Efficacy of Osimertinib in Lung Squamous Cell Carcinoma Patients with EGFR Gene Mutation–Case Report and a Literature Review
Abstract
:1. Introduction
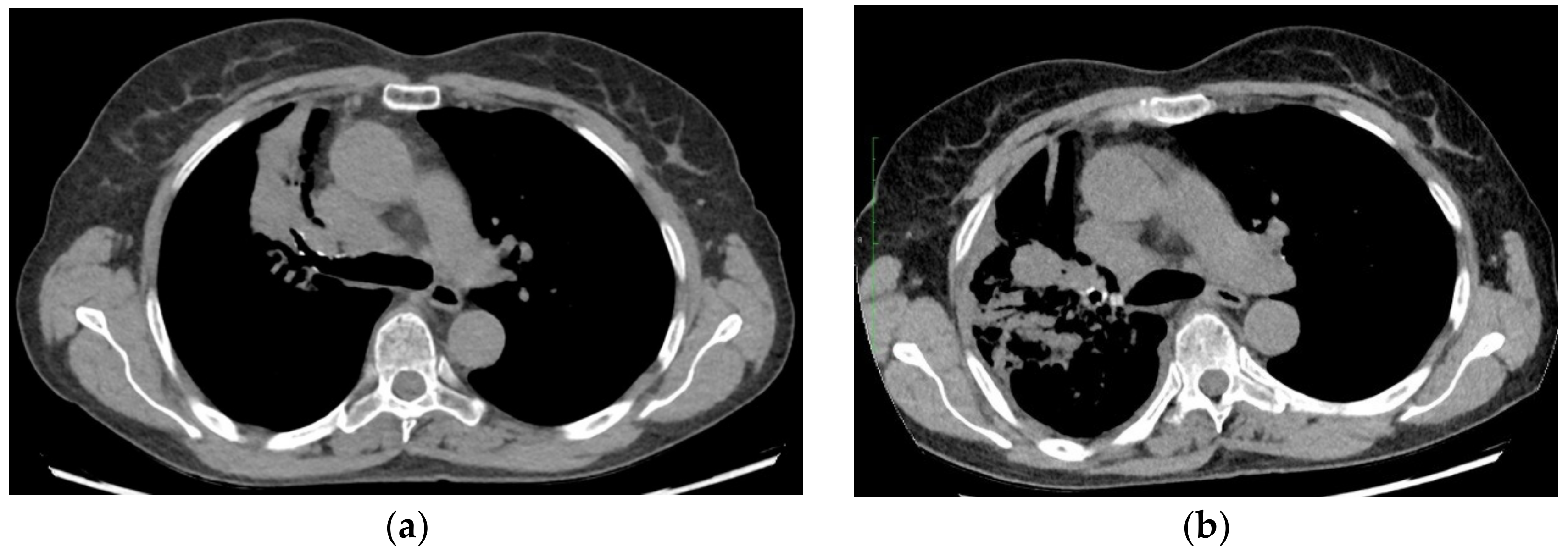
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ho, H.L.; Kao, H.L.; Yeh, Y.C.; Chou, T.Y. The importance of EGFR mutation testing in squamous cell carcinoma or non-small cell carcinoma favor squamous cell carcinoma diagnosed from small lung biopsies. Diagn Pathol. 2019, 14, 59. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, Y.; Matsumoto, Y.; Furukawa, R.; Ohara, S.; Usui, K. The clinical features of squamous cell lung carcinoma with sensitive EGFR mutations. Int. J. Clin. Oncol. 2018, 23, 452–457. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhu, L.; Zhang, J. Epidermal growth factor receptor gene mutation status in pure squamous-cell lung cancer in Chinese patients. BMC Cancer. 2015, 15, 88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, J.; Huang, Z.; Han, L.; Gong, Y.; Xie, C. Mechanisms and management of 3rd-generation EGFR-TKI resistance in advanced non-small cell lung cancer (Review). Int. J. Oncol. 2021, 59, 90. [Google Scholar] [CrossRef]
- Kim, K.; Kim, H.S.; Kim, J.Y.; Jung, H.; Sun, J.-M.; Ahn, J.S.; Ahn, M.-J.; Park, K.; Lee, S.-H.; Choi, J.K. Predicting clinical benefit of immunotherapy by antigenic or functional mutations affecting tumour immunogenicity. Nat. Commun. 2020, 11, 951. [Google Scholar] [CrossRef]
- Xia, L.; Yu, Y.; Lan, F.; Yan, J.; Li, J.; Li, W.; Xia, Y. Case Report: Tumor Microenvironment Characteristics in a Patient With HER2 Mutant Lung Squamous Cell Carcinoma Harboring High PD-L1 Expression Who Presented Hyperprogressive Disease. Front. Oncol. 2021, 11, 760703. [Google Scholar] [CrossRef]
- Wang, B.-Y.; Huang, J.-Y.; Chen, H.-C.; Lin, C.-H.; Lin, S.-H.; Hung, W.-H.; Cheng, Y.-F. The comparison between adenocarcinoma and squamous cell carcinoma in lung cancer patients. J. Cancer Res. Clin. Oncol. 2020, 146, 43–52. [Google Scholar] [CrossRef]
- Beau-Faller, M.; Prim, N.; Ruppert, A.-M.; Nanni-Metéllus, I.; Lacave, R.; Lacroix, L.; Escande, F.; Lizard, S.; Pretet, J.-L.; Rouquette, I.; et al. Rare EGFR exon 18 and exon 20 mutations in non-small-cell lung cancer on 10 117 patients: A multicentre observational study by the French ERMETIC-IFCT network. Ann. Oncol. 2014, 25, 126–131. [Google Scholar] [CrossRef]
- Lamb, Y.N. Osimertinib: A Review in Previously Untreated, EGFR Mutation-Positive, Advanced NSCLC. Targ. Oncol. 2021, 16, 687–695. [Google Scholar] [CrossRef]
