Abstract
The negative impact of fossil fuels and the increased demand for renewable energy sources has led to the use of novel raw material sources. Lignocellulosic biomass could serve as a possible raw material for anaerobic digestion and production of biogas. This work is aimed at using forest biomass, both softwood (spruce) and hardwood (birch), as a raw material for anaerobic digestion. We examined the effect of different operational conditions for the organosolv pretreatment (ethanol content, duration of treatment, and addition of acid catalyst) on the methane yield. In addition, we investigated the effect of addition of cellulolytic enzymes during the digestion. We found that inclusion of an acid catalyst during organosolv pretreatment improved the yields from spruce, but it did not affect the yields from birch. Shorter duration of treatment was advantageous with both materials. Methane yields from spruce were higher with lower ethanol content whereas higher ethanol content was more beneficial for birch. The highest yields obtained were 185 mL CH4/g VS from spruce and 259.9 mL CH4/g VS from birch. Addition of cellulolytic enzymes improved these yields to 266.6 mL CH4/g VS and 284.2 mL CH4/g VS, respectively.
1. Introduction
From the beginning of the industrial revolution, fossil fuels such as crude oil, coal, and gas were the main source of energy for transportation and for economic growth [1,2]. As has been stated previously, “the world’s energy market worth around 1.5 trillion dollars is still dominated by fossil fuels” [3] and according to the International Energy Agency (IEA), fossil fuels provide more than 81% of the world’s energy [4]. According to the same report, combustible biomass and waste contribute 10.3%, nuclear energy contributes 4.8%, and hydro-power contributes 2.4% to global energy [4]. During the last years, many studies have been conducted on the longevity of fossil fuels, and have indicated an imminent shortage of them [5]. With the consumption of fossil fuels still being on the increase, the next issue will be the rising price of these fuels [5,6]. Apart from the economic challenges involved, the use of fossil fuels also has an important negative effect on the environment. The impact of fossil fuels can be distinguished at the local, regional, and global level [3]. On a local level, fuel combustion pollutes urban air, which causes thousands of deaths annually around the world. At the regional level, the acidification that occurs from precursors of acid deposition from combustions, thousands of kilometres from their origin, can damage nature, crops, and buildings. At the global level, the release of carbon dioxide and other gases into the atmosphere results in the so-called “greenhouse effect” [3].
It is apparent that in order to avoid these negative effects, replacement of fossil fuels with renewable energy is an important priority. One of the solutions is the development of biofuels from renewable materials. Biofuels can be divided into three categories: solid (e.g., bio-char), liquid (e.g., biodiesel, ethanol, and vegetable oil), and gaseous (e.g., biohydrogen, biogas, and biosyngas) [1].
Biogas is a gaseous biofuel and it is one of the most versatile sources of renewable energy, which can be used for power and heat production and also as a fuel for vehicles [7]. It mostly consists of carbon dioxide (CO2) and methane (CH4), together with small amounts of other gases [8,9]. The CO2:CH4 ratio governs the energy efficiency of the biogas, which is between 18,630 and 26,081 kJ/m3 approximately [8]. Biogas can be produced on demand and its storage is relatively easy. The distribution of biogas can be done through the natural gas infrastructure that already exists. Moreover, the digested material, the so-called digestate, can be used as a biofertilizer in agriculture [10]. Production of biogas through anaerobic digestion is one of the most energy-efficient and environmentally beneficial technologies [7]. Anaerobic digestion is a complex biochemical process whereby a variety of anaerobic microorganisms that work synergistically in the absence of oxygen convert organic material into biogas. Anaerobic digestion can be divided into several steps: hydrolysis, acidogenesis, acetogenesis, and methanogenesis [11]. Different raw materials can be used for biogas production; these include organic wastes from households and the food industry, lignocellulosic material, animal manure, and wastewater sludge [12,13].
Lignocellulosic biomass is a widely available renewable raw material, rich in complex carbohydrates (55%–75% dry basis), and as such a suitable feedstock for bioenergy production [14]. Lignocellulosic biomass mainly consists of cellulose, hemicellulose, lignin, extractives, and inorganic materials [15] and its composition varies depending on the lignocellulosic species and origin [16]. Lignocellulose can be found in agricultural and wood residues, municipal solid waste, animal manure and dedicated energy crops [15,16].
From the above-mentioned sources of lignocellulose, wood sources have been the least examined in the literature even though they are very important—especially for countries with high volumes of forests, such as the Nordic countries. In Sweden, forest-based industry plays a very vital role in the national economy and it is currently under economic pressure, which would make the use of waste and by-products in the production of methane an important source of extra income. Depending on its structure, wood falls into two categories: hardwood and softwood. Softwoods contain around 40%–45% cellulose, 25%–29% hemicellulose, and 30%–60% lignin, with the hemicelluloses being mainly glucomannans. Hardwoods contain 25%–40% hemicellulose, 45%–47% cellulose, and 20%–25% lignin, with xylan being the main component of the hemicellulose [17]. Apart from differences in the chemical composition of hemicellulose, differences also occur in the lignins of the two species, with the softwood lignin consisting mainly of guaiacyl units, and hardwood lignin containing a mixture of guaiacyl and syringyl units [15]. The complex structure of lignocellulosic material makes it difficult to hydrolyze. The main factors that affect the hydrolysis of cellulose are: the crystallinity of cellulose, the amount of accessible surface area, the degree of cellulose polymerization, the amount of lignin and hemicellulose, and the degree of acetylation of hemicelluloses [13,18]. These factors have a negative effect on enzymatic hydrolysis, and to achieve efficient biodegradation and biogas production, a pretreatment step is required [15,19]. The purpose of pretreatment is to change the size and structure of lignocellulose, as well as the chemical composition, thus making the hydrolysis of carbohydrates to monomeric sugars more efficient and giving higher sugar yield and productivity [20]. During the pretreatment, different modifications of the structure of lignocellulose can occur, such as removal or modification of lignin, removal of hemicellulose, a reduction in the crystallinity and in the degree of polymerization, and an increase in pore volume [21]. There are several pretreatment processes available, and these fall into four categories: physical pretreatment, physicochemical pretreatment, chemical pretreatment, and biological pretreatment [22,23]. Different pretreatment methods were examined for obtaining methane from wood biomass, such as the use of steam explosion [24,25,26,27,28], supercritical water [29], ionic liquids [28,30,31], biological pretreatment [25], and organosolv [32,33].
Organosolv pretreatment is based on using organic or aqueous-organic solvents and cooking of lignocellulosic material at higher temperatures (100–250 °C) [34]. Lignocellulose is separated into its main fractions, with lignin and hemicellulose being dissolved, leaving cellulose in the solid phase [35]. A higher rate of delignification and higher yields of hemicellulose solubilization can be obtained when acid catalysts are added [36]. One important advantage of the process is the ability to recover the organic solvent by distillation and to reuse it again for pretreatment [35]. Many solvents have been used in organosolv pretreatment, such as methanol, ethanol, acetone, high-boiling-point alcohols, and organic acids [37]. Methanol and ethanol are often used as solvents for organosolv pretreatment, due to their low cost and their low boiling points, which makes the recovery of the solvent by distillation easier—with a lower energy requirement [35]. Kabir et al. [32] tested three different organic solvents (ethanol, methanol, and acetic acid) for pretreatment of forest residues. They obtained high yields of methane when acetic acid and ethanol were used rather than methanol. During the techno-economic evaluation that they performed, they found that treatment with methanol was more financially viable due to its lower cost. However, methanol is more toxic and flammable than ethanol [38]. Although organosolv can be used for efficient pretreatment of both softwood and hardwood biomass, it can also lead to the generation of inhibitory compounds such as furfural and HMF, produce low yields of hemicellulosic sugars, and could be regarded as an expensive method [39].
In this work, we investigated the use of two kinds of forest biomass, softwood (spruce) and hardwood (birch), as raw material for anaerobic digestion after subjecting them to different operational conditions for organosolv pretreatment. More specifically, the current work is a part of a broader project where the operational conditionals of organosolv are optimized to effectively fractionate hardwood and softwood biomass into lignin, cellulose and hemicellulose fractions; with the lignin and hemicellulose fraction to be further used for other processes. These processes can involve the conversion of lignin to advanced carbon materials [40], and conversion of hemicellulosic sugars through chemical of biochemical routes to value added chemicals such as furfural, furans, butanediol, ethanol and polymer intermediates [41,42]. The cellulose-rich solid fraction is evaluated as raw material for methane production through anaerobic digestion.
