Increased Circulating Epithelial Tumor Cells (CETC/CTC) over the Course of Adjuvant Radiotherapy Is a Predictor of Less Favorable Outcome in Patients with Early-Stage Breast Cancer
Abstract
1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BCS | Breast-conserving surgery |
| CETC/CTC | Circulating epithelial tumor cells |
| DFS | Disease-free survival |
| EpCAM | Epithelial cell adhesion molecule |
| FITC | Fluoroisothiocyanate |
| GAPDH | Glyceraldehyde 3-phosphate dehydrogenase |
| RT | Radiotherapy |
References
- Jagsi, R. Progress and controversies: Radiation therapy for invasive breast cancer. CA Cancer J. Clin. 2014, 64, 135–152. [Google Scholar] [CrossRef] [PubMed]
- McArdle, C.S.; McMillan, D.C.; Greenlaw, N.; Morrison, D.S. Adjuvant radiotherapy and chemotherapy in breast cancer: 30 year follow-up of survival. BMC Cancer 2010, 10, 398. [Google Scholar] [CrossRef] [PubMed]
- Early Breast Cancer Trialists Collaborative Group (EBCTCG); Darby, S.; McGale, P.; Correa, C.; Taylor, C.; Arriagada, R.; Clarke, M.; Cutter, D.; Davies, C.; Ewertz, M.; et al. Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: Meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet 2011, 378, 1707–1716. [Google Scholar] [CrossRef] [PubMed]
- Clarke, M.; Collins, R.; Darby, S.; Davies, C.; Elphinstone, P.; Evans, V.; Godwin, J.; Gray, R.; Hicks, C.; James, S.; et al. Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: An overview of the randomised trials. Lancet 2005, 366, 2087–2106. [Google Scholar] [CrossRef]
- Early Breast Cancer Trialists Collaborative Group (EBCTCG); Darby, S.; McGale, P.; Correa, C.; Taylor, C.; Arriagada, R.; Clarke, M.; Cutter, D.; Davies, C.; Ewertz, M.; et al. Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: Meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet 2014, 383, 2127–2135. [Google Scholar] [CrossRef] [PubMed]
- Keating, N.L.; Landrum, M.B.; Brooks, J.M.; Chrischilles, E.A.; Winer, E.P.; Wright, K.; Volya, R. Outcomes following local therapy for early-stage breast cancer in non-trial populations. Breast Cancer Res. Treat. 2011, 125, 803–813. [Google Scholar] [CrossRef]