- Zhang, Y.-L.; Yuan, J.-Q.; Wang, K.-F.; Fu, X.-H.; Han, X.-R.; Threapleton, D.; Yang, Z.-Y.; Mao, C.; Tang, J.-L. The prevalence of EGFR mutation in patients with non-small cell lung cancer: A systematic review and meta-analysis. Oncotarget 2016, 7, 78985–78993. [Google Scholar] [CrossRef] [Green Version]
- Krawczyk, P.; Reszka, K.; Ramlau, R.; Powrózek, T.; Pankowski, J.; Wojas-Krawczyk, K.; Kalinka-Warzocha, E.; Szczęsna, A.; Nicoś, M.; Jarosz, B.; et al. Prevalence of rare EGFR gene mutations in nonsmall-cell lung cancer: A multicenter study on 3856 Polish Caucasian patients. Ann. Oncol. 2016, 27, 358–359. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Zanwar, S.; Noronha, V.; Patil, V.M.; Chougule, A.; Kumar, R.; Janu, A.; Mahajan, A.; Kapoor, A.; Prabhash, K. EGFR mutation in squamous cell carcinoma of the lung: Does it carry the same connotation as in adenocarcinomas? OncoTargets Ther. 2017, 10, 1859–1863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lam, V.K.; Tran, H.T.; Banks, K.; Lanman, R.B.; Rinsurongkawong, W.; Peled, N.; Lewis, J.; Lee, J.J.; Roth, J.; Roarty, E.B.; et al. Targeted Tissue and Cell-Free Tumor DNA Sequencing of Advanced Lung Squamous-Cell Carcinoma Reveals Clinically Significant Prevalence of Actionable Alterations. Clin. Lung Cancer 2019, 20, 30–36.e3. [Google Scholar] [CrossRef] [Green Version]
- Sun, Y.; Yin, X.; Wen, M.; Zhang, J.; Wang, X.; Xia, J.; Zhang, Y.; Zhang, Z.; Li, X. EGFR mutations subset in Chinese lung squamous cell carcinoma patients. Mol. Med. Rep. 2018, 17, 7575–7584. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jones, G.S.; Baldwin, D.R. Recent advances in the management of lung cancer. Clin. Med. 2018, 18, s41–s46. [Google Scholar] [CrossRef]
- Zhuo, M.; Zheng, Q.; Zhao, J.; Wu, M.; An, T.; Wang, Y.; Li, J.; Wang, S.; Zhong, J.; Yang, X.; et al. Survival difference between EGFR Del19 and L858R mutant advanced non-small cell lung cancer patients receiving gefitinib: A propensity score matching analysis. Chin. J. Cancer Res. 2017, 29, 553–560. [Google Scholar] [CrossRef] [PubMed]
- McLoughlin, E.M.; Gentzler, R.D. Epidermal Growth Factor Receptor Mutations. Thorac. Surg. Clin. 2020, 30, 127–136. [Google Scholar] [CrossRef]
- Mok, T.S.; Wu, Y.-L.; Ahn, M.-J.; Garassino, M.C.; Kim, H.R.; Ramalingam, S.S.; Shepherd, F.A.; He, Y.; Akamatsu, H.; Theelen, W.S.; et al. Osimertinib or Platinum–Pemetrexed in EGFR T790M–Positive Lung Cancer. N. Engl. J. Med. 2017, 376, 629–640. [Google Scholar] [CrossRef] [Green Version]
