Experimental Investigations of Physical and Chemical Properties for Microalgae HTL Bio-Crude Using a Large Batch Reactor
Abstract
:1. Introduction
- Pumping the biomass slurry into a continuous reactor at high pressure and temperature remains a technological challenge at the industrial scale;
- Ability to easily switch between feedstocks; and
- For a factory, individual reactors can be taken out-of-service for maintenance.
2. Materials and Methods
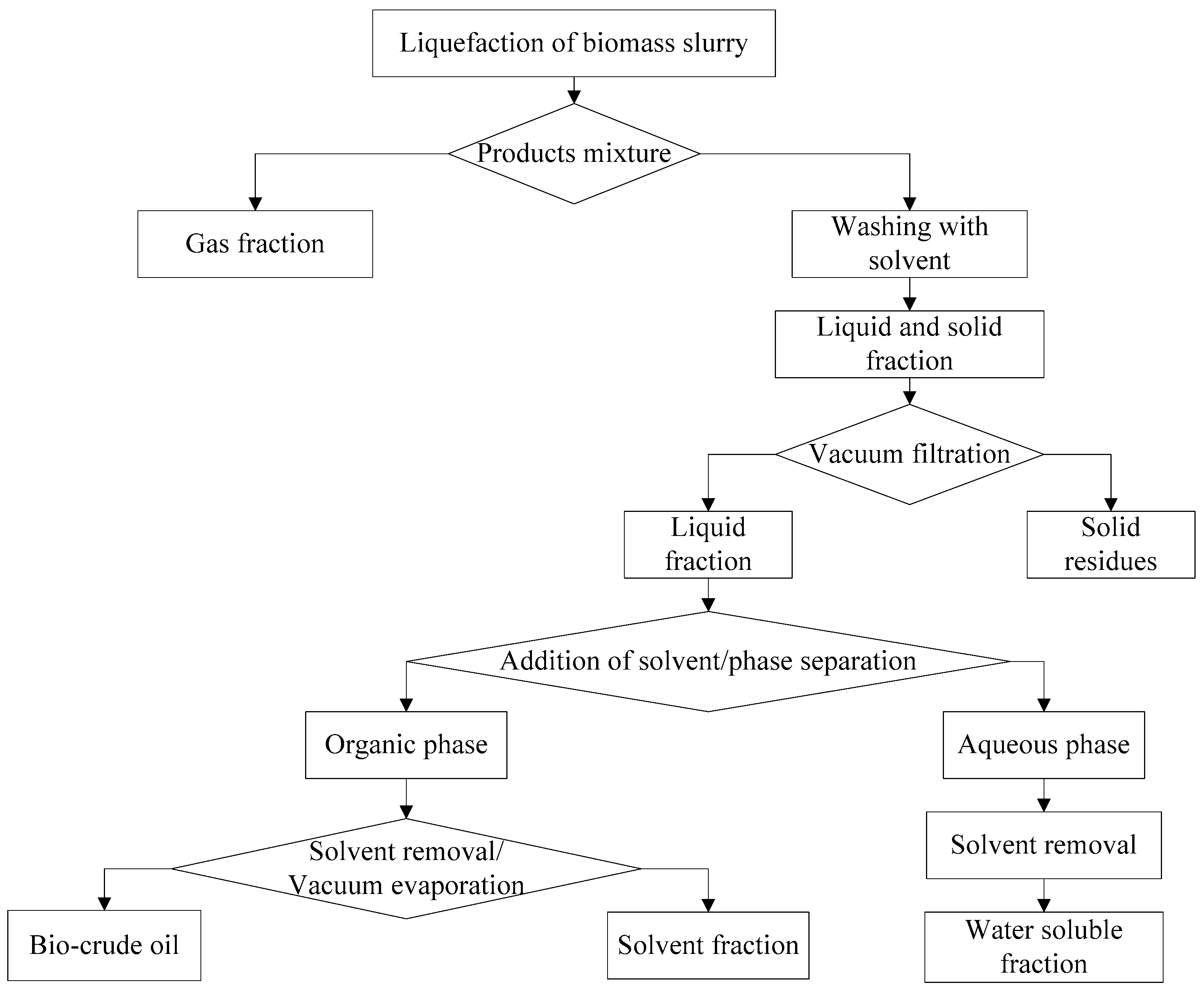
2.1. Hydrothermal Liquefaction (HTL)
2.2. Raw Materials
2.3. Analytical Methods
2.4. Bio-Crude Properties Measurements
3. Results and Discussion
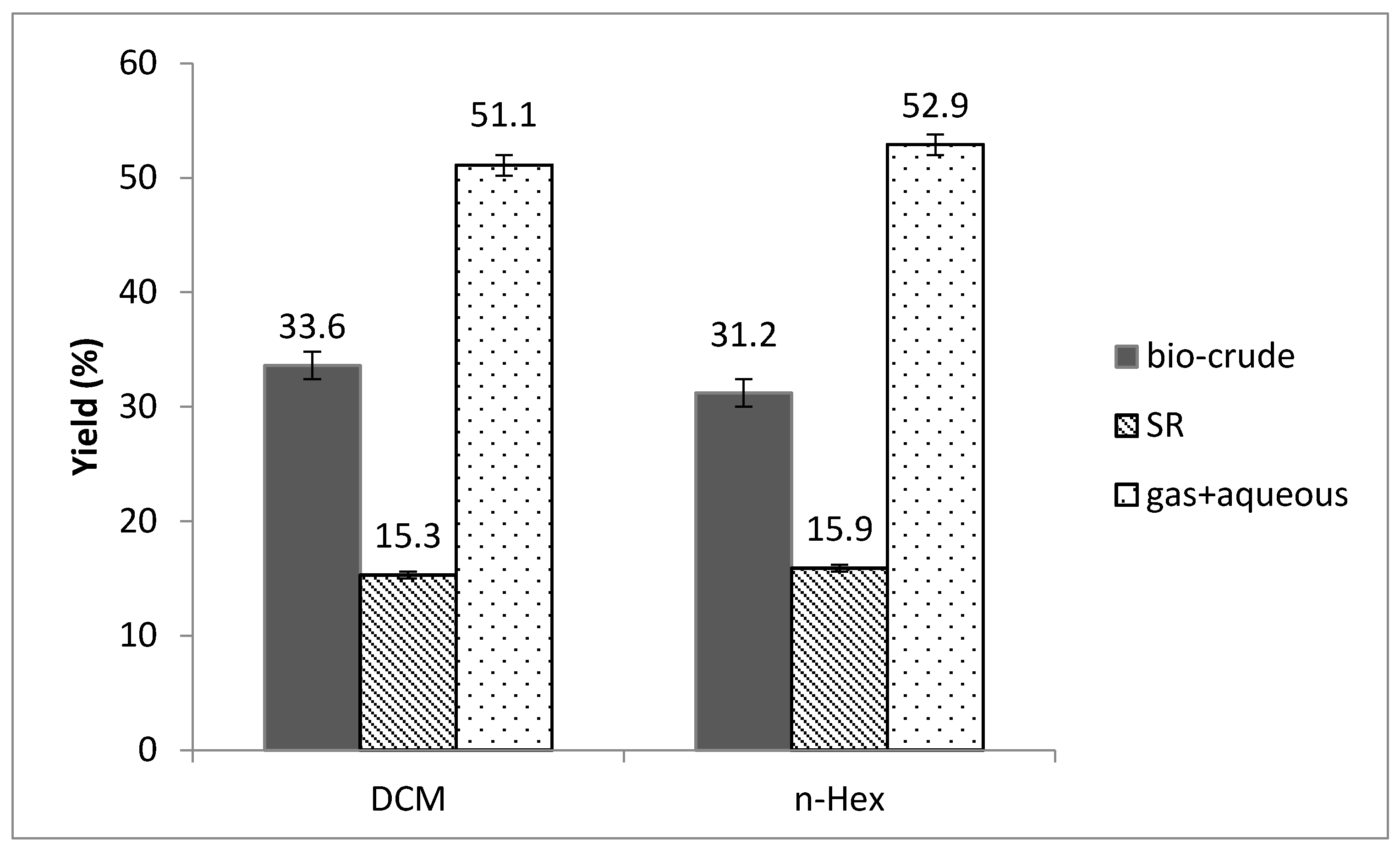
3.1. Influence of Solvents in Product Separation
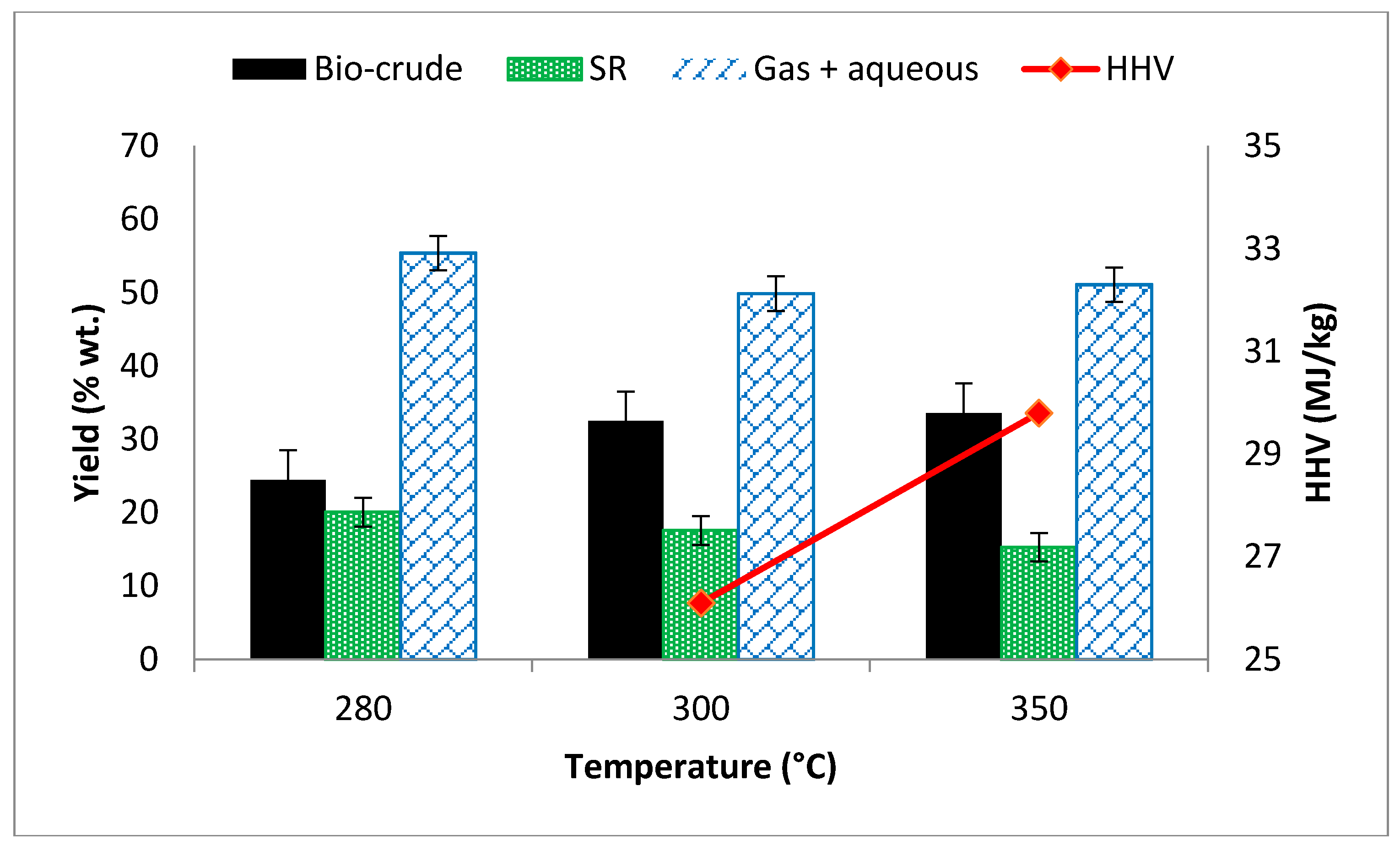
3.2. Effect of Reaction Temperature on Yield and HHV
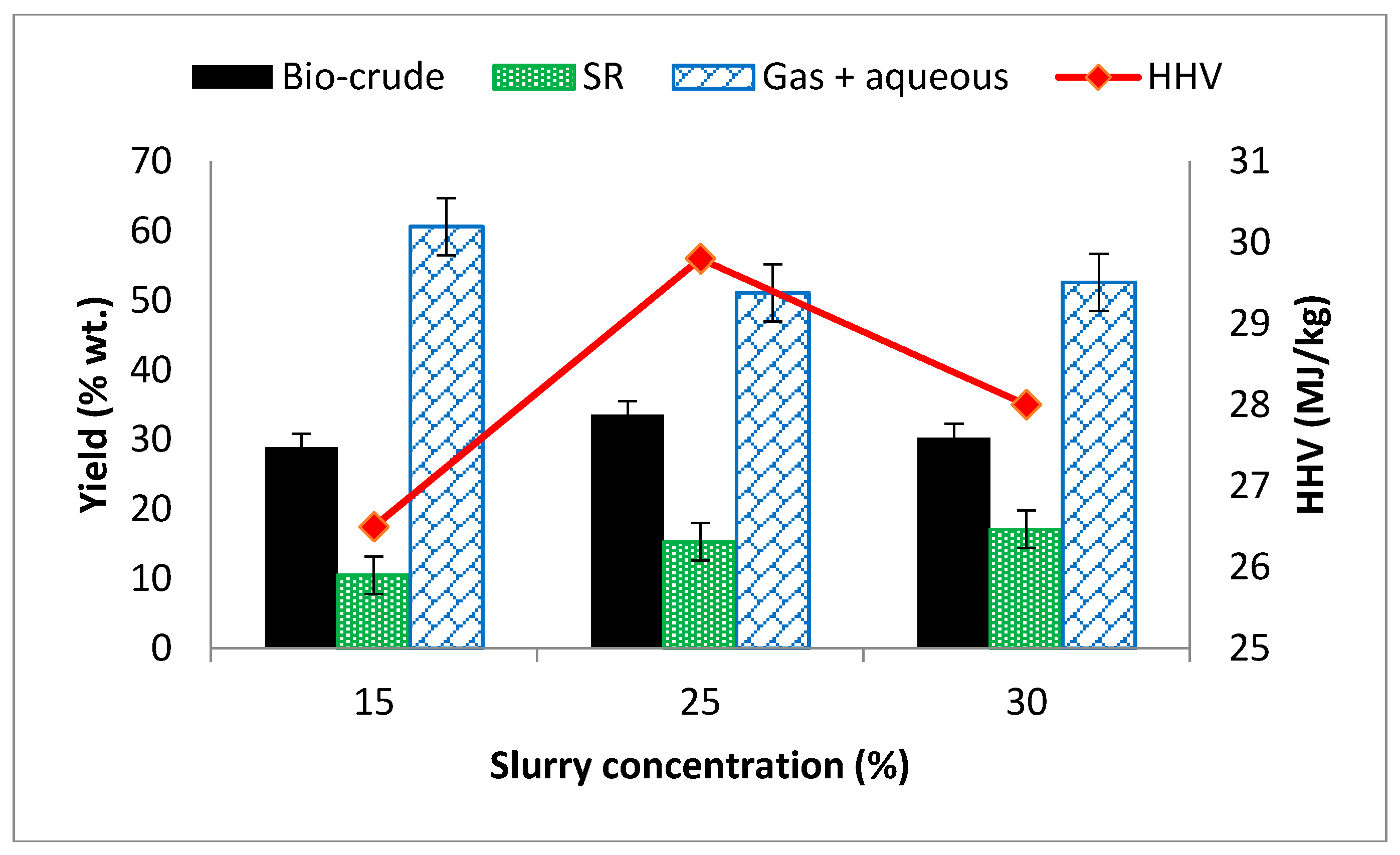
3.3. Effects of Solid Concentration on Yield and HHV
3.4. Chemical Characterisation of Bio-Crude Oil
3.5. Effect of Temperature and Concentration on Chemical and Physical Properties
3.5.1. Bio-Crude Chemical Composition
3.5.2. Viscosity
3.5.3. Density
3.5.4. Higher Heating Value
3.6. Comparison with Previous Studies
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Islam, M.; Magnusson, M.; Brown, R.; Ayoko, G.; Nabi, M.; Heimann, K. Microalgal species selection for biodiesel production based on fuel properties derived from fatty acid profiles. Energies 2013, 6, 5676–5702. [Google Scholar] [CrossRef]
- Eboibi, B.E.-O.; Lewis, D.M.; Ashman, P.J.; Chinnasamy, S. Hydrothermal liquefaction of microalgae for biocrude production: Improving the biocrude properties with vacuum distillation. Bioresour. Technol. 2014, 174, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Azad, A.K.; Rasul, M.G.; Khan, M.M.K.; Sharma, S.C.; Hazrat, M.A. Prospect of biofuels as an alternative transport fuel in Australia. Renew. Sustain. Energy Rev. 2015, 43, 331–351. [Google Scholar] [CrossRef]
- Oncel, S.S. Microalgae for a macroenergy world. Renew. Sustain. Energy Rev. 2013, 26, 241–264. [Google Scholar] [CrossRef]
- D’Ippolito, G.; Sardo, A.; Paris, D.; Vella, F.M.; Adelfi, M.G.; Botte, P.; Gallo, C.; Fontana, A. Potential of lipid metabolism in marine diatoms for biofuel production. Biotechnol. Biofuels 2015, 8, 28. [Google Scholar] [CrossRef] [PubMed]
- Kosinkova, J.; Doshi, A.; Maire, J.; Ristovski, Z.; Brown, R.; Rainey, T.J. Measuring the regional availability of biomass for biofuels and the potential for microalgae. Renew. Sustain. Energy Rev. 2015, 49, 1271–1285. [Google Scholar] [CrossRef]
