Liver Transplantation for Unresectable Colorectal Liver Metastases: A Scoping Review on Redefining Boundaries in Transplant Oncology
Simple Summary
Abstract
1. Introduction
2. Methodology
3. Liver Transplantation for Unresectable Colorectal Metastatic Disease
4. From SECA Trials to More Recent Protocols
5. Current Outcomes and Emerging Strategies for Unresectable Colorectal Liver Metastases
6. Patient Selection and Outcomes of Liver Transplantation for Unresectable Colorectal Liver Metastases
7. Immunosuppression Regimens After Liver Transplantation for Colorectal Metastases
8. Immunotherapy and Transplant Compatibility
9. Ethical and Allocation Considerations
10. Innovations in Organ Utilization
11. Biomarkers and Predictive Tools
12. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CRC | Colorectal cancer |
| CRLM | Colorectal liver metastases |
| LT | Liver transplantation |
| ST | Systemic therapy |
| OS | Overall survival |
| DFS | Disease-free survival |
| PFS | Progression-free survival |
| HCC | Hepatocellular carcinoma |
| NET | Neuroendocrine tumors |
| UNOS | United Network for Organ Sharing |
| SECA | Secondary cancer |
| IHPBA | International Hepato-Pancreatic Biliary Association |
| ECOG | Eastern Cooperative Oncology Group |
| CEA | Carcinoembruonic antigen |
| FCRS | Fong Clinical Risk Score |
| PET | Positron emission tomography |
| MTV | Metabolic tumor volume |
| MSI | Microsatellite instability |
| dMMR | Deficient mismatch repair |
| MELD | Model for end-stage liver disease |
| LDLT | Living donor liver transplantation |
| RAPID | Resection and partial liver transplantation with delayed total hepatectomy |
| MP | Machine perfusion |
| ECD | Extended criteria donor |
| HOPE | Hypothermic oxygenated perfusion |
| NMP | Normothermic machine perfusion |
| ctDNA | Circulating tumor DNA |
| cfDNA | Cell-free DNA |
| HR | Hazard ratio |
| RR | Relative risk |
| UCSF | University of California San Francisco |
| VITTAL | Viability testing and transplantation of marginal livers |
References
- Siegel, R.L.; Wagle, N.S.; Cercek, A.; Smith, R.A.; Jemal, A. Colorectal cancer statistics, 2023. CA Cancer J. Clin. 2023, 73, 233–254. [Google Scholar] [CrossRef]
- Zhou, H.; Liu, Z.; Wang, Y.; Wen, X.; Amador, E.H.; Yuan, L.; Ran, X.; Xiong, L.; Ran, Y.; Chen, W.; et al. Colorectal liver metastasis: Molecular mechanism and interventional therapy. Signal Transduct. Target. Ther. 2022, 7, 70. [Google Scholar] [CrossRef]
- Varley, R.; Tarazi, M.; Davé, M.; Mobarak, S.; Stott, M.C.; Baltatzis, M.; Satyadas, T. Liver Transplantation for Non-Resectable Liver Metastases from Colorectal Cancer: A Systematic Review and Meta-Analysis. World J. Surg. 2021, 45, 3404–3413. [Google Scholar] [CrossRef]
- Mc Fadden, N.; Perry, L.; Ghalambor, T.; Langan, R.; Gholami, S. Locoregional Liver-Directed Therapies to Treat Unresectable Colorectal Liver Metastases: A Review. Oncology 2022, 36, 108–114. [Google Scholar] [CrossRef]
