Present and Emerging Biomarkers in Immunotherapy for Metastatic Non-Small Cell Lung Cancer: A Review
Abstract
:1. Introduction
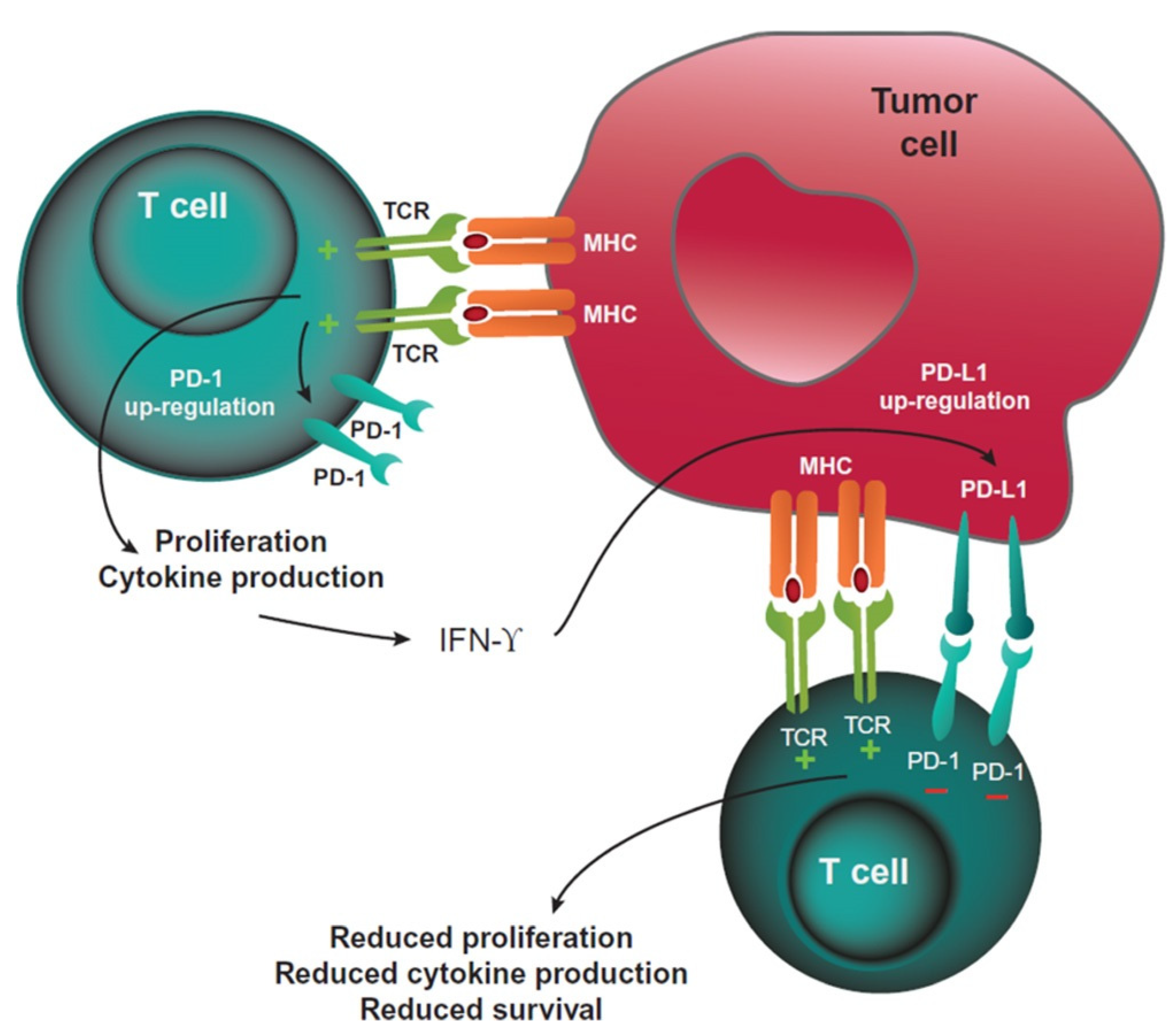
2. The Role of PD-L1 as Predictive Biomarker
3. Smoking History
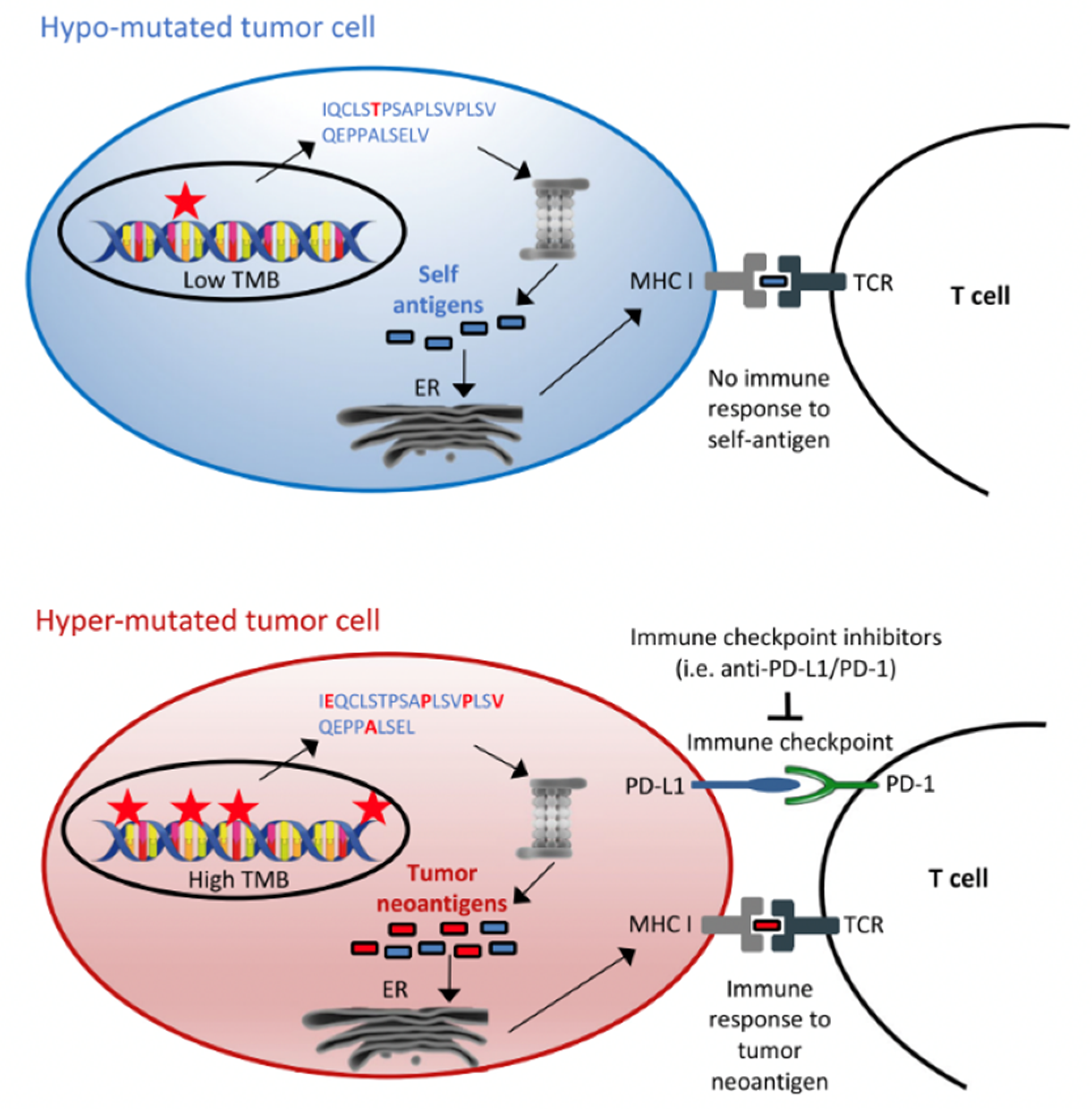
4. Tumour Mutational Burden
5. STK11, KEAP1
6. Host Microbiome
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hanahan, D.; Weinberg, R. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [Green Version]
- Choi, Y.; Shi, Y.; Haymaker, C.; Naing, A.; Ciliberto, G.; Joud, H. T-Cell Agonists in Cancer Immunotherapy. J. Immunother. Cancer 2020, 8, e000966. [Google Scholar] [CrossRef]
- Buchbinder, E.; Desai, A. CTLA-4 and PD-1 Pathways. Am. J. Clin. Oncol. 2016, 39, 98–106. [Google Scholar] [CrossRef] [Green Version]
- Brahmer, J.; Reckamp, K.; Baas, P.; Crino, L.; Eberhardt, W.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab vs. Docetaxel in Advanced Squamous Cell Non-Small-Cell-Lung Cancer. N. Engl. J. Med. 2015, 373, 123–135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borghaei, H.; Paz-Ares, L.; Horn, L.; Spigel, D.; Steins, M.; Ready, N.; Chow, L.; Vokes, E.E.; Felip, E.; Holgado, E.; et al. Nivolumab vs. Docetaxel in Advanced Non-Squamous Non-Small Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 1627–1639. [Google Scholar] [CrossRef]
- Herbst, R.; Baas, P.; Kim, D.W.; Felip, E.; Perez-Gracia, J.; Han, J.Y.; Molina, J.; Kim, J.; Arvis, C.D.; Ahn, M.; et al. Pembrolizumab vs. Docetaxel for Previously Treated, PD-L1 Positive, Advanced Non-Small-Cell Lung Cancer (KEYNOTE-010): A Randomized Controlled Trial. Lancet 2016, 387, 1540–1550. [Google Scholar] [CrossRef]
- Rittmeyer, A.; Barlesi, F.; Waterkamp, D.; Park, K.; Ciardiello, F.; Pawel, J.; Gadgeel, S.M.; Hida, T.; Kowalski, D.M.; Dols, M.C.; et al. Atezolizumab vs. Docetaxel inpatients with previously treated non-small-cell lung cancer (OAK): A phase 3, open-label, multi centre randomized controlled trial. Lancet 2017, 389, 255–265. [Google Scholar] [CrossRef]
- Reck, M.; Rodriguez-Abreu, D.; Robinson, A.; Hui, R.; Csoszi, T.; Fulop, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Updated Analysis of KEYNOTE-024: Pembrolizumab Vs. Platinum-Based Chemotherapy for Advanced Non-Small-Cell Lung Cancer With PD-L1 Tumor Proportion Score of 50% or Greater. J. Clin. Oncol. 2019, 37, 537–546. [Google Scholar] [CrossRef]
