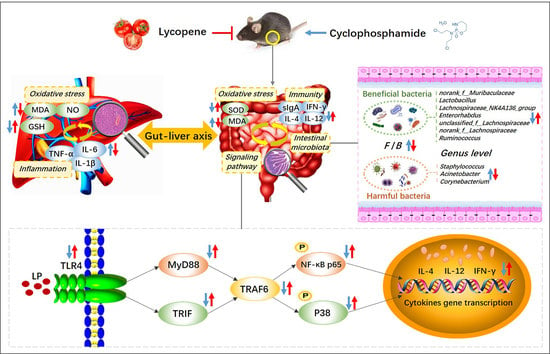
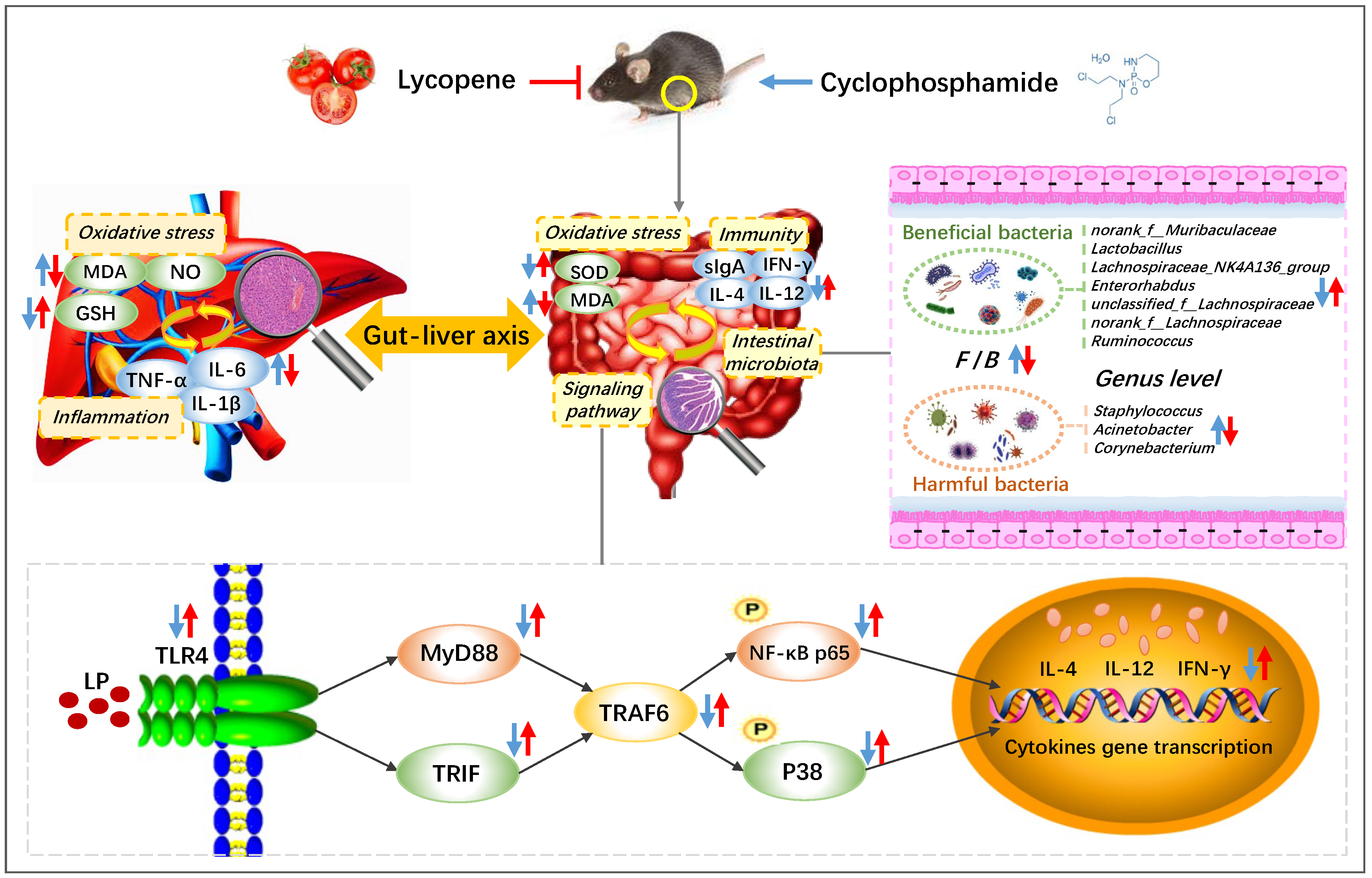
Preventive Mechanism of Lycopene on Intestinal Toxicity Caused by Cyclophosphamide Chemotherapy in Mice by Regulating TLR4-MyD88/TRIF-TRAF6 Signaling Pathway and Gut-Liver Axis
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. LP Intragastric Dose Selection and Preparation
2.2.2. Preparation of Animal Models
2.2.3. Visceral Index
2.2.4. Detection of Biochemical Indicators of the Small Intestine
2.2.5. Detection of sIgA Content in Intestinal Mucosa
2.2.6. Western Blot
2.2.7. Detection of Biochemical Indicators of the Liver
2.2.8. Determination of Intestinal Microbiota
2.2.9. Statistical Analysis
3. Results
3.1. Effects of LP on Spleen and Thymus Indexes in CYC Mice
3.2. Effects of LP on Intestinal Immune Function Injury Induced by CYC in Mice
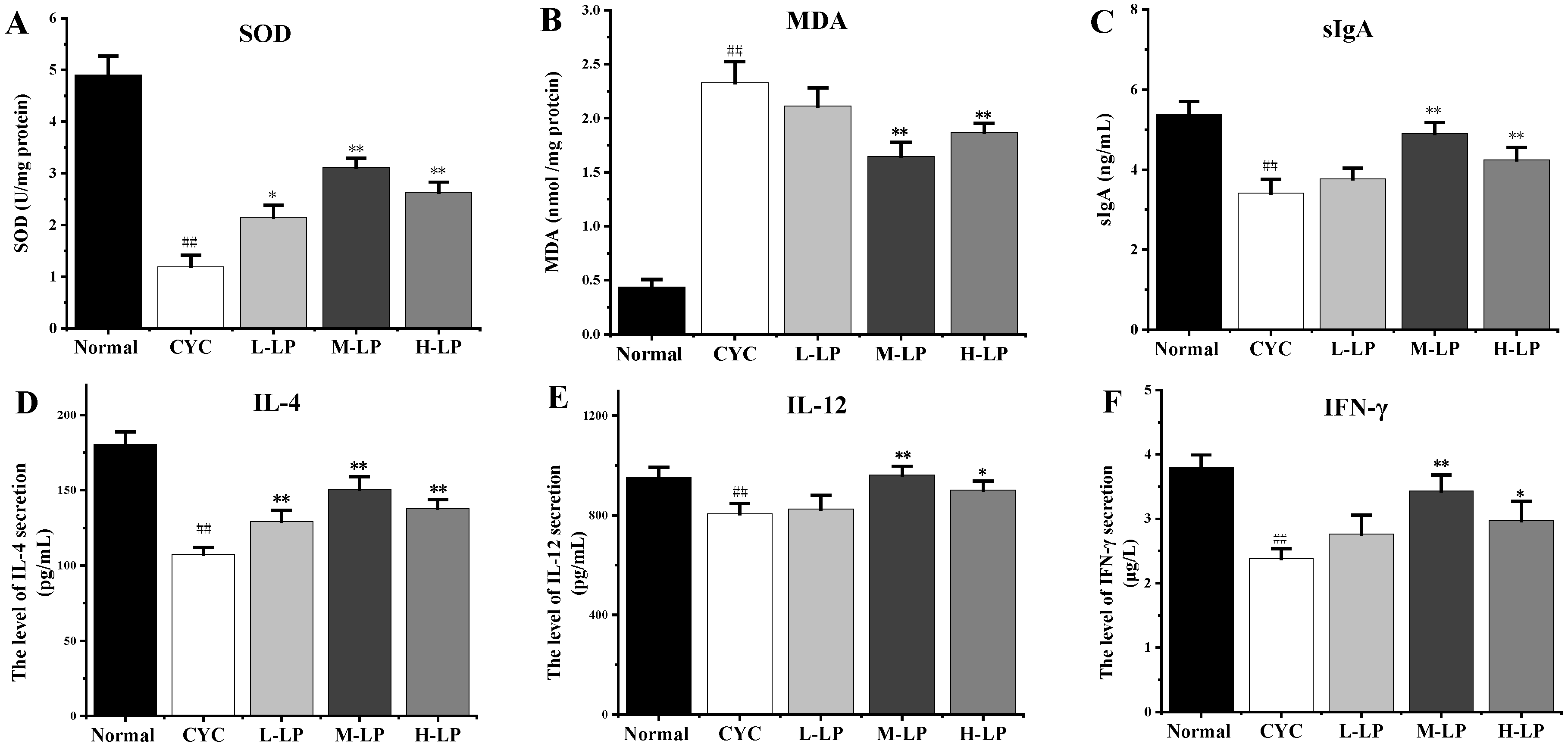
3.2.1. Effects of LP on SOD, MDA, sIgA, and Cytokine Levels in CYC Mice Intestinal Tissue
3.2.2. Effects of LP on Small Intestinal Morphology in CYC Mice
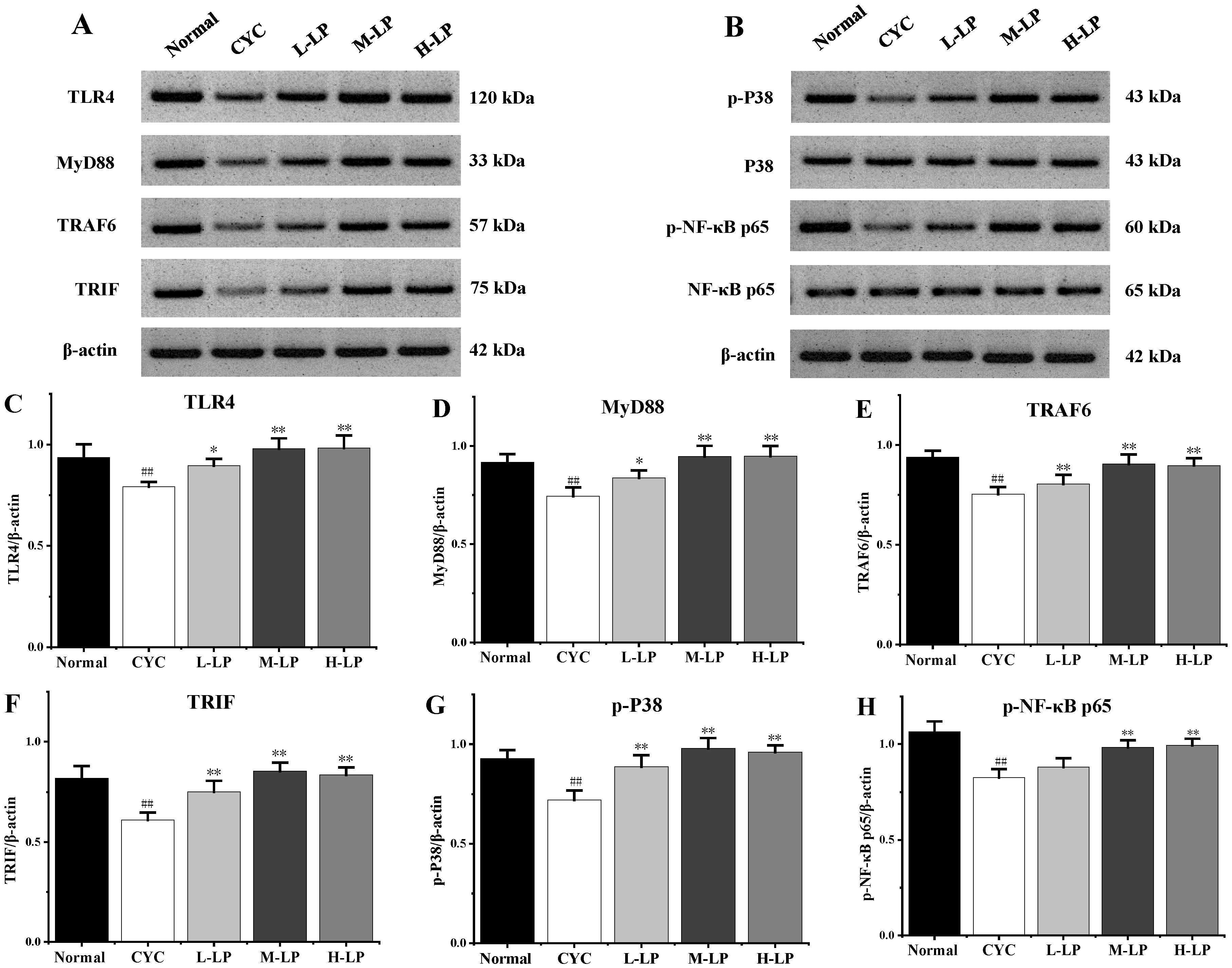
3.2.3. Effects of LP on Key Proteins in Small Intestine of CYC Mice
3.3. Effects of LP on CYC-Induced Liver Injury in Mice
3.4. Effects of LP on Intestinal Microbiota Induced by CYC in Mice
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| LP | Lycopene |
| CYC | Cyclophosphamide |
| sIgA | Secretory immunoglobulin A |
| TLR4 | Toll-like receptors 4 |
| MAPKs | Mitogen-activated protein kinases |
| NF-κB | Nuclear factor kappa-B |
| MyD88 | Myeloid differentiation factor88 |
| SOD | Superoxide dismutase |
| MDA | Lipid oxidation |
| NO | Nitric oxide |
| GSH | Glutathione |
| IL | Interleukin |
| IFN | Interferon |
| TNF | Tumor necrosis factor |
| TRAF6 | Tumor necrosis factor receptor-associated factor 6 |
| TRIF | Toll/IL-1receptor domain containing adaptor protein inducing IFN-β |
| P38 | p38 MAPK |
| NASH | Non-alcoholic steatohepatitis |
| F/B | The ratio of Firmicutes to Bacteroidota |
| BCS | Bacillus coagulans 13,002 |
| FOS | Fructo-oligosaccharides |
References
