The Potential of Proton Therapy for Locally Advanced Breast Cancer: Clinical and Technical Considerations
Abstract
1. Background
2. Current Challenges in Breast Radiotherapy Delivery
3. Rationale for Proton Therapy
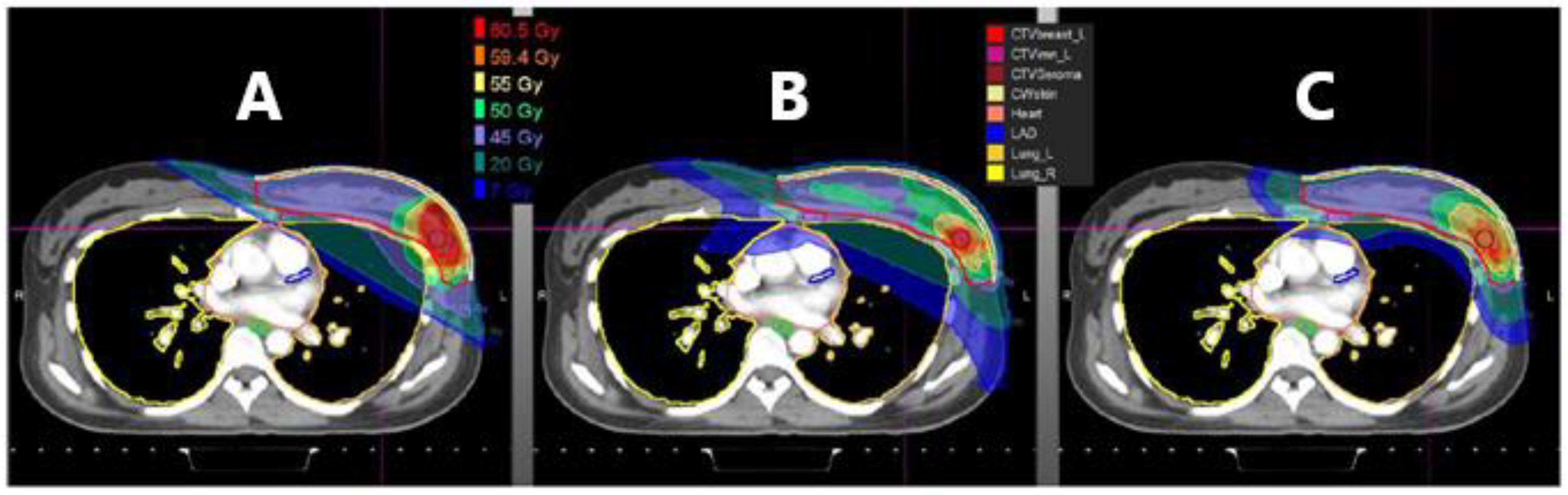
4. Areas of Uncertainty in Proton Radiation Implementation
5. Ongoing Trials
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Barton, M.B.; Jacob, S.; Shafiq, J.; Wong, K.; Thompson, S.R.; Hanna, T.P.; Delaney, G.P. Estimating the demand for radiotherapy from the evidence: A review of changes from 2003 to 2012. Radiother. Oncol. 2014, 112, 140–144. [Google Scholar] [CrossRef]
- Brown, A.; Suit, H. The centenary of the discovery of the Bragg peak. Radiother. Oncol. 2004, 73, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.R. Radiological Use of Fast Protons. Radiology 1946, 47, 487–491. [Google Scholar] [CrossRef]
- Lawrence, J.H. Proton irradiation of the pituitary. Cancer 1957, 10, 795–798. [Google Scholar] [CrossRef] [PubMed]
- Munzenrider, J.E.; Austin-Seymour, M.; Blitzer, P.J.; Gentry, R.; Goitein, M.; Gragoudas, E.S.; Johnson, K.; Koehler, A.M.; McNulty, P.; Moulton, G. Proton therapy at Harvard. Strahlentherapie 1985, 161, 756–763. [Google Scholar] [PubMed]
- Slater, J.M.; Archambeau, J.O.; Miller, D.W.; Notarus, M.I.; Preston, W.; Slater, J.D. The proton treatment center at Loma Linda University Medical Center: Rationale for and description of its development. IJROBP 1992, 22, 383–389. [Google Scholar] [CrossRef]
- Particle Therapy Co-Operative Group. Available online: https://www.ptcog.ch/index.php/other-news (accessed on 16 January 2023).