- Mok, T.; Wu, Y.L.; Kudaba, I.; Kowalski, D.; Cho, B.; Turna, H.; Castro, G., Jr.; Srimuninnimit, V.; Laktionov, K.K.; Bondarenko, I.; et al. Pembrolizumab vs. chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): A randomised, open-label, controlled, phase 3 trial. Lancet 2019, 393, 1819–1830. [Google Scholar] [CrossRef]
- Gandhi, L.; Rodríguez-Abreu, D.; Gadgeel, S.; Esteban, E.; Felip, E.; De Angelis, F.; Domine, M.; Clingan, P.; Hochmair, M.J.; Powell, S.F.; et al. Pembrolizumab plus Chemotherapy in Metastatic Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 2078–2092. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Luft, A.; Vicente, D.; Tafreshi, A.; Gümüş, M.; Mazières, J.; Hermes, B.; Çay Şenler, F.; Csőszi, T.; Fülöp, A.; et al. Pembrolizumab plus Chemotherapy for Squamous Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 379, 2040–2051. [Google Scholar] [CrossRef]
- West, H.; McCleod, M.; Hussein, M.; Morabito, A.; Rittmeyer, A.; Conter, H.; Kopp, H.-G.; Daniel, D.; McCune, S.; Mekhail, T.; et al. Atezolizumab in combination with Carboplatin plus nab-Paclitaxel chemotherapy compared with chemotherapy alone as first-line treatment for metastatic non-squamous non-small-cell lung cancer (IMpower130): A multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2019, 20, 924–937. [Google Scholar] [CrossRef]
- Socinski, M.A.; Nishio, M.; Jotte, R.M.; Cappuzzo, F.; Orlandi, F.; Stroyakovskiy, D.; Nogami, N.; Rodríguez-Abreu, D.; Moro-Sibilot, D.; Thomas, C.A.; et al. IMpower150 Final Overall Survival Analyses for Atezolizumab Plus Bevacizumab and Chemotherapy in First-Line Metastatic Nonsquamous NSCLC. J. Thorac. Oncol. 2021, 16, 1909–1924. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Ciuleamu, T.-E.; Cobo, M.; Schenker, M.; Zurawski, B.; de Menezes, J.J.; Richardet, E.; Bennouna, J.; Felip, E.; Juan-Vidal, O.; et al. First-line Nivolumab + Ipilimumab + 2 cycles of chemotherapy vs. chemotherapy alone (4 cycles) in patients with advanced non-small cell lung cancer: 2-year update from CheckMte 9LA. Proc. Am. Soc. Clin. Oncol. 2021, 17, A9000. [Google Scholar] [CrossRef]