- Tsuji, D.; Matsumoto, M.; Kawasaki, Y.; Kim, Y.-I.; Yamamoto, K.; Nakamichi, H.; Sahara, Y.; Makuta, R.; Yokoi, M.; Miyagi, T. Analysis of pharmacogenomic factors for chemotherapy-induced nausea and vomiting in patients with breast cancer receiving doxorubicin and cyclophosphamide chemotherapy. Cancer Chemother. Pharmacol. 2021, 87, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Emadi, A.; Jones, R.J.; Brodsky, R.A. Cyclophosphamide and cancer: Golden anniversary. Nat. Rev. Clin. Oncol. 2009, 6, 638–647. [Google Scholar] [CrossRef] [PubMed]
- Sitzia, J.; Huggins, L. Side effects of cyclophosphamide, methotrexate, and 5-fluorouracil (CMF) chemotherapy for breast cancer. Cancer Pract. 1998, 6, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Zhao, P.; Li, X.; Guo, L.; Gao, W. The potential roles of natural plant polysaccharides in inflammatory bowel disease: A review. Carbohydr. Polym. 2022, 277, 118821. [Google Scholar] [CrossRef] [PubMed]
- Cengiz, M.; Yildiz, S.C.; Demir, C.; Şahin, İ.K.; Teksoy, Ö.; Ayhanci, A. Hepato-preventive and anti-apoptotic role of boric acid against liver injury induced by cyclophosphamide. J. Trace Elem. Med. Biol. 2019, 53, 1–7. [Google Scholar] [CrossRef]
- Qian, L.; Yang, F.; Lin, X.; Jiang, S.; Zhang, Y.; Tang, Y. Pyrroloquinoline quinone ameliorates liver injury in mice induced by cyclophosphamide. Environ. Sci. Pollut. Res. 2022, 29, 30383–30393. [Google Scholar] [CrossRef]
- Marshall, J.C. The gut as a potential trigger of exercise-induced inflammatory responses. Can. J. Physiol. Pharmacol. 1998, 76, 479–484. [Google Scholar] [CrossRef]
- Hartmann, P.; Haimerl, M.; Mazagova, M.; Brenner, D.A.; Schnabl, B. Toll-like receptor 2-mediated intestinal injury and enteric tumor necrosis factor receptor I contribute to liver fibrosis in mice. Gastroenterology 2012, 143, 1330–1340.e1. [Google Scholar] [CrossRef]
- Jiang, J.-W.; Chen, X.-H.; Ren, Z.; Zheng, S.-S. Gut microbial dysbiosis associates hepatocellular carcinoma via the gut-liver axis. Hepatobiliary Pancreat. Dis. Int. 2019, 18, 19–27. [Google Scholar] [CrossRef]
- Wang, S.Z.; Yu, Y.J.; Adeli, K. Role of Gut Microbiota in Neuroendocrine Regulation of Carbohydrate and Lipid Metabolism via the Microbiota-Gut-Brain-Liver Axis. Microorganisms 2020, 8, 527. [Google Scholar] [CrossRef]