- Tasoudis, P.T.; Ziogas, I.A.; Alexopoulos, S.P.; Fung, J.J.; Tsoulfas, G. Role of liver transplantation in the management of colorectal liver metastases: Challenges and opportunities. World J. Clin. Oncol. 2021, 12, 1193–1201. [Google Scholar] [CrossRef]
- Bachler, J.L.; Khan, G.N.; Wollner, I.S.; Philip, P.A. Treatment of Unresectable and Resectable Stage IV Colorectal Cancer. Clin. Adv. Hematol. Oncol. 2024, 22, 455–463. [Google Scholar]
- Liu, W.; Liu, J.-M.; Wang, K.; Wang, H.-W.; Xing, B.-C. Recurrent colorectal liver metastasis patients could benefit from repeat hepatic resection. BMC Surg. 2021, 21, 327. [Google Scholar] [CrossRef] [PubMed]
- Ziemlewicz, T.J.; Critchfield, J.J.; Mendiratta-Lala, M.; Wiggermann, P.; Pech, M.; Serres-Créixams, X.; Lubner, M.; Wah, T.M.; Littler, P.; Davis, C.R.; et al. The #HOPE4LIVER single-arm Pivotal Trial for Histotripsy of Primary and Metastatic Liver Tumors: 1-year Update of Clinical Outcomes. Ann. Surg. 2025. [Google Scholar] [CrossRef]
- Tang, W.; Ren, L.; Liu, T.; Ye, Q.; Wei, Y.; He, G.; Lin, Q.; Wang, X.; Wang, M.; Liang, F.; et al. Bevacizumab Plus mFOLFOX6 Versus mFOLFOX6 Alone as First-Line Treatment for RAS Mutant Unresectable Colorectal Liver-Limited Metastases: The BECOME Randomized Controlled Trial. J. Clin. Oncol. 2020, 38, 3175–3184. [Google Scholar] [CrossRef]
- Smedman, T.M.; Line, P.; Hagness, M.; Syversveen, T.; Grut, H.; Dueland, S. Liver transplantation for unresectable colorectal liver metastases in patients and donors with extended criteria (SECA-II arm D study). BJS Open 2020, 4, 467–477. [Google Scholar] [CrossRef]
- Hagness, M.; Foss, A.; Line, P.-D.; Scholz, T.; Jørgensen, P.F.; Fosby, B.; Boberg, K.M.; Mathisen, Ø.; Gladhaug, I.P.; Egge, T.S.; et al. Liver Transplantation for Nonresectable Liver Metastases from Colorectal Cancer. Ann. Surg. 2013, 257, 800–806. [Google Scholar] [CrossRef]
- Ahearn, A. Ethical Dilemmas in Liver Transplant Organ Allocation: Is it Time for a New Mathematical Model? AMA J. Ethics 2016, 18, 126–132. [Google Scholar] [PubMed][Green Version]
- Clift, A.K.; Hagness, M.; Lehmann, K.; Rosen, C.B.; Adam, R.; Mazzaferro, V.; Frilling, A. Transplantation for metastatic liver disease. J. Hepatol. 2023, 78, 1137–1146. [Google Scholar] [CrossRef] [PubMed]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- Huang, G.; Song, W.; Zhang, Y.; Yu, J.; Lv, Y.; Liu, K. Liver transplantation for intrahepatic cholangiocarcinoma: A propensity score-matched analysis. Sci. Rep. 2023, 13, 10630. [Google Scholar] [CrossRef] [PubMed]
- Kulik, L.; Heimbach, J.K.; Zaiem, F.; Almasri, J.; Prokop, L.J.; Wang, Z.; Murad, M.H.; Mohammed, K. Therapies for patients with hepatocellular carcinoma awaiting liver transplantation: A systematic review and meta-analysis. Hepatology 2017, 67, 381–400. [Google Scholar] [CrossRef]
- Goldaracena, N.; Gorgen, A.; Sapisochin, G. Current status of liver transplantation for cholangiocarcinoma. Liver Transplant. 2018, 24, 294–303. [Google Scholar] [CrossRef]
- Citterio, D.; Coppa, J.; Sposito, C.; Busset, M.D.D.; Virdis, M.; Pezzoli, I.; Mazzaferro, V. The Role of Liver Transplantation in the Treatment of Liver Metastases from Neuroendocrine Tumors. Curr. Treat. Options Oncol. 2023, 24, 1651–1665. [Google Scholar] [CrossRef]
