Abstract
In this work, char gasification of two coals (i.e., Shenfu bituminous coal and Zunyi anthracite) and a petroleum coke under a steam and CO2 mixture (steam/CO2 partial pressures, 0.025–0.075 MPa; total pressures, 0.100 MPa) and CO2/steam chemisorption of char samples were conducted in a Thermogravimetric Analyzer (TGA). Two conventional kinetic models exhibited difficulties in exactly fitting the experimental data of char–steam–CO2 gasification. Hence, a modified model based on Langmuir–Hinshelwood model and assuming that char–CO2 and char–steam reactions partially shared active sites was proposed and had indicated high accuracy for estimating the interactions in char–steam–CO2 reaction. Moreover, it was found that two new model parameters (respectively characterized as the amount ratio of shared active sites to total active sites in char–CO2 and char–steam reactions) in the modified model hardly varied with gasification conditions, and the results of chemisorption indicate that these two new model parameters mainly depended on the carbon active sites in char samples.
1. Introduction
Gasification is an important technology for the clean and efficient utilization of coal, petroleum coke, and other solid fuels, and is available for large-scale industry due to its high efficiency, high production intensity, and near-zero pollution emission [1,2]. Fundamental research of gasification processes identified the rate of char gasification as a significant factor controlling gasification behaviors. This was mainly attributed to the relatively slow kinetics of char–CO2 reactions and char–steam reactions under gasification conditions [3]. Therefore, it is important to make a clear understanding of the gasification kinetics of char–CO2 and char–steam.
Many researchers [4,5,6,7] have developed reaction models of char–CO2 and char–steam respectively to determine their reaction rates. Moreover, the kinetics of char–CO2 gasification and char–steam gasification have also been studied in detail [8,9,10,11]. However, these models and studies, carried out in the system with a sole gasifying agent, could hardly be applied in systems where char–CO2 and char–steam reactions occur simultaneously. Under realistic gasification conditions, chars are reacted with the gasifying agent mixtures consisting of steam and CO2. Therefore, the researchers in [12] investigated the gasification reactivities of metallurgical coke in a mixture of steam and CO2, but it was hard to analyze the intrinsic char–CO2–steam reaction because the particle size of the tested samples was large (3–6 mm), which resulted in the effect of internal diffusion. Muhlen et al. [13] have also studied the mechanism of coal char gasification under a mixture of steam and CO2, and the proposed empirical approach could well describe the effect of the carbon conversion degree on the gasification rate.
Recently, some researchers [14,15,16,17,18] came to investigate interactions in the char–CO2–steam reaction using the Langmuir–Hinshelwood (L–H) model based on the theory of absorption and desorption. Roberts and Harris [14] found that char–CO2 and char–steam reactions occur at the common active sites and indicated that steam and CO2 competed for active sites of chars. However, some other researchers proposed an opposite view. Both Huang et al. [15] and Everson et al. [16] thought that the char–CO2 reaction and char–steam reaction proceeded at separate active sites and the overall gasification rate should be equal to the sum of the char–CO2 reaction rate and the char–steam reaction rate. Li et al. [17] also pointed out that the reaction mechanism of lignite char gasification was in accordance with the separate reactive site reaction mechanism under lower gasification pressures, whereas it was superior to the common reactive site reaction mechanism when gasification pressures further increased. On the other hand, Chen et al. [18] found that the overall gasification rate is not equal to the sum of the char–CO2 reaction rate and char–steam reaction rate, but there was no evidence proving that the two gasifying agents competed for active sites. An L–H model with two dimensionless parameters was proposed by Umemoto et al. [19] to evaluate the rate of char–CO2–steam gasification, and could estimate interactions more accurately than other models. However, the mechanism of the two proposed parameters is unclear.
Accordingly, a modified kinetic model based on the concept of active sites was proposed in this study to evaluate the interaction in char–CO2–steam gasification, and it was validated by char–CO2–steam gasification experiments of carbonaceous materials (two coals of different ranks and a petroleum coke) using a Thermogravimetric Analyzer (TGA). Additionally, CO2 and steam chemisorption of chars were also conducted in a TGA to measure chemisorption quantities so as to explore the correlation between kinetic parameters of the modified model and chemisorption quantities. This work can provide not only theoretical basis for simulation but also scientific basis for gasification industry.