- Vilalta, M.; Rafat, M.; Giaccia, A.J.; Graves, E.E. Recruitment of circulating breast cancer cells is stimulated by radiotherapy. Cell Rep. 2014, 8, 402–409. [Google Scholar] [CrossRef]
- Horton, J.K.; Jagsi, R.; Woodward, W.A.; Ho, A. Breast Cancer Biology: Clinical Implications for Breast Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 23–37. [Google Scholar] [CrossRef]
- Krug, D.; Baumann, R.; Budach, W.; Dunst, J.; Feyer, P.; Fietkau, R.; Haase, W.; Harms, W.; Piroth, M.D.; Sautter-Bihl, M.-L.; et al. Current controversies in radiotherapy for breast cancer. Radiat. Oncol. 2017, 12, 25. [Google Scholar] [CrossRef]
- Schirrmacher, V. Cancer metastasis: Experimental approaches, theoretical concepts, and impacts for treatment strategies. Adv. Cancer Res. 1985, 43, 1–73. [Google Scholar] [CrossRef]
- Liang, D.H.; Hall, C.; Lucci, A. Circulating Tumor Cells in Breast Cancer. Recent Results Cancer Res. 2020, 215, 127–145. [Google Scholar] [CrossRef] [PubMed]
- Martin, O.A.; Anderson, R.L.; Russell, P.A.; Cox, R.A.; Ivashkevich, A.; Swierczak, A.; Doherty, J.P.; Jacobs, D.H.; Smith, J.; Siva, S.; et al. Mobilization of viable tumor cells into the circulation during radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Schott, D.S.; Pizon, M.; Pachmann, U.; Pachmann, K.; Schobert, R.; Wittig, A.; Mäurer, M. Influence of adjuvant radiotherapy on circulating epithelial tumor cells and circulating cancer stem cells in primary non-metastatic breast cancer. Transl. Oncol. 2021, 14, 101009. [Google Scholar] [CrossRef] [PubMed]
- Pachmann, K.; Clement, J.H.; Schneider, C.P.; Willen, B.; Camara, O.; Pachmann, U.; Hoffken, K. Standardized quantification of circulating peripheral tumor cells from lung and breast cancer. Clin. Chem. Lab. Med. 2005, 43, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Goodman, C.R.; Seagle, B.L.; Friedl, T.W.P.; Rack, B.; Lato, K.; Fink, V.; Cristofanilli, M.; Donnelly, E.D.; Janni, W.; Shahabi, S.; et al. Association of Circulating Tumor Cell Status With Benefit of Radiotherapy and Survival in Early-Stage Breast Cancer. JAMA Oncol. 2018, 4, e180163. [Google Scholar] [CrossRef]