- Dueland, S.; Syversveen, T.; Solheim, J.M.; Solberg, S.; Grut, H.; Bjørnbeth, B.A.; Hagness, M.; Line, P.-D. Survival Following Liver Transplantation for Patients with Nonresectable Liver-only Colorectal Metastases. Ann. Surg. 2020, 271, 212–218. [Google Scholar] [CrossRef]
- Andres, A.; Oldani, G.; Berney, T.; Compagnon, P.; Line, P.-D.; Toso, C. Transplantation for colorectal metastases: On the edge of a revolution. Transl. Gastroenterol. Hepatol. 2018, 3, 74. [Google Scholar] [CrossRef] [PubMed]
- Adam, R.; Piedvache, C.; Chiche, L.; Adam, J.P.; Salamé, E.; Bucur, P.; Cherqui, D.; Scatton, O.; Granger, V.; Ducreux, M.; et al. Liver transplantation plus chemotherapy versus chemotherapy alone in patients with permanently unresectable colorectal liver metastases (TransMet): Results from a multicentre, open-label, prospective, randomised controlled trial. Lancet 2024, 404, 1107–1118. [Google Scholar] [CrossRef]
- Byrne, M.M.; Chávez-Villa, M.; Ruffolo, L.I.; Loria, A.; Endo, Y.; Niewiemski, A.; Jimenez-Soto, C.; Melaragno, J.I.; Ramaraju, G.A.; Farooq, P.D.; et al. Corrigendum to ‘The Rochester Protocol for living donor liver transplantation of unresectable colorectal liver metastasis: A 5-year report on selection, approval, and outcomes’. Am. J. Transplant. 2025, 25, 780–792. [Google Scholar] [CrossRef] [PubMed]
- Kaltenmeier, C.; Geller, D.A.; Ganesh, S.; Tohme, S.; Molinari, M.; Tevar, A.; Hughes, C.; Humar, A. Living donor liver transplantation for colorectal cancer liver metastases: Midterm outcomes at a single center in North America. Am. J. Transplant. 2023, 24, 681–687. [Google Scholar] [CrossRef]
- Dawood, Z.S.; Brown, Z.J.; Munir, M.M.; Waqar, U.; Rawicz-Pruszynski, K.; Endo, Y.; Gajjar, A.; Schenk, A.; Pawlik, T.M. Outcomes of liver transplant for colorectal liver metastasis: A systematic review and meta-analysis. J. Gastrointest. Surg. 2024, 28, 1943–1950. [Google Scholar] [CrossRef]
- Schepers, E.J.; Hartman, S.J.; Whitrock, J.N.; Quillin, R.C. Liver transplantation for colorectal liver metastases. Surg. Clin. 2024, 104, 227–242. [Google Scholar] [CrossRef] [PubMed]
- Sonbol, M.B.; Siddiqi, R.; Uson, P.L.S., Jr.; Pathak, S.; Firwana, B.; Botrus, G.; Almader-Douglas, D.; Ahn, D.H.; Borad, M.J.; Starr, J.; et al. The role of systemic therapy in resectable colorectal liver metastases: Systematic review and network meta-analysis. Oncologist 2022, 27, 1034–1040. [Google Scholar] [CrossRef]
- Krishnan, S.; Joshi, B.; Keith, J.; Frazier, R.; Kalinowska, B.; Choi, J.S.; Sanders, S.; Boi, S.; Le Tonqueze, O.; Kushner, N.; et al. Abstract 1331: MiNK-215, an IL-15 armored FAP-targeting CAR iNKT cell therapy, effectively treats human organoid models of treatment-refractory MSS colorectal cancer (CRC) liver metastases. Cancer Res. 2024, 84, 1331. [Google Scholar] [CrossRef]
- Wu, F.; Cao, G.; Lu, J.; Ye, S.; Tang, X. Correlation between 18F-FDG PET/CT metabolic parameters and microvascular invasion before liver transplantation in patients with hepatocellular carcinoma. Nucl. Med. Commun. 2024, 45, 1033–1038. [Google Scholar] [CrossRef]