2. Materials and Methods
2.1. Char Preparation
Two typical gasification coals (Shenfu bituminous coal (SF) and Zunyi anthracite (ZY) from China) and a petroleum coke (PC) from Sinopec Shanghai Gaoqiao Petrochemical Co., Ltd. (Shanghai, China) were chosen as raw materials in this study. During char preparation, all pulverized raw samples were heated at 25 °C/min to 850 °C and held 30 min under the high purity nitrogen atmosphere in a fixed bed reactor. After devolatilization, the prepared chars with particle sizes of <40 μm were used for further analysis. The proximate analysis, ultimate analysis, and specific surface area of char samples are listed in Table 1.

Table 1.
Properties of tested char samples.
2.2. Isothermal Gasification Tests
The tests of isothermal char gasification were carried out using a TGA (NETZSCH STA449-F3). In all cases, approximately 5 mg of char particles was placed in an alumina crucible and heated at 25 °C/min to the prescribed temperature (850–950 °C) under high purity nitrogen atmosphere (80 mL/min). Then, nitrogen was switched to gasifying agents to initiate the isothermal gasification. In char gasification tests, nitrogen was used to adjust the concentration of the gasifying agents. For char gasification with sole gasifying agent (steam or CO2), the partial pressures of the gasifying agents varied from 0.025 to 0.100 MPa. For char gasification with a mixture of steam and CO2, the partial pressures of steam and CO2 varied from 0.025 to 0.075 MPa, and the total pressure was always 0.100 MPa. The effects of external and internal diffusion were eliminated by previous tests [20].
The carbon conversion was expressed by the following equation [21]:
where w0 is the initial mass of char (wt. %); wt is the sample mass at gasification time of t (wt. %); and wash is the final sample mass at the end of gasification.
Gasification rate was defined as following:
2.3. CO2/Steam Chemisorption
CO2/steam chemisorption of chars was fulfilled by TGA, and the test procedures have been reported in previous literature [22,23,24]. Approximately 15 mg of char particles was first heated at 25 °C/min to 850 °C and then held for 30 min under a continuous high purity nitrogen flow, in order to remove any impurity which could be adsorbed during CO2 or steam chemisorption. Then, the temperature was decreased to 300 °C and stabilized for 10 min. Whereafter, nitrogen was switched to pure CO2 or steam to start the chemisorption test. After another 30 min of adsorption, pure CO2 or steam was finally switched to high purity nitrogen, and the char samples were degassed for 30 min, in order to remove all weakly chemisorbed CO2 or steam molecules. The flow rates of steam, CO2, and high-purity nitrogen were all set as 80 mL/min.
As a representative, the steam chemisorption curve of PC char is shown in Figure 1. Three sets of chemisorption data can be observed in Figure 1. The first one is the total quantity of chemisorption (Ct). The second one is the quantity of weak chemisorption (Cw). The weak chemisorption of steam or CO2 was desorbed from char surface during the process of degassing. The last one is the quantity of strong chemisorption (Cs). Strong chemisorption of steam or CO2 still stayed on the surface of the char after degassing.
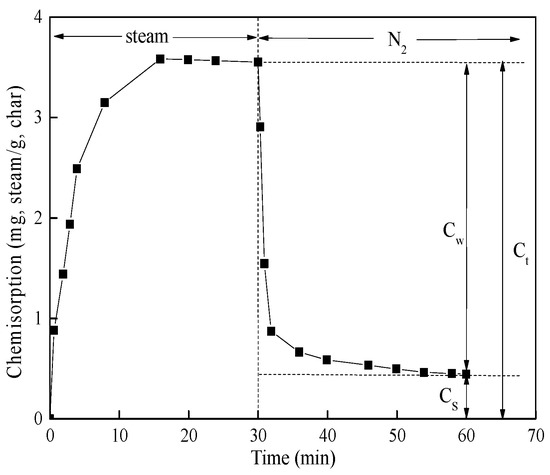
Figure 1.