- Fabisiewicz, A.; Szostakowska-Rodzos, M.; Zaczek, A.J.; Grzybowska, E.A. Circulating Tumor Cells in Early and Advanced Breast Cancer; Biology and Prognostic Value. Int. J. Mol. Sci. 2020, 21, 1671. [Google Scholar] [CrossRef]
- Pachmann, K.; Camara, O.; Kavallaris, A.; Krauspe, S.; Malarski, N.; Gajda, M.; Kroll, T.; Jorke, C.; Hammer, U.; Altendorf-Hofmann, A.; et al. Monitoring the response of circulating epithelial tumor cells to adjuvant chemotherapy in breast cancer allows detection of patients at risk of early relapse. J. Clin. Oncol. 2008, 26, 1208–1215. [Google Scholar] [CrossRef]
- Balzar, M.; Winter, M.J.; de Boer, C.J.; Litvinov, S.V. The biology of the 17-1A antigen (Ep-CAM). J. Mol. Med. 1999, 77, 699–712. [Google Scholar] [CrossRef]
- Huang, L.; Yang, Y.; Yang, F.; Liu, S.; Zhu, Z.; Lei, Z.; Guo, J. Functions of EpCAM in physiological processes and diseases (Review). Int. J. Mol. Med. 2018, 42, 1771–1785. [Google Scholar] [CrossRef]
- Harris, J.R. Fifty years of progress in radiation therapy for breast cancer. Am. Soc. Clin. Oncol. Educ. Book 2014, 34, 21–25. [Google Scholar] [CrossRef]
- Mäurer, M.; Pachmann, K.; Wendt, T.; Schott, D.; Wittig, A. Prospective Monitoring of Circulating Epithelial Tumor Cells (CETC) Reveals Changes in Gene Expression during Adjuvant Radiotherapy of Breast Cancer Patients. Curr. Oncol. 2021, 28, 3507–3524. [Google Scholar] [CrossRef] [PubMed]
- Pantel, K.; Muller, V.; Auer, M.; Nusser, N.; Harbeck, N.; Braun, S. Detection and clinical implications of early systemic tumor cell dissemination in breast cancer. Clin. Cancer Res. 2003, 9, 6326–6334. [Google Scholar] [PubMed]
- Pachmann, U.; Hekimian, K.; Pachmann, K. Comparing sequential steps for detection of circulating tumor cells: More specific or just less sensitive? WebmedCentral CANCER 2011, 2, WMC001490. [Google Scholar]
