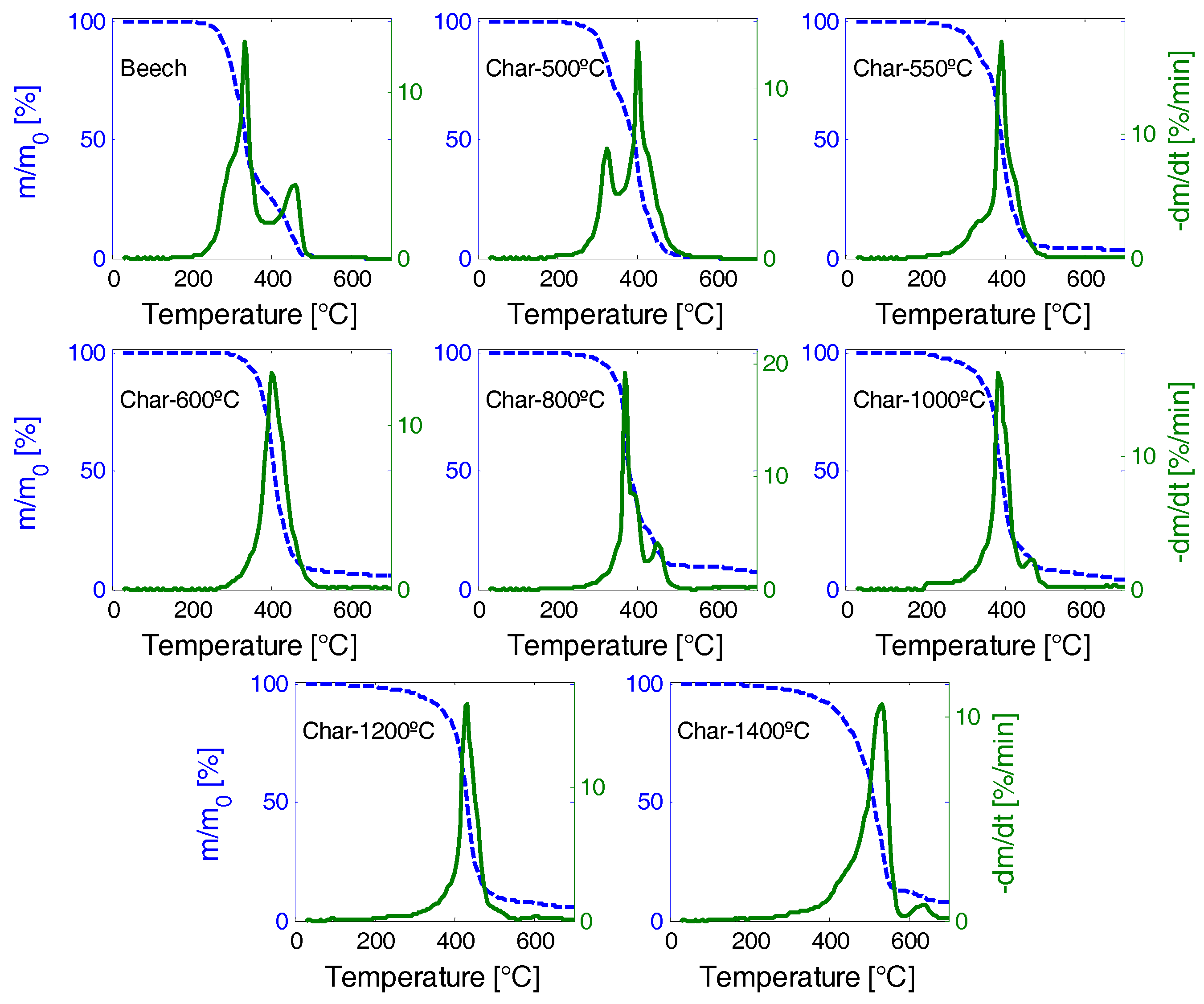
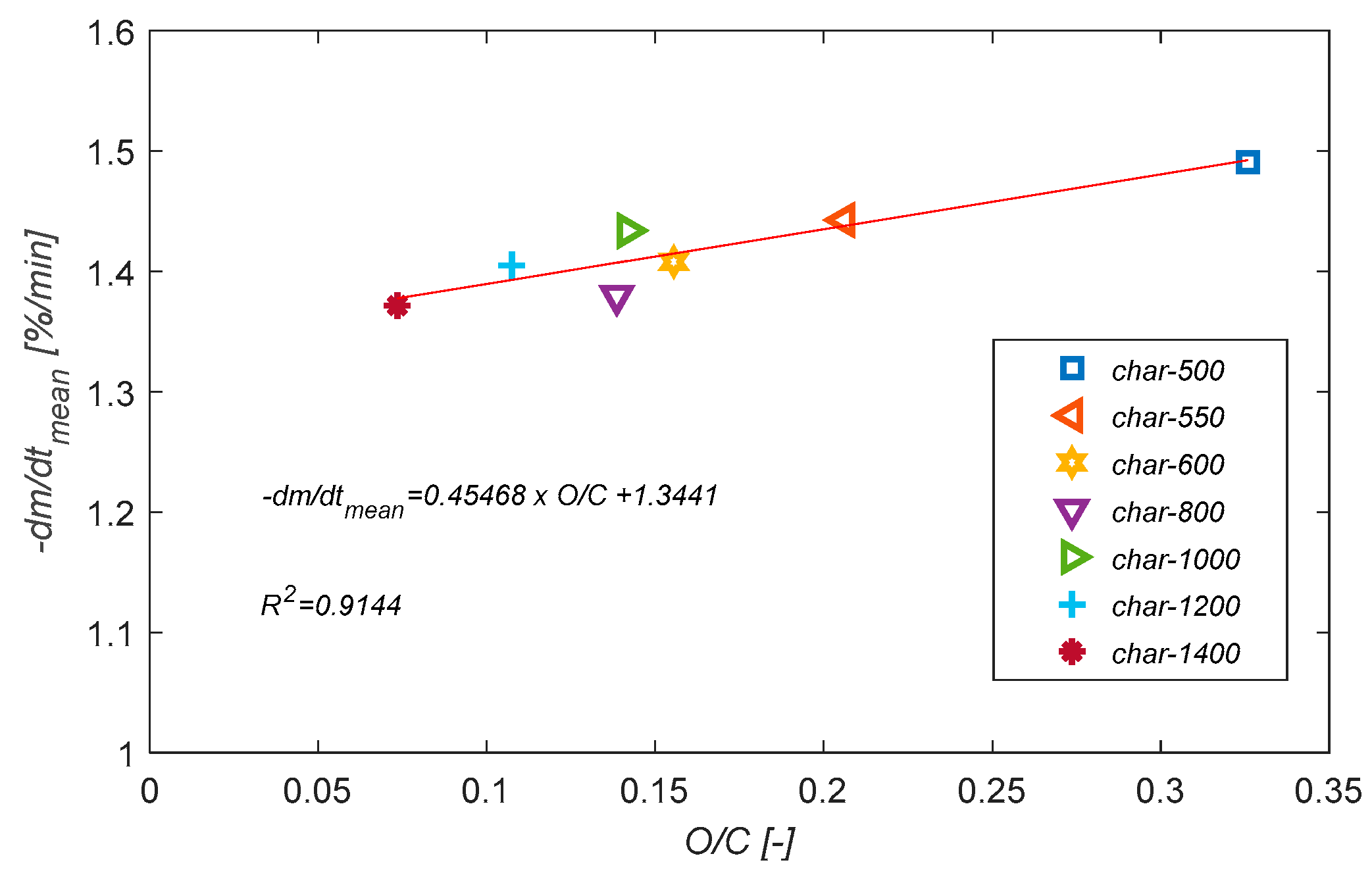
3.6. Char Reactivity towards O2
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Markovska, N.; Duić, N.; Vad Mathiesen, B.; Guzović, Z.; Piacentino, A.; Schlör, H.; Lund, H. Addressing the main challenges of energy security in the twenty-first century—Contributions of the conferences on Sustainable Development of Energy, Water and Environment Systems. Energy 2016, 115, 1504–1512. [Google Scholar] [CrossRef]
- Arodudu, O.; Helming, K.; Wiggering, H.; Voinov, A. Bioenergy from low-intensity agricultural systems: An energy efficiency analysis. Energies 2017, 10, 29. [Google Scholar] [CrossRef]
- Chiodo, V.; Zafarana, G.; Maisano, S.; Freni, S.; Urbani, F. Pyrolysis of different biomass: Direct comparison among Posidonia Oceanica, Lacustrine Alga and White-Pine. Fuel 2016, 164, 220–227. [Google Scholar] [CrossRef]
- No, S.-Y. Application of bio-oils from lignocellulosic biomass to transportation, heat and power generation—A review. Renew. Sustain. Energy Rev. 2014, 40, 1108–1125. [Google Scholar] [CrossRef]
- Guizani, C.; Escudero Sanz, F.J.; Salvador, S. Influence of temperature and particle size on the single and mixed atmosphere gasification of biomass char with H2O and CO2. Fuel Process. Technol. 2015, 134, 175–188. [Google Scholar] [CrossRef]
- Belhachemi, M.; Jeguirim, M.; Limousy, L.; Addoun, F. Comparison of NO2 removal using date pits activated carbon and modified commercialized activated carbon via different preparation methods: Effect of porosity and surface chemistry. Chem. Eng. J. 2014, 253, 121–129. [Google Scholar] [CrossRef]
- Zhu, L.; Shi, T.; Chen, Y. Preparation and characteristics of graphene oxide from the biomass carbon material using fir powder as precursor. Fuller. Nanotub. Carbon Nanostruct. 2015, 23, 961–967. [Google Scholar] [CrossRef]
- Lehmann, J.; Joseph, S. Biochar for Environmental Management: An Introduction; Routledge: Abingdon, UK, 2009; pp. 1–14. [Google Scholar]
- Brassard, P.; Godbout, S.; Raghavan, V.; Palacios, J.H.; Grenier, M.; Zegan, D. The production of engineered biochars in a vertical auger pyrolysis reactor for carbon sequestration. Energies 2017, 10, 288. [Google Scholar] [CrossRef]
