Stratification of Length of Stay Prediction following Surgical Cytoreduction in Advanced High-Grade Serous Ovarian Cancer Patients Using Artificial Intelligence; the Leeds L-AI-OS Score
Abstract
1. Introduction
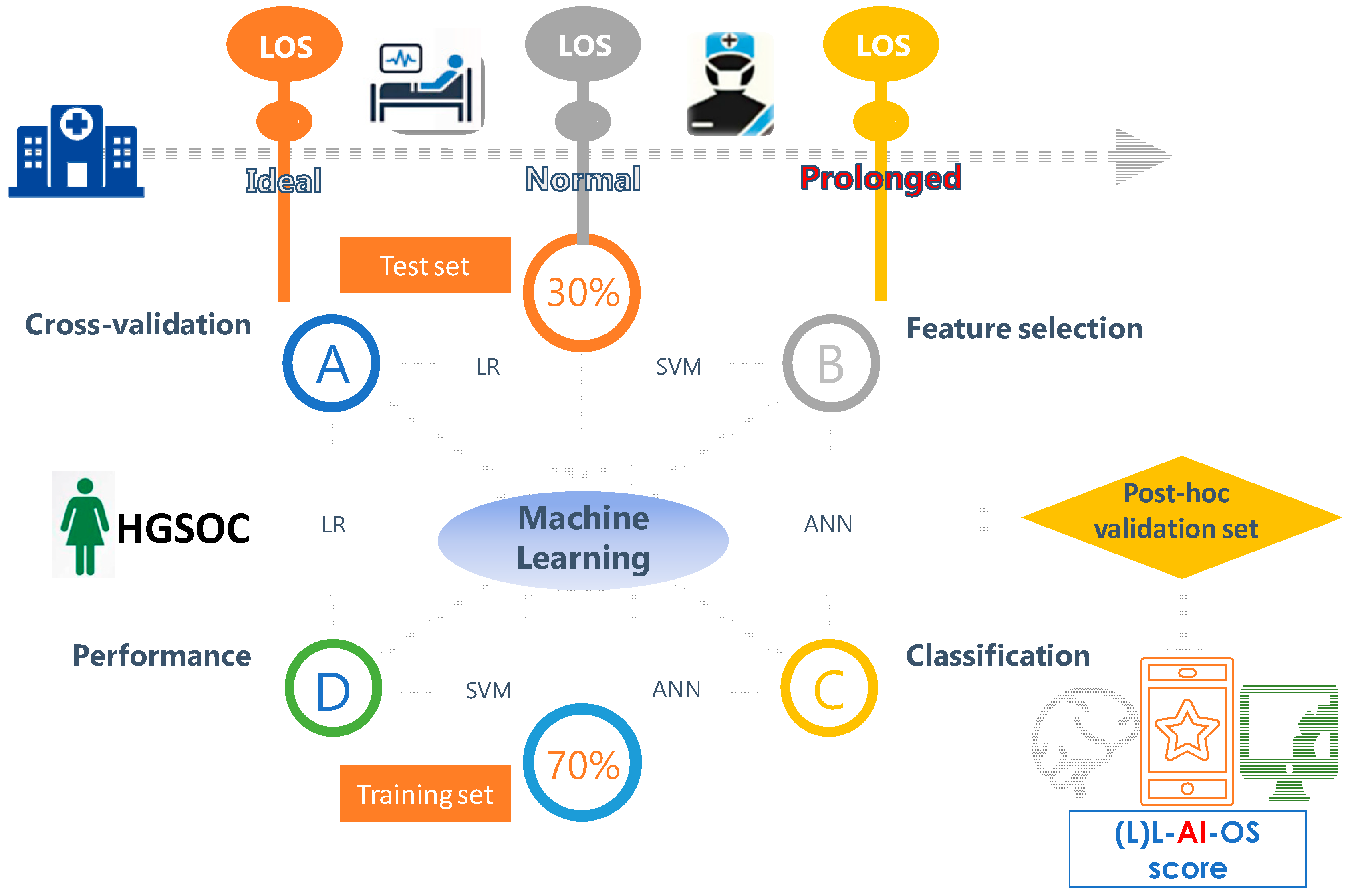
2. Materials and Methods
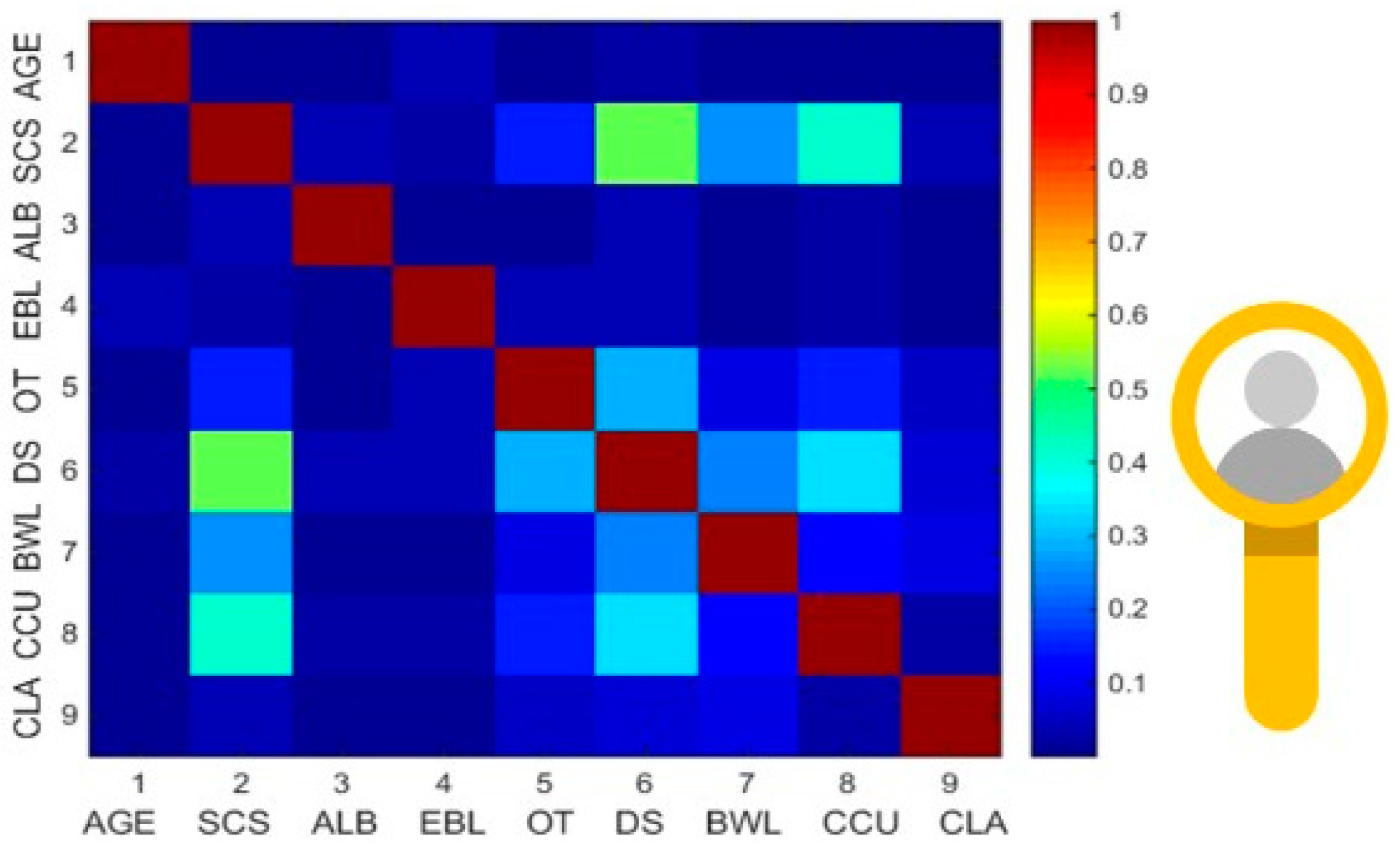
2.1. Feature Selection
2.2. Model Development
2.3. Multivariate Analysis
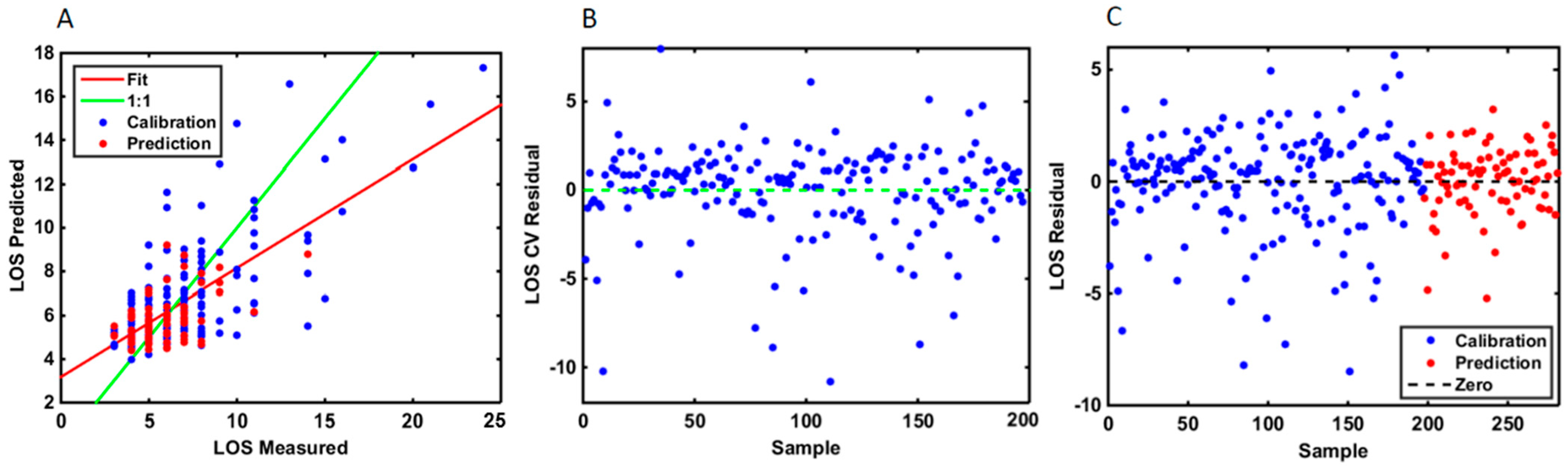
2.4. Model Performance
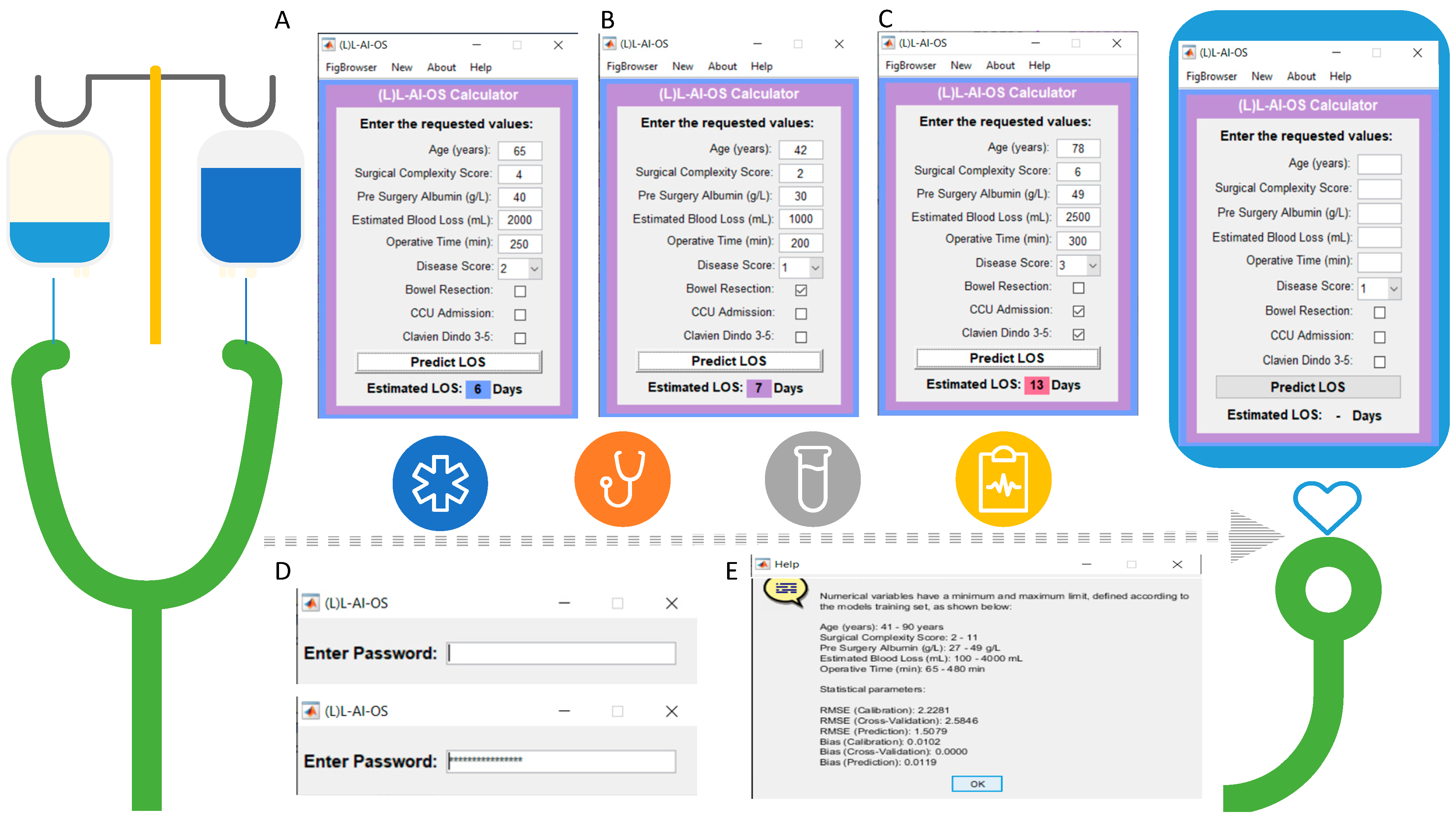
2.5. Development of the LEEDS L-AI-OS Score
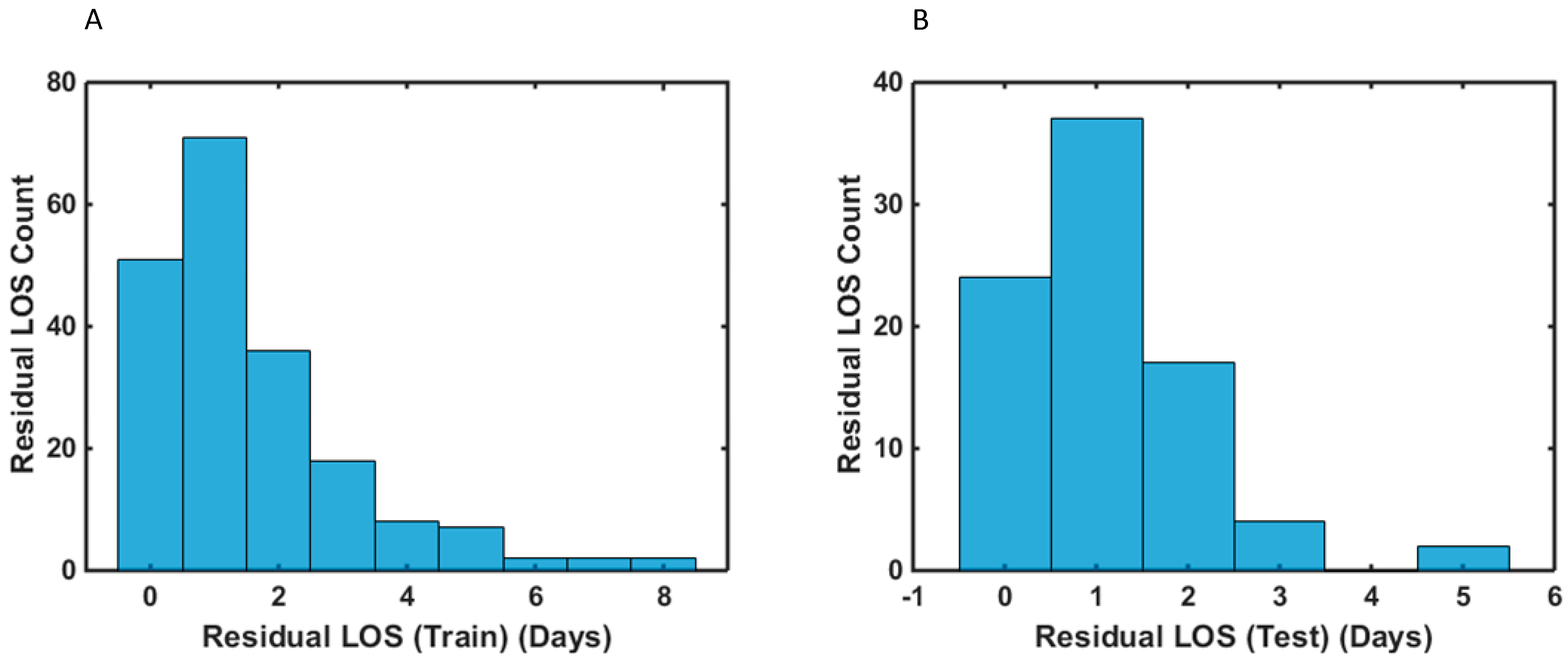
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Spanjersberg, W.R.; Reurings, J.; Keus, F.; van Laarhoven, C.J. Fast track surgery versus conventional recovery strategies for colorectal surgery. Cochrane Database Syst. Rev. 2011, 2, CD007635. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Iglesias, J.L.; Carbonell-Socias, M.; Pérez-Benavente, M.A.; Monreal Clua, S.; Manrique-Muñoz, S.; García Gorriz, M.; Burgos-Peláez, R.; Segurola Gurrutxaga, H.; Pamies Serrano, M.; Pilar Gutiérrez-Barceló, M.D.; et al. PROFAST: A randomised trial implementing enhanced recovery after surgery for highcomplexity advanced ovarian cancer surgery. Eur. J. Cancer 2020, 136, 149–158. [Google Scholar] [CrossRef]
- Nazzani, S.; Preisser, F.; Mazzone, E.; Tian, Z.; Mistretta, F.A.; Shariat, S.F.; Saad, F.; Graefen, M.; Tilki, D.; Montanari, E.; et al. In-hospital length of stay after major surgical oncological procedures. Eur. J. Surg. Oncol. 2018, 44, 969–974. [Google Scholar] [CrossRef] [PubMed]
- Brasel, K.J.; Lim, H.J.; Nirula, R.; Weigelt, J.A. Length of stay: An appropriate quality measure? Arch. Surg. 2007, 142, 461–465; discussion 465–466. [Google Scholar] [CrossRef]
- Smith, C.G.; Davenport, D.L.; Gorski, J.; McDowell, A.; Burgess, B.T.; Fredericks, T.I.; Baldwin, L.A.; Miller, R.W.; DeSimone, C.P.; Dietrich, C.S.; et al. Clinical Factors Associated with Longer Hospital Stay Following Ovarian Cancer Surgery. Healthcare 2019, 7, 85. [Google Scholar] [CrossRef]
- Allard, J.P.; Keller, H.; Teterina, A.; Jeejeebhoy, K.N.; Laporte, M.; Duerksen, D.R.; Gramlich, L.; Payette, H.; Bernier, P.; Davidson, B.; et al. Factors associated with nutritional decline in hospitalised medical and surgical patients admitted for 7 d or more: A prospective cohort study. Br. J. Nutr. 2015, 114, 1612–1622. [Google Scholar] [CrossRef]