- Malik, A.K.; Mahendran, B.; Lochan, R.; White, S.A. Liver Transplantation for Nonresectable Colorectal Liver Metastases (CRLM). Indian J. Surg. Oncol. 2023, 15, 255–260. [Google Scholar] [CrossRef]
- Toso, C.; Marques, H.P.; Andres, A.; Sousa, F.C.; Adam, R.; Kalil, A.; Clavien, P.; Furtado, E.; Barroso, E.; Bismuth, H.; et al. Liver transplantation for colorectal liver metastasis: Survival without recurrence can be achieved. Liver Transplant. 2017, 23, 1073–1076. [Google Scholar] [CrossRef]
- Hernandez-Alejandro, R.; Ruffolo, L.I.; Sasaki, K.; Tomiyama, K.; Orloff, M.S.; Pineda-Solis, K.; Nair, A.; Errigo, J.; Dokus, M.K.; Cattral, M.; et al. Recipient and Donor Outcomes After Living-Donor Liver Transplant for Unresectable Colorectal Liver Metastases. JAMA Surg. 2022, 157, 524–530. [Google Scholar] [CrossRef]
- Sasaki, K.; Ruffolo, L.I.; Kim, M.H.; Fujiki, M.; Hashimoto, K.; Imaoka, Y.; Melcher, M.L.; Aucejo, F.N.; Tomiyama, K.; Hernandez-Alejandro, R. The current state of liver transplantation for colorectal liver metastases in the United States: A call for standardized reporting. Ann. Surg. Oncol. 2023, 30, 2769. [Google Scholar] [CrossRef]
- Solheim, J.M.; Dueland, S.; Line, P.-D.; Hagness, M. Transplantation for Nonresectable Colorectal Liver Metastases: Long-Term Follow-Up of the First Prospective Pilot Study. Ann. Surg. 2022, 278, 239–245. [Google Scholar] [CrossRef]
- Bonney, G.K.; Chew, C.A.; Lodge, P.; Hubbard, J.; Halazun, K.J.; Trunecka, P.; Muiesan, P.; Mirza, D.F.; Isaac, J.; Laing, R.W.; et al. Liver transplantation for non-resectable colorectal liver metastases: The International Hepato-Pancreato-Biliary Association consensus guidelines. Lancet Gastroenterol. Hepatol. 2021, 6, 933–946. [Google Scholar] [CrossRef] [PubMed]
- Overman, M.J.; Gelsomino, F.; Aglietta, M.; Wong, M.; Miron, M.L.L.; Leonard, G.; García-Alfonso, P.; Hill, A.G.; Gracian, A.C.; Van Cutsem, E.; et al. Nivolumab plus relatlimab in patients with previously treated microsatellite instability-high/mismatch repair-deficient metastatic colorectal cancer: The phase II CheckMate 142 study. J. Immunother. Cancer 2024, 12, e008689. [Google Scholar] [CrossRef] [PubMed]
- Luft, F.C. Calcineurin inhibition, cardiovascular consequences, vascular resistance, and potential responses. Acta Physiol. 2024, 240, e14084. [Google Scholar] [CrossRef] [PubMed]
- Barreto, S.G.; Strasser, S.I.; McCaughan, G.W.; Fink, M.A.; Jones, R.; McCall, J.; Munn, S.; Macdonald, G.A.; Hodgkinson, P.; Jeffrey, G.P.; et al. Expansion of Liver Transplantation Criteria for Hepatocellular Carcinoma from Milan to UCSF in Australia and New Zealand and Justification for Metroticket 2.0. Cancers 2022, 14, 2777. [Google Scholar] [CrossRef] [PubMed]
- Coubeau, L.; Foguenne, M.; Marique, L.; Riani, E.B.; Ciccarelli, O. RAPID-Type Auxiliary Liver Transplantation for Unresectable Colorectal Liver Metastases: A First-Stage Surgical Video. Ann. Surg. Oncol. 2024, 32, 2300–2301. [Google Scholar] [CrossRef]