- Subramanian, S.; Barry, A.N.; Pieris, S.; Sayre, R.T. Comparative energetics and kinetics of autotrophic lipid and starch metabolism in chlorophytic microalgae: Implications for biomass and biofuel production. Biotechnol. Biofuels 2013, 6. [Google Scholar] [CrossRef] [PubMed]
- Guccione, A.; Biondi, N.; Sampietro, G.; Rodolfi, L.; Bassi, N.; Tredici, M.R. Chlorella for protein and biofuels: From strain selection to outdoor cultivation in a Green Wall Panel photobioreactor. Biotechnol. Biofuels 2014, 7, 84. [Google Scholar] [CrossRef] [PubMed]
- Naveena, B.; Armshaw, P.; Tony Pembroke, J. Ultrasonic intensification as a tool for enhanced microbial biofuel yields. Biotechnol. Biofuels 2015, 8. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.-P.; Duan, P.-G.; Wang, F. Hydrothermal processing of macroalgae for producing crude bio-oil. Fuel Process. Technol. 2015, 130, 268–274. [Google Scholar] [CrossRef]
- Bennion, E.P.; Ginosar, D.M.; Moses, J.; Agblevor, F.; Quinn, J.C. Lifecycle assessment of microalgae to biofuel: Comparison of thermochemical processing pathways. Appl. Energy 2015, 154, 1062–1071. [Google Scholar] [CrossRef]
- López Barreiro, D.; Prins, W.; Ronsse, F.; Brilman, W. Hydrothermal liquefaction (HTL) of microalgae for biofuel production: State of the art review and future prospects. Biomass Bioenergy 2013, 53, 113–127. [Google Scholar] [CrossRef]
- Peng, X.; Ma, X.; Lin, Y.; Wang, X.; Zhang, X.; Yang, C. Effect of process parameters on solvolysis liquefaction of Chlorella pyrenoidosa in ethanol–water system and energy evaluation. Energy Convers. Manag. 2016, 117, 43–63. [Google Scholar] [CrossRef]
- Amin, S. Review on biofuel oil and gas production processes from microalgae. Energy Convers. Manag. 2009, 50, 1834–1840. [Google Scholar] [CrossRef]
- Srirangan, K.; Akawi, L.; Moo-Young, M.; Chou, C.P. Towards sustainable production of clean energy carriers from biomass resources. Appl. Energy 2012, 100, 172–186. [Google Scholar] [CrossRef]
- Zhang, L.; Xu, C.; Champagne, P. Overview of recent advances in thermo-chemical conversion of biomass. Energy Convers. Manag. 2010, 51, 969–982. [Google Scholar] [CrossRef]
- Dote, Y.; Sawayama, S.; Inoue, S.; Minowa, T.; Yokoyama, S.-Y. Recovery of liquid fuel from hydrocarbon-rich microalgae by thermochemical liquefaction. Fuel 1994, 73, 1855–1857. [Google Scholar] [CrossRef]
- Vardon, D.R.; Sharma, B.K.; Blazina, G.V.; Rajagopalan, K.; Strathmann, T.J. Thermochemical conversion of raw and defatted algal biomass via hydrothermal liquefaction and slow pyrolysis. Bioresour. Technol. 2012, 109, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Mallick, N. Microalga Scenedesmus obliquus as a potential source for biodiesel production. Appl. Microbiol. Biotechnol. 2009, 84, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M.; Pourkhesalian, A.M.; Jahirul, M.I.; Stevanovic, S.; Pham, P.X.; Wang, H.; Masri, A.R.; Brown, R.J.; Ristovski, Z.D. Particle emissions from biodiesels with different physical properties and chemical composition. Fuel 2014, 134, 201–208. [Google Scholar] [CrossRef]