- Mahdi, H.; Jernigan, A.; Lockhart, D.; Moslemi-Kebria, M.; Rose, P.G. Racial disparity in 30-day morbidity and mortality after surgery for ovarian cancer. Int. J. Gynecol. Cancer 2015, 25, 55–62. [Google Scholar] [CrossRef]
- Bateni, S.B.; Meyers, F.J.; Bold, R.J.; Canter, R.J. Increased Rates of Prolonged Length of Stay, Readmissions, and Discharge to Care Facilities among Postoperative Patients with Disseminated Malignancy: Implications for Clinical Practice. PLoS ONE 2016, 11, e0165315. [Google Scholar] [CrossRef]
- Prescott, L.S.; Aloia, T.A.; Brown, A.J.; Taylor, J.S.; Munsell, M.F.; Sun, C.C.; Schmeler, K.M.; Levenback, C.F.; Bodurka, D.C. Perioperative blood transfusion in gynecologic oncology surgery: Analysis of the National Surgical Quality Improvement Program Database. Gynecol. Oncol. 2015, 136, 65–70. [Google Scholar] [CrossRef]
- Forsmo, H.M.; Pfeffer, F.; Rasdal, A.; Sintonen, H.; Körner, H.; Erichsen, C. Pre- and postoperative stoma education and guidance within an enhanced recovery after surgery (ERAS) programme reduces length of hospital stay in colorectal surgery. Int J. Surg. 2016, 36, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Badia, J.M.; Casey, A.L.; Petrosillo, N.; Hudson, P.M.; Mitchell, S.A.; Crosby, C. Impact of surgical site infection on healthcare costs and patient outcomes: A systematic review in six European countries. J. Hosp. Infect. 2017, 96, 1–15. [Google Scholar] [CrossRef]
- Mujagic, E.; Marti, W.R.; Coslovsky, M.; Soysal, S.D.; Mechera, R.; von Strauss, M.; Zeindler, J.; Saxer, F.; Mueller, A.; Fux, C.A.; et al. Associations of Hospital Length of Stay with Surgical Site Infections. World J. Surg. 2018, 42, 3888–3896. [Google Scholar] [CrossRef] [PubMed]
- Aletti, G.D.; Santillan, A.; Eisenhauer, E.L.; Hu, J.; Aletti, G.; Podratz, K.C.; Bristow, R.E.; Chi, D.S.; Cliby, W.A. A new frontier for quality of care in gynecologic oncology surgery: Multi-institutional assessment of short-term outcomes for ovarian cancer using a risk-adjusted model. Gynecol. Oncol. 2007, 107, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Sajobi, T.; Lucyk, K.; Lorenzetti, D.; Quan, H. Systematic review of risk adjustment models of hospital length of stay (LOS). Med. Care 2015, 53, 355–365. [Google Scholar] [CrossRef]
- Rowan, M.; Ryan, T.; Hegarty, F.; O’Hare, N. The use of artificial neural networks to stratify the length of stay of cardiac patients based on preoperative and initial postoperative factors. Artif. Intell. Med. 2007, 40, 211–221. [Google Scholar] [CrossRef]