- Arnold, M.; Morgan, E.; Rumgay, H.; Mafra, A.; Singh, D.; Laversanne, M.; Vignat, J.; Gralow, J.R.; Cardoso, F.; Siesling, S.; et al. Current and future burden of breast cancer: Global statistics for 2020 and 2040. Breast 2022, 66, 15–23. [Google Scholar] [CrossRef]
- Early Breast Cancer Trialists’ Collaborative Group (EBCTCG); Darby, S.; McGale, P.; Correa, C.; Taylor, C.; Arriagada, R.; Clarke, M.; Cutter, D.; Davies, C.; Ewertz, M.; et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: Meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet 2011, 378, 1707–1716. [Google Scholar]
- Njeh, C.F.; Saunders, M.W.; Langton, C.M. Accelerated Partial BReast Irradiation (APBI): A review of available techniques. Radiat. Oncol. 2010, 5, 90. [Google Scholar] [CrossRef]
- Bergom, C.; Currey, A.; Desai, N.; Tai, A.; Strauss, J. Deep Inspiration Breath Hold: Techniques and Advantages for Cardiac Sparing During Breast Cancer Irradiation. Front. Oncol. 2018, 8, 87. [Google Scholar] [CrossRef]
- Brunt, A.M.; Haviland, J.S.; Wheatley, D.A.; Sydenham, M.A.; Alhasso, A.; Bloomfield, D.J.; Chan, C.; Churn, M.; Cleator, S.; Coles, C.E.; et al. Hypofractionated breast radiotehrapy for 1 week versus 3 weeks (FAST-Forward): 5-year efficacy and late normal tissue effects results from a multicentre, non-inferiority, randomised, phase 3 trial. Lancet 2020, 395, 1613–1626. [Google Scholar] [CrossRef] [PubMed]
- Whelan, T.J.; Olivotto, I.A.; Parulekar, W.E.; Ackerman, I.; Chua, B.H.; Nabid, A.; Vallis, K.A.; White, J.R.; Rousseau, P.; Fortin, A.; et al. Regional Nodal Irradiation in Early Stage Breast Cancer. NEJM 2015, 373, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Poortmans, P.M.; Collette, S.; Kirkove, C.; Van Limbergen, E.; Budach, V.; Struikmans, H.; Collette, L.; Fourquet, A.; Maingon, P.; Valli, M.; et al. Internal mammary and medial supraclavicular irradiation in breast cancer. NEJM 2015, 373, 317–327. [Google Scholar] [CrossRef]
- Darby, S.C.; Ewertz, M.; McGale, P.; Bennet, A.M.; Blom-Goldman, U.; Brønnum, D.; Correa, C.; Cutter, D.; Gagliardi, G.; Gigante, B.; et al. Risk of Ischemic Heart Disease in Women after Radiotherapy for Breast Cancer. NEJM 2013, 368, 987–998. [Google Scholar] [CrossRef] [PubMed]
- van den Bogaard, V.A.B.; Ta, B.D.; van der Schaaf, A.; Bouma, A.B.; Middag, A.M.; Bantema-Joppe, E.J.; van Dijk, L.V.; van Dijk-Peters, F.B.; Marteijn, L.A.; de Bock, G.H.; et al. Validation and Modification of a Prediction Model for Acute Cardiac Events in Patients with Breast Cancer Treated with Radiotherapy Based on Three-Dimensional Dose Distributions to Cardiac Substructures. J. Clin. Oncol. 2017, 35, 1171–1178. [Google Scholar] [CrossRef]
- Grantzau, T.; Thomsen, M.S.; Vaeth, M.; Overgaard, J. Risk of second primary lung cancer in women after radiotherapy for breast cancer. Radiother. Oncol. 2014, 111, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Gokula, K.; Earnest, A.; Choung Wong, L. Meta-analysis of incidence of early lung toxicity in 3-dimensional conformal irradiation of breast carcinomas. Radiat. Oncol. 2013, 8, 268. [Google Scholar] [CrossRef]