- Demirbas, A. Progress and recent trends in biodiesel fuels. Energy Convers. Manag. 2009, 50, 14–34. [Google Scholar] [CrossRef]
- Hossain, F.M.; Rainey, T.J.; Ristovski, Z.; Brown, R.J. Performance and exhaust emissions of diesel engines using microalgae FAME and the prospects for microalgae HTL biocrude. Renew. Sustain. Energy Rev. 2017. submitted. [Google Scholar]
- Jena, U.; Das, K.C.; Kastner, J.R. Effect of operating conditions of thermochemical liquefaction on biocrude production from Spirulina platensis. Bioresour. Technol. 2011, 102, 6221–6229. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Savage, P.E. Characterization of biocrudes recovered with and without solvent after hydrothermal liquefaction of algae. Algal Res. 2014, 6, 1–7. [Google Scholar] [CrossRef]
- Eboibi, B.E.; Lewis, D.M.; Ashman, P.J.; Chinnasamy, S. Effect of operating conditions on yield and quality of biocrude during hydrothermal liquefaction of halophytic microalga Tetraselmis sp. Bioresour. Technol. 2014, 170, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Tang, X.; Asa-Awuku, A.; Jung, H.S. Characterization of a method for aerosol generation from heavy fuel oil (HFO) as an alternative to emissions from ship diesel engines. J. Aerosol Sci. 2010, 41, 1143–1151. [Google Scholar] [CrossRef]
- Anastasakis, K.; Ross, A.B. Hydrothermal liquefaction of the brown macro-alga Laminaria Saccharina: Effect of reaction conditions on product distribution and composition. Bioresour. Technol. 2011, 102, 4876–4883. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, J.A.; Brown, R.J.; Rainey, T.J. A review of hydrothermal liquefaction bio-crude properties and prospects for upgrading to transportation fuels. Energies 2015, 8, 6765–6794. [Google Scholar] [CrossRef]
- Smook, G. Handbook for Pulp and Paper Technologists; TAPPI Press: Quebec, QC, Canada, 1982; Chapter 8; p. 76. [Google Scholar]
- Biller, P.; Riley, R.; Ross, A. Catalytic hydrothermal processing of microalgae: Decomposition and upgrading of lipids. Bioresour. Technol. 2011, 102, 4841–4848. [Google Scholar] [CrossRef] [PubMed]
- Biller, P.; Ross, A. Potential yields and properties of oil from the hydrothermal liquefaction of microalgae with different biochemical content. Bioresour. Technol. 2011, 102, 215–225. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.M.; Duan, P.; Savage, P.E. Hydrothermal liquefaction and gasification of Nannochloropsis sp. Energy Fuels 2010, 24, 3639–3646. [Google Scholar] [CrossRef]