Steam chemisorption of petroleum coke (PC) char.
3. Results and Discussion
3.1. Choice of Kinetic Model of Char–CO2–Steam Gasification
The mechanism of the char–CO2 reaction is widely accepted by following description:
Based on the Langmuir–Hinshelwood (L–H) model derived from the theory of absorption and desorption, the char–CO2 reaction rate can be expressed as Equation (5):
The equation can also be written as:
Here, Ka, Kb, and Kc can be defined as following:
According to Equation (5), Ea = E1, Eb = E2 − E3, Ec = E1 − E3, Aa = A1, Ab = A2/A3, Ac = A1/A3. Similarly, the char–steam reaction rate can be expressed as Equation (8):
In this study, chars were gasified with a steam–CO2 mixture, so the mechanism of the char–steam–CO2 reaction should be defined. Interactions in a char–steam–CO2 reaction have been studied recently [14,16,19], and there were two kinds of mechanisms accepted by researchers. The first assumed that char–CO2 and char–steam reactions occur at separate active sites (Model A in Figure 2a). Based on this assumption, the overall reaction rate was equal to the sum of rates of these two reactions and can be described as following:
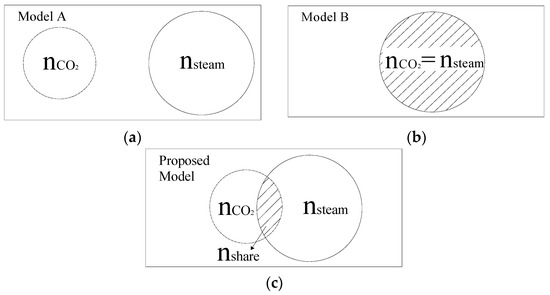
Figure 2.
Models of char–CO2–steam gasification: (a) Model A; (b) Model B; (c) Modified model.
The second mechanism assumed that these two reactions occur at common active sites (Model B in Figure 2b). This means that these two reactions compete for the same active sites, so the overall reaction rate was slower than the sum of reaction rates of these two reactions and can be described as following:
In general, a CO2 molecule can enter into pores with sizes larger than 1.5 nm, while a steam molecule can enter into pores with sizes larger than 0.6 nm. This led to a phenomenon where some active sites existed in pores with sizes smaller than 1.5 nm and could only be occupied by steam molecules. In addition, the catalytic effect of the inherent mineral on the char–CO2 reaction was quite different from that on the char–steam reaction [25]. Based on these reasons, it was conjectured that char–CO2 and char–steam reactions may share active sites partially, and the modified model was proposed as shown in Figure 2c. Some new parameters were introduced to quantify the amount of shared active sites for char–steam and char–CO2 reactions. They were defined as following:
Here, a and b are dimensionless parameters, representing the ratio of the amount of shared active sites to the total amount of active sites for the char–CO2 reaction and char–steam reaction, respectively. nshare is the amount of shared active sites for the char–steam and char–CO2 reactions. nCO2 and nsteam represent the total amount of active sites for the char–CO2 reaction and char–steam reaction, respectively.
Based on the assumption and these parameters, the overall reaction rate of the char–steam–CO2 reaction can be derived as:
It was noteworthy that the proposed model (Equation (13)) was the same as Model A (Equation (9)) in the case of a = b = 0 (nshare = 0), while the proposed model was the same as Model B (Equation (10)) in the case of a =b = 1 (nshare = nCO2 = nsteam).
3.2. Validation of the Modified Model
3.2.1. Kinetic Parameters of Char Gasification with Sole Gasifying Agent
In this study, the initial gasification rate (r0) was used as a representative. The concentrations or partial pressures of H2 and CO were very low and can be neglected at the initial stage of gasification. Therefore, PH2 and PCO were not shown in this study.
