Evolving Therapeutic Landscape of ROS1-Positive Non-Small Cell Lung Cancer: An Updated Review
Simple Summary
Abstract
1. Introduction
2. ROS1 Biology
2.1. ROS1 Gene
2.2. ROS1 Fusion Detection
3. Epidemiology
4. Efficacy of Conventional Treatments
5. Treatment of ROS-1-Positive NSCLCs
5.1. Crizotinib
5.2. Entrectinib
5.3. Ceritinib
5.4. Lorlatinib
5.5. Repotrectinib
5.6. Taletrectinib
5.7. Zidesamtinib
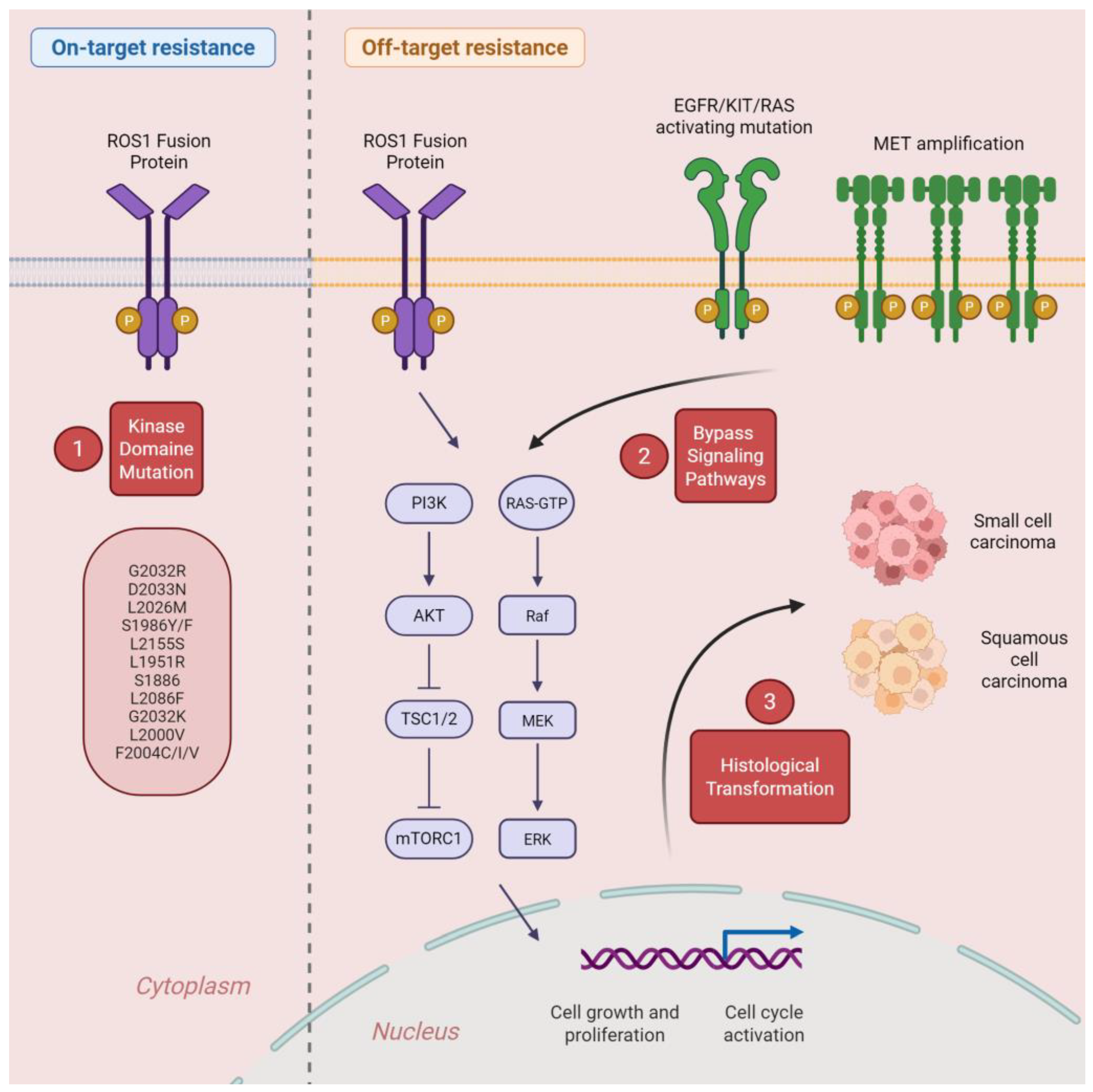
6. Mechanisms of Resistance
7. Therapeutic Strategies and Future Directions
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AE | Adverse event |
| ALK | Anaplastic lymphoma kinase |
| ALT | Alanine aminotransferase |
| AST | Aspartate aminotransferase |
| BMs | Brain metastases |
| CI | Confidence interval |
| CNS | Central nervous system |
| CPK | Creatine phosphokinase |
| DLT | Dose-limiting toxicity |
| DOR | Duration of response |
| EGFR | Epidermal growth factor receptor |
| EMA | European Medicines Agency |
| FDA | Food and Drug Administration |
| IC | Intracranial |
| IC-ORR | Intracranial overall response rate |
| ICI | Immune checkpoint inhibitor |
| IHC | Immunohistochemistry |
| ISH | In situ hybridization |
| MAPK | Mitogen-activated protein kinase |
| MET | Mesenchymal–epithelial transition factor |
| mPFS | Median progression-free survival |
| NGS | Next-generation sequencing |
| NR | Not reached |
| NSCLC | Non-small cell lung cancer |
| ORR | Overall response rate |
| OS | Overall survival |
| PCR | Polymerase chain reaction |
| PD-L1 | Programmed death-ligand 1 |
| PFS | Progression-free survival |
| QT | QT interval on electrocardiogram |
| RT-PCR | Reverse transcription polymerase chain reaction |
| TKI | Tyrosine kinase inhibitor |
| TRK | Tropomyosin receptor kinase |
References
- Bray, F.; Laversanne, M.; Sung, H.; Ferlay, J.; Siegel, R.L.; Soerjomataram, I.; Jemal, A. Global Cancer Statistics 2022: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2024, 74, 229–263. [Google Scholar] [CrossRef]
- Shiels, M.S.; Graubard, B.I.; McNeel, T.S.; Kahle, L.; Freedman, N.D. Trends in Smoking-Attributable and Smoking-Unrelated Lung Cancer Death Rates in the United States, 1991–2018. J. Natl. Cancer Inst. 2024, 116, 711–716. [Google Scholar] [CrossRef]
- Rikova, K.; Guo, A.; Zeng, Q.; Possemato, A.; Yu, J.; Haack, H.; Nardone, J.; Lee, K.; Reeves, C.; Li, Y.; et al. Global Survey of Phosphotyrosine Signaling Identifies Oncogenic Kinases in Lung Cancer. Cell 2007, 131, 1190–1203. [Google Scholar] [CrossRef]
- Gendarme, S.; Bylicki, O.; Chouaid, C.; Guisier, F. ROS-1 Fusions in Non-Small-Cell Lung Cancer: Evidence to Date. Curr. Oncol. 2022, 29, 641–658. [Google Scholar] [CrossRef] [PubMed]
- Charest, A.; Wilker, E.W.; McLaughlin, M.E.; Lane, K.; Gowda, R.; Coven, S.; McMahon, K.; Kovach, S.; Feng, Y.; Yaffe, M.B.; et al. ROS Fusion Tyrosine Kinase Activates a SH2 Domain-Containing Phosphatase-2/Phosphatidylinositol 3-Kinase/Mammalian Target of Rapamycin Signaling Axis to Form Glioblastoma in Mice. Cancer Res. 2006, 66, 7473–7481. [Google Scholar] [CrossRef] [PubMed]