- Matsui, T.-O.; Nishihara, A.; Ueda, C.; Ohtsuki, M.; Ikenaga, N.-O.; Suzuki, T. Liquefaction of micro-algae with iron catalyst. Fuel 1997, 76, 1043–1048. [Google Scholar] [CrossRef]
- Valdez, P.J.; Dickinson, J.G.; Savage, P.E. Characterization of product fractions from hydrothermal liquefaction of Nannochloropsis sp. and the influence of solvents. Energy Fuels 2011, 25, 3235–3243. [Google Scholar] [CrossRef]
- Jakob, G.; Wolf, J.; Bui, T.V.; Posten, C.; Kruse, O.; Stephens, E.; Ross, I.; Hankamer, B. Surveying a diverse pool of microalgae as a bioresource for future biotechnological applications. J. Phylogenet. Evol. Biol. 2013, 4, 1000153.1–1000153.8. [Google Scholar] [CrossRef]
- Demirbas, A. Calculation of higher heating values of biomass fuels. Fuel 1997, 76, 431–434. [Google Scholar] [CrossRef]
- Friedl, A.; Padouvas, E.; Rotter, H.; Varmuza, K. Prediction of heating values of biomass fuel from elemental composition. Anal. Chim. Acta 2005, 544, 191–198. [Google Scholar] [CrossRef]
- Yu, G.; Zhang, Y.; Schideman, L.; Funk, T.L.; Wang, Z. Hydrothermal liquefaction of low lipid content microalgae into bio-crude oil. Am. Soc. Agric. Eng. 2011, 54, 239–246. [Google Scholar]
- Boie, W. Fuel technology calculations. Energietechnik 1953, 3, 309–316. [Google Scholar]
- Xu, C.; Etcheverry, T. Hydro-liquefaction of woody biomass in sub-and super-critical ethanol with iron-based catalysts. Fuel 2008, 87, 335–345. [Google Scholar] [CrossRef]
- Garcia Alba, L.; Torri, C.; Samorì, C.; van der Spek, J.; Fabbri, D.; Kersten, S.R.; Brilman, D.W. Hydrothermal treatment (HTT) of microalgae: Evaluation of the process as conversion method in an algae biorefinery concept. Energy Fuels 2011, 26, 642–657. [Google Scholar] [CrossRef]
- Huang, H.; Yuan, X.; Zeng, G.; Wang, J.; Li, H.; Zhou, C.; Pei, X.; You, Q.; Chen, L. Thermochemical liquefaction characteristics of microalgae in sub-and supercritical ethanol. Fuel Process. Technol. 2011, 92, 147–153. [Google Scholar] [CrossRef]
- Toor, S.S.; Rosendahl, L.; Rudolf, A. Hydrothermal liquefaction of biomass: A review of subcritical water technologies. Energy 2011, 36, 2328–2342. [Google Scholar] [CrossRef]
- Peterson, A.A.; Vogel, F.; Lachance, R.P.; Fröling, M.; Antal, M.J., Jr.; Tester, J.W. Thermochemical biofuel production in hydrothermal media: A review of sub-and supercritical water technologies. Energy Environ. Sci. 2008, 1, 32–65. [Google Scholar] [CrossRef]