Figure 3 shows the initial rate of ZY char gasification with a sole gasifying agent as a function of the partial pressure of the gasifying agent (steam/CO2) and gasification temperature. It was evident that the initial gasification rate of ZY char depended strongly on temperature and steam/CO2 partial pressure. The initial gasification rate of ZY char increased as the temperature and steam/CO2 partial pressure increased. Moreover, it is shown in Figure 3 that the L–H model (the fitted line) fitted the experimental data well. In addition, the rate of steam gasification was faster than that of CO2 gasification when the gasification temperature was fixed. These phenomena were also observed for the other two chars. The kinetic parameters (Ki, Ai, Ei) were determined by the L–H model and the results are listed in Table 2. The activation energies of tested char samples were similar to those reported in the literature [15,16].
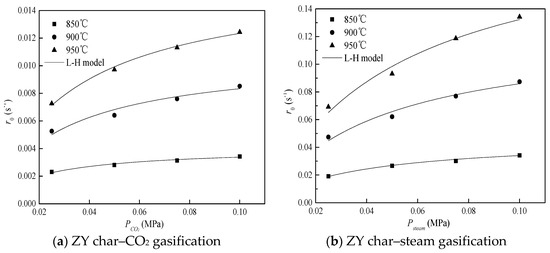
Figure 3.
Effect of steam–CO2 partial pressure on gasification rate.

Table 2.
Kinetic parameters of char–CO2 and char–steam reactions.
3.2.2. Kinetic Parameters of Char Gasification with Gasifying Agent Mixture
Initial rates of SF char gasification with a mixture of steam and CO2 are presented in Figure 4.
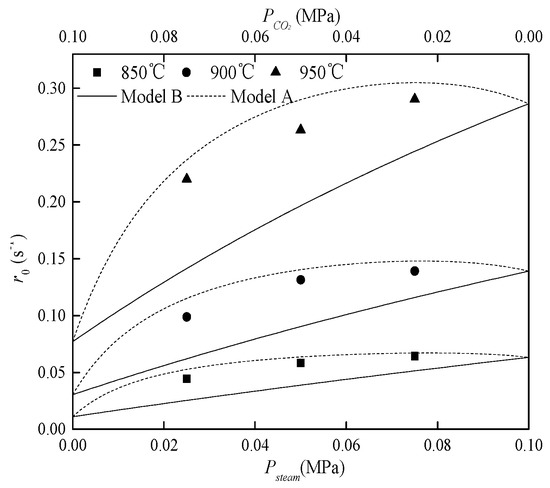
Figure 4.
Comparison of experimental data and two conventional models.
In each experiment, the total system pressure was 0.100 MPa. The initial gasification rate of SF char increased as steam partial pressure increased or as CO2 partial pressure decreased. In addition, the measured gasification rates were always a little lower than the gasification rates predicted by Model A and much higher than the gasification rates predicted by Model B. This means that char–CO2 and char–steam reactions neither absolutely occur at separate active sites nor absolutely occur at common active sites. Thus, it can be inferred that char–CO2 and char–steam reactions share active sites partially.
In order to evaluate the interactions by the char–CO2–steam reaction accurately, the modified model was employed. Comparisons between experimental data and predictions by the modified model are shown in Figure 5.
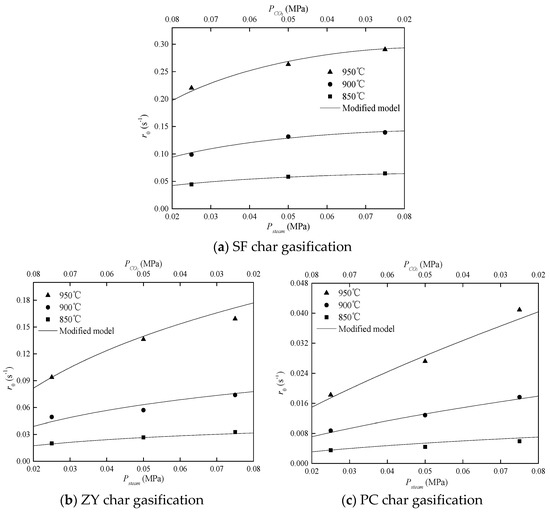
Figure 5.
Comparison of experimental data and the modified model.