- Nagahashi, M.; Sato, S.; Yuza, K.; Shimada, Y.; Ichikawa, H.; Watanabe, S.; Takada, K.; Okamoto, T.; Okuda, S.; Lyle, S.; et al. Common driver mutations and smoking history affect TMB in lung adenocarcinoma. J. Surg. Res. 2018, 230, 181–185. [Google Scholar] [CrossRef] [Green Version]
- Gainor, J.; Shaw, A.; Sequist, L.; Fu, X.; Azzoli, C.; Pitrowska, Z.; Huynh, T.G.; Zhao, L.; Fulton, L.; Schultz, K.R.; et al. EGFR Mutations and ALK Rearrangements Are Associated with Low Response Rates to PD1 Pathway Blockade in Non-Small Cell Lung Cancer: A Retrospective Analysis. Clin. Cancer Res. 2016, 22, 4585–4593. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lisberg, A.; Cummings, A.; Goldman, J.; Bornazyan, K.; Reese, N.; Wang, T.; Coluzzi, P.; Ledezma, B.; Mendenhall, M.; Hunt, J.; et al. A Phase II Study of Pembrolizumab in EGFR-Mutant, PD-L1+, Tyrosine Kinase Inhibitor Naïve Patients with Advanced NSCLC. J. Thorac. Oncol. 2018, 13, 1138–1145. [Google Scholar] [CrossRef] [Green Version]
- Nishio, M.; Barlesi, F.; West, H.; Ball, S.; Bordoni, R.; Cobo, M.; Longeras, P.D.; Goldschmidt, J.; Novello, S.; Orlandi, F.; et al. Atezolizumab Plus Chemotherapy for First-Line Treatment of Nonsquamous NSCLC: Results from the Randomized Phase 3 IMpower132 Trial. J. Thorac. Oncol. 2020, 16, 653–664. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Zheng, D.; Li, Y.; Cai, X.; Zheng, Z.; Jin, Y.; Hu, H.; Cheng, C.; Shen, L.; Wang, J.; et al. Unique distribution of programmed death ligand 1 (PD-L1) expression in East Asian non-small cell lung cancer. J. Thorac. Dis. 2017, 9, 2579–2586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fancello, L.; Gandini, S.; Pelicci, P.G.; Mazzarella, L. Tumor mutational burden quantification from targeted gene panels: Major advancements and challenges. J. Immunother. Cancer 2019, 7, 183. [Google Scholar] [CrossRef] [Green Version]