- Minguet, J.; Smith, K.H.; Bramlage, P. Targeted therapies for treatment of non-small cell lung cancer-Recent advances and future perspectives: Targeted Therapies for Treatment of NSCLC. Int. J. Cancer 2016, 138, 2549–2561. [Google Scholar] [CrossRef] [Green Version]
- Chang, Q.; Qiang, H.; Qian, J.; Lei, Y.; Lu, J.; Feng, H.; Zhao, Y.; Han, B.; Zhang, Y.; Chu, T. Epidermal Growth Factor Receptor Mutation Status and Response to Tyrosine Kinase Inhibitors in Advanced Chinese Female Lung Squamous Cell Carcinoma: A Retrospective Study. Front. Oncol. 2021, 11, 652560. [Google Scholar] [CrossRef]
- Mitsudomi, T.; Morita, S.; Yatabe, Y.; Negoro, S.; Okamoto, I.; Tsurutani, J.; Seto, T.; Satouchi, M.; Tada, H.; Hirashima, T.; et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): An open label, randomised phase 3 trial. Lancet Oncol. 2010, 11, 121–128. [Google Scholar] [CrossRef]
- Imyanitov, E.N.; Demidova, I.A.; Gordiev, M.G.; Filipenko, M.L.; Kekeyeva, T.V.; Moliaka, Y.K.; Gervas, P.A.; Kozhemyako, V.B.; Vodolazhskiy, D.I.; Sergeyeva, L.A.; et al. Distribution of EGFR Mutations in 10,607 Russian Patients with Lung Cancer. Mol. Diagn. Ther. 2016, 20, 401–406. [Google Scholar] [CrossRef] [PubMed]
- NCCN Guidelines for Patients: Metastatic Non-Small Cell Lung Cancer. 2021. Available online: https://www.nccn.org/patients/guidelines/content/PDF/lung-metastatic-patient.pdf (accessed on 25 April 2022).
- Metastatic Non-Small-Cell Lung Cancer. European Society for Medical Oncology. Available online: https://www.esmo.org/guidelines/lung-and-chest-tumours/clinical-practice-living-guidelines-metastatic-non-small-cell-lung-cancer (accessed on 25 April 2022).
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Jin, R.; Peng, L.; Shou, J.; Wang, J.; Jin, Y.; Liang, F.; Zhao, J.; Wu, M.; Li, Q.; Zhang, B.; et al. EGFR-Mutated Squamous Cell Lung Cancer and Its Association With Outcomes. Front. Oncol. 2021, 11, 680804. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Soejima, K.; Fukunaga, K.; Shintani, Y.; Sekine, I.; Shukuya, T.; Takayama, K.; Inoue, A.; Okamoto, I.; Kiura, K.; et al. Key prognostic factors for EGFR-mutated non-adenocarcinoma lung cancer patients in the Japanese Joint Committee of Lung Cancer Registry Database. Lung Cancer 2020, 146, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Skoczek, L.M.; Onkologiczny, O. Sequential Treatment with Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitors in a Non-Smoking Patient with Metastatic Non-Small-Cell Lung Cancer Harbouring EGFR Mutation. Available online: https://journals.viamedica.pl/onkologia_w_praktyce_klin_edu/article/view/61530 (accessed on 25 April 2022).