- Ayoub, I.; Sharshar, M.; Shoreem, H.; Osman, M.; El-Ella, K.A.; Uemoto, S. Outcome and challenges of left-lobe living-donor liver transplantation in adults. Egypt. J. Surg. 2021, 40, 1328–1337. [Google Scholar]
- Settmacher, U.; Ali-Deeb, A.; Coubeau, L.; Cillo, U.; Line, P.D.; Guba, M.; Nadalin, S.; Rauchfuß, F. Auxilliary liver transplantation according to the RAPID procedure in noncirrhotic patients: Technical aspects and early outcomes. Ann. Surg. 2023, 277, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Schlegel, A.; Mueller, M.; Muller, X.; Eden, J.; Panconesi, R.; von Felten, S.; Steigmiller, K.; Da Silva, R.X.S.; de Rougemont, O.; Mabrut, J.-Y.; et al. A multicenter randomized-controlled trial of hypothermic oxygenated perfusion (HOPE) for human liver grafts before transplantation. J. Hepatol. 2023, 78, 783–793. [Google Scholar] [CrossRef]
- Meszaros, A.T.; Hofmann, J.; Buch, M.L.; Cardini, B.; Dunzendorfer-Matt, T.; Nardin, F.; Blumer, M.J.; Fodor, M.; Hermann, M.; Zelger, B.; et al. Mitochondrial respiration during normothermic liver machine perfusion predicts clinical outcome. EBioMedicine 2022, 85, 104311. [Google Scholar] [CrossRef]
- Mergental, H.; Laing, R.W.; Kirkham, A.J.; Clarke, G.; Boteon, Y.L.; Barton, D.; Neil, D.A.; Isaac, J.R.; Roberts, K.J.; Abradelo, M.; et al. Discarded livers tested by normothermic machine perfusion in the VITTAL trial: Secondary end points and 5-year outcomes. Liver Transplant. 2023, 30, 30–45. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Zhao, A.; Cao, N.; Li, Z.; Zhang, G.; Liu, F. The value of circulation tumor DNA in predicting postoperative recurrence of colorectal cancer: A meta-analysis. Int. J. Color. Dis. 2020, 35, 1463–1475. [Google Scholar] [CrossRef]
- Stern, N.M.; Mikalsen, L.T.G.; Dueland, S.; Schulz, A.; Line, P.; Stokke, C.; Grut, H. The prognostic value of [18F]FDG PET/CT texture analysis prior to transplantation for unresectable colorectal liver metastases. Clin. Physiol. Funct. Imaging 2024, 45, e12908. [Google Scholar] [CrossRef]
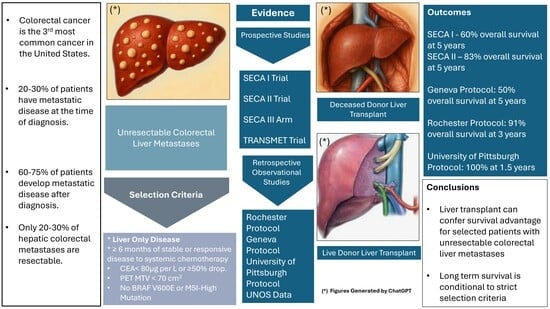
| Authors, Year of Publication, Reference | Time Period | Number of Patients | Study Design | Selection Criteria | Outcomes | Recurrence Rate | Comments |
|---|---|---|---|---|---|---|---|
| Hagness et al. 2013 SECA-I trial [11] | 2006–2011 | n = 21 | Prospective | · Resected primary CRC · ≥6 weeks of pre-LT chemotherapy · Liver-only CLMs · CEA 1–2000 µg/L · Largest tumor 28–130 mm · Metabolic tumor volume (MTV) at LT: 0–874 cm3 · No extrahepatic disease | 5-yr OS 60% | 19/21 (90%) | · No chemotherapy response requirement · Wide CEA range · Larger tumor size allowed · Excluded any extrahepatic disease |
| Dueland et al. 2020 SECA-II trial [19] | 2012–2016 | n = 15 | Prospective | · Resected primary CRC · Partial response after 6 weeks pre-LT chemo · Liver-only CLMs · Negative pre-LT colonoscopy · No lesion > 10 cm · CEA 1–30 µg/L · Largest tumor 3–47 mm · Metabolic tumor volume at LT: 0–140 cm3 | For patients with an Oslo score of 0 or 1, OS at 1, 3 and 5-year was 100%, 83% and 83% respectively. PFS was 53%, 44% and 35% respectively | 8/15 (53.3%) | · Required chemotherapy response · Tighter CEA range · Smaller maximum tumor size |