- Laios, A.; Gryparis, A.; DeJong, D.; Hutson, R.; Theophilou, G.; Leach, C. Predicting complete cytoreduction for advanced ovarian cancer patients using nearest-neighbor models. J. Ovarian Res. 2020, 13, 117. [Google Scholar] [CrossRef]
- Laios, A.; Katsenou, A.; Tan, Y.S.; Johnson, R.; Otify, M.; Kaufmann, A.; Munot, S.; Thangavelu, A.; Hutson, R.; Broadhead, T.; et al. Feature Selection is Critical for 2-Year Prognosis in Advanced Stage High Grade Serous Ovarian Cancer by Using Machine Learning. Cancer Control 2021, 28, 10732748211044678. [Google Scholar] [CrossRef]
- Intensive Care Society. Levels of Critical Care for Adult Patients 2009. 2018. Available online: www.scribd.com/document/222104382/ICS-Levels-of-Critical-Care-for-Adult-Patients-2009 (accessed on 25 January 2022).
- Older, P.; Hall, A.; Hader, R. Cardiopulmonary exercise testing as a screening test for perioperative management of major surgery in the elderly. Chest 1999, 116, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Clavien, P.A.; Barkun, J.; de Oliveira, M.L.; Vauthey, J.N.; Dindo, D.; Schulick, R.D.; de Santibañes, E.; Pekolj, J.; Slankamenac, K.; Bassi, C.; et al. The Clavien-Dindo classification of surgical complications: Five-year experience. Ann. Surg. 2009, 250, 187–196. [Google Scholar] [CrossRef]
- Armitage, J.N.; van der Meulen, J.H.; Royal College of Surgeons Co-morbidity Consensus Group. Identifying co-morbidity in surgical patients using administrative data with the Royal College of Surgeons Charlson Score. Br. J. Surg. 2010, 97, 772–781. [Google Scholar] [CrossRef]
- Aletti, G.D.; Dowdy, S.C.; Podratz, K.C.; Cliby, W.A. Relationship among surgical complexity, short-term morbidity, and overall survival in primary surgery for advanced ovarian cancer. Am. J. Obstet. Gynecol. 2007, 197, 676.e1–676.e7. [Google Scholar] [CrossRef]
- Walker, K.; Finan, P.J.; van der Meulen, J.H. Model for risk adjustment of postoperative mortality in patients with colorectal cancer. Br. J. Surg. 2015, 102, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Ballabio, D.; Consonni, V. Classification tools in chemistry. Part 1: Linear models. PLS-DA. Anal. Methods 2013, 5, 3790–3798. [Google Scholar] [CrossRef]
- Cortes, C.; Vapnik, V. Support-vector networks. Mach. Learn. 1995, 20, 273–297. [Google Scholar] [CrossRef]
- Laios, A.; De Oliveira Silva, R.V.; Dantas De Freitas, D.L.; Tan, Y.S.; Saalmink, G.; Zubayraeva, A.; Johnson, R.; Kaufmann, A.; Otify, M.; Hutson, R.; et al. Machine Learning-Based Risk Prediction of Critical Care Unit Admission for Advanced Stage High Grade Serous Ovarian Cancer Patients Undergoing Cytoreductive Surgery: The Leeds-Natal Score. J. Clin. Med. 2021, 11, 87. [Google Scholar] [CrossRef]