- Friedlaender, A.; Drilon, A.; Weiss, G.J.; Banna, G.L.; Addeo, A. KRAS as a druggable target in NSCLC: Rising like a phoenix after decades of development failures. Cancer Treat. Rev. 2020, 85, 101978. [Google Scholar] [CrossRef]
- Ferrer, I.; Zugazagoitia, J.; Herbertz, S.; John, W.; Paz-Ares, L.; Schmid-Bindert, G. KRAS-Mutant non-small cell lung cancer: From biology to therapy. Lung Cancer 2018, 124, 53–64. [Google Scholar] [CrossRef] [Green Version]
- Acker, F.; Stratmann, J.; Aspacher, L.; Nguyen, N.T.T.; Wagner, S.; Serve, H.; Wild, P.J.; Sebastian, M. KRAS Mutations in Squamous Cell Carcinomas of the Lung. Front. Oncol. 2021, 11, 788084. [Google Scholar] [CrossRef]
- Huang, L.; Guo, Z.; Wang, F.; Fu, L. KRAS mutation: From undruggable to druggable in cancer. Signal Transduct. Target. Ther. 2021, 6, 386. [Google Scholar] [CrossRef]
- Cortiula, F.; De Maglio, G.; Cangi, M.G.; Gerratana, L.; Lisanti, C.; Bonura, S.; Fasola, G.; Follador, A. Third-generation tyrosine kinase inhibitor in the treatment of epidermal growth factor receptor mutated squamous cell lung cancer: A tailored therapy approach. Ann. Transl. Med. 2019, 7, 14. [Google Scholar] [CrossRef]
- Fassunke, J.; Müller, F.; Keul, M.; Michels, S.; Dammert, M.A.; Schmitt, A.; Plenker, D.; Lategahn, J.; Heydt, C.; Brägelmann, J.; et al. Overcoming EGFRG724S-mediated osimertinib resistance through unique binding characteristics of second-generation EGFR inhibitors. Nat. Commun. 2018, 9, 4655. [Google Scholar] [CrossRef] [PubMed]
- Peng, M.; Wen, Q.; Wu, X.; Yu, F.; Liu, W. Osimertinib for compound EGFR exon 19 deletion/T790M mutated lung squamous cell carcinoma. Thorac. Cancer 2020, 11, 2704–2708. [Google Scholar] [CrossRef] [PubMed]
- Shoji, S.; Watanabe, S.; Takamura, K.; Umezu, H.; Kikuchi, T. First-line osimertinib treatment in patients with lung squamous cell carcinoma harboring active epidermal growth factor receptor mutations. Lung Cancer 2020, 140, 113–115. [Google Scholar] [CrossRef] [PubMed]
- Yoshimatsu, Y.; Ebi, N.; Ooi, R.; Sueyasu, T.; Nishizawa, S.; Munechika, M.; Yoshimine, K.; Ko, Y.; Ide, H.; Tsuruno, K.; et al. Osimertinib for Lung Squamous Cell Carcinoma: A Case Report and Literature Review. Intern. Med. 2021, 60, 1067–1071. [Google Scholar] [CrossRef] [PubMed]
- Travis, W.D.; Brambilla, E.; Noguchi, M.; Nicholson, A.G.; Geisinger, K.R.; Yatabe, Y.; Beer, D.G.; Powell, C.; Riely, G.J.; Van Schil, P.E.; et al. International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society International Multidisciplinary Classification of Lung Adenocarcinoma. J. Thorac. Oncol. 2011, 6, 244–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cheung, A.H.-K.; Tong, J.H.-M.; Chung, L.-Y.; Chau, S.-L.; Ng, C.S.-H.; Wan, I.Y.; To, K.-F. EGFR mutation exists in squamous cell lung carcinoma. Pathology 2020, 52, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Layfield, L.J.; Pearson, L.; Walker, B.S.; White, S.K.; Schmidt, R.L. Diagnostic Accuracy of Fine-Needle Aspiration Cytology for Discrimination of Squamous Cell Carcinoma from Adenocarcinoma in Non-Small Cell Lung Cancer: A Systematic Review and Meta-Analysis. Acta Cytol. 2018, 62, 318–326. [Google Scholar] [CrossRef]
