Diagnosis, Treatment and Prognosis of Primary Pulmonary NUT Carcinoma: A Literature Review
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Clinical Features
3.2. Imaging Examination Findings
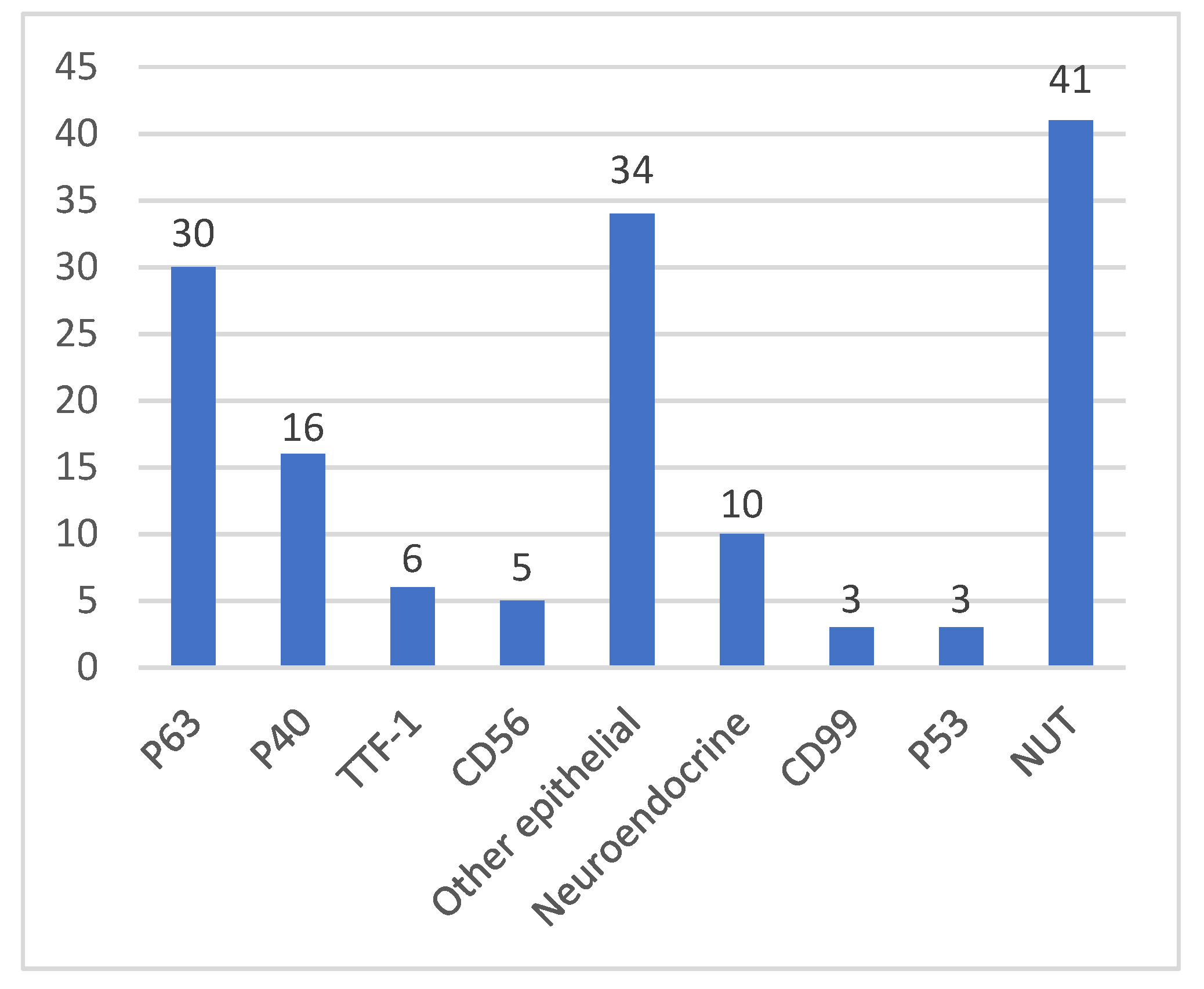
3.3. Pathological Characteristics
3.4. Treatments and Outcomes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- French, C.A.; Ramirez, C.L.; Kolmakova, J.; Hickman, T.T.; Cameron, M.J.; Thyne, M.E.; Kutok, J.L.; Toretsky, J.A.; Tadavarthy, A.K.; Kees, U.R.; et al. BRD-NUT oncoproteins: A family of closely related nuclear proteins that block epithelial differentiation and maintain the growth of carcinoma cells. Oncogene 2008, 27, 2237–2242. [Google Scholar] [CrossRef] [PubMed]
- Bauer, D.E.; Mitchell, C.M.; Strait, K.M.; Lathan, C.S.; Stelow, E.B.; Lüer, S.C.; Muhammed, S.; Evans, A.G.; Sholl, L.M.; Rosai, J.; et al. Clinicopathologic features and long-term outcomes of NUT midline carcinoma. Clin. Cancer Res. 2012, 18, 5773–5779. [Google Scholar] [CrossRef] [PubMed]
- Aryal, S.C.; Zia, S.; Rodgers, S.; Shen, Y.; Perry, K.; Yuan, L. BRD3-NUTM1-expressing NUT carcinoma of lung on endobronchial ultrasound-guided transbronchial needle aspiration cytology, a diagnostic pitfall. Diagn. Cytopathol. 2022, 50, E47–E53. [Google Scholar] [CrossRef] [PubMed]
- Baras, A.S.; Naidoo, J.; Hann, C.L.; Illei, P.B.; Reninger, C.W.; Lauring, J. Rediagnosis of Lung Cancer as NUT Midline Carcinoma Based on Clues From Tumor Genomic Profiling. J. Natl. Compr. Canc. Netw. 2018, 16, 467–472. [Google Scholar] [CrossRef]
- Benito Bernáldez, C.; Romero Muñoz, C.; Almadana Pacheco, V. NUT midline carcinoma of the lung, a rare form of lung cancer. Arch. Bronconeumol. 2016, 52, 619–621. [Google Scholar] [CrossRef]