- Ou, S.-H.I.; Tan, J.; Yen, Y.; Soo, R.A. ROS1 as a “druggable” Receptor Tyrosine Kinase: Lessons Learned from Inhibiting the ALK Pathway. Expert Rev. Anticancer Ther. 2012, 12, 447–456. [Google Scholar] [CrossRef] [PubMed]
- Dugay, F.; Llamas-Gutierrez, F.; Gournay, M.; Medane, S.; Mazet, F.; Chiforeanu, D.C.; Becker, E.; Lamy, R.; Léna, H.; Rioux-Leclercq, N.; et al. Clinicopathological Characteristics of ROS1- and RET-Rearranged NSCLC in Caucasian Patients: Data from a Cohort of 713 Non-Squamous NSCLC Lacking KRAS/EGFR/HER2/BRAF/PIK3CA/ALK Alterations. Oncotarget 2017, 8, 53336–53351. [Google Scholar] [CrossRef]
- Bergethon, K.; Shaw, A.T.; Ou, S.-H.I.; Katayama, R.; Lovly, C.M.; McDonald, N.T.; Massion, P.P.; Siwak-Tapp, C.; Gonzalez, A.; Fang, R.; et al. ROS1 Rearrangements Define a Unique Molecular Class of Lung Cancers. J. Clin. Oncol. 2012, 30, 863–870. [Google Scholar] [CrossRef]
- Ou, S.-H.I.; Nagasaka, M. A Catalog of 5’ Fusion Partners in ROS1-Positive NSCLC Circa 2020. JTO Clin. Res. Rep. 2020, 1, 100048. [Google Scholar] [CrossRef]
- Roth, J.A.; Swisher, S.G.; Merritt, J.A.; Lawrence, D.D.; Kemp, B.L.; Carrasco, C.H.; El-Naggar, A.K.; Fossella, F.V.; Glisson, B.S.; Hong, W.K.; et al. Gene Therapy for Non-Small Cell Lung Cancer: A Preliminary Report of a Phase I Trial of Adenoviral P53 Gene Replacement. Semin. Oncol. 1998, 25, 33–37. [Google Scholar]
- Lin, J.J.; Ritterhouse, L.L.; Ali, S.M.; Bailey, M.; Schrock, A.B.; Gainor, J.F.; Ferris, L.A.; Mino-Kenudson, M.; Miller, V.A.; Iafrate, A.J.; et al. ROS1 Fusions Rarely Overlap with Other Oncogenic Drivers in Non-Small Cell Lung Cancer. J. Thorac. Oncol. 2017, 12, 872–877. [Google Scholar] [CrossRef]
- Lindeman, N.I.; Cagle, P.T.; Aisner, D.L.; Arcila, M.E.; Beasley, M.B.; Bernicker, E.H.; Colasacco, C.; Dacic, S.; Hirsch, F.R.; Kerr, K.; et al. Updated Molecular Testing Guideline for the Selection of Lung Cancer Patients for Treatment with Targeted Tyrosine Kinase Inhibitors: Guideline from the College of American Pathologists, the International Association for the Study of Lung Cancer, and the Association for Molecular Pathology. Arch. Pathol. Lab. Med. 2018, 142, 321–346. [Google Scholar] [CrossRef]
- Bubendorf, L.; Büttner, R.; Al-Dayel, F.; Dietel, M.; Elmberger, G.; Kerr, K.; López-Ríos, F.; Marchetti, A.; Öz, B.; Pauwels, P.; et al. Testing for ROS1 in Non-Small Cell Lung Cancer: A Review with Recommendations. Virchows Arch. 2016, 469, 489–503. [Google Scholar] [CrossRef]
- Wilcock, D.M.; Schmidt, R.L.; Furtado, L.V.; Matynia, A.P.; Deftereos, G.; Sirohi, D. Histologic and Molecular Characterization of Non-Small Cell Lung Carcinoma with Discordant ROS1 Immunohistochemistry and Fluorescence In Situ Hybridization. Appl. Immunohistochem. Mol. Morphol. 2022, 30, 19–26. [Google Scholar] [CrossRef]
- Zito Marino, F.; Rossi, G.; Cozzolino, I.; Montella, M.; Micheli, M.; Bogina, G.; Munari, E.; Brunelli, M.; Franco, R. Multiplex Fluorescence in Situ Hybridisation to Detect Anaplastic Lymphoma Kinase and ROS Proto-Oncogene 1 Receptor Tyrosine Kinase Rearrangements in Lung Cancer Cytological Samples. J. Clin. Pathol. 2020, 73, 96–101. [Google Scholar] [CrossRef]