- Rekhtman, N.; Paik, P.K.; Arcila, M.E.; Tafe, L.J.; Oxnard, G.R.; Moreira, A.L.; Travis, W.D.; Zakowski, M.F.; Kris, M.G.; Ladanyi, M. Clarifying the Spectrum of Driver Oncogene Mutations in Biomarker-Verified Squamous Carcinoma of Lung: Lack of EGFR/KRA S and Presence of PIK3CA/AKT1 Mutations. Clin. Cancer Res. 2012, 18, 1167–1176. [Google Scholar] [CrossRef] [Green Version]
- Schwitter, M.; Rodriguez, R.; Schneider, T.; Kluckert, T.; Brutsche, M.; Früh, M. Epidermal Growth Factor Receptor Mutation in a Patient with Squamous Cell Carcinoma of the Lung: Who Should Be Tested. Case Rep. Oncol. 2013, 6, 263–268. [Google Scholar] [CrossRef]
- Wu, F.; Fan, J.; He, Y.; Xiong, A.; Yu, J.; Li, Y.; Zhang, Y.; Zhao, W.; Zhou, F.; Li, W.; et al. Single-cell profiling of tumor heterogeneity and the microenvironment in advanced non-small cell lung cancer. Nat. Commun. 2021, 12, 2540. [Google Scholar] [CrossRef]
- Jamal-Hanjani, M.; Wilson, G.A.; McGranahan, N.; Birkbak, N.J.; Watkins, T.B.K.; Veeriah, S.; Shafi, S.; Johnson, D.H.; Mitter, R.; Rosenthal, R.; et al. Tracking the Evolution of Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 376, 2109–2121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Testa, U.; Castelli, G.; Pelosi, E. Lung Cancers: Molecular Characterization, Clonal Heterogeneity and Evolution, and Cancer Stem Cells. Cancers 2018, 10, 248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rekowska, A.; Rola, P.; Wójcik-Superczyńska, M.; Chmielewska, I.; Krawczyk, P.; Milanowski, J. Efficacy of Osimertinib in Lung Squamous Cell Carcinoma Patients with EGFR Gene Mutation–Case Report and a Literature Review. Curr. Oncol. 2022, 29, 3531-3539. https://doi.org/10.3390/curroncol29050285
Rekowska A, Rola P, Wójcik-Superczyńska M, Chmielewska I, Krawczyk P, Milanowski J. Efficacy of Osimertinib in Lung Squamous Cell Carcinoma Patients with EGFR Gene Mutation–Case Report and a Literature Review. Current Oncology. 2022; 29(5):3531-3539. https://doi.org/10.3390/curroncol29050285
Chicago/Turabian StyleRekowska, Anna, Piotr Rola, Magdalena Wójcik-Superczyńska, Izabela Chmielewska, Paweł Krawczyk, and Janusz Milanowski. 2022. "Efficacy of Osimertinib in Lung Squamous Cell Carcinoma Patients with EGFR Gene Mutation–Case Report and a Literature Review" Current Oncology 29, no. 5: 3531-3539. https://doi.org/10.3390/curroncol29050285
APA StyleRekowska, A., Rola, P., Wójcik-Superczyńska, M., Chmielewska, I., Krawczyk, P., & Milanowski, J. (2022). Efficacy of Osimertinib in Lung Squamous Cell Carcinoma Patients with EGFR Gene Mutation–Case Report and a Literature Review. Current Oncology, 29(5), 3531-3539. https://doi.org/10.3390/curroncol29050285