2. Results and Discussion
2.1. Effect of Organosolv Pretreatment on Enzymatic Hydrolysis
As it was previously discussed, lignocellulose is a very stable and complex structure, which makes it resistant to enzymatic saccharification [43]. To make cellulose more susceptible to enzymatic hydrolysis to its constituent sugars, a pretreatment stage is required. Vivekanand et al. [44] tested the effect of different steam explosion parameters on the enzymatic hydrolysis of birch. The glucose concentration after 24-h enzymatic hydrolysis trials increased until a severity factor of 4.5, but treatment at higher severity did not increase glucose concentrations any further. On the contrary, the highest severities tested even led to a reduction in glucose concentration. According to the authors, one possible reason for this finding may be the modification of the lignin and the formation of pseudo-lignin, which inhibits the action of the enzymes on cellulose. Kang et al. [45] also tested different severities of SO2-catalyzed steam explosion on the digestibility of softwood biomass (Loblolly pine). They showed that with increasing severity of pretreatment, non-selective degradation of carbohydrates occurs. Pretreatment with the highest severity resulted in the lowest glucan content, due to the unspecifically hydrolyzed cellulose.
In the current work, we evaluated the effects of different parameters of organosolv pretreatment on the release of sugars after enzymatic hydrolysis for 24 h and 48 h. These parameters varied regarding the duration of the pretreatment, the concentration of ethanol, and the addition of sulfuric acid as a catalyst. The different conditions that were examined resulted in varied severities of pretreatment, and to be able to compare the different treatment combinations, the combined severity factor (CSF) was calculated according to the following equation:
where t is the reaction time (min), T is the pretreatment temperature (°C) and TR is a reference temperature (100 °C) [21]. The values of CSF for the treatment combinations varied between −0.21 and 1.84.
CSF = log (texp[T − TR/14.75]) − pH
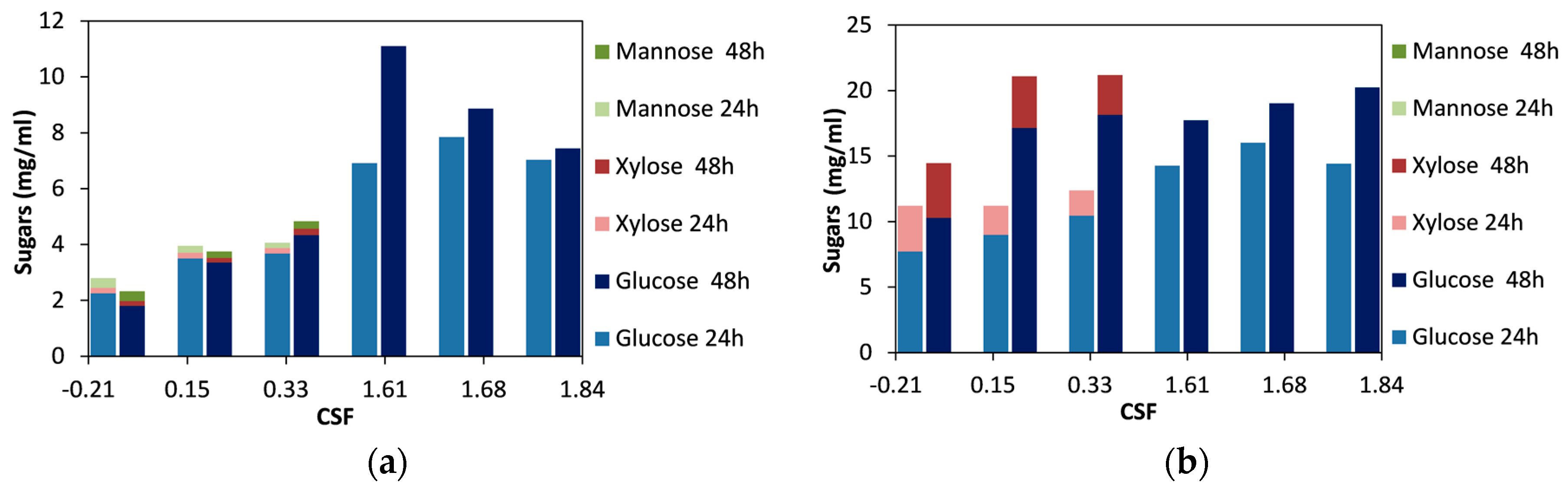
For spruce biomass, we observed that with increasing severity, the concentration of sugars after 48 h of hydrolysis steadily increased up to a CSF value of 1.61 (Figure 1a). Higher severities resulted in a decrease in the concentration of sugars released. Moreover, hemicellulosic sugars (xylose and mannose) were only detected in the lower range of CSF (for values between −0.21 and 0.33). The highest amount of sugars released was obtained for the material treated for 60 min with 60% ethanol and acid catalyst (CSF 1.61), and this can be attributed to the high cellulose content of the material, which reached 69.1% w/w and was the highest among all the materials used in this work [46]. The duration of the enzymatic hydrolysis did not significantly affect the release of sugars, as all the materials (except the one treated at a CSF of 1.61) reached the highest sugar concentration after 24 h of hydrolysis, and prolonging hydrolysis to 48 h did not have a significant effect. In contrast, spruce treated at a CSF of 1.61 continued to be hydrolyzed up to 48 h, and this was probably due to the high cellulose content.
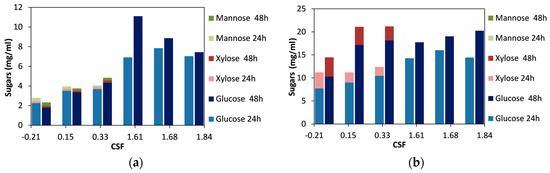
Figure 1.
Effect of the combined severity factor (CSF) of organosolv pretreatment, on the release of sugars after enzymatic hydrolysis for 24 h and 48 h: (a) spruce; (b) birch. The difference between the replicates was less than 10% in all the samples.
For the birch biomass, there was a clear trend of an increase in sugar release after 24 h of hydrolysis up to a CSF of 1.68, and for higher severities the concentration of the sugars decreased (Figure 1b). When the enzymatic hydrolysis was prolonged to 48 h, the sugar concentration increased up to a CSF value of 0.33; for higher CSF values, the concentration of the sugars was less. This decrease in the concentration of total sugars was due to the fact that no xylose was detected for materials treated at CSF 1.61 and higher. On the other hand, the glucose concentration was stable for CSF 0.15 and higher (Figure 1b). Moreover, it can be noted that for materials treated without any acid catalyst (CSF values between −0.21 and 0.33), prolongation of enzymatic hydrolysis to 48 h was important, whereas for the materials treated with acid (CSF values between 1.61 and 1.84), most of the sugars were released during the first 24 h of hydrolysis. This indicates that although the glucose concentration was not affected by the severity of the treatment, the productivity of glucose release was higher under more severe conditions.
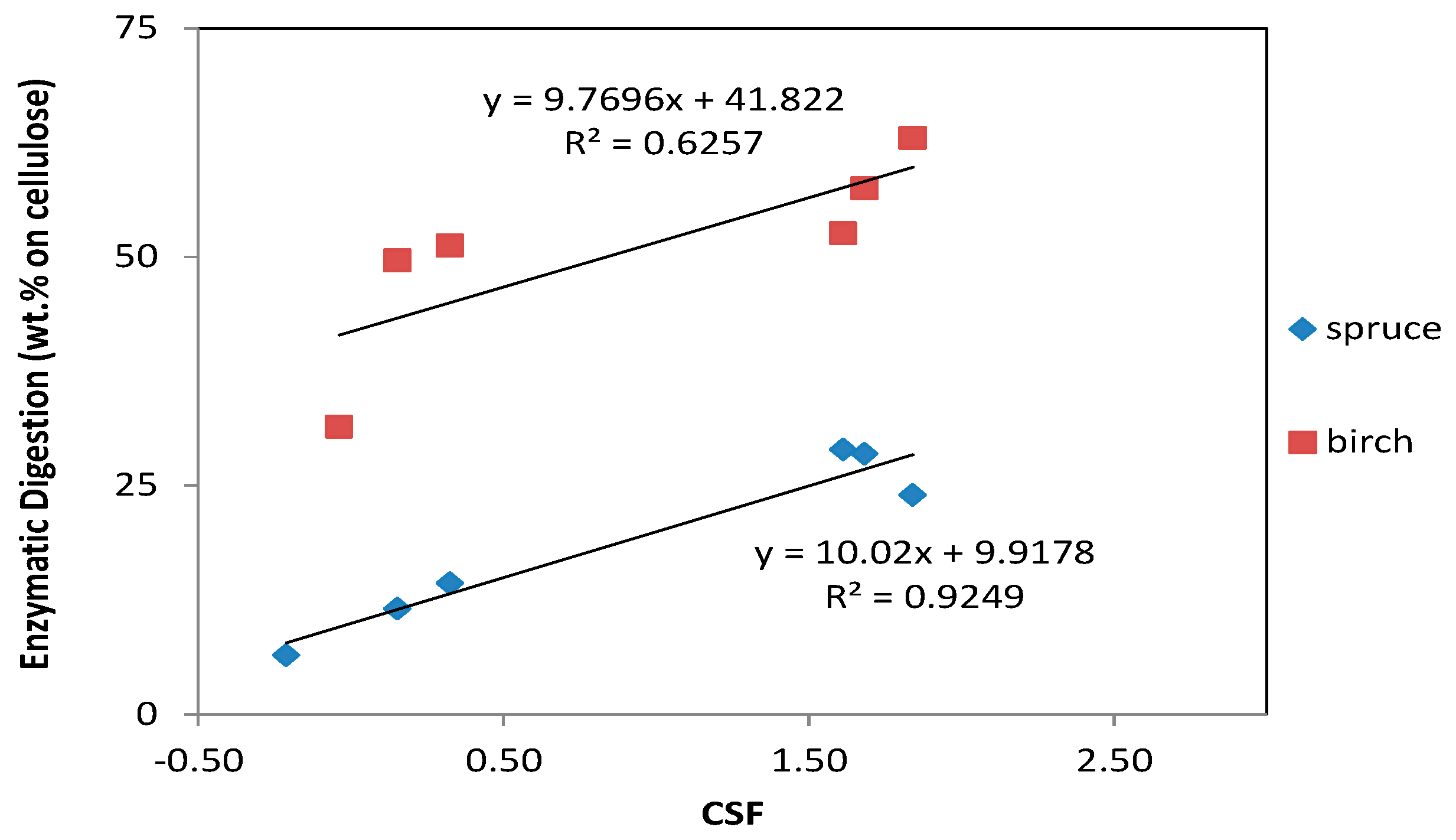
During this study, we observed that when we increased the CSF, the enzymatic digestibility of cellulose also increased in both materials (Figure 2). In both, there was a strong positive correlation between CSF and enzymatic digestion; with the spruce showing a slightly higher positive effect of increasing the CSF. The greatest amount of saccharification of cellulose for birch and spruce was 63% and 28.9%, respectively. This high conversion of cellulose to glucose makes the materials promising feedstock for microbial conversion processes. Regardless of the CSF, birch generally had higher cellulose conversion yields than spruce. This is a result of the difference in structure between hardwoods and softwoods: the softwoods have harder material, and more severe conditions are necessary to break them down.