- Dantas De Freitas, D.L.; Câmara, I.M.; Silva, P.P.; Wanderley, N.R.S.; Alves, M.B.C.; Morais, C.L.M.; Martin, F.L.; Lajus, T.B.P.; Lima, K.M.G. Spectrochemical analysis of liquid biopsy harnessed to multivariate analysis towards breast cancer screening. Sci. Rep. 2020, 10, 12818. [Google Scholar] [CrossRef]
- Luo, W.; Phung, Q.-D.; Tran, T.; Gupta, S.; Rana, S.; Karmakar, C.; Shilton, A.; Yearwood, J.L.; Dimitrova, N.; Ho, T.B.; et al. Guidelines for Developing and Reporting Machine Learning Predictive Models in Biomedical Research: A Multidisciplinary View. J. Med. Internet Res. 2016, 18, e323. [Google Scholar] [CrossRef]
- Khuri, S.F. The NSQIP: A new frontier in surgery. Surgery 2005, 138, 837–843. [Google Scholar] [CrossRef]
- Chiang, A.C.; Lake, J.; Sinanis, N.; Brandt, D.; Kanowitz, J.; Kidwai, W.; Kortmansky, J.; LaSala, J.; Orell, J.; Sabbath, K.; et al. Measuring the Impact of Academic Cancer Network Development on Clinical Integration, Quality of Care, and Patient Satisfaction. J. Oncol. Pract. 2018, 12, e823–e833. [Google Scholar] [CrossRef]
- Ladbury, C.; Zarinshenas, R.; Semwal, H.; Tam, A.; Vaidehi, N.; Rodin, A.; Liu, A.; Glaser, S.; Salgia, R.; Amini, A. Utilization of model-agnostic explainable artificial intelligence frameworks in oncology: A narrative review. Transl. Cancer Res. 2022, 11, 3853–3868. [Google Scholar] [CrossRef] [PubMed]
- Laios, A.; Kalampokis, E.; Johnson, R.; Thangavelu, A.; Tarabanis, C.; Nugent, D.; De Jong, D. Explainable Artificial Intelligence for Prediction of Complete Surgical Cytoreduction in Advanced-Stage Epithelial Ovarian Cancer. J. Pers. Med. 2022, 12, 607. [Google Scholar] [CrossRef]
- Laios, A.; Kalampokis, E.; Johnson, R.; Munot, S.; Thangavelu, A.; Hutson, R.; Broadhead, T.; Theophilou, G.; Leach, C.; Nugent, D.; et al. Factors Predicting Surgical Effort Using Explainable Artificial Intelligence in Advanced Stage Epithelial Ovarian Cancer. Cancers 2022, 14, 3447. [Google Scholar] [CrossRef] [PubMed]
- Aravani, A.; Samy, E.F.; Thomas, J.D.; Quirke, P.; Morris, E.J.A.; Finan, P.J. A retrospective observational study of length of stay in hospital after colorectal cancer surgery in England (1998–2010). Medicine 2016, 95, e5064. [Google Scholar] [CrossRef] [PubMed]