- Coumans, F.A.W.; Doggen, C.J.M.; Attard, G.; de Bono, J.S.; Terstappen, L. All circulating EpCAM+CK+CD45- objects predict overall survival in castration-resistant prostate cancer. Ann. Oncol. 2010, 21, 1851–1857. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Somasundaram, R.; Nederhof, R.F.; Dijkstra, G.; Faber, K.N.; Peppelenbosch, M.P.; Fuhler, G.M. Impact of human granulocyte and monocyte isolation procedures on functional studies. Clin. Vaccine Immunol. 2012, 19, 1065–1074. [Google Scholar] [CrossRef] [PubMed]
- Mehrishi, J.N.; Bakács, T. A novel method of CD34+ cell separation from umbilical cord blood. Transfusion 2013, 53, 2675–2680. [Google Scholar] [CrossRef]
- Mani, S.A.; Guo, W.; Liao, M.J.; Eaton, E.N.; Ayyanan, A.; Zhou, A.Y.; Brooks, M.; Reinhard, F.; Zhang, C.C.; Shipitsin, M.; et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008, 133, 704–715. [Google Scholar] [CrossRef]
- Bankó, P.; Lee, S.Y.; Nagygyörgy, V.; Zrínyi, M.; Chae, C.H.; Cho, D.H.; Telekes, A. Technologies for circulating tumor cell separation from whole blood. J. Hematol. Oncol. 2019, 12, 48. [Google Scholar] [CrossRef]
- Pachmann, K.; Dengler, R.; Lobodasch, K.; Frohlich, F.; Kroll, T.; Rengsberger, M.; Schubert, R.; Pachmann, U. An increase in cell number at completion of therapy may develop as an indicator of early relapse: Quantification of circulating epithelial tumor cells (CETC) for monitoring of adjuvant therapy in breast cancer. J. Cancer Res. Clin. Oncol. 2008, 134, 59–65. [Google Scholar] [CrossRef]
- Hickey, B.E.; Francis, D.P.; Lehman, M. Sequencing of chemotherapy and radiotherapy for early breast cancer. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef]
- Hilsenbeck, S.G.; Ravdin, P.M.; de Moor, C.A.; Chamness, G.C.; Osborne, C.K.; Clark, G.M. Time-dependence of hazard ratios for prognostic factors in primary breast cancer. Breast Cancer Res. Treat. 1998, 52, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Pachmann, K.; Schuster, S. The Value of Monitoring the Behavior of Circulating Tumor Cells at the End of Endocrine Therapy in Breast Cancer Patients. Cancers 2018, 10, 407. [Google Scholar] [CrossRef] [PubMed]