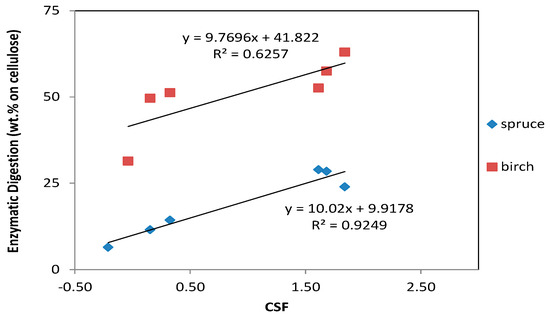
Figure 2.
Correlation between the combined severity factor (CSF) and the enzymatic digestibility of cellulose.
It was also evident that addition of the acid catalyst generally improved the enzymatic digestibility of both spruce and birch (the values with the highest CSF correspond to the pretreatment with the addition of acid catalyst). Addition of an acid catalyst during organosolv pretreatment of lignocellulosic biomass with ethanol is commonly used to enhance enzyme accessibility. For example, Pan et al. [47] used an acid-ethanol pretreatment of lodgepole pine, which resulted in a conversion of 93%–97% of the cellulose to glucose. Of the different options of acid catalysts, sulfuric acid is the most frequently used one because of its strong reactivity and high efficiency [47,48], and it has been used in numerous feedstocks [21]. Park et al. [49] evaluated three different types of catalysts (sulfuric acid, magnesium chloride, and sodium hydroxide) for organosolv pretreatment of pitch pine, followed by a digestibility assay of the pretreated material. With 1% sulfuric acid, the digestibility was approximately 55%–60% and with 1% magnesium chloride the digestibility was almost 60%. Pretreatment with 1% sodium hydroxide had no effect on the digestibility, which, however, improved to 85% when the concentration of the sodium hydroxide was increased to 2%.
2.2. Effect of Pretreatment Time on Methane Yield
As previously discussed, the aim of this work was to evaluate the effect of organosolv pretreatment conditions on the methane yield. Pretreatment duration plays a crucial role in the pretreatment, and increasing the treatment duration can improve the digestibility of the biomass. In addition, a prolonged duration of treatment can result in decomposition of the biomass and loss of sugars, and also generation of inhibitory compounds. To investigate the effect of duration of pretreatment, the materials were treated for 60 min and for 103 min in the presence or absence of acid catalyst, as previously reported [46].
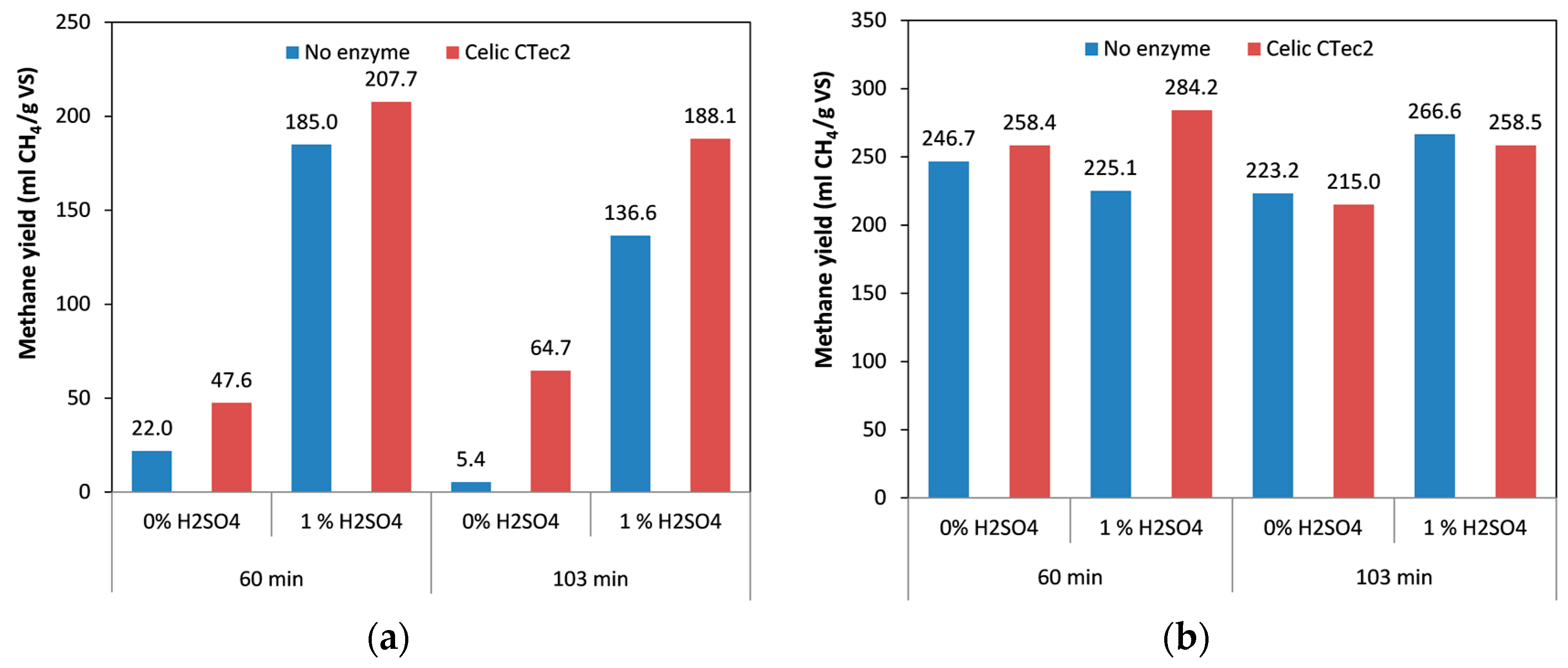
For spruce biomass, we found that with prolongation of the duration from 60 min to 103 min, the methane yields were reduced—both with and without addition of the acid catalyst (Figure 3a). This decrease was between 26.1% and 75.5%, the highest decrease being observed in the presence of the acid. With prolongation of the pretreatment time an increase in the hydrolysis of carbohydrates into monomeric pentose and hexose sugars, is achieved [46]. When the treatment conditions become more severe, these sugars can be degraded into different inhibitors (e.g., furfural, hydroxymethyfurfural, formic acid, or levulinic acid) that might negatively affect the microorganisms during anaerobic digestion. However, as the materials were thoroughly washed after the pretreatment, the inhibitors generated during the pretreatment were removed from the solid fraction. As only the solid fraction was used for the digestion, no inhibitors were present, indicating that it is not possible that the decrease in the methane yield could be attributed to the presence of inhibitors during the digestion. The negative impact of increased pretreatment time on methane yield can be associated with the degradation of the substrate and the loss of sugars. Another possible reason is the modification of lignin and the formation of pseudolignin on the biomass surface [44]. Moreover, in the presence of the acid catalyst, the lignin concentration increased from 31.7% w/w to 37.8% w/w [46] when the duration of pretreatment was increased, and this increase in lignin content could affect the methane yield, as lignin “protects” the cellulose from microbial attacks. Moreover, cellulases tend to become irreversibly adsorbed to lignin, reducing their adsorption to cellulose [50,51]. The fact that a shorter duration of treatment resulted in a higher methane yield is more favorable for industrial application, as less energy is required to treat the material, thus reducing costs. We also noted that introduction of the acidic catalyst in the pretreatment lead to an increase of the methane yields by 8.4–25.3 times compared to the non-catalytically pretreated samples. This positive effect is the result of the improved hemicellulose removal that is achieved by the introduction of the acid catalyst [46] that increases the accessibility of the biomass to enzymes and microorganisms. This was also evidenced by the higher biomass solubilization during the pretreatment experiments with an increase from 25%–30% to 50%–55% when the acid catalyst was included [46]. The highest yield of methane obtained during this part of the work was 185 mL CH4/g VS; which in comparison with the biogas production from hydrothermally treated spruce without the use of enzymes [52], the biogas production yield from organosolv treated spruce was between approximately 26% and 94% higher (depending on the severity of the hydrothermally treated spruce).