- Fagotti, A.; Ferrandina, G.; Vizzielli, G.; Fanfani, F.; Gallotta, V.; Chiantera, V.; Costantini, B.; Margariti, P.A.; Gueli Alletti, S.; Cosentino, F.; et al. Phase III randomised clinical trial comparing primary surgery versus neoadjuvant chemotherapy in advanced epithelial ovarian cancer with high tumour load (SCORPION trial): Final analysis of peri-operative outcome. Eur. J. Cancer. 2016, 59, 22–33. [Google Scholar] [CrossRef]
- Smart, N.J.; White, P.; Allison, A.S.; Ockrim, J.B.; Kennedy, R.H.; Francis, N.K. Deviation and failure of enhanced recovery after surgery following laparoscopic colorectal surgery: Early prediction model. Colorectal Dis. 2012, 10, e727–e734. [Google Scholar] [CrossRef]
- Menachemi, N.; Rahurkar, S.; Harle, C.A.; Vest, J.R. The benefits of health information exchange: An updated systematic review. J. Am. Med. Inform. Assoc. 2018, 25, 1259–1265. [Google Scholar] [CrossRef]
- Weiser, M.R.; Gonen, M.; Usiak, S.; Pottinger, T.; Samedy, P.; Patel, D.; Seo, S.; Smith, J.J.; Guillem, J.G.; Temple, L.; et al. Memorial Sloan Kettering Multidisciplinary Surgical-Site Infection Reduction Team. Effectiveness of a multidisciplinary patient care bundle for reducing surgical-site infections. Br. J. Surg. 2018, 105, 1680–1687. [Google Scholar] [CrossRef]
- Ayyoubzadeh, S.M.; Ghazisaeedi, M.; Rostam Niakan Kalhori, S.; Hassaniazad, M.; Baniasadi, T.; Maghooli, K.; Kahnouji, K. A study of factors related to patients’ length of stay using data mining techniques in a general hospital in southern Iran. Health Inf. Sci. Syst. 2020, 8, 9. [Google Scholar] [CrossRef]
- Rashidi, L.; Long, K.; Hawkins, M.; Menon, R.; Bellevue, O. Stoma creation: Does onset of ostomy care education delay hospital length of stay? Am. J. Surg. 2016, 211, 954–957. [Google Scholar] [CrossRef]
- Laky, B.; Janda, M.; Kondalsamy-Chennakesavan, S.; Cleghorn, G.; Obermair, A. Pretreatment malnutrition and quality of life—Association with prolonged length of hospital stay among patients with gynecological cancer: A cohort study. BMC Cancer 2010, 10, 232. [Google Scholar] [CrossRef] [PubMed]
- Kathiresan, A.S.; Brookfield, K.F.; Schuman, S.I.; Lucci, J.A., 3rd. Malnutrition as a predictor of poor postoperative outcomes in gynecologic cancer patients. Arch. Gynecol. Obstet. 2011, 284, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, J.; Cull, W.; Henderson, W.; Daley, J.; Hur, K.; Khuri, S.F. Preoperative serum albumin level as a predictor of operative mortality and morbidity: Results from the National VA Surgical Risk Study. Arch. Surg. 1999, 134, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Alphs, H.H.; Zahurak, M.L.; Bristow, R.E.; Díaz-Montes, T.P. Predictors of surgical outcome and survival among elderly women diagnosed with ovarian and primary peritoneal cancer. Gynecol. Oncol. 