- Shi, Y.; He, X.; Yang, S.; Ai, B.; Zhang, C.; Huang, D.; Dong, M.; Liu, P.; Zhou, S.; Han, X. Ramosetron versus ondansetron in the prevention of chemotherapy-induced gastrointestinal side effects: A prospective randomized controlled study. Chemotherapy 2007, 53, 44–50. [Google Scholar] [CrossRef]
- Souza, A.; Silva, L.; Fayh, A. Nutritional Intervention Contributes to the Improvement of Symptoms Related to Quality of Life in Breast Cancer Patients Undergoing Neoadjuvant Chemotherapy: A Randomized Clinical Trial. Nutrients 2021, 13, 598. [Google Scholar] [CrossRef]
- Xie, H.; Fang, J.; Farag, M.A.; Li, Z.; Sun, P.; Shao, P. Dendrobium officinale leaf polysaccharides regulation of immune response and gut microbiota composition in cyclophosphamide-treated mice. Food Chem. X 2022, 13, 100235. [Google Scholar] [CrossRef]
- Li, C.; Duan, S.; Li, Y.; Pan, X.; Han, L. Polysaccharides in natural products that repair the damage to intestinal mucosa caused by cyclophosphamide and their mechanisms: A review. Carbohydr. Polym. 2021, 261, 117876. [Google Scholar] [CrossRef]
- Li, N.; Wang, C.; Georgiev, M.I.; Bajpai, V.K.; Qiao, X. Advances in dietary polysaccharides as anticancer agents: Structure-activity relationship. Trends Food Sci. Technol. 2021, 111, 360–377. [Google Scholar] [CrossRef]
- Liu, F.; Li, P.; Chen, M.; Luo, Y.; Prabhakar, M.; Zheng, H.; He, Y.; Qi, Q.; Long, H.; Zhang, Y. Fructooligosaccharide (FOS) and galactooligosaccharide (GOS) increase Bifidobacterium but reduce butyrate producing bacteria with adverse glycemic metabolism in healthy young population. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Caseiro, M.; Ascenso, A.; Costa, A.; Creagh-Flynn, J.; Johnson, M.; Simões, S. Lycopene in human health. LWT 2020, 127, 109323. [Google Scholar] [CrossRef]
- Grabowska, M.; Wawrzyniak, D.; Rolle, K.; Chomczyński, P.; Oziewicz, S.; Jurga, S.; Barciszewski, J. Let food be your medicine: Nutraceutical properties of lycopene. Food Funct. 2019, 10, 3090–3102. [Google Scholar] [CrossRef]
- Pinto, C.; Rodriguez-Galdon, B.; Cestero, J.J.; Macias, P. Processed tomatoes improves the antioxidant status of carbon tetrachloride-intoxicated rat tissues. Eur. Food Res. Technol. 2018, 244, 1843–1852. [Google Scholar] [CrossRef]