- Keiluweit, M.; Nico, P.S.; Johnson, M.; Kleber, M. Dynamic molecular structure of plant biomass-derived black carbon (biochar). Environ. Sci. Technol. 2010, 44, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Rutherford, D.W.; Wershaw, R.L.; Cox, L.G. Changes in Composition and Porosity Occurring During the Thermal Degradation of Wood and Wood Components; Scientific Investigations Report 2004-5292; United States Geological Survey: Reston, WV, USA, 2004.
- Azargohar, R.; Nanda, S.; Kozinski, J.A.; Dalai, A.K.; Sutarto, R. Effects of temperature on the physicochemical characteristics of fast pyrolysis bio-chars derived from Canadian waste biomass. Fuel 2014, 125, 90–100. [Google Scholar] [CrossRef]
- Guerrero, M.; Ruiz, M.P.; Alzueta, M.U.; Bilbao, R.; Millera, A. Pyrolysis of eucalyptus at different heating rates: Studies of char characterization and oxidative reactivity. J. Anal. Appl. Pyrolysis 2005, 74, 307–314. [Google Scholar] [CrossRef]
- Rollinson, A.N. Gasifier reactor engineering approach to understanding the formation of biochar properties. Proc. A R. Soc. 2016, 472. [Google Scholar] [CrossRef] [PubMed]
- McDonald-Wharry, J.; Manley-Harris, M.; Pickering, K. Carbonisation of biomass-derived chars and the thermal reduction of a graphene oxide sample studied using Raman spectroscopy. Carbon 2013, 59, 383–405. [Google Scholar] [CrossRef]
- Keown, D.M.; Li, X.; Hayashi, J.I.; Li, C.Z. Characterization of the structural features of char from the pyrolysis of cane trash using Fourier transform-Raman spectroscopy. Energy Fuels 2007, 21, 1816–1821. [Google Scholar] [CrossRef]
- Guizani, C.; Haddad, K.; Limousy, L.; Jeguirim, M. Document New insights on the structural evolution of biomass char upon pyrolysis as revealed by the Raman spectroscopy and elemental analysis. Carbon 2017, 119, 519–521. [Google Scholar] [CrossRef]
- Trubetskaya, A.; Jensen, P.A.; Jensen, A.D.; Steibel, M.; Spliethoff, H.; Glarborg, P. Influence of fast pyrolysis conditions on yield and structural transformation of biomass chars. Fuel Process. Technol. 2015, 140, 205–214. [Google Scholar] [CrossRef]