- Makarem, M.; Ezeife, D.A.; Smith, A.C.; Li, J.J.N.; Law, J.H.; Tsao, M.-S.; Leighl, N.B. Reflex ROS1 IHC Screening with FISH Confirmation for Advanced Non-Small Cell Lung Cancer—A Cost-Efficient Strategy in a Public Healthcare System. Curr. Oncol. 2021, 28, 3268–3279. [Google Scholar] [CrossRef]
- Rojo, F.; Conde, E.; Torres, H.; Cabezón-Gutiérrez, L.; Bautista, D.; Ramos, I.; Carcedo, D.; Arrabal, N.; García, J.F.; Galán, R.; et al. Clinical and Economic Impact of “ROS1-Testing” Strategy Compared to a “No-ROS1-Testing” Strategy in Advanced NSCLC in Spain. BMC Cancer 2022, 22, 292. [Google Scholar] [CrossRef] [PubMed]
- Stroyakovskiy, D.; Shilo, P.; Danilova, A.; Andreiashkina, I.; Zhukova, L.; Smolin, S.; Fedyanin, M.; Pokataev, I.; Akhmadiyarova, Y.; Sokolov, N. Real-World Data on the Treatment of Patients with ROS1-Positive Non-Small Cell Lung Cancer. J. Clin. Oncol. 2025, 43, e20685. [Google Scholar] [CrossRef]
- Patil, T.; Smith, D.E.; Bunn, P.A.; Aisner, D.L.; Le, A.T.; Hancock, M.; Purcell, W.T.; Bowles, D.W.; Camidge, D.R.; Doebele, R.C. The Incidence of Brain Metastases in Stage IV ROS1-Rearranged Non-Small Cell Lung Cancer and Rate of Central Nervous System Progression on Crizotinib. J. Thorac. Oncol. 2018, 13, 1717–1726. [Google Scholar] [CrossRef] [PubMed]
- Digumarthy, S.R.; Mendoza, D.P.; Lin, J.J.; Chen, T.; Rooney, M.M.; Chin, E.; Sequist, L.V.; Lennerz, J.K.; Gainor, J.F.; Shaw, A.T. Computed Tomography Imaging Features and Distribution of Metastases in ROS1-Rearranged Non-Small-Cell Lung Cancer. Clin. Lung Cancer 2020, 21, 153–159.e3. [Google Scholar] [CrossRef]
- Riess, J.W.; Padda, S.K.; Bangs, C.D.; Das, M.; Neal, J.W.; Adrouny, A.R.; Cherry, A.; Wakelee, H.A. A Case Series of Lengthy Progression-Free Survival with Pemetrexed-Containing Therapy in Metastatic Non–Small-Cell Lung Cancer Patients Harboring ROS1 Gene Rearrangements. Clin. Lung Cancer 2013, 14, 592–595. [Google Scholar] [CrossRef]
- Chen, Y.-F.; Hsieh, M.-S.; Wu, S.-G.; Chang, Y.-L.; Yu, C.-J.; Yang, J.C.-H.; Yang, P.-C.; Shih, J.-Y. Efficacy of Pemetrexed-Based Chemotherapy in Patients with ROS1 Fusion-Positive Lung Adenocarcinoma Compared with in Patients Harboring Other Driver Mutations in East Asian Populations. J. Thorac. Oncol. 2016, 11, 1140–1152. [Google Scholar] [CrossRef]
- Mazières, J.; Zalcman, G.; Crinò, L.; Biondani, P.; Barlesi, F.; Filleron, T.; Dingemans, A.-M.C.; Léna, H.; Monnet, I.; Rothschild, S.I.; et al. Crizotinib Therapy for Advanced Lung Adenocarcinoma and a ROS1 Rearrangement: Results from the EUROS1 Cohort. J. Clin. Oncol. 2015, 33, 992–999. [Google Scholar] [CrossRef]
- Lee, C.K.; Man, J.; Lord, S.; Links, M.; Gebski, V.; Mok, T.; Yang, J.C.-H. Checkpoint Inhibitors in Metastatic EGFR-Mutated Non-Small Cell Lung Cancer—A Meta-Analysis. J. Thorac. Oncol. 2017, 12, 403–407. [Google Scholar] [CrossRef] [PubMed]