For these three chars, the good agreement between experimental data and predictions confirmed the validity of the modified model. Parameters [a,b] were obtained by the fitting results and were equal to [0.47,0.25], [0.83,0.21], and [1.00,0.15] for SF char, ZY char, and PC char, respectively. The results shows that parameters [a,b] were constants and hardly varied with gasification temperature. For PC char, parameter a was equal to 1, but parameter b was not. This means that all active sites for char–CO2 gasification were shared by steam gasification, but many active sites for char–steam gasification were not shared by CO2 gasification. It is also noteworthy that the value of parameter a was always higher than that of parameter b for each char. It can be inferred that the total amount of active sites for steam gasification was larger than that for CO2 gasification, based on the assumption of Equations (11) and (12). This result can also explain why rates of steam gasification were always faster than those of CO2 gasification.
3.3. Correlations between Chemisorption Quantity and Model Parameters [a,b]
Indeed, active sites play a pivotal role in gasification [26,27], so an experimental method of chemisorption was proposed to quantity the amount of active sites in previous work [23]. Molina et al. [23] found that Cw was related to the presence of the organic components (carbon active sites) of the char, and Cs was related to the presence of the inorganic matter (catalytic active sites) of the char. As shown in Figure 1, Cw, Cs, and Ct can be obtained by the chemisorption experiment. Table 3 lists the chemisorption parameters of the tested chars. For each char, the value of Cs was higher than that of Cw for CO2 chemisorption, and the opposite trend was observed for steam chemisorption. It was indicated that the catalytic (mineral) matter possessed strong ability to withhold CO2 molecules. In addition, for all these chars, the values of Ct and Cw for steam chemisorption were always higher than those for CO2 chemisorption. This means that the total quantity of active sites and the quantity of carbon active sites for char–steam reactions were always more than those for char–CO2 reactions. This result was in accordance with the result obtained by the modified model.

Table 3.
Chemisorption parameters of tested chars.
In order to further study the correlation between chemisorption quantity and parameters [a,b], an equation was derived from the assumption of Equations (11) and (12). It can be expressed by following.
According to this equation, the ratio of the total amount of active sites for steam gasification to that for CO2 gasification can be calculated. The ratio values for SF char, ZY char, and PC char were 1.88, 3.95, and 6.67, respectively. The ratio of Cw of steam chemisorption to that of CO2 chemisorption can also be seen in Table 3. The result shows that values of Cw(H2O)/Cw(CO2) for SF char, ZY char, and PC char were 1.84, 4.39, and 7.03, respectively. These values were nearly the same as the values of a/b. However, the values of Cs(H2O)/Cs(CO2) or Ct(H2O)/Ct(CO2) were absolutely not equal to those of a/b. This phenomenon indicates that parameters [a,b] depended on the presence of the organic components (carbon active sites) of the char and showed little relationship with others factors, including the gasification process and conditions. Based on the principle of gasification, all reactions were initiated at the carbon surface. In addition, carbon active sites consisted of three aspects: nascent sites attached to the aromatic clusters, carbon atoms bonded to heteroatoms, and the edge carbon atoms [24], which were all chemically unstable and could easily be reacted with gasifying agents. This indicates that the gasifying agents can easily occupy the carbon active sites and react with the char on the carbon active sites. Hence, it was concluded that the kinetic parameters [a,b] mainly depended on the carbon active sites of the char. Moreover, the results also validate the modified model proposed in this study.
4. Conclusions
The gasification of three different chars under a mixture of steam and CO2 was carried out using a TGA. Initial gasification rates of these three chars all increased as steam partial pressure increased or as CO2 partial pressure decreased. Two conventional gasification models were used to fit the experimental data but showed great difficulties in exactly describing the interactions in the char–steam–CO2 reaction. A modified model was proposed based on shared active sites and can exactly predict the gasification rates of these three char gasification with a mixture of steam and CO2. The results indicate that char–CO2 and char–steam reactions shared active sites partially and also show that two new parameters in the modified model were constants and did not vary with gasification conditions. In addition, the results from chemisorption tests show that these two parameters just depended on the carbon active sites of the char and thus validated the modified model.
Acknowledgments
This work has been supported by the National Natural Science Foundation of China (21376081; 21676091).
Author Contributions
Xia Liu and Juntao Wei designed the experiments and prepared the manuscript; Wei Huo performed the experiments and analyzed the data; Guangsuo Yu led the project and research.