- Brewer, C.E.; Schmidt-rohr, K.; Satrio, J.A.; Brown, R.C. Characterization of biochar from fast pyrolysis and gasification systems. Environ. Prog. Sustain. Energy 2009, 28, 386–396. [Google Scholar] [CrossRef]
- Morin, M.; Pécate, S.; Hémati, M.; Kara, Y. Pyrolysis of biomass in a batch fluidized bed reactor: Effect of the pyrolysis conditions and the nature of the biomass on the physicochemical properties and the reactivity of char. J. Anal. Appl. Pyrolysis 2016, 122, 511–523. [Google Scholar] [CrossRef]
- Zaida, A.; Bar-Ziv, E.; Radovic, L.R.; Lee, Y.-J. Further development of Raman Microprobe spectroscopy for characterization of char reactivity. Proc. Combust. Inst. 2007, 31, 1881–1887. [Google Scholar] [CrossRef]
- Sheng, C. Char structure characterised by Raman spectroscopy and its correlations with combustion reactivity. Fuel 2007, 86, 2316–2324. [Google Scholar] [CrossRef]
- Billaud, J.; Valin, S.; Peyrot, M.; Salvador, S. Influence of H2O, CO2 and O2 addition on biomass gasification in entrained flow reactor conditions: Experiments and modelling. Fuel 2016, 166, 166–178. [Google Scholar] [CrossRef]
- Chen, L. Fast Pyrolysis of Millimetric Wood Particles between 800 °C and 1000 °C. Ph.D. Thesis, Claude Bernard Lyon I, Villeurbanne, France, December 2009. [Google Scholar]
- Billaud, J.; Valin, S.; Ratel, G.; Thiery, S.; Salvador, S. Biomass gasification between 800 and 1400 °C in the presence of O2: Drop tube reactor experiments and simulation. Chem. Eng. Trans. 2014, 37, 163–168. [Google Scholar] [CrossRef]
- Paethanom, A.; Yoshikawa, K. Influence of pyrolysis temperature on rice husk char characteristics and its tar adsorption capability. Energies 2012, 5, 4941–4951. [Google Scholar] [CrossRef]
- Crombie, K.; Mašek, O.; Sohi, S.P.; Brownsort, P.; Cross, A. The effect of pyrolysis conditions on biochar stability as determined by three methods. GCB Bioenergy 2013, 5, 122–131. [Google Scholar] [CrossRef]
- Guizani, C.; Valin, S.; Billaud, J.; Peyrot, M.; Salvador, S. Biomass fast pyrolysis in a drop tube reactor for bio oil production: Experiments and modeling. Fuel 2017, in press. [Google Scholar]
- Smith, M.W.; Dallmeyer, I.; Johnson, T.J.; Brauer, C.S.; McEwen, J.S.; Espinal, J.F.; Garcia-Perez, M. Structural analysis of char by Raman spectroscopy: Improving band assignments through computational calculations from first principles. Carbon 2016, 100, 678–692. [Google Scholar] [CrossRef]