- Hastings, K.; Yu, H.A.; Wei, W.; Sanchez-Vega, F.; DeVeaux, M.; Choi, J.; Rizvi, H.; Lisberg, A.; Truini, A.; Lydon, C.A.; et al. EGFR Mutation Subtypes and Response to Immune Checkpoint Blockade Treatment in Non-Small-Cell Lung Cancer. Ann. Oncol. 2019, 30, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Yan, H.; Zeng, L.; Xu, Q.; Guo, W.; Lin, S.; Jiang, W.; Wang, Z.; Deng, L.; Qin, H.; et al. Efficacy of Immune Checkpoint Inhibitor Plus Chemotherapy in Patients with ROS1-Rearranged Advanced Lung Adenocarcinoma: A Multicenter, Retrospective Cohort Study. JCO Precis. Oncol. 2023, 7, e2200614. [Google Scholar] [CrossRef]
- Lee, J.; Park, C.K.; Yoon, H.-K.; Sa, Y.J.; Woo, I.S.; Kim, H.R.; Kim, S.Y.; Kim, T.-J. PD-L1 Expression in ROS1-Rearranged Non-Small Cell Lung Cancer: A Study Using Simultaneous Genotypic Screening of EGFR, ALK, and ROS1. Thorac. Cancer 2019, 10, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, N.J.; Schneider, J.L.; Patil, T.; Zhu, V.W.; Goldman, D.A.; Yang, S.-R.; Falcon, C.J.; Do, A.; Nie, Y.; Plodkowski, A.J.; et al. Response to Immune Checkpoint Inhibition as Monotherapy or in Combination with Chemotherapy in Metastatic ROS1-Rearranged Lung Cancers. JTO Clin. Res. Rep. 2021, 2, 100187. [Google Scholar] [CrossRef]
- Shaw, A.T.; Ou, S.-H.I.; Bang, Y.-J.; Camidge, D.R.; Solomon, B.J.; Salgia, R.; Riely, G.J.; Varella-Garcia, M.; Shapiro, G.I.; Costa, D.B.; et al. Crizotinib in ROS1-Rearranged Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2014, 371, 1963–1971. [Google Scholar] [CrossRef]
- Shaw, A.T.; Riely, G.J.; Bang, Y.-J.; Kim, D.-W.; Camidge, D.R.; Solomon, B.J.; Varella-Garcia, M.; Iafrate, A.J.; Shapiro, G.I.; Usari, T.; et al. Crizotinib in ROS1-Rearranged Advanced Non–Small-Cell Lung Cancer (NSCLC): Updated Results, Including Overall Survival, from PROFILE 1001. Ann. Oncol. 2019, 30, 1121–1126. [Google Scholar] [CrossRef]
- Wu, Y.-L.; Yang, J.C.-H.; Kim, D.-W.; Lu, S.; Zhou, J.; Seto, T.; Yang, J.-J.; Yamamoto, N.; Ahn, M.-J.; Takahashi, T.; et al. Phase II Study of Crizotinib in East Asian Patients with ROS1-Positive Advanced Non-Small-Cell Lung Cancer. J. Clin. Oncol. 2018, 36, 1405–1411. [Google Scholar] [CrossRef]
- Moro-Sibilot, D.; Cozic, N.; Pérol, M.; Mazières, J.; Otto, J.; Souquet, P.J.; Bahleda, R.; Wislez, M.; Zalcman, G.; Guibert, S.D.; et al. Crizotinib in C-MET- or ROS1-Positive NSCLC: Results of the AcSé Phase II Trial. Ann. Oncol. 2019, 30, 1985–1991. [Google Scholar] [CrossRef] [PubMed]
- Michels, S.; Massutí, B.; Schildhaus, H.-U.; Franklin, J.; Sebastian, M.; Felip, E.; Grohé, C.; Rodriguez-Abreu, D.; Abdulla, D.S.Y.; Bischoff, H.; et al. Safety and Efficacy of Crizotinib in Patients with Advanced or Metastatic ROS1-Rearranged Lung Cancer (EUCROSS): A European Phase II Clinical Trial. J. Thorac. Oncol. 2019, 14, 1266–1276. [Google Scholar] [CrossRef] [PubMed]
- Landi, L.; Chiari, R.; Tiseo, M.; D’Incà, F.; Dazzi, C.; Chella, A.; Delmonte, A.; Bonanno, L.; Giannarelli, D.; Cortinovis, D.L.; et al. Crizotinib in MET-Deregulated or ROS1-Rearranged Pretreated Non-Small Cell Lung Cancer (METROS): A Phase II, Prospective, Multicenter, Two-Arms Trial. Clin. Cancer Res. 2019, 25, 7312–7319. [Google Scholar] [CrossRef] [PubMed]