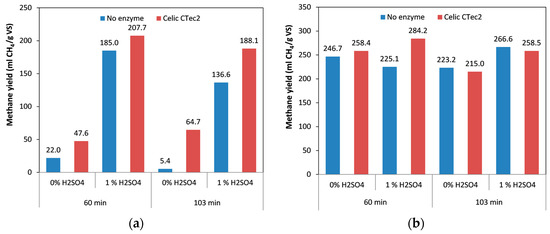
Figure 3.
Effect of duration of pretreatment, in combination with addition of acid catalyst and cellulolytic enzymes, on methane yield: (a) spruce; (b) birch. The difference between the replicates was less than 10% in all the samples.
Finally, the effect of cellulolytic enzymes during the anaerobic digestion was also evaluated. The methane yield obtained from spruce pretreated with additional enzyme was higher comparing to when no enzyme was added; this positive effect was observed in all treatment combinations (i.e., higher by a factor of between 1.1 and 12 times) (Figure 3a). This improvement was more pronounced when spruce was treated for 103 min, especially in the absence of the acid catalyst. The highest methane yield was obtained from the spruce pretreated at 60 min in the presence of catalyst and enzymes, and reached 207.7 mL CH4/g VS (approximately 12% higher than for the digestion without enzymes). The improvement in methane yields when cellulolytic enzymes were included can be attributed to the fact that the enzymes lead to better solubilization of cellulose. The hydrolysis of carbohydrates to monomeric sugars is considered to be the rate-limiting step in anaerobic digestion [13,52]. The same positive effect of enzymes on methane yields was also found in our previous work with hydrothermally pretreated spruce [52], and in some other studies in the literature. For example, Sonakya et al. [53] used wheat grain and treated it with Tizyme Trizyme? (a mixture of cellulase, α-amylase, and protease) prior the anaerobic digestion, which improved the methane yield by 7%–14%. Antonopoulou and Lyberatos [54] also showed an increase in carbohydrate solubilization and in turn, an increase in methane yield, when they used sweet sorghum as a substrate.
Similar trends to those with spruce could also be observed in birch, although the negative effect of a prolonged duration of treatment was not as apparent as with spruce. Increase in treatment time from 60 min to 103 min decreased the methane yield by 9.5% when the acid catalyst was not included (Figure 3b). On the other hand, an increase of approximately 18.4% is observed when birch was treated for 103 min with sulfuric acid. Even though prolonged treatment reduced the methane yields when the catalyst was not included, for both biomass types, the decrease was more pronounced in the case of spruce. The addition of acid catalyst improved the methane yield by approximately 19% when the material was treated for 103 min. In the material that was treated for 60 min, use of the catalyst resulted in a small reduction in the methane yield, of 8.6%.
Finally, the use of cellulolytic enzymes improved the methane yield, both for the 60-min pretreatment and for the 103-min pretreatment, with an improvement ranging between 9.1% and 16.8% (with only the material treated without acid catalyst for 103 min resulting in a minor decrease in methane yield) (Figure 3b). One important observation was that under all the conditions tested, the methane yield from birch was not significantly different between the different treatment conditions. This indicates that the conditions used were already severe enough for the efficient conversion of birch into methane, even at lower CSF values. The highest yield obtained from birch was 284.2 mL/g VS. This yield was close to that obtained in our previous study (305 mL CH4/g VS) from birch that was given hydothermal pretreatment with additional SO2 as a catalyst and cellulolytic enzymes [52]. One major difference was that during our previous work, the whole pretreated slurry was used, which also contained sugars from hemicellulose, whereas in the current work only the cellulose-rich fiber fraction of the pretreatment was used. The reason for using only the fiber fraction, lays in the fact that the lignin and hemicellulose fraction, which are recovered as separate fraction during organosolv pretreatment, can be further utilized in other process. That is also the main benefit of the organosolv pretreatment in comparison with other traditional pretreatment techniques, as it allows the recovery of the different fraction and their separate use in various processes.
When we compared birch and spruce, the methane obtained from birch was higher under all the treatment conditions, with the methane yield ranging from 18% to 98% higher. For both materials, the shorter treatment duration (60 min) was more beneficial for higher methane yield and the highest yields were obtained in the presence of both acid catalyst and enzymes.
2.3. Effect of Ethanol Content on Methane Yield
After evaluating the effect of duration of pretreatment on the methane yield, we examined the effect of ethanol concentration on methane yield. Ethanol concentration is an important factor during organosolv pretreatments, and it has been shown to affect both the delignification and the fragmentation of the lignin obtained. When using low ethanol concentration, acid-catalyzed cleavage of α- and β-ether linkages in the lignin resulted in the production of lignin fragments of smaller molecular weight that were soluble [38]. On the other hand, when higher ethanol concentrations were used, solubilization of lignin itself was also increased without the need for fragmentation [38]. Pan et al. [47] also observed that at low ethanol concentrations, acid-catalyzed cleavage of lignin was promoted whereas higher ethanol concentrations enhanced dissolution of depolymerized lignin fragments. In the materials used in the present work, we generally observed that increase in ethanol from 50% to 60% resulted in higher solubilization of lignin and, in turn, lower lignin content [46].
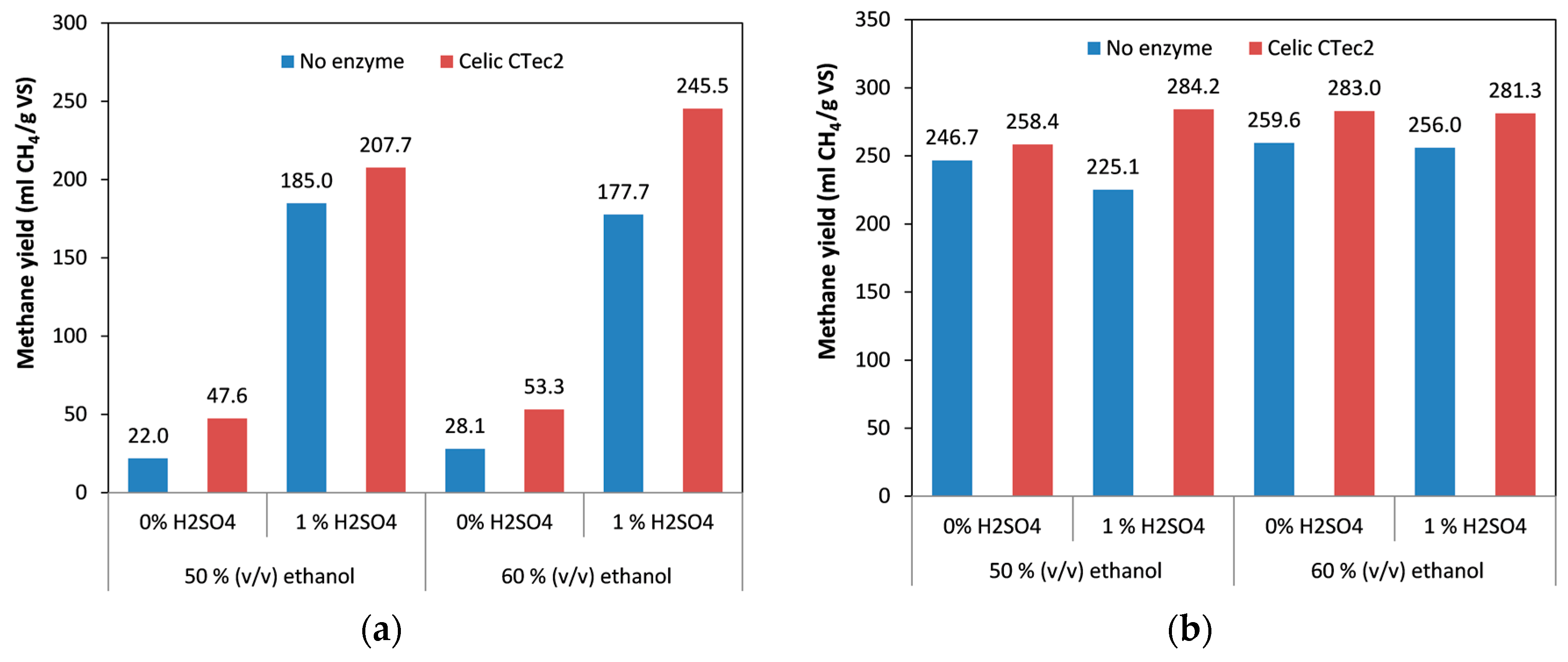
Although the lignin content of the spruce pretreated with 60% ethanol was lower than in spruce pretreated with 50% ethanol, the methane yields obtained from these materials were the same (Figure 4a), with a minor decrease in the material treated with acid catalyst and a minor increase in the material with no acid catalyst. This could be attributed to the fact that although the major effect of ethanol during pretreatment was removal of lignin and hemicellulose, it also had a (non-uniform) effect on cellulose and could sometimes result in more recalcitrant cellulose [55,56,57].