2006, 103, 1048–1053. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.W.; Yin, T.; Cao, W.X.; Gu, Z.D.; Wang, X.J.; Yan, M.; Liu, B.Y. Clinical application of subjective global assessment in Chinese patients with gastrointestinal cancer. World J. Gastroenterol. 2009, 15, 3542–3549. [Google Scholar] [CrossRef]
- Arends, J.; Baracos, V.; Bertz, H.; Bozzetti, F.; Calder, P.C.; Deutz, N.E.P.; Erickson, N.; Laviano, A.; Lisanti, M.P.; Lobo, D.N.; et al. ESPEN expert group recommendations for action against cancer-related malnutrition. Clin. Nutr. 2017, 36, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Milne, A.C.; Potter, J.; Vivanti, A.; Avenell, A. Protein and energy supplementation in elderly people at risk from malnutrition. Cochrane Database Syst. Rev. 2009, 2009, CD003288. [Google Scholar] [CrossRef]
- Sanger, P.C.; van Ramshorst, G.H.; Mercan, E.; Huang, S.; Hartzler, A.L.; Armstrong, C.A.; Lordon, R.J.; Lober, W.B.; Evans, H.L. A Prognostic Model of Surgical Site Infection Using Daily Clinical Wound Assessment. J. Am. Coll. Surg. 2016, 223, 259–270.e2. [Google Scholar] [CrossRef]
- Dean, M.M.; Finan, M.A.; Kline, R.C. Predictors of complications and hospital stay in gynecologic cancer surgery. Obstet Gynecol. 2001, 97 Pt 1, 721–724. [Google Scholar]
- Iglesias, D.A.; Westin, S.N.; Rallapalli, V.; Huang, M.; Fellman, B.; Urbauer, D.; Frumovitz, M.; Ramirez, P.T.; Soliman, P.T. The effect of body mass index on surgical outcomes and survival following pelvic exenteration. Gynecol. Oncol. 2012, 125, 336–342. [Google Scholar] [CrossRef]
- Ross, M.S.; Burriss, M.E.; Winger, D.G.; Edwards, R.P.; Courtney-Brooks, M.; Boisen, M.M. Unplanned postoperative intensive care unit admission for ovarian cancer cytoreduction is associated with significant decrease in overall survival. Gynecol. Oncol. 2018, 150, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Earle, C.C.; Park, E.R.; Lai, B.; Weeks, J.C.; Ayanian, J.Z.; Block, S. Identifying potential indicators of the quality of end-of-life cancer care from administrative data. J. Clin. Oncol. 2003, 21, 1133–1138. [Google Scholar] [CrossRef] [PubMed]