- Carbone, D.P.; Reck, M.; Paz-Ares, L.; Creelan, B.; Horn, L.; Steins, M.; Felip, E.; van den Heuvel, M.M.; Ciuleanu, T.E.; Badin, F.; et al. First-line Nivolumab in stage IV or recurrent non-small-cell lung cancer. N. Engl. J. Med. 2017, 376, 2415–2426. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Paz-Ares, L.; Bernabe Caro, R.; Zurawski, B.; Kim, S.-W.; Carcereny Costa, E.; Park, K.; Alexandru, A.; Lupinacci, L.; de la Mora Jimenez, E.; et al. Nivolumab plus Ipilimumab in advanced non-small-cell lung cancer. N. Engl. J. Med. 2019, 381, 2020–2031. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, N.; Cho, B.; Reimuth, N.; Lee, H.; Luft, A. Durvalumab With or Without Tremelimumab vs Standard Chemotherapy in First-line Treatment of Metastatic Non–Small Cell Lung Cancer The MYSTIC Phase 3 Randomized Clinical Trial. JAMA Oncol. 2020, 6, 661–674. [Google Scholar] [CrossRef] [Green Version]
- Mok, T.; Aren, O.; Arrieta, O.; Schmid, P.; Gottfried, M.; Jazieh, A.R. NEPTUNE: A global, phase 3 study of Durvalumab (MEDI4736) plus Tremelimumab combination therapy vs. standard of care (SoC) platinum-based chemotherapy in the first-line treatment of patients (pts) with advanced or metastatic NSCLC. J. Thorac. Oncol. 2016, 11, S140–S141. [Google Scholar] [CrossRef] [Green Version]
- Arbour, K.; Shen, R.; Plodkowski, A.; Rizvi, H.; Ni, A.; Long, N.; Halpenny, D.; Sanchez-Vega, F.; Rudin, C.; Riely, G.; et al. Concurrent mutations in STK11 and KEAP1 is associated with resistance to PD-(L)1 blockade in patients with NSCLC despite high TMB. J. Thorac. Oncol. 2018, 13, S424. [Google Scholar] [CrossRef] [Green Version]
- Papillon-Cavanagh, S.; Doshi, P.; Dobrin, R.; Szutakowski, J.; Walsh, A. STK11 and KEAP1 mutations as prognostic biomarkers in an observational real-world lung adenocarcinoma cohort. ESMO Open 2020, 5, e000706. [Google Scholar] [CrossRef] [Green Version]
- Arbour, K.C.; Jordan, E.; Kim, H.R.; Dienstag, J.; Yu, H.A.; Sanchez-Vega, F.; Lito, P.; Berger, M.; Solit, D.B.; Hellmann, M.; et al. Effects of Co-occurring Genomic Alterations on Outcomes in Patients with KRAS-Mutant Non-Small Cell Lung Cancer. Clin. Cancer Res. 2018, 24, 334–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Skoulidis, F.; Goldbert, M.; Greenawalt, D.; Hellmann, M.; Awad, M.; Gainor, J. STK11/LKB1 Mutations and PD-1 Inhibitor Resistance in KRAS-Mutant Lung Adenocarcinoma. Cancer Discov. 2018, 8, 822–835. [Google Scholar] [CrossRef] [Green Version]