- Varatharajan, K.; Cheralathan, M. Influence of fuel properties and composition on NOx emissions from biodiesel powered diesel engines: A review. Renew. Sustain. Energy Rev. 2012, 16, 3702–3710. [Google Scholar] [CrossRef]
- Giakoumis, E.G. A statistical investigation of biodiesel physical and chemical properties, and their correlation with the degree of unsaturation. Renew. Energy 2013, 50, 858–878. [Google Scholar] [CrossRef]
- Pandey, R.K.; Rehman, A.; Sarviya, R.M. Impact of alternative fuel properties on fuel spray behavior and atomization. Renew. Sustain. Energy Rev. 2012, 16, 1762–1778. [Google Scholar] [CrossRef]
- Kegl, B. Effects of biodiesel on emissions of a bus diesel engine. Bioresour Technol. 2008, 99, 863–873. [Google Scholar] [CrossRef] [PubMed]
- Vajda, B.; Lešnik, L.; Bombek, G.; Biluš, I.; Žunič, Z.; Škerget, L.; Hočevar, M.; Širok, B.; Kegl, B. The numerical simulation of biofuels spray. Fuel 2015, 144, 71–79. [Google Scholar] [CrossRef]
- Nabi, M.N. Theoretical investigation of engine thermal efficiency, adiabatic flame temperature, NOx emission and combustion-related parameters for different oxygenated fuels. Appl. Therm. Eng. 2010, 30, 839–844. [Google Scholar] [CrossRef]
- Senneca, O. Kinetics of pyrolysis, combustion and gasification of three biomass fuels. Fuel Process. Technol. 2007, 88, 87–97. [Google Scholar] [CrossRef]
- Yuliansyah, A.T.; Hirajima, T. Efficacy of Hydrothermal Treatment for Production of Solid Fuel from Oil Palm Wastes; INTECH: Rijeka, Croatia, 2012. [Google Scholar]
- Tyson, K.S.; McCormick, R.L. Biodiesel Handling and Use Guidelines; DIANE Publishing: Collingdale, PA, USA, 2006. [Google Scholar]
- Boelhouwer, J.; Nederbragt, G.; Verberg, G. Viscosity data of organic liquids. Appl. Sci. Res. 1951, 2, 249–268. [Google Scholar] [CrossRef]
- Vihar, R.; Seljak, T.; Rodman Oprešnik, S.; Katrašnik, T. Combustion characteristics of tire pyrolysis oil in turbo charged compression ignition engine. Fuel 2015, 150, 226–235. [Google Scholar] [CrossRef]
- Lang, X.; Dalai, A.K.; Bakhshi, N.N.; Reaney, M.J.; Hertz, P. Preparation and characterization of bio-diesels from various bio-oils. Bioresour. Technol. 2001, 80, 53–62. [Google Scholar] [CrossRef]
- Abramzon, B.; Sazhin, S. Convective vaporization of a fuel droplet with thermal radiation absorption. Fuel 2006, 85, 32–46. [Google Scholar] [CrossRef]
- Li, D.; Chen, L.; Xu, D.; Zhang, X.; Ye, N.; Chen, F.; Chen, S. Preparation and characteristics of bio-oil from the marine brown alga Sargassum patens C. Agardh. Bioresour. Technol. 2012, 104, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Vardon, D.R. Hydrothermal Liquefaction for Energy Recovery from High-Moisture Waste Biomass. Master’s Thesis, University of Illinois at Urbana Champaign, Champaign, IL, USA, 2012. [Google Scholar]