- Birkmeyer, J.D.; Dimick, J.B.; Birkmeyer, N.J. Measuring the quality of surgical care: Structure, process, or outcomes? J. Am. Coll. Surg. 2004, 198, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves-Bradley, D.C.; Lannin, N.A.; Clemson, L.; Cameron, I.D.; Shepperd, S. Discharge planning from hospital. Cochrane Database Syst. Rev. 2022, 2, CD000313. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.; Neuburger, J.; Walker, K.; Cromwell, D.; Groene, O. How is feedback from national clinical audits used? Views from English National Health Service trust audit leads. J. Health Serv. Res. Policy 2016, 21, 91–100. [Google Scholar] [CrossRef]
| Variable | Type | p-Value |
|---|---|---|
| Age | Numerical | 0.049 |
| BMI | Numerical | 0.317 |
| Performance Status | Numerical | 0.293 |
| CCI | Numerical | 0.985 |
| Type of Surgery | Categorical | 0.124 |
| SCS | Numerical | 0.000 |
| Disease Score | Categorical | 0.002 |
| CA 125 | Numerical | 0.458 |
| Albumin | Numerical | 0.001 |
| EBL | Numerical | 0.000 |
| Operative Time | Numerical | 0.000 |
| Bowel Resection | Categorical | 0.000 |
| Residual | Numerical | 0.363 |
| R0 | Categorical | 0.262 |
| CCU Admission | Categorical | 0.000 |
| Clavien-Dindo complications | Categorical | 0.000 |
| Variable | Age (Years) | Surgical Complexity Score (SCS) | Disease Score (DS) | Pre Surgery Alb (ALB) | Estimated Blood Loss (EBL) (mL) | Operative Time (OT) (min) | LOS (Days) |
|---|---|---|---|---|---|---|---|
| Mean | 64 | 4 | 2 | 39 | 484 | 181 | 6 |
| Standard Deviation | 10 | 2 | 1 | 4 | 411 | 76 | 4 |
| Minimum | 41 | 2 | 1 | 27 | 100 | 65 | 3 |
| Maximum | 90 | 11 | 3 | 49 | 4000 | 480 | 24 |
| Tenth Percentile | 50 | 2 | 2 | 34 | 200 | 105 | 4 |
| Lower Quartile | 56 | 2 | 2 | 36 | 250 | 120 | 5 |
| Median | 65 | 3 | 2 | 38 | 400 | 160 | 5 |
| Upper Quartile | 73 | 4 | 2 | 41 | 500 | 225 | 7 |
| 90th centile | 77 | 6 | 3 | 43 | 900 | 285 | 9 |
| LOS (1 ≤ 5 d)—Ideal (2 ≥ 6 d)—Prolonged 1 (3 ≥ 9 d)—Prolonged 2 | Model | Set | Accuracy | Sensitivity | Specificity | F-Score | G-Score |
|---|---|---|---|---|---|---|---|
| 1 | ANN | TRAIN | 93% | 93% | 93% | 93% | 93% |
| 1 | ANN | CV | 62% | 62% | 62% | 62% | 62% |
| 1 | ANN | TEST | 64% | 67% | 59% | 63% | 63% |
| 1 | SVM | TRAIN | 79% | 91% | 68% | 78% | 79% |
| 1 | SVM | CV | 72% | 79% | 65% | 71% | 72% |
| 1 | SVM | TEST | 69% | 89% | 45% | 60% | 63% |
| 1 | LR | TRAIN | 73% | 78% | 68% | 73% | 73% |
| 1 | LR | TEST | 70% | 91% | 45% | 60% | 64% |
| 2 | ANN | TRAIN | 98% | 99% | 98% | 98% | 98% |
| 2 | ANN | CV | 71% | 64% | 76% | 69% | 70% |
| 2 | ANN | TEST | 76% | 45% | 87% | 59% | 63% |