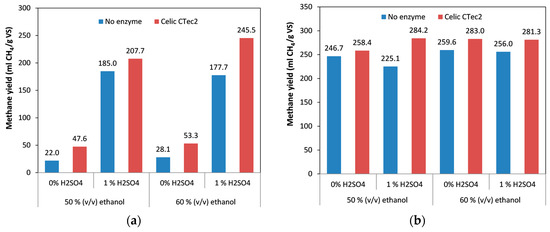
Figure 4.
Effect of ethanol content, in combination with addition of acid catalyst and cellulolytic enzymes, on methane yield: (a) spruce; (b) birch. The difference between the replicates was less than 10% in all the samples.
It was again found that adding catalyst during the pretreatment significantly increased the methane yield, by a factor of 4.3–8.4 times, with the highest positive effect being observed when 50% ethanol was used (Figure 4a). Finally, addition of enzymes during anaerobic digestion improved methane yield under every set of conditions, increasing the methane yield by 11% to 54%. We also found that when enzymes were included, the materials that were treated with the higher ethanol concentration gave higher methane yields. This was even more apparent in the spruce treated with a combination of ethanol and acid catalyst, which was probably a result of the increased cellulose content of this material with 60% ethanol (69% w/w) relative to the one treated with 50% ethanol (56% w/w) [46]. Although this positive effect on methane yield was not observed without the use of enzymes (in which case the methane yields were similar), it appears that the presence of enzymes facilitated the hydrolysis of the more recalcitrant cellulose to glucose, which in turn was converted to methane. The highest yield obtained for spruce during the current work was 245.5 mL CH4/g VS (Figure 4a) which is higher than other yields from obtained fiber fractions reported in the literature, such as fiber fraction from steam refining with SO2 which demonstrated a biogas yield of 200 mL/g (organic dry matter) [58]. One should also take into account that this is the total biogas yield and the expected methane content is normally around 60% so the difference in the methane yield is even higher. Other methods of pretreatment such as ionic liquids have also been employed for the pretreatment of spruce. In the study of Gao et al. [59] the yield obtained was considerably lower compared to our work, reaching a biogas yield of 122 mL/g VS (with approximately 60% methane content). On the other hand, Teghammar et al. [30] obtained similar results (245 mL CH4/g VS) with our work from ionic liquid pretreated spruce. Finally, alkaline pretreatment resulted also in lower results of methane yields, such as 50 mL/g VS after pretreatment with 7% w/w NaOH [60], whereas addition of 5.5% w/w thiourea resulted in a methane yield of 210 mL/g VS [61]. It is apparent from the above that the organosolv pretreatment is one of the most efficient techniques to pretreat spruce biomass to achieve high mehane yields during anaerobic digestion.
As was noted previously, birch gave higher yields of methane than spruce with all the variables tested during this work. Increase in ethanol concentration from 50% to 60% had a positive effect on the methane yields, with an increase of between 5.2% (without catalyst) and 13.7% (with the catalyst). Unlike the situation with spruce, addition of the acid catalyst had no effect or even a slight negative effect, indicating that the treatment conditions were severe enough for effective treatment of birch biomass. When birch was used, addition of the catalysts did not have any significant effect on the methane yields (in contrast to spruce), probably due to the presence of acetic acid formed from the birch itself, which in turn acted as a catalyst. Finally, addition of cellulolytic enzymes had a positive effect, which resulted in an increase in the methane yields by up to 21%. The highest yield obtained from birch was 284.2 mL CH4/g VS, which is comparable to that in some studies reported in the literature. Although the results were slight lower compared to the yield obtained from hydrothermally pretreated birch (305 mL CH4/g VS) [52], during the current work only the fiber fraction was used, whereas the whole slurry containing the solubilized sugars was used for the hydrothermally pretreated birch, which resulted in the higher methane yield. As was previously discussed, the main advantage of the organosolv pretreatment is the ability to fractionate biomass and use the other components (lignin and hemicellulose) for other process. Use of the whole slurry results in the loss of valuable compounds such as lignin. The methane yield obtained from the organosolv pretreatment was also higher compared to the methane yield obtained after ionic liquids treatment (232 mL CH4/g VS) [62]. In another work with alkaline pretreatment, the obtained methane yield was higher than this work (reached 360 mL CH4/g VS), but to achieve this, high concentration of sodium hydroxide (7% w/w) and thiourea (5.5% w/w) were used. Moreover, use of alkaline solutions require an extra step of neutralization prior to digestion and re-use of the alkali is not easy. Apart from this, alkaline treatments fail to recover lignin which can be further used for other processes.
2.4. Correlation between Combined Severity Factor (CSF) and the Methane Yield
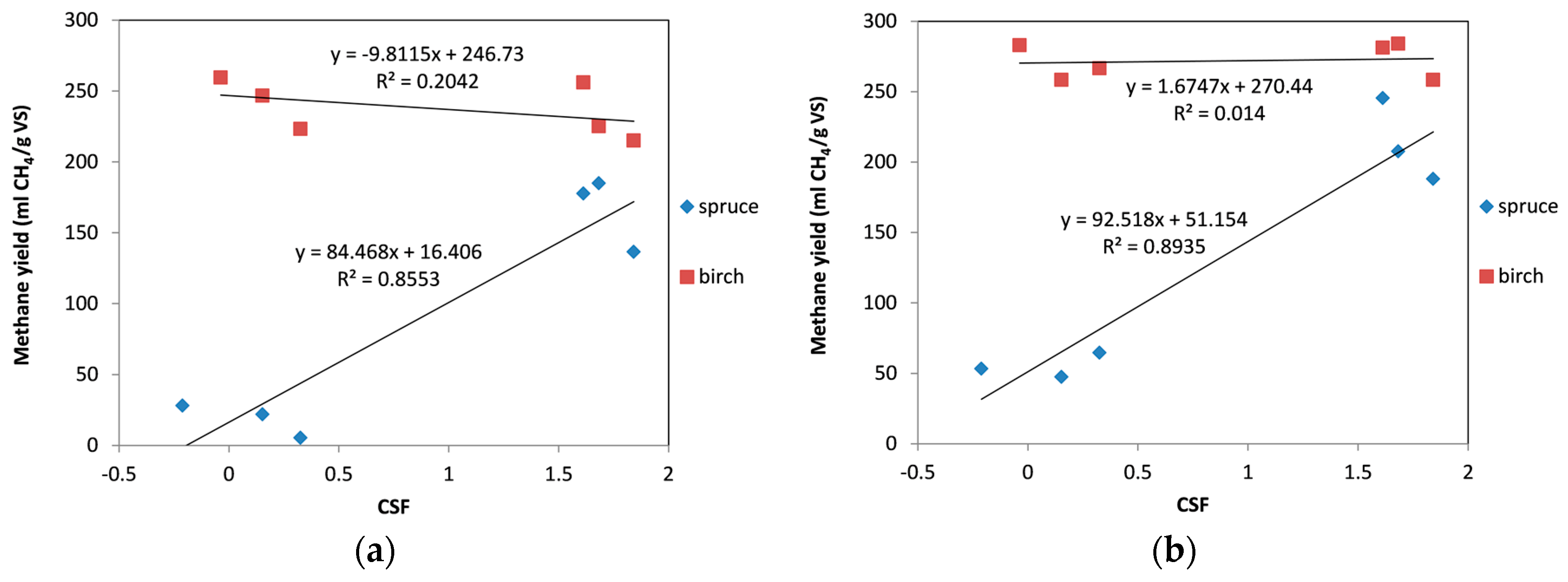
The effect of the operational conditions of the organosolv pretreatment expressed as CSF on the methane yields was also evaluated. Figure 5a shows the correlations between CSF and methane yield for both materials. There was a very good correlation between CSF and methane yield for spruce, and little or almost none for birch. In spruce, we could see a clear trend of increase in methane yield with increasing severity of pretreatment. Contrarily, for birch a slight decrease in the methane yield could be seen. This decrease was very minor, however, which indicates that even the less severe pretreatment conditions were adequate for the treatment of birch biomass. It can also be noted that for the same CSF, higher yields were obtained for birch than for spruce, as spruce is a softwood and more difficult to hydrolyze in comparison with hardwoods (i.e., birch) [60]. A similar correlation was observed between the CSF values and the methane yields when cellulolytic enzymes were included in the digestion (Figure 5b). A major difference, however, was that when enzymes were added during the digestion of birch, the CSF had no effect on the methane yield, with the results obtained to be identical in the whole range of tested CSF values.