Conflicts of Interest
The authors declare no conflict of interest.
Nomenclature
| Ai | frequency factor for L–H model, s−1MPa−1 (I = a,d), MPa−1 (I = b,c,e,f) |
| A | parameter for the modified model, dimensionless |
| b | parameter for the modified model, dimensionless |
| Cs | the quantity of strong chemisorption, mg/mg-char |
| Ct | the total quantity of chemisorption, mg/mg-char |
| Cw | the quantity of weak chemisorption, mg/mg-char |
| Ei | activation energy for L–H model, kJ/mol |
| Ki | adsorption constant for L–H model, s−1MPa−1 (I = a,d), MPa−1 (I = b,c,e,f) |
| nCO2 | the total amount of active sites for char–CO2 reaction |
| nshare | the amount of shared active sites for char–steam and char–CO2 reactions |
| nsteam | the total amount of active sites for char–steam reaction |
| P | the partial pressure of H2/CO/steam/CO2, MPa |
| r0 | initial gasification rate, s−1 |
| T | the thermodynamic temperature, K |
| w0 | the initial mass of char, wt. % |
| wash | the sample mass at the end of char gasification, wt. % |
| wt | the sample mass at the gasification time of t, wt. % |
| x | carbon conversion |
References
- Kumar, A.; Jones, D.D.; Hanna, M.A. Thermochemical biomass gasification: A review of the current status of the technology. Energies 2009, 2, 556–581. [Google Scholar] [CrossRef]
- Zaccariello, L.; Mastellone, M.L. Fluidized-bed gasification of plastic waste, wood, and their blends with coal. Energies 2015, 8, 8052–8068. [Google Scholar] [CrossRef]
- Roberts, D.G.; Hodge, E.M.; Harris, D.J.; Stubington, J.F. Kinetics of Char Gasification with CO2 under Regime II Conditions: Effects of Temperature, Reactant, and Total Pressure. Energy Fuels 2010, 24, 5300–5308. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Perlmutter, D.D. A random pore model for fluid-solid reactions: I. Isothermal, kinetic control. AIChE J. 1980, 26, 379–385. [Google Scholar] [CrossRef]
- Bhatia, S.K.; Perlmutter, D.D. A random pore model for fluid-solid reactions: II. Diffusion and transport effects. AIChE J. 1981, 27, 247–254. [Google Scholar] [CrossRef]
- Gavals, G.R. A random capillary model with application to char gasification at chemically controlled rates. AIChE J. 1980, 26, 577–585. [Google Scholar] [CrossRef]
- Wen, C.Y. Noncatalytic heterogeneous solid-fluid reaction models. Ind. Eng. Chem. 1968, 60, 34–54. [Google Scholar] [CrossRef]
- Bliek, A.; Lont, J.C.; Van Swaaij, W.P.M. Gasification of coal-derived chars in synthesis gas mixtures under intraparticle mass-transfer-controlled conditions. Chem. Eng. Sci. 1986, 41, 1895–1909. [Google Scholar] [CrossRef]
- Kirtania, K.; Joshua, J.; Kassim, M.A.; Bhattacharya, S. Comparison of CO2 and steam gasification reactivity of algal and woody biomass chars. Fuel Process. Technol. 2014, 117, 44–52. [Google Scholar] [CrossRef]
- Ren, L.; Yang, J.; Gao, F.; Yan, J. Laboratory study on gasification reactivity of coals and petcokes in CO2/steam at high temperatures. Energy Fuels 2013, 27, 5054–5068. [Google Scholar] [CrossRef]
- Jayaraman, K.; Gökalp, I.; Jeyakumar, S. Estimation of synergetic effects of CO2 in high ash coal-char steam gasification. Appl. Therm. Eng. 2017, 110, 991–998. [Google Scholar] [CrossRef]
- Koba, K.; Ida, S. Gasification reactivities of metallurgical cokes with carbon dioxide, steam and their mixtures. Fuel 1980, 59, 59–63. [Google Scholar] [CrossRef]