- Zhou, D.; Zhang, L.; Zhang, S.; Fu, H.; Chen, J. Hydrothermal liquefaction of macroalgae enteromorpha prolifera to bio-oil. Energy Fuels 2010, 24, 4054–4061. [Google Scholar] [CrossRef]
- Duan, P.; Chang, Z.; Xu, Y.; Bai, X.; Wang, F.; Zhang, L. Hydrothermal processing of duckweed: Effect of reaction conditions on product distribution and composition. Bioresour. Technol. 2013, 135, 710–719. [Google Scholar] [CrossRef] [PubMed]
- Duan, P.; Bai, X.; Xu, Y.; Zhang, A.; Wang, F.; Zhang, L.; Miao, J. Catalytic upgrading of crude algal oil using platinum/gamma alumina in supercritical water. Fuel 2013, 109, 225–233. [Google Scholar] [CrossRef]
- Elliott, D.C.; Hart, T.R.; Neuenschwander, G.G.; Rotness, L.J.; Roesijadi, G.; Zacher, A.H.; Magnuson, J.K. Hydrothermal processing of macroalgal feedstocks in continuous-flow reactors. ACS Sustain. Chem. Eng. 2013, 2, 207–215. [Google Scholar] [CrossRef]
- Elliott, D.C.; Hart, T.R.; Schmidt, A.J.; Neuenschwander, G.G.; Rotness, L.J.; Olarte, M.V.; Zacher, A.H.; Albrecht, K.O.; Hallen, R.T.; Holladay, J.E. Process development for hydrothermal liquefaction of algae feedstocks in a continuous-flow reactor. Algal Res. 2013, 2, 445–454. [Google Scholar] [CrossRef]
| Proximate Analyses | Ultimate Analyses | ||
|---|---|---|---|
| Composition | Percentage of Weight | Element | Percentage of Weight |
| Fixed carbon | 24.8 | C | 46.3 |
| Volatile matter | 67.3 | H | 6.9 |
| Ash | 3.2 | O | 32.3 |
| Moisture | 85 | N | 7.3 |
| - | - | S | 2.3 |
| RT (Retention Times, min) | Name of Compound | Area % | |
|---|---|---|---|
| DCM | n-Hexane | ||
| 4.5 | 4-hydroxy-4-methyl-2-pentanone | 5.03 | - |
| 4.82 | Ethylbenzene | 8.86 | 3.38 |
| 4.98 | 1,4-dimethyl benzene | 11.99 | 6.34 |
| 5.44 | 1,3-dimethyl benzene | 3.44 | 5.12 |
| 5.82 | 2,5-dimethyl pyrazine | 4.09 | - |
| 6.91 | 2,6-dimethyl-4-heptanone | - | 4.74 |
| 7.54 | Trimethyl pyrazine | 2.76 | 4.3 |
| 7.79 | 2,3,5-trimethyl-1H-pyrrole | - | 3.06 |
| 8.18 | 2,3-dimethyl-2-cyclopenten-1-one | 5.32 | 4.36 |
| 8.49 | 3,7-dimethyl undecane | - | 2.82 |
| 8.77 | 4-methyl phenol | 3.53 | 4.45 |
| 9.06 | 1-acetate-1,2,3-propanetriol | 2.99 | 7.11 |
| 9.21 | Undecane | - | 3.91 |
| 9.32 | 1-ethyl-2-pyrrolidinone | 10.38 | - |
| 10.08 | 2,2,5,5-tetramethyl-3-cyclopenten-1-one | - | 5.15 |
| 11.42 | 1-butyl-2-pyrrolidinone | 4.25 | 2.72 |
| 12.93 | 1-pentyl piperidine | 3.78 | 4.84 |
| 13.49 | 2-methyl-3-hydroxy-2,4,4-trimethylpentyl ester, propanoic acid | - | 5.11 |
| 24.91 | Di(2-propylpentyl) ester, phthalic acid | 10.78 | - |
| Total | - | 77.2 | 67.41 |
| RT (min) | Name of Compound | Area (%) | |||||