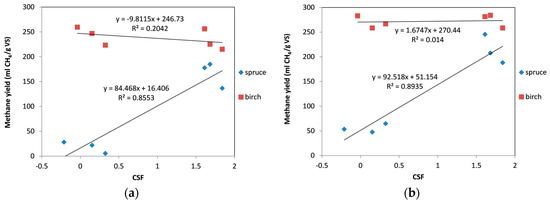
Figure 5.
Correlation between combined severity factor (CSF) and methane yield: (a) without enzymes; (b) with addition of enzymes.
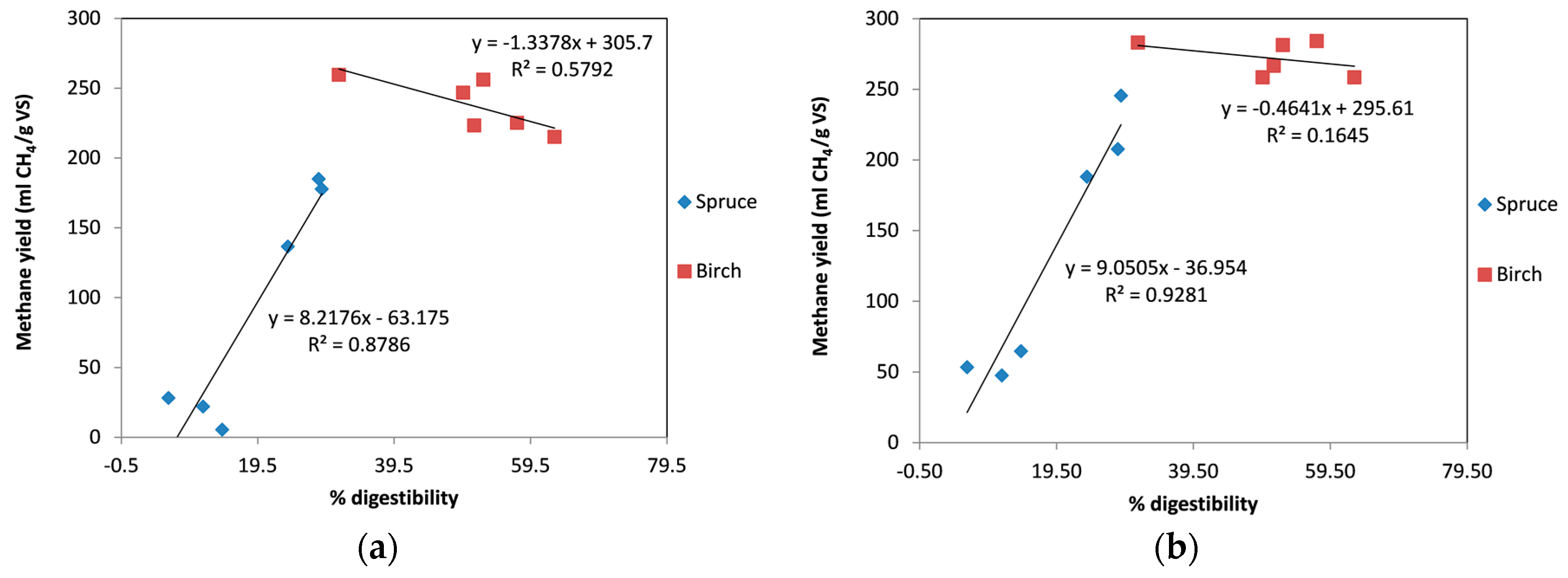
Previous studies have also found a correlation between methane yield and the yield of sugars during enzymatic saccharification trials [26,44]. The same correlation of the yield during enzymatic hydrolysis (expressed as cellulose conversion to glucose after 48 h of hydrolysis) with the methane yield was also observed during our work using spruce without the addition of enzymes (Figure 6a) and with the addition of enzymes (Figure 6b). This indicates that for a material such as spruce, instead of performing a time-consuming anaerobic digestion trial, it is possible to perform a 2-day enzymatic digestion trial and obtain a very good indication of the methane yield that can be obtained during anaerobic digestion. In contrast, for birch biomass there is no good correlation between enzymatic digestibility and methane yield. We even observed that increased enzymatic yields resulted in slightly reduced methane yields. Once again, addition of enzymes during the digestion gave similar results regarding methane yield, irrespective of differences in enzymatic digestibility.
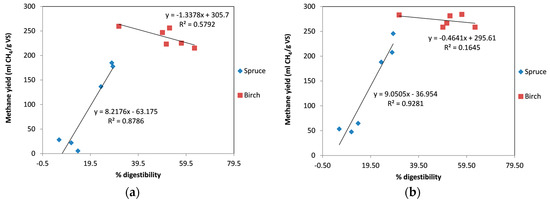
Figure 6.
Correlation between digestibility and methane yield: (a) without enzymes; (b) with addition of enzymes.
3. Materials and Methods
3.1. Feedstock, Inoculum, and Enzymes
Wood chips of Norway spruce and silver birch, from the northern part of Sweden, were used in the current work. The materials were air-dried, milled in a cutting mill (RETSCH SM300, Haan, Germany) to a size of less than 1 mm and stored in plastic bags at room temperature for further use. During anaerobic digestion, thermophilic anaerobic sludge was used as inoculum. The sludge was collected from a biogas-producing plant located in Boden, Sweden. The plant operates under thermophilic conditions (55 °C) for the co-digestion of food waste with sewage sludge.
The enzymes used for enzymatic treatment during methane production were the commercial enzyme solution Cellic® CTec2 from Novozymes A/S (Bagsærd, Denmark), with an enzyme activity of 238 FPU/mL and a protein concentration of 278 mg/mL, as measured with the Lowry protocol [63].
3.2. Pretreatment of Forest Material
Organosolv pretreatment was carried out in an air-heated multidigester system (AATO) with six 2.5-L batch autoclave reactors at the Biochemical Process Engineering laboratories (Division of Chemical Engineering, Luleå University of Technology) as previously described [46]. More specifically, milled forest biomass was loaded into the autoclave reactors and different ethanol concentrations (50% or 60% v/v) were added at a liquid-to-solid ratio of 10. Additionally, acid catalyst (H2SO4; 1% w/w of biomass) was added if required. The pretreatment took place at 182 °C, with a holding time of either 60 min or 103 min. At the end of the pretreatment, the materials were collected and filtered to separate the solid phase from the liquid phase and washed three times with distilled water to remove the inhibitors and residual ethanol. The solid phase was used for the biochemical methane potential (BMP) test.
3.3. Enzymatic Hydrolysis
Enzymatic hydrolysis trials were performed in 2-mL Eppendorf tubes containing 1.5 mL of solution with 5% w/w solids. The reactions were performed at pH 5 (50 mM citrate-phosphate buffer) with the addition of 0.02% sodium azide to prevent microbial contaminations. The enzymatic solution Cellic® CTec2 was added at a concentration equal to 10 mg enzyme/g solids and the incubation took place for 48 h at 50 °C and 1200 rpm in a ThermoMixer C (Eppendorf, Hamburg, Germany). Samples were withdrawn at 0, 24, and 48 h and were immediately stored at −20 °C. Before analysis, the samples were centrifuged to remove solids and the liquid was analyzed for sugar concentration using an HPLC apparatus. All the enzymatic hydrolysis trials were performed in duplicate.
3.4. Biochemical Methane Potential (BMP) Test
BMP test was performed in the Automatic Methane Potential Test System (AMPTS II) of Bioprocess Control AB (Lund, Sweden). The anaerobic digestion was conducted in 500-mL glass bottles, with a total sample weight (substrate and inoculum) of 400 g. The inoculum-to-substrate ratio (I/S ratio) in terms of volatile solids (VS) was 2. Before use, the sludge was filtered to remove large particles. A mixture of minerals and trace elements, with a composition that has been described elsewhere [54], was added to each flask at the start of the digestion. The glass bottles were sealed with rubber stoppers that had a metallic stirrer. These were connected to micro-motors to gently mix the sludge. Mixing of the sludge occurred at intervals of 10 min of mixing and 1 min of rest. Each incubation bottle was connected to a 100-mL glass bottle called a “CO2-absorption unit”, which contained approximately 80 mL of 3 M NaOH and thymolphthalein as pH indicator. This unit removed all other gases except methane. Finally, the volume of methane was measured with a gas-flow meter unit that was connected to the CO2-absorption unit.
All the anaerobic digestion trials were done in duplicate and control digestions were also included in each batch of experiments. Control digestions contained only anaerobic sludge in order to determine the production of methane from the organic material present in the microbial sludge. This amount of methane was then removed from the total amount of methane produced during the actual digestions. Finally, to evaluate the effect of the addition of cellulolytic enzymes on the methane yield, another series of experiments took place by adding cellulolytic enzymes at a concentration equivalent to 15 FPU/g VS. Control digestions with enzymes and sludge were also included in order to subtract the methane produced by the digestion of the enzymes from the overall yield.