- Pizon, M.; Schott, D.; Pachmann, U.; Pachmann, K. The number of tumorspheres cultured from peripheral blood is a predictor for presence of metastasis in patients with breast cancer. Oncotarget 2016, 7, 48143–48154. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pizon, M.; Schott, D.; Pachmann, U.; Schobert, R.; Pizon, M.; Wozniak, M.; Bobinski, R.; Pachmann, K. Chick Chorioallantoic Membrane (CAM) Assays as a Model of Patient-Derived Xenografts from Circulating Cancer Stem Cells (cCSCs) in Breast Cancer Patients. Cancers 2022, 14, 1476. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.F.; Chang, L.; Li, W.H.; Xiao, M.Y.; Wang, Y.; He, W.J.; Xia, Y.X.; Wang, L.; Chen, Y. Radiotherapy concurrent versus sequential with endocrine therapy in breast cancer: A meta-analysis. Breast 2016, 27, 93–98. [Google Scholar] [CrossRef]
- Qi, X.S.; Pajonk, F.; McCloskey, S.; Low, D.A.; Kupelian, P.; Steinberg, M.; Sheng, K. Radioresistance of the breast tumor is highly correlated to its level of cancer stem cell and its clinical implication for breast irradiation. Radiother. Oncol. 2017, 124, 455–461. [Google Scholar] [CrossRef]
- Leeman, J.E.; Schoenfeld, J.D. Radiation Therapy and Immune Modulation. Hematol./Oncol. Clin. N. Am. 2019, 33, 233–248. [Google Scholar] [CrossRef]
- Lewin, N.L.; Luetragoon, T.; Andersson, B.A.; Oliva, D.; Nilsson, M.; Strandeus, M.; Lofgren, S.; Rutqvist, L.E.; Lewin, F. The Influence of Single Nucleotide Polymorphisms and Adjuvant Radiotherapy on Systemic Inflammatory Proteins, Chemokines and Cytokines of Patients With Breast Cancer. Anticancer. Res. 2019, 39, 1287–1292. [Google Scholar] [CrossRef]
- Bentzen, S.M. Preventing or reducing late side effects of radiation therapy: Radiobiology meets molecular pathology. Nat. Rev. Cancer 2006, 6, 702–713. [Google Scholar] [CrossRef]