|---|---|---|---|---|---|---|---|
| Slurry Concentration at Fixed 350 °C Temperature | Temperature at Fixed 25% Slurry Concentration | ||||||
| 15% | 25% | 30% | 280 °C | 300 °C | 350 °C | ||
| 4.01 | 2-methyl pyrimidine | 3.92 | - | 4.05 | 4.94 | 8.18 | - |
| 4.41 | 4-hydroxy-4-methyl-2-pentanone | - | 5.03 | - | 23.8 | 4.15 | 4.03 |
| 4.82 | Ethylbenzene | 0.68 | 8.86 | - | 1.34 | 1.75 | 8.86 |
| 4.97 | 1,4-dimethyl benzene | - | 11.9 | - | 2.48 | 2.77 | 11.3 |
| 5.67 | 2-methyl-2-cyclopenten-1-one | 4.71 | - | 2.72 | 9.19 | 10.0 | - |
| 5.79 | 2,5-dimethyl pyrazine | 4.84 | 4.09 | 3.53 | 12.1 | 12.4 | 4.09 |
| 6.8 | 3-methyl-2-cyclopenten-1-one | 6.66 | 4.86 | 2.31 | 2.7 | 6.0 | 4.5 |
| 7.1 | Phenol | 3.24 | 3.33 | 2.16 | - | 2.43 | 3.32 |
| 8.15 | 2,3-dimethyl-2-cyclopenten-1-one | 12.8 | 12.0 | 5.32 | 2.29 | 5.73 | 6.05 |
| 9.03 | 1-acetate-1,2,3-Propanetriol | 4.72 | 5.09 | 5.63 | 2.58 | 4.22 | 5.19 |
| 9.27 | Undecane | 9.20 | 11.1 | 12.6 | 2.49 | 5.9 | 8.99 |
| 9.94 | 1,3-diethyl-3-methyl-2,5-pyrrolidinedione | 2.27 | 2.5 | 2.72 | - | 2.48 | 2.51 |
| 10.69 | 1-propyl-2-pyrrolidinone | 3.04 | 2.32 | 3.48 | - | - | 2.30 |
| 11.39 | 1-butyl-2-pyrrolidinone | 3.3 | 4.25 | 3.16 | - | - | 4.20 |
| 24.91 | Di(2-propylpentyl) ester, phthalic acid | - | 10.7 | 8.25 | - | - | 10.7 |
| Component (wt. %) | 25% Slurry Concentration at Different Temperature | 350 °C Temperature at Slurry Concentration | ||||
|---|---|---|---|---|---|---|
| 280 °C | 300 °C | 350 °C | 15% | 25% | 30% | |
| C | 68.1 | 70.4 | 75.6 | 74.1 | 75.6 | 73.7 |
| H | 9.3 | 8.9 | 10.1 | 9.7 | 10.1 | 9.8 |
| O | 15.7 | 12.1 | 10.3 | 11.2 | 10.3 | 10.8 |
| N | 6.9 | 8.6 | 4.0 | 5.0 | 4.0 | 5.7 |
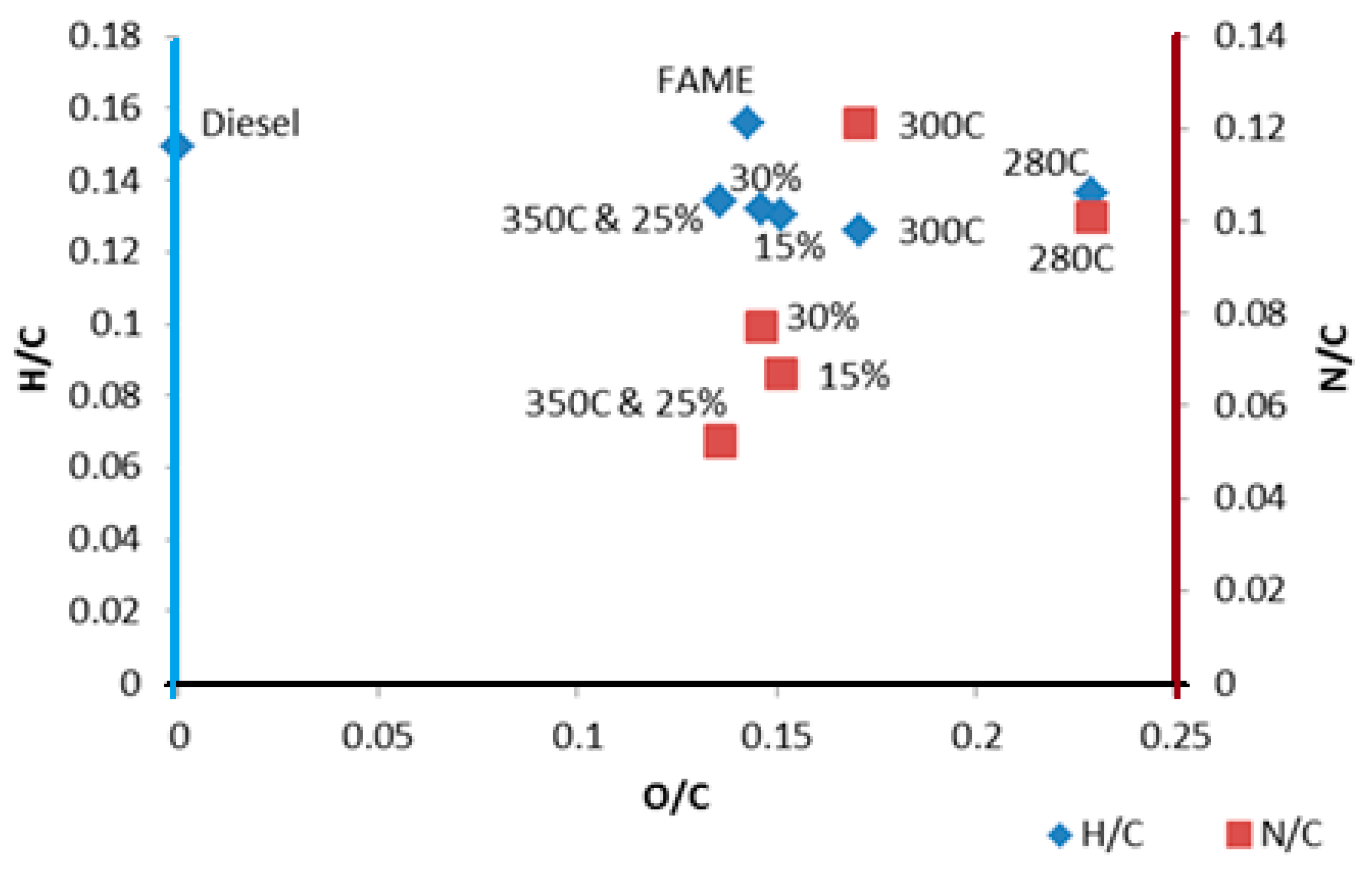
| H:C | 0.14 | 0.13 | 0.13 | 0.13 | 0.13 | 0.13 |
| O:C | 0.23 | 0.17 | 0.14 | 0.15 | 0.14 | 0.15 |
| N:C | 0.10 | 0.12 | 0.05 | 0.07 | 0.05 | 0.08 |
| HHV (Cal.), MJ·kg−1 | 33.5 | 34.3 | 37.4 | 36.4 | 37.5 | 36.5 |
| HHV(Meas.), MJ·kg−1 | -- | 26.1 | 29.8 | 26.5 | 29.8 | 28.0 |
| Name of the Properties | HTL Scenedesmus sp. Biocrude Oil | FAME Crypthecodinium cohnii Biodiesel [52] | Biodiesel Standards EN 14214 | Petroleum Diesel | Marine Fuels ISO 8217 |
|---|---|---|---|---|---|
| Kinematic viscosity@40 °C (mm2·s−1) | 70.7–73.8 | 5.06 | 3.5–5 | 2.64 | 1.4–11.0 |
| Density @15 °C (kg·L−1) | 0.97 * | 0.91 | 0.86–0.9 | 0.84 | 0.96–0.99 |
| HHV (MJ·kg−1) | 29.7 | 39.8 | - | 44 | 44–45 |
| Oxygen content (wt. %) | 10.3 | 10.4 | - | 0 | - |
| Hydrogen Content (wt. %) | 10.1 | 11.1 | - | 13.8 | - |
| Carbon Content (wt. %) | 75.5 | 78.4 | - | 86.1 | - |
| Nitrogen Content (wt. %) | 3.97 | 0 | - | 0 | - |