- Muhlen, H.J.; Van Heek, K.H.; Juntgen, H. Kinetic studies of steam gasification of char in the presence of H2, CO2 and CO. Fuel 1985, 64, 944–949. [Google Scholar] [CrossRef]
- Roberts, D.G.; Harris, D.J. Char gasification in the mixtures of CO2 and H2O: Competition and inhibition. Fuel 2007, 86, 2672–2678. [Google Scholar] [CrossRef]
- Huang, Z.M.; Zhang, J.S.; Zhao, Y.; Zhang, H.; Yue, G.X.; Suda, T.; Narukawa, M. Kinetic studies of char gasification by steam and CO2 in the presence of H2 and CO. Fuel Process. Technol. 2010, 91, 843–847. [Google Scholar] [CrossRef]
- Everson, R.C.; Neomagus, H.W.J.P.; Kasaini, H.; Njapha, D. Reaction kinetics of pulverized coal-chars derived from inertinite-rich coal discards: Gasification with carbon dioxide and steam. Fuel 2006, 85, 1076–1082. [Google Scholar] [CrossRef]
- Li, F.H.; Yan, Q.X.; Huang, J.J.; Zhao, J.T.; Fang, Y.T.; Wang, J.F. Lignite-char gasification mechanism in mixed atmospheres of steam and CO2 at different pressures. Fuel Process. Technol. 2015, 138, 555–563. [Google Scholar] [CrossRef]
- Chen, C.; Wang, J.; Liu, W.; Zhang, S.; Yin, J.S.; Luo, G.Q.; Yao, H. Effect of pyrolysis conditions on the char gasification with the mixtures of CO2 and H2O. Proc. Combust. Inst. 2013, 34, 2453–2460. [Google Scholar] [CrossRef]
- Umemoto, S.; Kajitani, S.; Hara, S. Modeling of coal char gasification in coexistence of CO2 and H2O considering sharing of active sites. Fuel 2013, 103, 14–21. [Google Scholar] [CrossRef]
- Wei, J.T.; Guo, Q.H.; Ding, L.; Yoshikawa, K.; Yu, G.S. Synergy mechanism analysis of petroleum coke and municipal solid waste (MSW)-derived hydrochar co-gasification. Appl. Energy 2017, 206, 1354–1363. [Google Scholar] [CrossRef]
- Guizani, C.; Jeguirim, M.; Valin, S.; Limousy, L.; Salvador, S. Biomass chars: The effects of pyrolysis conditions on their morphology, structure, chemical properties and reactivity. Energies 2017, 10, 796. [Google Scholar] [CrossRef]
- Jing, X.L.; Wang, Z.Q.; Zhang, Q.; Yu, Z.L.; Li, C.Y.; Huang, J.J.; Fang, Y.T. Evaluation of CO2 gasification reactivity of different coal rank chars by physicochemical properties. Energy Fuels 2013, 27, 7287–7293. [Google Scholar] [CrossRef]
- Molina, A.; Montoya, A.; Mondragon, F. CO2 strong chemisorption as an estimate of coal char gasification reactivity. Fuel 1999, 78, 971–977. [Google Scholar] [CrossRef]
- Xu, K.; Hu, S.; Su, S.; Xu, C.F.; Sun, L.S.; Shuai, C.; Jiang, L.; Xiang, J. Study on char surface active sites and their relationship to gasification reactivity. Energy Fuels 2013, 27, 118–125. [Google Scholar] [CrossRef]
- Takarada, T.; Ida, N.; Hioki, A.; Kanbara, S.; Yamamoto, M.; Kato, K. Estimation of gasification of coal chars in steam-nitrogen and carbon dioxide–nitrogen atmospheres. J. Fuel Soc. Jpn. 1988, 67, 1061–1069. [Google Scholar] [CrossRef]
- Huttinger, K.J.; Nill, J.S. A method for the determination of active sites and true activation energies in carbon gasification: (II) Experimental results. Carbon 1990, 4, 457–465. [Google Scholar] [CrossRef]
- Miura, K.; Hashimoto, K.; Silveston, P.L. Factors affecting the reactivity of coal chars during gasification, and indexes representing reactivity. Fuel 1989, 68, 1461–1475. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).