- Jarosz-Biej, M.; Smolarczyk, R.; Cichon, T.; Kulach, N. Tumor Microenvironment as A “Game Changer” in Cancer Radiotherapy. Int. J. Mol. Sci. 2019, 20, 3212. [Google Scholar] [CrossRef]
- Meltzer, A. Dormancy and breast cancer. J. Surg. Oncol. 1990, 43, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Al-Hajj, M.; Wicha, M.S.; Benito-Hernandez, A.; Morrison, S.J.; Clarke, M.F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl. Acad. Sci. USA 2003, 100, 3983–3988. [Google Scholar] [CrossRef] [PubMed]
- Alieva, M.; van Rheenen, J.; Broekman, M.L.D. Potential impact of invasive surgical procedures on primary tumor growth and metastasis. Clin. Exp. Metastasis 2018, 35, 319–331. [Google Scholar] [CrossRef] [PubMed]
- Hekimian, K.; Meisezahl, S.; Trompelt, K.; Rabenstein, C.; Pachmann, K. Epithelial cell dissemination and readhesion: Analysis of factors contributing to metastasis formation in breast cancer. ISRN Oncol. 2012, 2012, 601810. [Google Scholar] [CrossRef]
- Pachmann, K. Current and potential use of MAINTRAC method for cancer diagnosis and prediction of metastasis. Expert Rev. Mol. Diagn. 2015, 15, 597–605. [Google Scholar] [CrossRef]
- Pizon, M.; Zimon, D.; Carl, S.; Pachmann, U.; Pachmann, K.; Camara, O. Heterogeneity of circulating epithelial tumour cells from individual patients with respect to expression profiles and clonal growth (sphere formation) in breast cancer. Ecancermedicalscience 2013, 7, 343. [Google Scholar] [CrossRef]
- Mäurer, M.A. Analysen zirkulierender epithelialer Tumorzellen im periphervenösen Blut bei Patientinnen mit primär nicht metastasiertem Mammakarzinom unter adjuvanter Radiotherapie. Ph.D. Thesis, Friedrich-Schiller-Universität Jena, Jena, Germany, 2014. [Google Scholar]
- Schott, D.; Pizon, M.; Pachamnn, U.; Pachmann, K. Successful mutational profiling of single circulating epithelial tumor cells (CETCs) from patients with solid cancers for guiding therapy. J. Clin. Oncol. 2020, 38, e15527. [Google Scholar] [CrossRef]