- Ardini, E.; Menichincheri, M.; Banfi, P.; Bosotti, R.; De Ponti, C.; Pulci, R.; Ballinari, D.; Ciomei, M.; Texido, G.; Degrassi, A.; et al. Entrectinib, a Pan-TRK, ROS1, and ALK Inhibitor with Activity in Multiple Molecularly Defined Cancer Indications. Mol. Cancer Ther. 2016, 15, 628–639. [Google Scholar] [CrossRef]
- Drilon, A.; Chiu, C.-H.; Fan, Y.; Cho, B.C.; Lu, S.; Ahn, M.-J.; Krebs, M.G.; Liu, S.V.; John, T.; Otterson, G.A.; et al. Long-Term Efficacy and Safety of Entrectinib in ROS1 Fusion-Positive NSCLC. JTO Clin. Res. Rep. 2022, 3, 100332. [Google Scholar] [CrossRef]
- Huang, E.J.; Reichardt, L.F. Neurotrophins: Roles in Neuronal Development and Function. Annu. Rev. Neurosci. 2001, 24, 677–736. [Google Scholar] [CrossRef]
- Lim, S.M.; Kim, H.R.; Lee, J.-S.; Lee, K.H.; Lee, Y.-G.; Min, Y.J.; Cho, E.K.; Lee, S.S.; Kim, B.-S.; Choi, M.Y.; et al. Open-Label, Multicenter, Phase II Study of Ceritinib in Patients with Non-Small-Cell Lung Cancer Harboring ROS1 Rearrangement. J. Clin. Oncol. 2017, 35, 2613–2618. [Google Scholar] [CrossRef]
- Crinò, L.; Ahn, M.-J.; De Marinis, F.; Groen, H.J.M.; Wakelee, H.; Hida, T.; Mok, T.; Spigel, D.; Felip, E.; Nishio, M.; et al. Multicenter Phase II Study of Whole-Body and Intracranial Activity with Ceritinib in Patients with ALK-Rearranged Non-Small-Cell Lung Cancer Previously Treated with Chemotherapy and Crizotinib: Results from ASCEND-2. J. Clin. Oncol. 2016, 34, 2866–2873. [Google Scholar] [CrossRef]
- Shaw, A.T.; Solomon, B.J.; Chiari, R.; Riely, G.J.; Besse, B.; Soo, R.A.; Kao, S.; Lin, C.-C.; Bauer, T.M.; Clancy, J.S.; et al. Lorlatinib in Advanced ROS1-Positive Non-Small-Cell Lung Cancer: A Multicentre, Open-Label, Single-Arm, Phase 1-2 Trial. Lancet Oncol. 2019, 20, 1691–1701. [Google Scholar] [CrossRef]
- Ahn, B.C.; Kim, Y.J.; Kim, D.-W.; Lee, K.H.; Lee, Y.; Han, J.-Y. Lorlatinib in TKI Naïve, Advanced ROS1-Positive Non-Small-Cell Lung Cancer: A Multicenter, Open-Label, Single-Arm, Phase 2 Trial. J. Clin. Oncol. 2024, 42, 8519. [Google Scholar] [CrossRef]
- Girard, N.; Galland-Girodet, S.; Avrillon, V.; Besse, B.; Duruisseaux, M.; Cadranel, J.; Otto, J.; Prevost, A.; Roch, B.; Bennouna, J.; et al. Lorlatinib for Advanced ROS1+ Non-Small-Cell Lung Cancer: Results of the IFCT-1803 LORLATU Study. ESMO Open 2022, 7, 100418. [Google Scholar] [CrossRef]
- Lin, J.J.; Choudhury, N.J.; Yoda, S.; Zhu, V.W.; Johnson, T.W.; Sakhtemani, R.; Dagogo-Jack, I.; Digumarthy, S.R.; Lee, C.; Do, A.; et al. Spectrum of Mechanisms of Resistance to Crizotinib and Lorlatinib in ROS1 Fusion-Positive Lung Cancer. Clin. Cancer Res. 2021, 27, 2899–2909. [Google Scholar] [CrossRef]
- Ou, S.-H.I.; Hagopian, G.G.; Zhang, S.S.; Nagasaka, M. Comprehensive Review of ROS1 Tyrosine Kinase Inhibitors-Classified by Structural Designs and Mutation Spectrum (Solvent Front Mutation [G2032R] and Central β-Sheet 6 [Cβ6] Mutation [L2086F]). J. Thorac. Oncol. 2024, 19, 706–718. [Google Scholar] [CrossRef]