- Skoulidis, F.; Byers, L.A.; Diao, L.; Papadimitrakopoulou, V.A.; Tong, P.; Izzo, J.; Behrens, C.; Kadara, H.; Parra, E.R.; Canales, J.R.; et al. Co-occurring genomic alterations define major subsets of KRAS- mutant lung adenocarcinoma with distinct biology, immune profiles, and therapeutic vulnerabilities. Cancer Discov. 2015, 5, 860–877. [Google Scholar] [CrossRef] [Green Version]
- Routy, B.; Chatelier, E.; Derosa, L.; Duong, C.; Alou, M.; Daillère, R.; Fluckiger, A.; Messaoudene, M.; Rauber, C.; Roberti, M.P.; et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 2018, 359, 91–97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chalabi, M.; Cardona, A.; Nagarkar, D.; Scala, A.D.; Gandara, D.; Rittmeyer, A.; Albert, M.; Powles, T.; Kok, M.; Herrera, F. Efficacy of chemotherapy and Atezolizumab in patients with non-small-cell lung cancer receiving antibiotics and proton pump inhibitors: Pooled post hoc analyses of the OAK and POPLAR trials. Ann. Oncol. 2020, 31, 525–531. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pasello, G.; Pavan, A.; Attili, I.; Bortolami, A.; Bonanno, L.; Menis, J.; Conte, P.; Guarneri, V. Real world data in the era of Immune Checkpoint Inhibitors (ICIs): Increasing evidence and future applications in lung cancer. Cancer Treat. Rev. 2020, 87, 102031. [Google Scholar] [CrossRef] [PubMed]

| Trial | Line of Therapy | Immunohistochemistry Antibody and Positivity | Agents | HR for mOS (95% CI) by PDL-1 (%) |
|---|---|---|---|---|
| CheckMate 017 (squamous) | Second | 1%, 5%, 10% Dako 28-8 | Nivolumab vs. docetaxel | HR = 0.62 (0.48–0.79) |
| CheckMate 057 (non-squamous) | Second | 1%, 5%, 10% Dako 28-8 | Nivolumab vs. docetaxel | HR = 0.70 (0.58–0.83) |
| CheckMate 017/057 (pooled) | Second | Nivolumab vs. Docdetaxel | >1%: HR = 0.61 (0.49–0.76) <1% HR = 0.76 (0.61–0.96) | |
| KEYNOTE-010 | Second | >1% Dako 22C3 | Pembrolizumab 2 mg/kg and 10 mg/kg (pooled analysis) vs. docetaxel | >50% HR = 0.53 (0.42–0.66) >1% HR = 0.69 (0.60–0.80) |
| KEYNOTE-024 | First | >50% Dako 22C3 | Pembrolizumab vs. platinum-based chemotherapy | >50% HR = 0.63 (0.47–0.86) |
| KEYNOTE-042 | First | >1% Dako 22C3 | Pembrolizumab vs. platinum-based chemotherapy | >50% HR = 0.69 (0.56–0.85) 1–49% HR = 0.92 (0.77–1.10) |