3.5. Analytical Methods
Total solids (TS) were determined gravimetrically after drying the biomass in an oven at 110 °C overnight. Ash content was determined gravimetrically after burning the biomass in a furnance at 550 °C for 3 h. To determine volatile solids (VS), ash content was subtracted from the TS content. The values of TS and VS for the materials used during this work are presented in Table 1.

Table 1.
Volatile solids (VS) and total solids (TS) from pretreated spruce and birch.
The concentration of sugars during the enzymatic hydrolysis trials was determined with an Series 200 HPLC apparatus (Perkin-Elmer, Waltham, MA, USA) equipped with a Series 200 refractive index detector (Perkin-Elmer, Waltham, MA, USA) and an Aminex® HPX-87P column (BioRad, Hercules, CA, USA). The mobile phase was double-distilled H2O with a flow rate of 0.6 mL/min. During the analysis, the column was kept at 85 °C.
4. Conclusions
We have demonstrated that organosolv pretreatment can be a useful method for treatment of wood material (both spruce and birch) before using it for anaerobic digestion. The effect of different operational conditions of the organosolv pretreatment (duration time, ethanol content and addition of an acid catalyst) as well as the addition of cellulolytic enzymes on the methane yield were examined. Initially the effect of organosolv pretreatment in the enzymatic digestibility was examined where from spruce material it was found that with increasing CSF of the pretreatment up to a value of 1.61, the enzymatic digestibility was improved. On the other hand, for birch biomass the enzymatic digestibility improved up to a CSF value of 0.33.
In the subsequent anaerobic digestion trials, we found that a shorter duration of the pretreatment improved the methane yield from both spruce and birch. Addition of acid catalyst improved the methane yield from spruce, but did not affect the methane yield from birch. Finally, methane yields from spruce were higher with lower ethanol content, in contrast to birch where higher ethanol content was more beneficial. The highest yields obtained were 185 mL CH4/g VS from spruce and 259.9 mL CH4/g VS from birch. Addition of cellulolytic enzymes improved these yields to 266.6 mL CH4/g VS and 284.2 mL CH4/g VS, respectively. These results were high in comparison with other results reported in the literature, especially when only solid fraction is used for the digestion.
In the last part of this work, an attempt to obtain a correlation between the methane yields and either the CSF or the digestibility was performed. For the spruce, a very good correlation of the methane yields was obtained with both CSF and digestibility, indicating that instead of performing a time consuming anaerobic digestion trials, a 2-days enzymatic digestibility trial can provide with a very good picture of the potentials for the production of methane. On the other hand, the correlations obtained with the birch were poorer.
Acknowledgments
We thank Bio4Energy, a strategic research environment appointed by the Swedish government, for financial support.
Author Contributions
L.M., P.C. and U.R. conceived and designed the experiments; D.V. and L.M. performed the experiments; C.N. and D.V. pretreated the spruce and birch biomass; L.M. and C.N. analyzed the data; and D.V. drafted the manuscript. All the authors have read, corrected, and approved the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Demirbas, A. Political, economic and environmental impacts of biofuels: A review. Appl. Energy 2009, 86, S108–S117. [Google Scholar] [CrossRef]
- Höök, M.; Tang, X. Depletion of fossil fuels and anthropogenic climate change—A review. Energy Policy 2013, 52, 797–809. [Google Scholar] [CrossRef]
- Goldemberg, J. The promise of clean energy. Energy Policy 2006, 34, 2185–2190. [Google Scholar] [CrossRef]
- International Energy Agency. Key World Energy Statistics; International Education Association (IEA): Paris, France, 2016. [Google Scholar]
- Shafiee, S.; Topal, E. When will fossil fuel reserves be diminished? Energy Policy 2009, 37, 181–189. [Google Scholar] [CrossRef]
- Owen, N.A.; Inderwildi, O.R.; King, D.A. The status of conventional world oil reserves—Hype or cause for concern? Energy Policy 2010, 38, 4743–4749. [Google Scholar] [CrossRef]
- Weiland, P. Biogas production: Current state and perspectives. Appl. Microbiol. Biotechnol. 2010, 85, 849–860. [Google Scholar] [CrossRef] [PubMed]
- Romano, R.T.; Zhang, R.; Teter, S.; McGarvey, J.A. The effect of enzyme addition on anaerobic digestion of Jose Tall Wheat Grass. Bioresour. Technol. 2009, 100, 4564–4571. [Google Scholar] [CrossRef] [PubMed]
- Rasi, S.; Veijanen, A.; Rintala, J. Trace compounds of biogas from different biogas production plants. Energy 2007, 32, 1375–1380. [Google Scholar] [CrossRef]
- Holm-Nielsen, J.B.; Al Seadi, T.; Oleskowicz-Popiel, P. The future of anaerobic digestion and biogas utilization. Bioresour. Technol. 2009, 100, 5478–5484. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Park, S.Y.; Zhu, J. Solid-state anaerobic digestion for methane production from organic waste. Renew. Sustain. Energy Rev. 2011, 15, 821–826. [Google Scholar] [CrossRef]
- Börjesson, P.; Berglund, M. Environmental systems analysis of biogas systems—Part I: Fuel-cycle emissions. Biomass Bioenergy 2006, 30, 469–485. [Google Scholar] [CrossRef]
- Parawira, W. Enzyme research and applications in biotechnological intensification of biogas production. Crit. Rev. Biotechnol. 2012, 32, 172–186. [Google Scholar] [CrossRef] [PubMed]
- Wan, C.; Li, Y. Fungal pretreatment of lignocellulosic biomass. Biotechnol. Adv. 2012, 30, 1447–1457. [Google Scholar] [CrossRef] [PubMed]
- Taherzadeh, M.J.; Karimi, K. Pretreatment of lignocellulosic wastes to improve ethanol and biogas production: A review. Int. J. Mol. Sci. 2008, 9, 1621–1651. [Google Scholar] [CrossRef] [PubMed]
- Mancini, G.; Papirio, S.; Lens, P.N.L.; Esposito, G. Solvent Pretreatments of Lignocellulosic Materials to Enhance Biogas Production: A Review. Energy Fuels 2016, 30, 1892–1903. [Google Scholar] [CrossRef]
- Limayem, A.; Ricke, S.C. Lignocellulosic biomass for bioethanol production: Current perspectives, potential issues and future prospects. Prog. Energy Combust. Sci. 2012, 38, 449–467. [Google Scholar] [CrossRef]
- Kim, S.; Holtzapple, M.T. Effect of structural features on enzyme digestibility of corn stover. Bioresour. Technol. 2006, 97, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Matsakas, L.; Christakopoulos, P. Ethanol Production from Enzymatically Treated Dried Food Waste Using Enzymes Produced On-Site. Sustainability 2015, 7, 1446–1458. [Google Scholar] [CrossRef]
- Kumar, P.; Barrett, D.M.; Delwiche, M.J.; Stroeve, P. Methods for Pretreatment of Lignocellulosic Biomass for Efficient Hydrolysis and Biofuel Production. Ind. Eng. Chem. Res. 2009, 48, 3713–3729. [Google Scholar] [CrossRef]
- Sannigrahi, P.; Ragauskas, A.J. Fundamentals of Biomass Pretreatment by Fractionation. In Aqueous Pretreatment of Plant Biomass for Biological and Chemical Conversion to Fuels and Chemicals; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2013; pp. 201–222. [Google Scholar]
- Mosier, N.; Wyman, C.; Dale, B.; Elander, R.; Lee, Y.Y.; Holtzapple, M.; Ladisch, M. Features of promising technologies for pretreatment of lignocellulosic biomass. Bioresour. Technol. 2005, 96, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Xiros, C.; Topakas, E.; Christakopoulos, P. Hydrolysis and fermentation for cellulosic ethanol production. Wiley Interdiscip. Rev. Energy Environ. 2013, 2, 633–654. [Google Scholar] [CrossRef]
- Nakamura, Y.; Mtui, G. Anaerobic fermentation of woody biomass treated by various methods. Biotechnol. Bioprocess Eng. 2003, 8, 179–182. [Google Scholar] [CrossRef]
- Take, H.; Andou, Y.; Nakamura, Y.; Kobayashi, F.; Kurimoto, Y.; Kuwahara, M. Production of methane gas from Japanese cedar chips pretreated by various delignification methods. Biochem. Eng. J. 2006, 28, 30–35. [Google Scholar] [CrossRef]