- Abd Al Haleem, E.N.; Ahmed, H.I.; El-Naga, R.N. Lycopene and Chrysin through Mitigation of Neuroinflammation and Oxidative Stress Exerted Antidepressant Effects in Clonidine-Induced Depression-like Behavior in Rats. J. Diet. Suppl. 2021, 1–20. [Google Scholar] [CrossRef]
- Gajowik, A.; Dobrzynska, M. Lycopene-antioxidant with radioprotective and anticancer properties. A review. Rocz. Państwowego Zakładu Hig. 2014, 65, 263–271. [Google Scholar]
- Stojiljkovic, N.; Ilic, S.; Jakovljevic, V.; Stojanovic, N.; Stojnev, S.; Kocic, H.; Stojanovic, M.; Kocic, G. The Encapsulation of Lycopene in Nanoliposomes Enhances Its Protective Potential in Methotrexate-Induced Kidney Injury Model. Oxid. Med. Cell Longev. 2018, 2018, 2627917. [Google Scholar] [CrossRef] [PubMed]
- Rajput, S.A.; Liang, S.J.; Wang, X.Q.; Yan, H.C. Lycopene Protects Intestinal Epithelium from Deoxynivalenol-Induced Oxidative Damage via Regulating Keap1/Nrf2 Signaling. Antioxidants 2021, 10, 1493. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Pan, X.; Yin, M.; Li, C.; Han, L. Preventive Effect of Lycopene in Dextran Sulfate Sodium-Induced Ulcerative Colitis Mice through the Regulation of TLR4/TRIF/NF-κB Signaling Pathway and Tight Junctions. J. Agric. Food Chem. 2021, 69, 13500–13509. [Google Scholar] [CrossRef] [PubMed]
- Han, L.; Yu, J.; Chen, Y.; Cheng, D.; Wang, X.; Wang, C. Immunomodulatory Activity of Docosahexenoic Acid on RAW264.7 Cells Activation through GPR120-Mediated Signaling Pathway. J. Agric. Food Chem. 2018, 66, 926–934. [Google Scholar] [CrossRef]
- Imran, M.; Ghorat, F.; Ul-Haq, I.; Ur-Rehman, H.; Aslam, F.; Heydari, M.; Shariati, M.A.; Okuskhanova, E.; Yessimbekov, Z.; Thiruvengadam, M. Lycopene as a natural antioxidant used to prevent human health disorders. Antioxidants 2020, 9, 706. [Google Scholar] [CrossRef]
- Zhao, B.; Wu, J.; Li, J.; Bai, Y.; Luo, Y.; Ji, B.; Xia, B.; Liu, Z.; Tan, X.; Lv, J. Lycopene alleviates DSS-induced colitis and behavioral disorders via mediating microbes-gut–brain axis balance. J. Agric. Food Chem. 2020, 68, 3963–3975. [Google Scholar] [CrossRef]
- Kim, J.; Chan, J.J. Cyclophosphamide in dermatology. Australas. J. Dermatol. 2017, 58, 5–17. [Google Scholar] [CrossRef]
- Han, L.; Lei, H.; Tian, Z.; Wang, X.; Cheng, D.; Wang, C. The immunomodulatory activity and mechanism of docosahexenoic acid (DHA) on immunosuppressive mice models. Food Funct. 2018, 9, 3254–3263. [Google Scholar] [CrossRef]