| 2 | SVM | TRAIN | 100% | 100% | 100% | 100% | 100% |
| 2 | SVM | CV | 72% | 67% | 75% | 71% | 71% |
| 2 | SVM | TEST | 68% | 39% | 79% | 52% | 56% |
| 2 | LR | TRAIN | 75% | 51% | 90% | 65% | 68% |
| 2 | LR | TEST | 75% | 35% | 90% | 50% | 56% |
| 3 | ANN | TRAIN | 97% | 96% | 97% | 96% | 96% |
| 3 | ANN | CV | 80% | 54% | 84% | 66% | 67% |
| 3 | ANN | TEST | 89% | 50% | 90% | 64% | 67% |
| 3 | SVM | TRAIN | 97% | 81% | 100% | 90% | 90% |
| 3 | SVM | CV | 86% | 35% | 94% | 51% | 57% |
| 3 | SVM | TEST | 94% | 50% | 95% | 66% | 69% |
| 3 | LR | TRAIN | 90% | 46% | 97% | 63% | 67% |
| 3 | LR | TEST | 98% | 0% | 100% | 0% | 0% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Laios, A.; De Freitas, D.L.D.; Saalmink, G.; Tan, Y.S.; Johnson, R.; Zubayraeva, A.; Munot, S.; Hutson, R.; Thangavelu, A.; Broadhead, T.; et al. Stratification of Length of Stay Prediction following Surgical Cytoreduction in Advanced High-Grade Serous Ovarian Cancer Patients Using Artificial Intelligence; the Leeds L-AI-OS Score. Curr. Oncol. 2022, 29, 9088-9104. https://doi.org/10.3390/curroncol29120711
Laios A, De Freitas DLD, Saalmink G, Tan YS, Johnson R, Zubayraeva A, Munot S, Hutson R, Thangavelu A, Broadhead T, et al. Stratification of Length of Stay Prediction following Surgical Cytoreduction in Advanced High-Grade Serous Ovarian Cancer Patients Using Artificial Intelligence; the Leeds L-AI-OS Score. Current Oncology. 2022; 29(12):9088-9104. https://doi.org/10.3390/curroncol29120711
Chicago/Turabian StyleLaios, Alexandros, Daniel Lucas Dantas De Freitas, Gwendolyn Saalmink, Yong Sheng Tan, Racheal Johnson, Albina Zubayraeva, Sarika Munot, Richard Hutson, Amudha Thangavelu, Tim Broadhead, and et al. 2022. "Stratification of Length of Stay Prediction following Surgical Cytoreduction in Advanced High-Grade Serous Ovarian Cancer Patients Using Artificial Intelligence; the Leeds L-AI-OS Score" Current Oncology 29, no. 12: 9088-9104. https://doi.org/10.3390/curroncol29120711
APA StyleLaios, A., De Freitas, D. L. D., Saalmink, G., Tan, Y. S., Johnson, R., Zubayraeva, A., Munot, S., Hutson, R., Thangavelu, A., Broadhead, T., Nugent, D., Kalampokis, E., de Lima, K. M. G., Theophilou, G., & De Jong, D. (2022). Stratification of Length of Stay Prediction following Surgical Cytoreduction in Advanced High-Grade Serous Ovarian Cancer Patients Using Artificial Intelligence; the Leeds L-AI-OS Score. Current Oncology, 29(12), 9088-9104. https://doi.org/10.3390/curroncol29120711