| KEYNOTE-189 | First | Dako 22C3 | Pembrolizumab and platinum/pemetrexed vs. platinum/pemetrexed | >50% HR = 0.64 (0.37–1.10) 1–49% HR = 0.57 (0.36–0.90) <1% HR = 0.61 (0.38–0.98) |
| KEYONTE-407 | First | Dako 22C3 | Pembrolizumab and carboplatin/paclitaxel or nab-paclitaxel vs. carboplatin/paclitaxel or nab-paclitaxel | >50% HR 0.64 (0.37–1.10) 1–49% HR 0.57 (0.36–0.90) <1% HR 0.61 (0.38–0.98) |
| IMpower130 | First | Ventana SP142 | Atezolizumab and carboplatin/nab-paclitaxel vs. carboplatin/nab-paclitaxel | TC3/TC3 HR = 0.84 (0.51–1.39) TC1-2/IC1-2 HR = 0.70 (0.45–1.08) TC0/IC0 HR = 0.81 (0.61–1.08) |
| IMpower150 | First | Ventana SP142 | Atezolizumab/bevacizumab and carboplatin/paclitaxel vs. carboplatin/paclitaxel and bevacizumab | TC3/IC3 HR = 0.39 (0.25–0.60) TC1-2/IC1-2 HR = 0.56 (0.41–0.77) TC0/IC0 HR = 0.77 (0.61–0.99) |
| CheckMate 227 | First | Dako 28-8 | Nivolumab/ipilimumab vs. chemotherapy | >1% 17.1 months vs. 14.9 months, p = 0.007 <1% 17.2 months vs. 12.2 months |
| CheckMate 9LA | First | Dako 28-8 | Nivolumab/ipilimumab and 2 cycles of platinum-based chemotherapy vs. 4 cycles of platinum-based chemotherapy | >50% HR = 0.67 (0.46–0.97) 1–49% HR = 0.70 (0.56–0.89) <1% HR = 0.67 (0.51–0.88) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khwaja, R.M.; Chu, Q.S.-C. Present and Emerging Biomarkers in Immunotherapy for Metastatic Non-Small Cell Lung Cancer: A Review. Curr. Oncol. 2022, 29, 479-489. https://doi.org/10.3390/curroncol29020043
Khwaja RM, Chu QS-C. Present and Emerging Biomarkers in Immunotherapy for Metastatic Non-Small Cell Lung Cancer: A Review. Current Oncology. 2022; 29(2):479-489. https://doi.org/10.3390/curroncol29020043
Chicago/Turabian StyleKhwaja, Raida M., and Quincy S.-C. Chu. 2022. "Present and Emerging Biomarkers in Immunotherapy for Metastatic Non-Small Cell Lung Cancer: A Review" Current Oncology 29, no. 2: 479-489. https://doi.org/10.3390/curroncol29020043
APA StyleKhwaja, R. M., & Chu, Q. S.-C. (2022). Present and Emerging Biomarkers in Immunotherapy for Metastatic Non-Small Cell Lung Cancer: A Review. Current Oncology, 29(2), 479-489. https://doi.org/10.3390/curroncol29020043