- Xiang, X.W.; Zheng, H.Z.; Wang, R.; Chen, H.; Xiao, J.X.; Zheng, B.; Liu, S.L.; Ding, Y.T. Ameliorative Effects of Peptides Derived from Oyster (Crassostrea gigas) on Immunomodulatory Function and Gut Microbiota Structure in Cyclophosphamide-Treated Mice. Mar. Drugs 2021, 19, 546. [Google Scholar] [CrossRef]
- Chen, S.; Wang, J.; Fang, Q.; Dong, N.; Nie, S. Polysaccharide from natural Cordyceps sinensis ameliorated intestinal injury and enhanced antioxidant activity in immunosuppressed mice. Food Hydrocoll. 2019, 89, 661–667. [Google Scholar] [CrossRef]
- Shang, L.; Liu, Y.; Li, J.; Pan, G.; Zhou, F.; Yang, S. Emodin Protects Sepsis Associated Damage to the Intestinal Mucosal Barrier Through the VDR/Nrf2/HO-1 Pathway. Front. Pharmacol. 2021, 12, 724511. [Google Scholar] [CrossRef]
- Cai, G.; Wu, Y.; Wusiman, A.; Gu, P.; Mao, N.; Xu, S.; Zhu, T.; Feng, Z.; Liu, Z.; Wang, D. Alhagi honey polysaccharides attenuate intestinal injury and immune suppression in cyclophosphamide-induced mice. Food Funct. 2021, 12, 6863–6877. [Google Scholar] [CrossRef]
- Fu, Y.-P.; Feng, B.; Zhu, Z.-K.; Feng, X.; Chen, S.-F.; Li, L.-X.; Yin, Z.-Q.; Huang, C.; Chen, X.-F.; Zhang, B.-Z. The polysaccharides from Codonopsis pilosula modulates the immunity and intestinal microbiota of cyclophosphamide-treated immunosuppressed mice. Molecules 2018, 23, 1801. [Google Scholar] [CrossRef]
- Rajanna, M.; Bharathi, B.; Shivakumar, B.; Deepak, M.; Prabakaran, D.; Vijayabhaskar, T.; Arun, B. Immunomodulatory effects of Andrographis paniculata extract in healthy adults–An open-label study. J. Ayurveda Integr. Med. 2021, 12, 529–534. [Google Scholar] [CrossRef]
- Tian, C.; Liu, X.; Chang, Y.; Wang, R.; Yang, M.; Liu, M. Rutin prevents inflammation induced by lipopolysaccharide in RAW 264.7 cells via conquering the TLR4-MyD88-TRAF6-NF-κB signalling pathway. J. Pharm. Pharmacol. 2021, 73, 110–117. [Google Scholar] [CrossRef]
- Duan, Y.; Dai, H.; An, Y.; Cheng, L.; Shi, L.; Lv, Y.; Li, H.; Wang, C.; He, C.; Zhang, H. Mulberry Leaf Flavonoids Inhibit Liver Inflammation in Type 2 Diabetes Rats by Regulating TLR4/MyD88/NF-κB Signaling Pathway. Evid. Based Complement. Altern. Med. 2022, 2022, 3354062. [Google Scholar] [CrossRef]