- Asadullah, M.; Zhang, S.; Li, C.Z. Evaluation of structural features of chars from pyrolysis of biomass of different particle sizes. Fuel Process. Technol. 2010, 91, 877–881. [Google Scholar] [CrossRef]
- Elmay, Y.; Le Brech, Y.; Delmotte, L.; Dufour, A.; Brosse, N.; Gadiou, R. Characterization of miscanthus pyrolysis by DRIFTs, UV Raman spectroscopy and mass spectrometry. J. Anal. Appl. Pyrolysis 2015, 113, 402–411. [Google Scholar] [CrossRef]
- Asadullah, M.; Zhang, S.; Min, Z.; Yimsiri, P.; Li, C.Z. Effects of biomass char structure on its gasification reactivity. Bioresour. Technol. 2010, 101, 7935–7943. [Google Scholar] [CrossRef] [PubMed]



| Proximate Analysis | Value |
|---|---|
| Moisture [wt % ar *] | 8.7 |
| Volatile Matter [wt % db **] | 84.3 |
| Fixed carbon [wt % db] | 15.2 |
| Ash (815 °C) [wt % db] | 0.5 |
| Ultimate analysis [wt % db] | |
| C | 49.1 |
| H | 5.7 |
| N | 0.15 |
| S | 0.045 |
| O (by difference) | 44.5 |
| Reactor Temperature [°C] | [g/min] | Gas Residence Time [s] | Particle Residence Time [s] |
|---|---|---|---|
| 500 | 1 ± 0.1 | 16.6 ± 0.2 | 2.26 |
| 550 | 1 ± 0.1 | 16.6 ± 0.2 | 4.03 |
| 600 | 1 ± 0.1 | 16.6 ± 0.2 | 5.12 |
| 800 | 1 ± 0.1 | 4.3 ± 0.1 | 2.33 |
| 1000 | 1 ± 0.1 | 4.3 ± 0.1 | 2.40 |
| 1200 | 1 ± 0.1 | 4.3 ± 0.1 | 2.45 |
| 1400 | 1 ± 0.1 | 4.3 ± 0.1 | 2.52 |
| Sample | C [mol % afb] | H [mol % afb] | O [mol % afb] |
|---|---|---|---|
| char-500 | 47.1 ± 2.3 | 37.5 ± 0.8 | 15.4 ± 1.5 |
| char-550 | 56.1 ± 1.3 | 32.4 ± 0.8 | 11.6 ± 0.5 |
| char-600 | 63.4 ± 2.7 | 26.8 ± 1.1 | 9.9 ± 1.5 |
| char-800 | 64.2 ± 1.5 | 26.9 ± 0.9 | 8.9 ± 0.6 |
| char-1000 | 74.4 ± 3.1 | 15.0 ± 0.2 | 10.6 ± 2.8 |
| char-1200 | 77.6 ± 2.6 | 14.0 ± 0.3 | 8.3 ± 2.2 |
| char-1400 | 84.0 ± 2.7 | 9.9 ± 0.3 | 6.2 ± 2.4 |
| Sample | Dposition [cm−1] | Gposition [cm−1] | ||||
|---|---|---|---|---|---|---|
| char-500 | 410.87 ± 10.23 | 1363 ± 4 | 1576 ± 7 | 0.76 ± 0.05 | 0.82 ± 0.05 | 1.08 ± 0.04 |
| char-550 | 418.53 ± 8.01 | 1351 ± 5 | 1576 ± 5 | 0.82 ± 0.03 | 0.74 ± 0.05 | 0.91 ± 0.05 |
| char-600 | 368.25 ± 12.67 | 1357 ± 5 | 1588 ± 6 | 0.81 ± 0.07 | 0.65 ± 0.05 | 0.80 ± 0.03 |