- Taylor, A.; Powell, M.E.B. Intensity-modulated radiotherapy—What is it? Cancer Imaging 2004, 4, 68–73. [Google Scholar] [CrossRef]
- Stick, L.B.; Yu, J.; Maraldo, M.V.; Aznar, M.C.; Pedersen, A.N.; Bentzen, S.M.; Vogelius, I.R. Joint Estimation of Cardiac Toxicity and Recurrence Risks After Comprehensive Nodal Photon Versus Proton Therapy for Breast Cancer. IJROBP 2017, 97, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, R.B.; Hickey, S.; Depauw, N.; Yeap, B.Y.; Batin, E.; Gadd, M.A.; Specht, M.; Isakoff, S.J.; Smith, B.L.; Liao, E.; et al. Phase II Study of Proton Beam Radiation Therapy for Patients with Breast Cancer Requiring Regional Nodal Irradiation. J. Clin. Oncol. 2019, 37, 2778–2785. [Google Scholar] [CrossRef]
- Hassan, M.Z.O.; Awadalla, M.; Tan, T.C.; Scherrer-Crosbie, M.; Bakar, R.B.; Drobni, Z.D.; Zarif, A.; Gilman, H.K.; Supraja, S.; Nikolaidou, S.; et al. Serial Measurement of Global Longitudinal Strain Among Women with Breast Cancer Treated with Proton Radiation Therapy: A Prospective Trial for 70 Patients. IJROBP 2022, 115, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Erven, K.; Jurcut, R.; Weltens, C.; Giusca, S.; Ector, J.; Wildiers, H.; Bogaert, W.V.D.; Voigt, J.-U. Acute radiation effects on cardiac function detected by strain rate imaging in breast cancer patients. IJROBP 2011, 79, 1444–1451. [Google Scholar] [CrossRef]
- Skytta, T.; Tuohinen, S.; Boman, E.; Virtanen, V.; Raatikainen, P.; Kellokumpu-Lehtinen, P.-L. Troponin trelease associates with cardiac radiation doses during adjuvant left-sided breast cancer radiotherapy. Radiat. Oncol. 2015, 10, 141. [Google Scholar] [CrossRef] [PubMed]
- The Differential Impact of Proton Beam Irradiation Versus Conventional Radiation on Organs-at-Risk in Stage II–III Breast Cancer Patients. Available online: https://clinicaltrials.gov/ct2/show/NCT03270072 (accessed on 26 January 2023).
- Paganetti, H.; DePauw, N.; Johnson, A.; Forman, R.B.; Lau, J.; Jimenez, R. The risk for developing a secondary cancer after breast radiation therapy: Comparison of photon and proton techniques. Radiother. Oncol. 2020, 149, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Fattahi, S.; Mullikin, T.C.; Aziz, K.A.; Afzal, A.; Smith, N.L.; Francis, L.N.; Harmsen, W.S.; Routman, D.M.; Remmes, N.B.; Ahmed, S.K.; et al. Proton therapy for the treatment of inflammatory breast cancer. Radiother. Oncol. 2022, 171, 77–83. [Google Scholar] [CrossRef]
- Garda, A.E.; Hunzeker, A.E.; Michel, A.K.; Fattahi, S.; Shiraishi, S.; Remmes, N.B.; Schultz, H.L.; Harmsen, W.S.; Shumway, D.A.; Yan, E.S.; et al. Intensity Modulated Proton Therapy for Bilateral Breast or Chest Wall and Comprehensive Nodal Irradiation for Synchronous Bilateral Breast Cancer: Initial Clinical Experience and Dosimetric Comparison. Adv. Radiat. Oncol. 2022, 7, 100901. [Google Scholar] [CrossRef]
- Panchal, H.; Pilewskie, M.L.; Sheckter, C.C.; Albornoz, C.R.; Razdan, S.N.; Disa, J.J.; Cordeiro, P.G.; Mehrara, B.J.; Matros, E. National trends in contralateral prophylactic mastectomy in women with locally advanced breast cancer. J. Surg. Oncol. 2019, 119, 79–87. [Google Scholar] [CrossRef]