- Choi, J.; Lim, J.W.; Kim, H. Lycopene Inhibits Toll-Like Receptor 4-Mediated Expression of Inflammatory Cytokines in House Dust Mite-Stimulated Respiratory Epithelial Cells. Molecules 2021, 26, 3127. [Google Scholar] [CrossRef]
- Zhao, Y.; Yan, Y.; Zhou, W.; Chen, D.; Huang, K.; Yu, S.; Mi, J.; Lu, L.; Zeng, X.; Cao, Y. Effects of polysaccharides from bee collected pollen of Chinese wolfberry on immune response and gut microbiota composition in cyclophosphamide-treated mice. J. Funct. Foods 2020, 72, 104057. [Google Scholar] [CrossRef]
- Ying, M.; Yu, Q.; Zheng, B.; Wang, H.; Wang, J.; Chen, S.; Nie, S.; Xie, M. Cultured Cordyceps sinensis polysaccharides modulate intestinal mucosal immunity and gut microbiota in cyclophosphamide-treated mice. Carbohydr. Polym. 2020, 235, 115957. [Google Scholar] [CrossRef]
- Martín-Mateos, R.; Albillos, A. The role of the gut-liver axis in metabolic dysfunction-associated fatty liver disease. Front. Immunol. 2021, 12, 660179. [Google Scholar] [CrossRef] [PubMed]
- Habibi, E.; Shokrzadeh, M.; Chabra, A.; Naghshvar, F.; Keshavarz-Maleki, R.; Ahmadi, A. Protective effects of Origanum vulgare ethanol extract against cyclophosphamide-induced liver toxicity in mice. Pharm. Biol. 2015, 53, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, A.; Qu, Y.; Wang, Z.; Zhang, Y.; Liu, Y.; Wang, N.; Teng, L.; Wang, D. Ameliorative effects of Antrodia cinnamomea polysaccharides against cyclophosphamide-induced immunosuppression related to Nrf2/HO-1 signaling in BALB/c mice. Int. J. Biol. Macromol. 2018, 116, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Temel, Y.; Kucukler, S.; Yıldırım, S.; Caglayan, C.; Kandemir, F.M. Protective effect of chrysin on cyclophosphamide-induced hepatotoxicity and nephrotoxicity via the inhibition of oxidative stress, inflammation, and apoptosis. Naunyn-Schmiedeberg’s Arch. Pharmacol. 2019, 393, 325–337. [Google Scholar] [CrossRef] [PubMed]
- Stice, C.P.; Xia, H.; Wang, X.D. Tomato lycopene prevention of alcoholic fatty liver disease and hepatocellular carcinoma development. Chronic Dis. Transl. Med. 2018, 4, 211–224. [Google Scholar] [CrossRef]