| Smedman et al. 2020 SECA-III Arm D [10] | 2014–2018 | n = 10 | Prospective | · Same as SECA-II (resected primary; response to 6 weeks chemo; liver-only; neg. colonoscopy; no tumor > 10 cm) · Allowed resectable lung metastases · CEA 2–4346 µg/L · MTV at LT: 0–201 cm3 | 2-yr OS 43% | 8/10 (80%) | · Included resectable lung metastases · Much higher allowable CEA range |
| Toso et al. 2017 [30] | 1995–2015 | n = 12 | Retrospective | · Resected primary CRC · ≥1/12 had partial response to pre-LT chemo; one patient received intraoperative chemotherapy · Median CEA 16.9 µg/L · Two patients with lesions > 5 cm · Presumed liver-only disease | 5-yr OS 50% | 6/12 (50%) | · One patient received intraoperative chemotherapy · No strict size cutoff (lesions > 5 cm allowed) |
| Hernández-Alejandro et al. 2022 [31] | 2017–2021 | n = 10 | Retrospective | · Followed IHPBA LT guidelines · Resected primary CRC · Median CEA 1.6–56.4 µg/L · Included KRAS-mutated cases (3 patients) · Liver-only disease; no explicit size thresholds | 1.5-yr OS 100% | 3/10 (30%) | · IHPBA consensus criteria · No size or response cutoffs · KRAS mutations allowed |
| Sasaki et al. 2023 [32] | 2017–2022 | n = 46 | Retrospective | · Resected primary CRC · Unresectable liver-only metastases · No other selection criteria specified | 3-yr OS 60.4% | 10/46 (22%) | · Broad inclusion: only “liver-only” requirement |
| Kaltenmeier et al. 2023 [31] | 2019–2022 | n = 10 | Retrospective | · Resected primary CRC · 6–12 wk pre-LT chemo with stable disease or partial response · Negative pre-LT colonoscopy · CEA < 100 µg/L · 5 patients had lesions > 5 cm · Liver-only disease | 1.5-yr OS 100% | 3/10 (30%) | · Required chemotherapy response · Allowed some lesions > 5 cm if chemo-responsive · Moderate CEA cutoff |
| Solheim et al. 2023 (SECA-I/II; 10-year follow-up results) [33] | 2006–2012 | n = 23 | Prospective | · Unresectable liver-only CRC metastases, complete radical resection of primary tumor · ECOG 0–1 · ≥6 weeks of chemo · No extrahepatic disease on CT/PET-CT · Exclusions: >10% weight loss; LT contraindications; other malignancies; BMI > 30 | 5-yr OS 75%; 10-yr OS 50% | 23/23 (100%) | · Only unresectable liver-only requirement · Added performance and BMI/exclusion criteria |
| Adam et al. 2024 TRANSMET trial [21] | 2016–2021 | n = 94 | Prospective | · Resected primary CRC · ECOG 0–1 · No local recurrence on colonoscopy within past 12 months · No extrahepatic disease · No BRAF mutations · CEA < 80 µg/L or ≥50% decrease from baseline · Liver-only metastases | 5-yr OS 73% | 28/47 (60%) | · Added colonoscopy requirement · Excluded BRAF-mutated tumors · CEA < 80 µg/L or ≥50% decrease from baseline criteria |