- Horn, S.J.; Estevez, M.M.; Nielsen, H.K.; Linjordet, R.; Eijsink, V.G.H. Biogas production and saccharification of Salix pretreated at different steam explosion conditions. Bioresour. Technol. 2011, 102, 7932–7936. [Google Scholar] [CrossRef] [PubMed]
- Estevez, M.M.; Linjordet, R.; Morken, J. Effects of steam explosion and co-digestion in the methane production from Salix by mesophilic batch assays. Bioresour. Technol. 2012, 104, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Khoshnevisan, B.; Shafiei, M.; Rajaeifar, M.A.; Tabatabaei, M. Biogas and bioethanol production from pinewood pre-treated with steam explosion and N-methylmorpholine-N-oxide (NMMO): A comparative life cycle assessment approach. Energy 2016, 114, 935–950. [Google Scholar] [CrossRef]
- Yoshida, K.; Miyafuji, H.; Saka, S. Methane production from organic acids obtained by supercritical water treatment of Japanese beech. J. Wood Sci. 2010, 56, 160–165. [Google Scholar] [CrossRef]
- Teghammar, A.; Karimi, K.; Sárvári Horváth, I.; Taherzadeh, M.J. Enhanced biogas production from rice straw, triticale straw and softwood spruce by NMMO pretreatment. Biomass Bioenergy 2012, 36, 116–120. [Google Scholar] [CrossRef]
- Kabir, M.M.; Niklasson, C.; Taherzadeh, M.J.; Horváth, I.S. Biogas production from lignocelluloses by N-methylmorpholine-N-oxide (NMMO) pretreatment: Effects of recovery and reuse of NMMO. Bioresour. Technol. 2014, 161, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Kabir, M.M.; Rajendran, K.; Taherzadeh, M.J.; Sárvári Horváth, I. Experimental and economical evaluation of bioconversion of forest residues to biogas using organosolv pretreatment. Bioresour. Technol. 2015, 178, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Mirmohamadsadeghi, S.; Karimi, K.; Zamani, A.; Amiri, H.; Horváth, I.S. Enhanced solid-state biogas production from lignocellulosic biomass by organosolv pretreatment. Biomed. Res. Int. 2014, 2014, 350414. [Google Scholar] [CrossRef] [PubMed]
- McDonough, T. The chemistry of organosolv delignification. Tappi J. 1993, 76, 186–193. [Google Scholar]
- Zhao, X.; Cheng, K.; Liu, D. Organosolv pretreatment of lignocellulosic biomass for enzymatic hydrolysis. Appl. Microbiol. Biotechnol. 2009, 82, 815–827. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Cheng, J. Hydrolysis of lignocellulosic materials for ethanol production: A review. Bioresour. Technol. 2002, 83, 1–11. [Google Scholar] [CrossRef]
- Agbor, V.B.; Cicek, N.; Sparling, R.; Berlin, A.; Levin, D.B. Biomass pretreatment: Fundamentals toward application. Biotechnol. Adv. 2011, 29, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Harrison, M.D.; Rackemann, D.W.; Doherty, W.O.S.; O’Hara, I.M. Organosolv pretreatment of plant biomass for enhanced enzymatic saccharification. Green Chem. 2016, 18, 360–381. [Google Scholar] [CrossRef]
- Eggeman, T.; Elander, R.T. Process and economic analysis of pretreatment technologies. Bioresour. Technol. 2005, 96, 2019–2025. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Saito, T. Lignin-derived advanced carbon materials. ChemSusChem 2015, 8, 3941–3958. [Google Scholar] [CrossRef] [PubMed]
- Mamman, A.S.; Lee, J.M.; Kim, Y.C.; Hwang, I.T.; Park, N.J.; Hwang, Y.K.; Chang, J.S.; Hwang, J.S. Furfural: Hemicellulose/xylose-derived biochemical. Biofuels Bioprod. Biorefin. 2008, 2, 438–454. [Google Scholar] [CrossRef]
- Saha, B.C. Hemicellulose bioconversion. J. Ind. Microbiol. Biotechnol. 2003, 30, 279–291. [Google Scholar] [CrossRef] [PubMed]
- Matsakas, L.; Rova, U.; Christakopoulos, P. Strategies for Enhanced Biogas Generation through Anaerobic Digestion of Forest Material—An Overview. BioResources 2016, 11, 1–18. [Google Scholar]
- Vivekanand, V.; Olsen, E.F.; Eijsink, V.G.H.; Horn, S.J. Effect of different steam explosion conditions on methane potential and enzymatic saccharification of birch. Bioresour. Technol. 2013, 127, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Bansal, P.; Realff, M.J.; Bommarius, A.S. SO2-catalyzed steam explosion: The effects of different severity on digestibility, accessibility, and crystallinity of lignocellulosic biomass. Biotechnol. Prog. 2013, 29, 909–916. [Google Scholar] [CrossRef] [PubMed]
- Nitsos, C.; Stoklosa, R.; Karnaouri, A.; Vörös, D.; Lange, H.; Hodge, D.; Crestini, C.; Rova, U.; Christakopoulos, P. Isolation and Characterization of Organosolv and Alkaline Lignins from Hardwood and Softwood Biomass. ACS Sustain. Chem. Eng. 2016, 4, 5181–5193. [Google Scholar] [CrossRef]
- Pan, X.; Xie, D.; Yu, R.W.; Lam, D.; Saddler, J.N. Pretreatment of lodgepole pine killed by mountain pine beetle using the ethanol organosolv process: Fractionation and process optimization. Ind. Eng. Chem. Res. 2007, 46, 2609–2617. [Google Scholar] [CrossRef]
- Araque, E.; Parra, C.; Freer, J.; Contreras, D.; Rodríguez, J.; Mendonça, R.; Baeza, J. Evaluation of organosolv pretreatment for the conversion of Pinus radiata D. Don to ethanol. Enzym. Microb. Technol. 2008, 43, 214–219. [Google Scholar] [CrossRef]
- Park, N.; Kim, H.-Y.; Koo, B.-W.; Yeo, H.; Choi, I.-G. Organosolv pretreatment with various catalysts for enhancing enzymatic hydrolysis of pitch pine (Pinus rigida). Bioresour. Technol. 2010, 101, 7046–7053. [Google Scholar] [CrossRef] [PubMed]
- Saini, J.K.; Patel, A.K.; Adsul, M. Cellulase adsorption on lignin: A roadblock for economic hydrolysis of biomass. Renew. Energy 2016, 98, 29–42. [Google Scholar] [CrossRef]
- Li, Y.; Sun, Z.; Ge, X.; Zhang, J. Effects of lignin and surfactant on adsorption and hydrolysis of cellulases on cellulose. Biotechnol. Biofuels 2016, 9, 20. [Google Scholar] [CrossRef] [PubMed]
- Matsakas, L.; Rova, U.; Christakopoulos, P. Sequential parametric optimization of methane production from different sources of forest raw material. Front. Microbiol. 2015, 6, 1163. [Google Scholar] [CrossRef] [PubMed]
- Sonakya, V.; Raizada, N.; Kalia, V.C. Microbial and enzymatic improvement of anaerobic digestion of waste biomass. Biotechnol. Lett. 2001, 23, 1463–1466. [Google Scholar] [CrossRef]
- Antonopoulou, G.; Lyberatos, G. Effect of Pretreatment of Sweet Sorghum Biomass on Methane Generation. Waste Biomass Valoriz. 2013, 4, 583–591. [Google Scholar] [CrossRef]
- Pan, X.; Xie, D.; Yu, R.W.; Saddler, J.N. The bioconversion of mountain pine beetle-killed lodgepole pine to fuel ethanol using the organosolv process. Biotechnol. Bioeng. 2008, 101, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Van Heiningen, A.R.P. The swelling of pulp fibers derived from the ethanol-based organosolv process. Tappi J. 1997, 80, 211–213. [Google Scholar]
- Pan, X.; Xie, D.; Kang, K.-Y.; Yoon, S.-L.; Saddler, J.N. Effect of organosolv ethanol pretreatment variables on physical characteristics of hybrid poplar substrates. Appl. Biochem. Biotechnol. 2007, 137–140, 367–377. [Google Scholar] [CrossRef] [PubMed]
- Janzon, R.; Schütt, F.; Oldenburg, S.; Fischer, E.; Körner, I.; Saake, B. Steam pretreatment of spruce forest residues: Optimal conditions for biogas production and enzymatic hydrolysis. Carbohydr. Polym. 2014, 100, 202–210. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Chen, L.; Yuan, K.; Huang, H.; Yan, Z. Ionic liquid pretreatment to enhance the anaerobic digestion of lignocellulosic biomass. Bioresour. Technol. 2013, 150, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Mirahmadi, K.; Kabir, M.M.; Jeihanipour, A.; Karimi, K.; Taherzadeh, M. Alkaline pretreatment of spruce and birch to improve bioethanol and biogas production. BioResources 2010, 5, 928–938. [Google Scholar]
- Mohsenzadeh, A.; Jeihanipour, A.; Karimi, K.; Taherzadeh, M.J. Alkali pretreatment of softwood spruce and hardwood birch by NaOH/thiourea, NaOH/urea, NaOH/urea/thiourea, and NaOH/PEG to improve ethanol and biogas production. J. Chem. Technol. Biotechnol. 2012, 87, 1209–1214. [Google Scholar] [CrossRef]
- Goshadrou, A.; Karimi, K.; Taherzadeh, M.J. Ethanol and biogas production from birch by NMMO pretreatment. Biomass Bioenergy 2013, 49, 95–101. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).