- Smith, N.L.; Jethwa, K.R.; Viehman, J.K.; Harmsen, W.S.; Gonuguntla, K.; Elswick, S.M.; Grauberger, J.N.; Amundson, A.C.; Whitaker, T.J.; Remmes, N.B.; et al. Post-mastectomy intensity modulated proton therapy after immediate breast reconstruction: Initial report of reconstruction outcomes and predictors of complications. Radiother. Oncol. 2019, 140, 76–83. [Google Scholar] [CrossRef]
- Macdonald, S.M.; Jimenez, R.; Paetzold, P.; Adams, J.; Beatty, J.; Delaney, T.F.; Kooy, H.; Taghian, A.G.; Lu, H.-M. Proton radiotherapy for chest wall and regional lymphatic radiation; dose comparisons and treatment delivery. Radiat. Oncol. 2013, 8, 71. [Google Scholar] [CrossRef]
- Mutter, R.W.; Choi, J.I.; Jimenez, R.B.; Kirova, Y.M.; Fagundes, M.; Haffty, B.G.; Amos, R.A.; Bradley, J.A.; Chen, P.Y.; Ding, X.; et al. Proton Therapy for Breast Cancer: A Consensus Statement From the Particle Therapy Cooperative Group Breast Cancer Subcommittee. IJROBP 2021, 111, 337–359. [Google Scholar] [CrossRef]
- Depauw, N.; Batin, E.; Daartz, J.; Rosenfeld, A.; Adams, J.; Kooy, H.; MacDonald, S.; Lu, H.-M. A novel approach to postmastectomy radiation therapy using scanned proton beams. IJROBP 2015, 91, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Paganetti, H.; Blakely, E.; Carabe-Fernandez, A.; Carlson, D.J.; Das, I.J.; Dong, L.; Grosshans, D.; Held, K.D.; Mohan, R.; Moiseenko, V.; et al. Report of the AAPM TG-256 on the relative biological effectiveness of proton beams in radiation therapy. Med Phys. 2019, 46, 53–78. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.C.; McNamara, A.L.; Shin, J.; Schuemann, J.; Grassberger, C.; Taghian, A.G.; Jimenez, R.B.; MacDonald, S.M.; Paganetti, H. End-of-Range Radiobiological Effect on Rib Fractures in Patients Receiving Proton Therapy for Breast Cancer. IJROBP 2020, 107, 449–454. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zheng, D.; Bradley, J.A.; Vega, R.B.M.; Zhang, Y.; Indelicato, D.J.; Mendenhall, N.; Liang, X. Incorporation of the LETd-weighted biological dose in the evaluation of breast intensity-modulated proton therapy plans. Acta Oncol. 2021, 60, 252–259. [Google Scholar] [CrossRef]
- Guan, F.; Geng, C.; Ma, B.D.; Bronk, L.; Kerr, B.M.; Li, Y.; Gates, M.D.; Kroger, B.B.; Sahoo, N.; Titt, U.; et al. RBE Model-based biological dose optimization for proton radiobiology studies. Int. J. Part. Ther. 2018, 5, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Pragmatic Phase III Randomized Trial of Proton vs. Photon Therapy for Patients with Non-Metastatic Breast Cancer Receiving Comprehensive Nodal Radiation: A Radiotherapy Comparative Effectiveness (RADCOMP) Consortium. Available online: https://clinicaltrials.gov/ct2/show/NCT02603341 (accessed on 26 January 2023).
- The DBCG Proton Trial: Photon Versus Proton Radiation Therapy for Early Breast Cancer. Available online: https://clinicaltrials.gov/ct2/show/NCT04291378 (accessed on 26 January 2023).