- Drilon, A.; Ou, S.-H.I.; Cho, B.C.; Kim, D.-W.; Lee, J.; Lin, J.J.; Zhu, V.W.; Ahn, M.-J.; Camidge, D.R.; Nguyen, J.; et al. Repotrectinib (TPX-0005) Is a Next-Generation ROS1/TRK/ALK Inhibitor That Potently Inhibits ROS1/TRK/ALK Solvent-Front Mutations. Cancer Discov. 2018, 8, 1227–1236. [Google Scholar] [CrossRef]
- Drilon, A.; Camidge, D.R.; Lin, J.J.; Kim, S.-W.; Solomon, B.J.; Dziadziuszko, R.; Besse, B.; Goto, K.; de Langen, A.J.; Wolf, J.; et al. Repotrectinib in ROS1 Fusion-Positive Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2024, 390, 118–131. [Google Scholar] [CrossRef]
- Nagasaka, M.; Ohe, Y.; Zhou, C.; Choi, C.-M.; Yang, N.; Liu, G.; Felip, E.; Pérol, M.; Besse, B.; Nieva, J.; et al. TRUST-II: A Global Phase II Study of Taletrectinib in ROS1-Positive Non-Small-Cell Lung Cancer and Other Solid Tumors. Future Oncol. 2023, 19, 123–135. [Google Scholar] [CrossRef]
- Li, W.; Xiong, A.; Yang, N.; Fan, H.; Yu, Q.; Zhao, Y.; Wang, Y.; Meng, X.; Wu, J.; Wang, Z.; et al. Efficacy and Safety of Taletrectinib in Chinese Patients with ROS1+ Non-Small Cell Lung Cancer: The Phase II TRUST-I Study. J. Clin. Oncol. 2024, 42, 2660–2670. [Google Scholar] [CrossRef] [PubMed]
- Pérol, M.; Li, W.; Lai, R.K.; Zhou, C. Reply to: “Baseline Brain Metastasis Imbalance in TRUST: Implications for Interpreting Intracranial Efficacy of Taletrectinib” and “Taletrectinib in ROS1 Fusion–Positive Non–Small Cell Lung Cancer: Promising Yet Premature?”. J. Clin. Oncol. 2025, 43, 3227–3228. [Google Scholar] [CrossRef] [PubMed]
- Pérol, M.; Li, W.; Pennell, N.A.; Liu, G.; Ohe, Y.; De Braud, F.; Nagasaka, M.; Felip, E.; Xiong, A.; Zhang, Y.; et al. Taletrectinib in ROS1+ Non-Small Cell Lung Cancer: TRUST. J. Clin. Oncol. 2025, 43, 1920–1929. [Google Scholar] [CrossRef] [PubMed]
- Perol, M.; Li, W.; Pennell, N.A.; Liu, G.; De Braud, F.G.M.; Nagasaka, M.; Felip, E.; Xiong, A.; Zhang, Y.; Fan, H.; et al. Comparable Efficacy and Safety of Taletrectinib for Advanced ROS1+ Non–Small Cell Lung Cancer across Pivotal Studies and between Races and World Regions. J. Clin. Oncol. 2025, 43, 8643. [Google Scholar] [CrossRef]
- Drilon, A.; Horan, J.C.; Tangpeerachaikul, A.; Besse, B.; Ou, S.-H.I.; Gadgeel, S.M.; Camidge, D.R.; van der Wekken, A.J.; Nguyen-Phuong, L.; Acker, A.; et al. NVL-520 Is a Selective, TRK-Sparing, and Brain-Penetrant Inhibitor of ROS1 Fusions and Secondary Resistance Mutations. Cancer Discov. 2023, 13, 598–615. [Google Scholar] [CrossRef]
- Besse, B.; Drilon, A.E.; Cho, B.C.; Camidge, D.R.; Neal, J.; Lin, C.-C.; Liu, S.V.; Nagasaka, M.; Kao, S.C.-H.; Felip, E.; et al. 1256MO Phase I/II ARROS-1 Study of Zidesamtinib (NVL-520) in ROS1 Fusion-Positive Solid Tumours. Ann. Oncol. 2024, 35, S804–S805. [Google Scholar] [CrossRef]
- Nuvalent Inc. Nuvalent Presents Pivotal Data from ARROS-1 Clinical Trial of Zidesamtinib for TKI Pre-Treated Patients with Advanced ROS1-Positive NSCLC at WCLC 2025. Available online: https://www.prnewswire.com/news-releases/nuvalent-presents-pivotal-data-from-arros-1-clinical-trial-of-zidesamtinib-for-tki-pre-treated-patients-with-advanced-ros1-positive-nsclc-at-wclc-2025-302548220.html (accessed on 17 September 2025).