| char-800 | 438.93 ± 15.86 | 1366 ± 6 | 1599 ± 8 | 0.90 ± 0.08 | 0.62 ± 0.05 | 0.69 ± 0.07 |
| char-1000 | 411.66 ± 14.34 | 1336 ± 5 | 1588 ± 6 | 1.01 ± 0.05 | 0.62 ± 0.05 | 0.62 ± 0.05 |
| char-1200 | 362.75 ± 16.22 | 1326 ± 6 | 1590 ± 8 | 1.20 ± 0.06 | 0.63 ± 0.05 | 0.53 ± 0.05 |
| char-1400 | 254.78 ± 12.35 | 1327 ± 7 | 1596 ± 4 | 1.44 ± 0.04 | 0.39 ± 0.05 | 0.27 ± 0.03 |
| Sample | [°C] | [°C] | [°C] | [°C] | [%/min] | [%/min] | [min−1] | [min−1] |
|---|---|---|---|---|---|---|---|---|
| Beech | 254.0 | 337.3 | 472.7 | 332.1 | 12.86 | 1.488 | 0.196 | 0.0216 |
| char-500 | 269.6 | 394.6 | 488.3 | 399.8 | 17.53 | 1.488 | 0.198 | 0.0191 |
| char-550 | 264.4 | 389.4 | 483.1 | 389.4 | 13.31 | 1.440 | 0.142 | 0.0265 |
| char-600 | 321.7 | 405.0 | 545.6 | 399.8 | 19.20 | 1.405 | 0.119 | 0.0198 |
| char-800 | 285.2 | 379.0 | 613.3 | 368.5 | 16.27 | 1.378 | 0.074 | 0.0299 |
| char-1000 | 243.5 | 389.4 | 623.8 | 384.2 | 16.35 | 1.430 | 0.077 | 0.0247 |
| char-1200 | 238.3 | 431.0 | 597.7 | 431.0 | 10.62 | 1.402 | 0.074 | 0.0232 |
| char-1400 | 280.0 | 509.2 | 639.4 | 530.0 | 12.86 | 1.369 | 0.067 | 0.0132 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guizani, C.; Jeguirim, M.; Valin, S.; Limousy, L.; Salvador, S. Biomass Chars: The Effects of Pyrolysis Conditions on Their Morphology, Structure, Chemical Properties and Reactivity. Energies 2017, 10, 796. https://doi.org/10.3390/en10060796
Guizani C, Jeguirim M, Valin S, Limousy L, Salvador S. Biomass Chars: The Effects of Pyrolysis Conditions on Their Morphology, Structure, Chemical Properties and Reactivity. Energies. 2017; 10(6):796. https://doi.org/10.3390/en10060796
Chicago/Turabian StyleGuizani, Chamseddine, Mejdi Jeguirim, Sylvie Valin, Lionel Limousy, and Sylvain Salvador. 2017. "Biomass Chars: The Effects of Pyrolysis Conditions on Their Morphology, Structure, Chemical Properties and Reactivity" Energies 10, no. 6: 796. https://doi.org/10.3390/en10060796
APA StyleGuizani, C., Jeguirim, M., Valin, S., Limousy, L., & Salvador, S. (2017). Biomass Chars: The Effects of Pyrolysis Conditions on Their Morphology, Structure, Chemical Properties and Reactivity. Energies, 10(6), 796. https://doi.org/10.3390/en10060796