- Proton Beam Therapy in Patients with Breast Cancer: Evaluating Early and Late Effects. Available online: https://www.isrctn.com/ISRCTN14220944 (accessed on 26 January 2023).
- A Trial of 15 Fraction vs. 25 Fraction Pencil Beam Scanning Proton Radiotherapy after Mastectomy in Patients Requiring Regional Nodal Irradiation. Available online: https://www.mayo.edu/research/clinical-trials/cls-20206558 (accessed on 26 January 2023).
- Proton Radiation Therapy for Invasive Breast Cancer Following Mastectomy. Available online: https://clinicaltrials.gov/ct2/show/NCT01340495 (accessed on 26 January 2023).

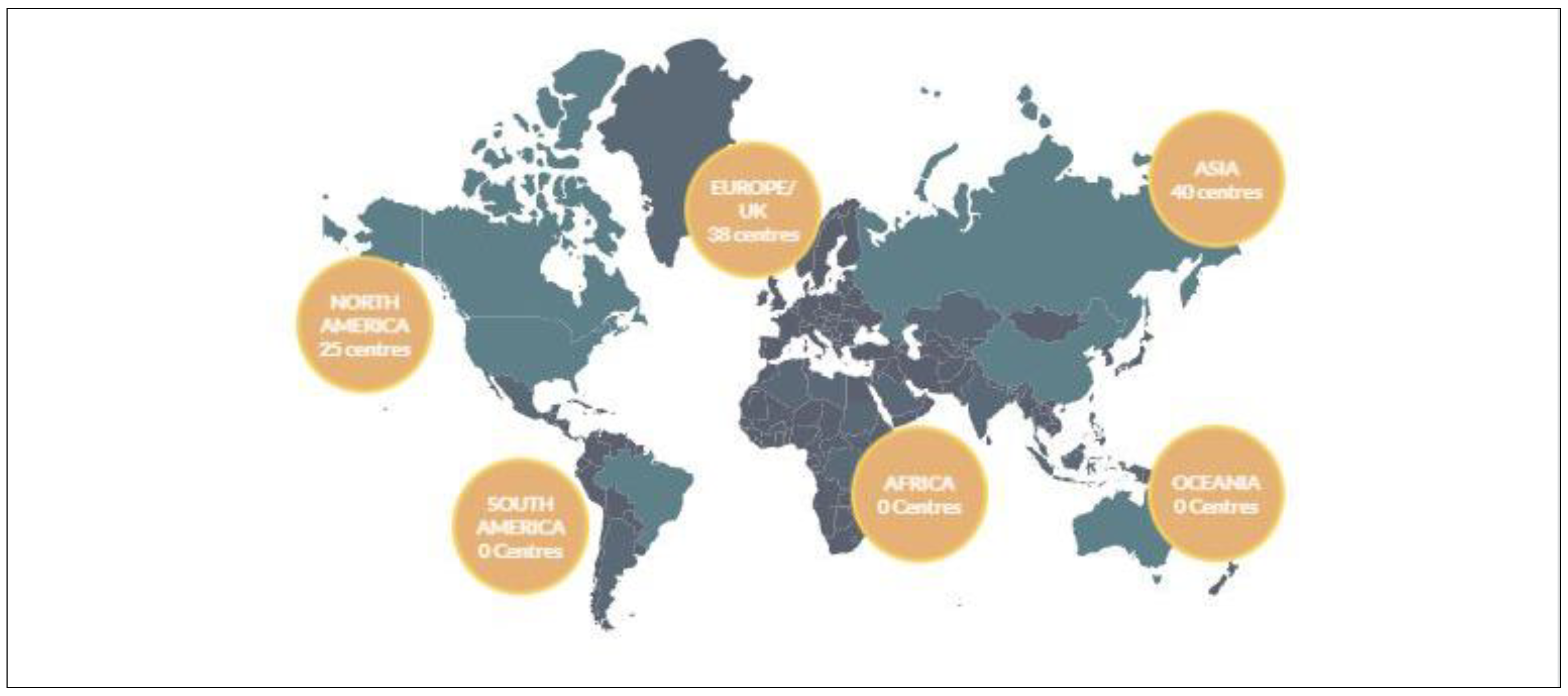

| Region | Number of Facilities in Current Operation |
|---|---|
| North America | 25 |
| Europe/UK | 38 |
| Asia | 40 |
| South America | 0 |
| Africa | 0 |
| Oceana | 0 |
| INCLUSION CRITERIA | RADCOMP | DBCG | UK |
|---|---|---|---|
| Age range | ≥21 years old | ≥18 years old | ≥18 years old |
| Gender | Females and males | Females and males | Females and males |
| Histology | Invasive mammary carcinoma | Invasive breast cancer or DCIS | Histologically proven invasive breast carcinoma |
| Stage | Clinical or pathological stage I-III; stage yp 0-III; loco-regional recurrence; Non-metastatic (AJCC 7th) | pTis-4, pN0-N3, M0 | |
| Surgical treatment | Post lumpectomy or any type of mastectomy; any axillary surgery; with or without any type of reconstruction | Post lumpectomy or any type of mastectomy; any axillary surgery; with or without any type of reconstruction (except metal implants) | wide local excision or mastectomy, and any type of axillary surgery |
| Laterality | Right, left, and bilateral | Right, left, and bilateral | |
| Radiotherapy | Undergoes adjuvant loco-regional radiotherapy including IMN |
|
|
| Days since last history and physical examination | Within 90 days prior to enrollment | Not specified | |
| ECOG performance status | 0–2 within 90 days prior to enrollment | Not specified | |
| Health coverage | Any but not through this trial | Not specified | |
| Others |
|
|
|
| EXCLUSION CRITERIA |
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lalani, N.; Alqarni, S.; Jimenez, R.B. The Potential of Proton Therapy for Locally Advanced Breast Cancer: Clinical and Technical Considerations. Curr. Oncol. 2023, 30, 2869-2878. https://doi.org/10.3390/curroncol30030219
Lalani N, Alqarni S, Jimenez RB. The Potential of Proton Therapy for Locally Advanced Breast Cancer: Clinical and Technical Considerations. Current Oncology. 2023; 30(3):2869-2878. https://doi.org/10.3390/curroncol30030219
Chicago/Turabian StyleLalani, N., S. Alqarni, and R. B. Jimenez. 2023. "The Potential of Proton Therapy for Locally Advanced Breast Cancer: Clinical and Technical Considerations" Current Oncology 30, no. 3: 2869-2878. https://doi.org/10.3390/curroncol30030219
APA StyleLalani, N., Alqarni, S., & Jimenez, R. B. (2023). The Potential of Proton Therapy for Locally Advanced Breast Cancer: Clinical and Technical Considerations. Current Oncology, 30(3), 2869-2878. https://doi.org/10.3390/curroncol30030219