- Dagogo-Jack, I.; Rooney, M.; Nagy, R.J.; Lin, J.J.; Chin, E.; Ferris, L.A.; Ackil, J.; Lennerz, J.K.; Lanman, R.B.; Gainor, J.F.; et al. Molecular Analysis of Plasma from Patients with ROS1-Positive NSCLC. J. Thorac. Oncol. 2019, 14, 816–824. [Google Scholar] [CrossRef]
- McCoach, C.E.; Le, A.T.; Gowan, K.; Jones, K.; Schubert, L.; Doak, A.; Estrada-Bernal, A.; Davies, K.D.; Merrick, D.T.; Bunn, P.A.; et al. Resistance Mechanisms to Targeted Therapies in ROS1 + and ALK + Non–Small Cell Lung Cancer. Clin. Cancer Res. 2018, 24, 3334–3347. [Google Scholar] [CrossRef]
- Gainor, J.F.; Tseng, D.; Yoda, S.; Dagogo-Jack, I.; Friboulet, L.; Lin, J.J.; Hubbeling, H.G.; Dardaei, L.; Farago, A.F.; Schultz, K.R.; et al. Patterns of Metastatic Spread and Mechanisms of Resistance to Crizotinib in ROS1-Positive Non-Small-Cell Lung Cancer. JCO Precis. Oncol. 2017, 2017, PO.17.00063. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Zhang, X.; Chen, H.; Bao, H.; Wu, X.; Wang, H.; Bao, H.; Pang, J.; Wang, S.; Wang, J. Mechanisms of Resistance to Tyrosine Kinase Inhibitors in ROS1 Fusion-Positive Nonsmall Cell Lung Cancer. Clin. Chem. 2024, 70, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Tyler, L.C.; Le, A.T.; Chen, N.; Nijmeh, H.; Bao, L.; Wilson, T.R.; Chen, D.; Simmons, B.; Turner, K.M.; Perusse, D.; et al. MET Gene Amplification Is a Mechanism of Resistance to Entrectinib in ROS1 + NSCLC. Thorac. Cancer 2022, 13, 3032–3041. [Google Scholar] [CrossRef]
- Cargnelutti, M.; Corso, S.; Pergolizzi, M.; Mévellec, L.; Aisner, D.L.; Dziadziuszko, R.; Varella-Garcia, M.; Comoglio, P.M.; Doebele, R.C.; Vialard, J.; et al. Activation of RAS Family Members Confers Resistance to ROS1 Targeting Drugs. Oncotarget 2015, 6, 5182–5194. [Google Scholar] [CrossRef]
- Dziadziuszko, R.; Le, A.T.; Wrona, A.; Jassem, J.; Camidge, D.R.; Varella-Garcia, M.; Aisner, D.L.; Doebele, R.C. An Activating KIT Mutation Induces Crizotinib Resistance in ROS1-Positive Lung Cancer. J. Thorac. Oncol. 2016, 11, 1273–1281. [Google Scholar] [CrossRef]
- Sato, H.; Schoenfeld, A.J.; Siau, E.; Lu, Y.C.; Tai, H.; Suzawa, K.; Kubota, D.; Lui, A.J.W.; Qeriqi, B.; Mattar, M.; et al. MAPK Pathway Alterations Correlate with Poor Survival and Drive Resistance to Therapy in Patients with Lung Cancers Driven by ROS1 Fusions. Clin. Cancer Res. 2020, 26, 2932–2945. [Google Scholar] [CrossRef]
- Lin, J.J.; Langenbucher, A.; Gupta, P.; Yoda, S.; Fetter, I.J.; Rooney, M.; Do, A.; Kem, M.; Chang, K.P.; Oh, A.Y.; et al. Small Cell Transformation of ROS1 Fusion-Positive Lung Cancer Resistant to ROS1 Inhibition. npj Precis. Oncol. 2020, 4, 21. [Google Scholar] [CrossRef]
- Waliany, S.; Do, A.; Peterson, J.; Liang, J.; Hata, A.N.; Dagogo-Jack, I.; Gainor, J.F.; Lin, J.J. Shifting Landscape of Resistance to Next-Generation ALK Inhibitors with Evolving Treatment Paradigm in ALK+ Lung Cancer. J. Clin. Oncol. 2025, 43, 8607. [Google Scholar] [CrossRef]
- An, Z.; Yang, X.; Zhu, Y.; Zhao, D.; Chen, C.; Dai, C. Outcomes After Neoadjuvant Targeted Therapy in Patients with ALK or ROS1 Rearrangement Locally Advanced Non-Small Cell Lung Cancer. Ann. Surg. Oncol. 2025, 32, 7874–7882. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Shi, Y.; Li, R. Surgical Treatment of IIIb (N2), ROS1(+) Non-Small Cell Lung Cancer after Neoadjuvant Chemotherapy Combined with Targeted Therapy: A Case Report. AME Case Rep. 2025, 9, 83. [Google Scholar] [CrossRef]
- Chen, A.-G.; Chen, D.-S.; Li, S.; Zhao, L.; Xiao, M.-Z. Case Report: Adjuvant Crizotinib Therapy Exerted Favorable Survival Benefit in a Resectable Stage IIIA NSCLC Patient with Novel LDLR-ROS1 Fusion. Front. Oncol. 2022, 12, 837219. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, J.B.; Urbanska, E.; Melchior, L.; Santoni-Rugiu, E. P46.03 Targeting ROS1 Gene Rearrangement by Crizotinib as Neoadjuvant Treatment Before Definitive Radiotherapy in Locally Advanced NSCLC. J. Thorac. Oncol. 2021, 16, S1094. [Google Scholar] [CrossRef]