| HTL Bio-Crude | Reactor Type | Reactor Volume (mL) | Operating Conditions | Yield wt. % | Physicochemical Properties | References | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Temperature (°C) | Time (min) | Chemical Composition % | HHV (MJ/kg) | Kinematic Viscosity (mm2/s) | Density (kg/L) | |||||||||
| C | H | N | O | Calculated | Measured | |||||||||
| Scenedesmus sp. | Batch reactor | 1800 | 350 | 60 | 33.6 | 75.6 | 10.1 | 3.97 | 10.3 | 37.4 | 29.8 | 70.7–73.8 | 0.97 | Current study |
| 500 | 300 | 30 | 45 | 72.6 | 9.0 | 6.5 | 10.5 | 35.5 | - | - | - | [59] | ||
| Enteromorpha prolifera | 25 | 300 | 30 | 23 | 64.5 | 7.7 | 5.4 | 22.4 | 30.8 | - | - | - | [60] | |
| Lemna sp. | 25 | 350 | 30 | 17.5 | 72.1 | 7.8 | 4.6 | 15.5 | 32.8 | - | - | - | [61] | |
| Laminaria saccharina | 75 | 350 | 15 | 19.3 | 82.0 | 7.1 | 4.9 | 6.0 | 37.4 | - | - | - | [27] | |
| Chlorella pyrenoidosa | 17.2 | 350 | 60 | 41 | 75.1 | 9.9 | 7.3 | 7.7 | 38.1 | - | - | - | [62] | |
| Nannochloropsis sp. | 35 | 350 | 60 | 43 | 76.0 | 10.3 | 3.9 | 9.0 | 39 | - | - | - | [32] | |
| Enteromorpha prolifera | 25 | 370 | 40 | 31.7 | 77.9 | 9.6 | 5.6 | 6.9 | 39.4 | - | - | - | [10] | |
| Sargassum patens C. Agardh | 1000 | 340 | 10 | 32.1 | 64.6 | 7.4 | 2.5 | 25.5 | - | 27.1 | - | - | [58] | |
| Saccharina sp. | Continuous-flow reactor | 1000 | 340 | 87 | 58.8 | 79.4 | 8.0 | 4.1 | 8.5 | 37.5 | - | - | - | [63] |
| NB238 | 1000 | 350 | - | 38 | 78.6 | 10.4 | 4.2 | 5.3 | - | - | - | - | [64] | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hossain, F.M.; Kosinkova, J.; Brown, R.J.; Ristovski, Z.; Hankamer, B.; Stephens, E.; Rainey, T.J. Experimental Investigations of Physical and Chemical Properties for Microalgae HTL Bio-Crude Using a Large Batch Reactor. Energies 2017, 10, 467. https://doi.org/10.3390/en10040467
Hossain FM, Kosinkova J, Brown RJ, Ristovski Z, Hankamer B, Stephens E, Rainey TJ. Experimental Investigations of Physical and Chemical Properties for Microalgae HTL Bio-Crude Using a Large Batch Reactor. Energies. 2017; 10(4):467. https://doi.org/10.3390/en10040467
Chicago/Turabian StyleHossain, Farhad M., Jana Kosinkova, Richard J. Brown, Zoran Ristovski, Ben Hankamer, Evan Stephens, and Thomas J. Rainey. 2017. "Experimental Investigations of Physical and Chemical Properties for Microalgae HTL Bio-Crude Using a Large Batch Reactor" Energies 10, no. 4: 467. https://doi.org/10.3390/en10040467
APA StyleHossain, F. M., Kosinkova, J., Brown, R. J., Ristovski, Z., Hankamer, B., Stephens, E., & Rainey, T. J. (2017). Experimental Investigations of Physical and Chemical Properties for Microalgae HTL Bio-Crude Using a Large Batch Reactor. Energies, 10(4), 467. https://doi.org/10.3390/en10040467