- Yizhak, K.; Aguet, F.; Kim, J.; Hess, J.M.; Kübler, K.; Grimsby, J.; Frazer, R.; Zhang, H.; Haradhvala, N.J.; Rosebrock, D.; et al. RNA sequence analysis reveals macroscopic somatic clonal expansion across normal tissues. Science 2019, 364, eaaw0726. [Google Scholar] [CrossRef]
- Masuda, N.; Lee, S.J.; Ohtani, S.; Im, Y.H.; Lee, E.S.; Yokota, I.; Kuroi, K.; Im, S.A.; Park, B.W.; Kim, S.B.; et al. Adjuvant Capecitabine for Breast Cancer after Preoperative Chemotherapy. N. Engl. J. Med. 2017, 376, 2147–2159. [Google Scholar] [CrossRef]
- Folprecht, G.; Reinacher-Schick, A.; Weitz, J.; Lugnier, C.; Kraeft, A.L.; Wisser, S.; Aust, D.E.; Weiss, L.; von Bubnoff, N.; Kramer, M.; et al. The CIRCULATE Trial: Circulating Tumor DNA Based Decision for Adjuvant Treatment in Colon Cancer Stage II Evaluation (AIO-KRK-0217). Clin. Colorectal. Cancer 2022, 21, 170–174. [Google Scholar] [CrossRef]
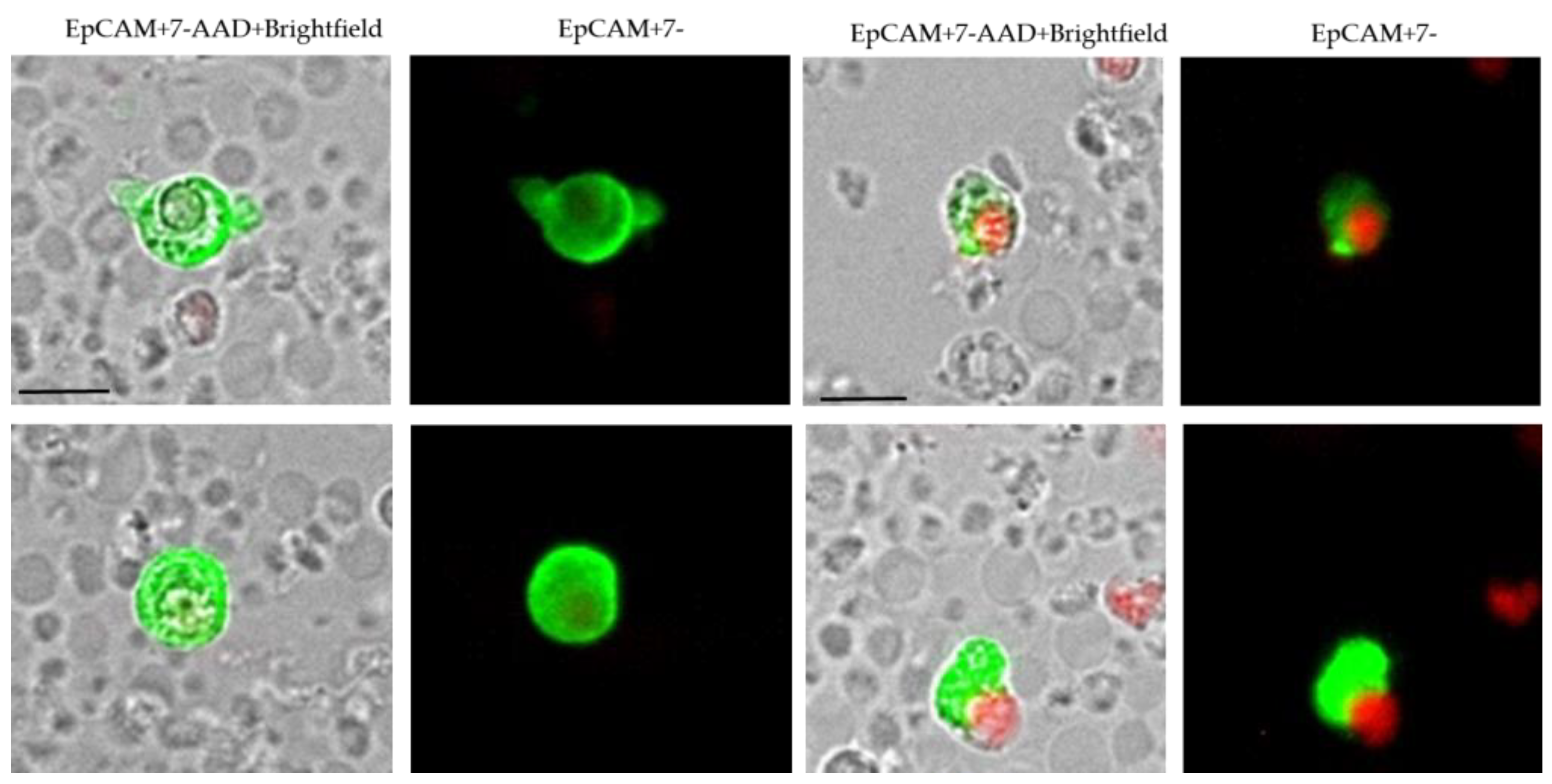

| Patient Number (%) | |
|---|---|
| Sex | |
| Male | 0 (0.0%) |
| Female | 165 (100.0%) |
| Age | Mean 56.5 (min. 27–max. 89) years |
| T stage | |
| T1 | 98 (59.4%) |
| T2 | 56 (34.0%) |
| T3 | 6 (3.6%) |
| T4 | 5 (3.0%) |
| N stage | |
| N0 | 112 (67.9%) |
| N1 | 40 (24.2%) |
| N2 | 6 (3.6%) |
| N3 | 7 (4.3) |
| Grading | |
| 1 | 12 (7.3%) |
| 2 | 66 (40.0%) |
| 3 | 82 (49.7%) |
| Not available | 5 (3.0%) |
| Histologic finding | |
| IDC 1 | 122 (73.9%) |
| ILC 2/mixed | 23 (13.9%) |
| Other | 20 (12.2%) |
| ER 3 status | |
| Positive | 115 (69.7%) |
| Negative | 48 (29.1%) |
| Not available | 2 (1.2%) |
| PR 4 status | |
| Positive | 108 (65.5%) |
| Negative | 55 (33.3) |
| Not available | 2 (1.2%) |
| Her2/neu status | |
| Positive | 53 (32.1%) |
| Negative | 107 (64.9%) |
| Not available | 5 (3.0%) |
| Chemotherapy | |
| Neoadjuvant | 44 (28.5%) |
| Adjuvant | 83 (52.1%) |
| None | 39 (19.4%) |
| Endocrine treatment | |
| Yes | 114 (69.1%) |
| No | 51 (30.9%) |
| CETC/CTC Behavior | Patients | Events | Local Relapse | Distant Metastasis | DFS |
|---|---|---|---|---|---|