- Wang, J.; Li, M.; Gao, Y.; Li, H.; Fang, L.; Liu, C.; Liu, X.; Min, W. Effects of Exopolysaccharides from Lactiplantibacillus plantarum JLAU103 on Intestinal Immune Response, Oxidative Stress, and Microbial Communities in Cyclophosphamide-Induced Immunosuppressed Mice. J. Agric. Food Chem. 2022, 70, 2197–2210. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, X. Effects of cyclophosphamide on immune system and gut microbiota in mice. Microbiol. Res. 2015, 171, 97–106. [Google Scholar] [CrossRef]
- Smith, B.J.; Miller, R.A.; Ericsson, A.C.; Harrison, D.C.; Strong, R.; Schmidt, T.M. Changes in the gut microbiome and fermentation products concurrent with enhanced longevity in acarbose-treated mice. BMC Microbiol. 2019, 19, 1–16. [Google Scholar] [CrossRef]
- Huang, J.; Huang, J.; Li, Y.; Wang, Y.; Wang, F.; Qiu, X.; Liu, X.; Li, H. Sodium Alginate Modulates Immunity, Intestinal Mucosal Barrier Function, and Gut Microbiota in Cyclophosphamide-Induced Immunosuppressed BALB/c Mice. J. Agric. Food Chem. 2021, 69, 7064–7073. [Google Scholar] [CrossRef]
- Chen, X.; Sun, W.; Xu, B.; Wu, E.; Cui, Y.; Hao, K.; Zhang, G.; Zhou, C.; Xu, Y.; Li, J.; et al. Polysaccharides From the Roots of Millettia Speciosa Champ Modulate Gut Health and Ameliorate Cyclophosphamide-Induced Intestinal Injury and Immunosuppression. Front. Immunol. 2021, 12, 766296. [Google Scholar] [CrossRef]
- Zhu, H.; He, Y.S.; Ma, J.; Zhou, J.; Kong, M.; Wu, C.Y.; Mao, Q.; Lin, G.; Li, S.L. The dual roles of ginsenosides in improving the anti-tumor efficiency of cyclophosphamide in mammary carcinoma mice. J. Ethnopharmacol. 2021, 265, 113271. [Google Scholar] [CrossRef]
- Han, R.; Ma, Y.; Xiao, J.; You, L.; Pedisic, S.; Liao, L. The possible mechanism of the protective effect of a sulfated polysaccharide from Gracilaria Lemaneiformis against colitis induced by dextran sulfate sodium in mice. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2021, 149, 112001. [Google Scholar] [CrossRef]
- Zhao, S.; Peng, X.; Zhou, Q.Y.; Huang, Y.Y.; Rao, X.; Tu, J.L.; Xiao, H.Y.; Liu, D.M. Bacillus coagulans 13002 and fructo-oligosaccharides improve the immunity of mice with immunosuppression induced by cyclophosphamide through modulating intestinal-derived and fecal microbiota. Food Res. Int. 2021, 140, 109793. [Google Scholar] [CrossRef]