| Byrne et al. 2024 Rochester Protocol [22] | 2019–2024 | Retrospective Study | Resection of primary tumor No signs of local recurrence on colonoscopy within 12 months before LT. No radiological signs of extra-hepatic disease (CT, MRI, PET at least 6 weeks before LT). ECOG 0–1 Absence of BRAF V600E mutations Absence of high microsatellite instability Right-sided primary tumors and patients with KRAS and TP53 mutations are observed for 18 months. Serum CEA levels at listing < 80 µg/dL Rising serum CEA levels at the time of LT or progressive disease are exclusion criteria | 1-yr OS 100%, 3-yr OS 91% 1-yr DFS 100% 3-yr DFS 40% | A total of 225 patients referred for LT; 206 patients with unresectable CRM completed the initial evaluation; median age was 47 years (IQR 42–54 years); median size of the largest metastasis was 4.5 cm (IQR 2.6–7.4 cm); 135 patients were potential candidates; 23 patients underwent living donor liver transplant (LDLT). |
| Study/Source | Induction Regimen | Maintenance Regimen | Notes |
|---|---|---|---|
| SECA/Oslo experience [19] | Basiliximab + steroids + mycophenolate mofetil (MMF) + tacrolimus (first 4–6 weeks) | Convert tacrolimus → sirolimus (mTOR) with defined trough goals; steroid taper by 6 months | No adjuvant chemo post-LT in early reports |
| LDLT (multi-center U.S.) [23,31] | Tacrolimus + steroids + basiliximab | Everolimus or sirolimus (often with CNI minimization or withdrawal) | mTOR chosen for potential anti-tumor effects |
| TransMet program [21] | Steroids + tacrolimus + MMF | Taper/reduce over time per protocol | Trial protocol; details on exact timing limited |
| Narrative/consensus guidance [34] | Steroids + IL-2RA induction (basiliximab) | Tacrolimus + MMF, then switch to mTOR or CNI + mTOR combination | Recommended to minimize CNI exposure in oncologic LT cases |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demirors, B.; Sethi, V.; Abdullah, A.; Elias, C.; Spitz, F.; Mial-Anthony, J.; Packiaraj, G.; Subedi, S.; Han, S.; Fokken, T.; et al. Liver Transplantation for Unresectable Colorectal Liver Metastases: A Scoping Review on Redefining Boundaries in Transplant Oncology. Curr. Oncol. 2025, 32, 481. https://doi.org/10.3390/curroncol32090481
Demirors B, Sethi V, Abdullah A, Elias C, Spitz F, Mial-Anthony J, Packiaraj G, Subedi S, Han S, Fokken T, et al. Liver Transplantation for Unresectable Colorectal Liver Metastases: A Scoping Review on Redefining Boundaries in Transplant Oncology. Current Oncology. 2025; 32(9):481. https://doi.org/10.3390/curroncol32090481
Chicago/Turabian StyleDemirors, Berkay, Vrishketan Sethi, Abiha Abdullah, Charbel Elias, Francis Spitz, Jason Mial-Anthony, Godwin Packiaraj, Sabin Subedi, Shwe Han, Timothy Fokken, and et al. 2025. "Liver Transplantation for Unresectable Colorectal Liver Metastases: A Scoping Review on Redefining Boundaries in Transplant Oncology" Current Oncology 32, no. 9: 481. https://doi.org/10.3390/curroncol32090481
APA StyleDemirors, B., Sethi, V., Abdullah, A., Elias, C., Spitz, F., Mial-Anthony, J., Packiaraj, G., Subedi, S., Han, S., Fokken, T., & Molinari, M. (2025). Liver Transplantation for Unresectable Colorectal Liver Metastases: A Scoping Review on Redefining Boundaries in Transplant Oncology. Current Oncology, 32(9), 481. https://doi.org/10.3390/curroncol32090481