| No Chemo | |||||
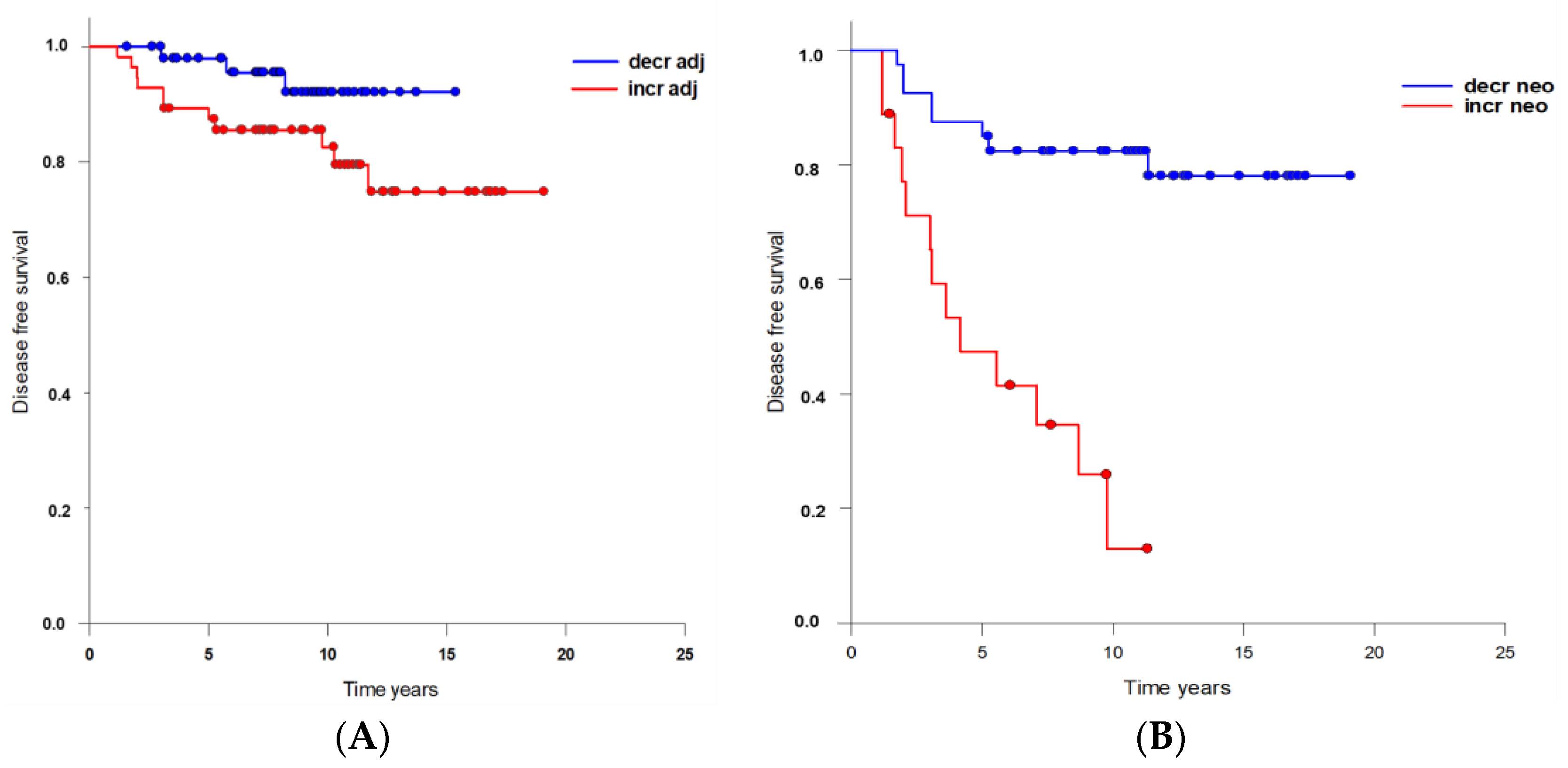
| Increase | 18 (44.4%) | 2 | 1 | 1 | p = 0.123 ns |
| Decrease | 21 (55.6%) | 0 | 0 | 0 | |
| Adjuvant Chemo | |||||
| Increase | 52 (54.9%) | 11 | 4 | 7 | p = 0.172 ns |
| Decrease | 31 (45.1%) | 3 | 1 | 2 | |
| Neoadjuvant Chemo | |||||
| Increase | 13 (38.3%) | 10 | 2 | 8 | p < 0.001 |
| Decrease | 31 (61.7%) | 5 | 3 | 2 | |
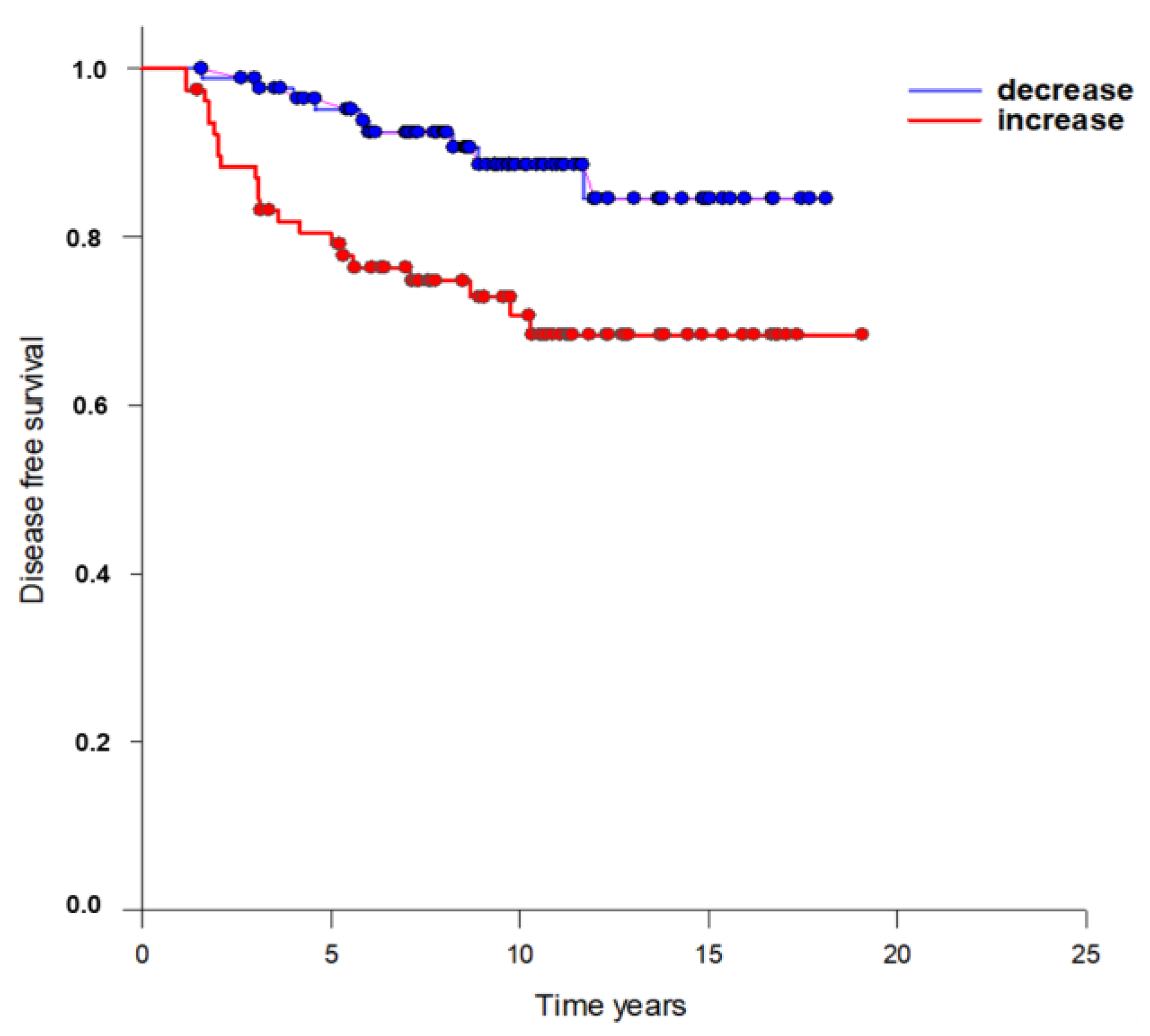
| Total Increase | 82 (47.3%) | 23 | 7 | 16 | p = 0.003 |
| Decrease | 83 (52.7%) | 8 | 4 | 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mäurer, M.; Schott, D.; Pizon, M.; Drozdz, S.; Wendt, T.; Wittig, A.; Pachmann, K. Increased Circulating Epithelial Tumor Cells (CETC/CTC) over the Course of Adjuvant Radiotherapy Is a Predictor of Less Favorable Outcome in Patients with Early-Stage Breast Cancer. Curr. Oncol. 2023, 30, 261-273. https://doi.org/10.3390/curroncol30010021
Mäurer M, Schott D, Pizon M, Drozdz S, Wendt T, Wittig A, Pachmann K. Increased Circulating Epithelial Tumor Cells (CETC/CTC) over the Course of Adjuvant Radiotherapy Is a Predictor of Less Favorable Outcome in Patients with Early-Stage Breast Cancer. Current Oncology. 2023; 30(1):261-273. https://doi.org/10.3390/curroncol30010021
Chicago/Turabian StyleMäurer, Matthias, Dorothea Schott, Monika Pizon, Sonia Drozdz, Thomas Wendt, Andrea Wittig, and Katharina Pachmann. 2023. "Increased Circulating Epithelial Tumor Cells (CETC/CTC) over the Course of Adjuvant Radiotherapy Is a Predictor of Less Favorable Outcome in Patients with Early-Stage Breast Cancer" Current Oncology 30, no. 1: 261-273. https://doi.org/10.3390/curroncol30010021
APA StyleMäurer, M., Schott, D., Pizon, M., Drozdz, S., Wendt, T., Wittig, A., & Pachmann, K. (2023). Increased Circulating Epithelial Tumor Cells (CETC/CTC) over the Course of Adjuvant Radiotherapy Is a Predictor of Less Favorable Outcome in Patients with Early-Stage Breast Cancer. Current Oncology, 30(1), 261-273. https://doi.org/10.3390/curroncol30010021