- Jhang, S.Y.; Lee, S.H.; Lee, E.B.; Choi, J.H.; Bang, S.; Jeong, M.; Jang, H.H.; Kim, H.J.; Lee, H.J.; Jeong, H.C.; et al. Effects of Platycodon grandiflorum on Gut Microbiome and Immune System of Immunosuppressed Mouse. Metabolites 2021, 11, 817. [Google Scholar] [CrossRef]
- Wiese, M.; Bashmakov, Y.; Chalyk, N.; Nielsen, D.S.; Krych, L.; Kot, W.; Klochkov, V.; Pristensky, D.; Bandaletova, T.; Chernyshova, M.; et al. Prebiotic Effect of Lycopene and Dark Chocolate on Gut Microbiome with Systemic Changes in Liver Metabolism, Skeletal Muscles and Skin in Moderately Obese Persons. Biomed. Res. Int. 2019, 2019, 4625279. [Google Scholar] [CrossRef]



| Groups | Thymus Index (mg/g) | Spleen Index (mg/g) |
|---|---|---|
| Normal | 2.20 ± 0.33 | 4.45 ± 0.29 |
| Cyclophosphamide (CYC) | 0.93 ± 0.19 ## | 3.22 ± 0.24 ## |
| Low-lycopene (L-LP) | 1.12 ± 0.24 | 3.39 ± 0.33 |
| Medium-lycopene (M-LP) | 1.55 ± 0.15 ** | 3.88 ± 0.26 * |
| High-lycopene (H-LP) | 1.24 ± 0.15 | 3.56 ± 0.23 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, X.; Niu, X.; Li, Y.; Yao, Y.; Han, L. Preventive Mechanism of Lycopene on Intestinal Toxicity Caused by Cyclophosphamide Chemotherapy in Mice by Regulating TLR4-MyD88/TRIF-TRAF6 Signaling Pathway and Gut-Liver Axis. Nutrients 2022, 14, 4467. https://doi.org/10.3390/nu14214467
Pan X, Niu X, Li Y, Yao Y, Han L. Preventive Mechanism of Lycopene on Intestinal Toxicity Caused by Cyclophosphamide Chemotherapy in Mice by Regulating TLR4-MyD88/TRIF-TRAF6 Signaling Pathway and Gut-Liver Axis. Nutrients. 2022; 14(21):4467. https://doi.org/10.3390/nu14214467
Chicago/Turabian StylePan, Xiao, Xiaoyan Niu, Yaping Li, Yupei Yao, and Lirong Han. 2022. "Preventive Mechanism of Lycopene on Intestinal Toxicity Caused by Cyclophosphamide Chemotherapy in Mice by Regulating TLR4-MyD88/TRIF-TRAF6 Signaling Pathway and Gut-Liver Axis" Nutrients 14, no. 21: 4467. https://doi.org/10.3390/nu14214467
APA StylePan, X., Niu, X., Li, Y., Yao, Y., & Han, L. (2022). Preventive Mechanism of Lycopene on Intestinal Toxicity Caused by Cyclophosphamide Chemotherapy in Mice by Regulating TLR4-MyD88/TRIF-TRAF6 Signaling Pathway and Gut-Liver Axis. Nutrients, 14(21), 4467. https://doi.org/10.3390/nu14214467