- Wu, Y.-L.; Dziadziuszko, R.; Ahn, J.S.; Barlesi, F.; Nishio, M.; Lee, D.H.; Lee, J.-S.; Zhong, W.; Horinouchi, H.; Mao, W.; et al. Alectinib in Resected ALK-Positive Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2024, 390, 1265–1276. [Google Scholar] [CrossRef]
| Drug [Reference] | Targets | n | TKI-Naive ORR (%) | mPFS (Months) | IC-ORR (%) |
|---|---|---|---|---|---|
| Crizotinib [29] | ROS1, ALK, MET | 50 | 72 | 19.2 | / |
| Entrectinib [36] | ROS1, TRK, ALK | 168 | 68 | 15.7 | 80 a |
| Ceritinib [38] | ROS1, ALK | 30 | 67 | 19.3 | 25 b |
| Lorlatinib [40] | ROS1, ALK | 21 | 62 | 21.0 | 64 c |
| Repotrectinib [46] | ROS1, TRK, ALK | 71 | 79 | 35.7 | 89 d |
| Taletrectinib [50] | ROS1 (TRK-sparing) | 160 | 88.8 | 45.6 | 76.5 e |
| Drug [Reference] | Notable AEs | Main Grade ≥ 3 AEs (%) | Discontinuation for AEs (%) |
|---|---|---|---|
| Crizotinib [29] | Visual disturbances (82%), diarrhea (44%), nausea (40%), peripheral edema (40%) | Hypophosphatemia (10%), neutropenia (10%) | <1 |
| Entrectinib [36] | Dysgeusia (40%), dizziness (37%), constipation (32%), paresthesia (18%) | Weight gain (11%) | 5 |
| Lorlatinib [40] | Hypercholesterolemia (80%), cognitive effects (27%), mood disorders (16%), dizziness (12%), weight gain (23%) | Hypercholesterolemia (15%), hypertriglyceridemia (19%), weight gain (7%) | 1 |
| Repotrectinib [46] | Dizziness (58%), dysgeusia (50%), paresthesia (30%) | Dizziness (3%), anemia (4%) | 7 |
| Taletrectinib [50] | Increased AST/ALT (70%), diarrhea (61%), nausea (44%), vomiting (41%), dizziness (15%), QT prolongation (18%) | Increased AST/ALT (9%), diarrhea (3%), anemia (3%), QT prolongation (3%), neutropenia (4%) | 2 |
| Zidesamtinib a | Peripheral edema (36%), constipation (17%), CPK elevation (16%), fatigue (16%), dyspnea (15%) | CPK elevation (3%), dyspnea (3%) | 2 |
| Drug [Reference] | Targets | n | TKI-Pretreated ORR (%) | mPFS (Months) | IC-ORR (%) | ORR if G2032R Mutation (%) |
|---|---|---|---|---|---|---|
| Lorlatinib [40] | ROS1, ALK | 40 | 62 | 8.5 | 50 | 0 |
| Repotrectinib [46] | ROS1, TRK, ALK | 56 | 38 | 9.0 | 38 | 59 |
| Taletrectinib [50] | ROS1 (TRK-sparing) | 113 | 55.8 | 9.7 | 65.6 | 67 |
| Zidesamtinib a | ROS1 (TRK-sparing) | 117 | 44 | 9.7 | 48 | 83 |
| Trial | Population | Phase | Planned Enrollment (n) | Study Drugs | Primary Endpoint | Status |
|---|---|---|---|---|---|---|
| NCT06564324 (TRUST-III) | TKI-naive | III | 138 | Taletrectinib vs. Crizotinib | PFS | Recruiting |
| NCT06140836 (TRIDENT-3) | TKI-naive | III | 230 | Repotrectinib vs. Crizotinib | PFS | Recruiting |
| NCT04603807 (MO41552) | TKI-naive | III | 220 | Entrectinib vs. Crizotinib | PFS | Recruiting |
| NCT04621188 (ALBATROS) | TKI-pretreated | II | 54 | Lorlatinib (single arm) | ORR | Active, not recruiting |
| NCT05297890 | TKI-pretreated | II | 70 | Lorlatinib (single arm) | ORR | Active, not recruiting |
| NCT06128148 | NR | I | 54 | JYP0322 (single arm) | DLT | Recruiting |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bischoff, H.; Gendarme, S.; Somme, L.; Chouaid, C.; Schott, R. Evolving Therapeutic Landscape of ROS1-Positive Non-Small Cell Lung Cancer: An Updated Review. Curr. Oncol. 2025, 32, 626. https://doi.org/10.3390/curroncol32110626
Bischoff H, Gendarme S, Somme L, Chouaid C, Schott R. Evolving Therapeutic Landscape of ROS1-Positive Non-Small Cell Lung Cancer: An Updated Review. Current Oncology. 2025; 32(11):626. https://doi.org/10.3390/curroncol32110626
Chicago/Turabian StyleBischoff, Hervé, Sébastien Gendarme, Laura Somme, Christos Chouaid, and Roland Schott. 2025. "Evolving Therapeutic Landscape of ROS1-Positive Non-Small Cell Lung Cancer: An Updated Review" Current Oncology 32, no. 11: 626. https://doi.org/10.3390/curroncol32110626
APA StyleBischoff, H., Gendarme, S., Somme, L., Chouaid, C., & Schott, R. (2025). Evolving Therapeutic Landscape of ROS1-Positive Non-Small Cell Lung Cancer: An Updated Review. Current Oncology, 32(11), 626. https://doi.org/10.3390/curroncol32110626