- Cao, J.; Chen, D.; Yang, F.; Yao, J.; Zhu, W.; Zhao, C. NUT midline carcinoma as a primary lung tumor: A case report. J. Thorac. Dis. 2017, 9, E1045–E1049. [Google Scholar] [CrossRef]
- Claudia, G.; Alexandra, G. Challenging Diagnosis in NUT Carcinoma. Int. J. Surg. Pathol. 2021, 29, 722–725. [Google Scholar] [CrossRef]
- Fekkar, A.; Emprou, C.; Lefebvre, C.; Toffart, A.C.; Rousseaux, S.; Khochbin, S.; Lantuejoul, S.; Valmary-Degano, S. Thoracic NUT carcinoma: Common pathological features despite diversity of clinical presentations. Lung Cancer 2021, 158, 55–59. [Google Scholar] [CrossRef]
- Fujioka, N.; French, C.A.; Cameron, M.J.; Kratzke, R.A. Long-term survival of a patient with squamous cell carcinoma harboring NUT gene rearrangement. J. Thorac. Oncol. 2010, 5, 1704–1705. [Google Scholar] [CrossRef]
- Costa, B.A.; Maraveyas, A.; Wilkoff, M.H.; Correia, G.S.C.; Tallón de Lara, P.; Rohs, N.C.; Salonia, J. Primary Pulmonary NUT Carcinoma: Case Illustration and Updated Review of Literature. Clin. Lung Cancer 2022, 23, e296–e300. [Google Scholar] [CrossRef]
- Gasljevic, G.; Matter, M.S.; Blatnik, O.; Unk, M.; Dirnhofer, S. NUT Carcinoma: A Clinical, Morphological and Immunohistochemical Mimicker-The Role of RNA Sequencing in the Diagnostic Procedure. Int. J. Surg. Pathol. 2022, 30, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Mumaw, D.; Antonios, B.; Anusim, N.; Dhulipalla, S.P.; Stender, M.; Huben, M.; Jaiyesimi, I. NUT midline lung cancer: A rare case report with literature review. AME Case Rep. 2022, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Harms, A.; Herpel, E.; Pfarr, N.; Penzel, R.; Heussel, C.P.; Herth, F.J.; Dienemann, H.; Weichert, W.; Warth, A. NUT carcinoma of the thorax: Case report and review of the literature. Lung Cancer 2015, 90, 484–491. [Google Scholar] [CrossRef]
- Jiang, J.; Ren, Y.; Xu, C.; Lin, X. NUT midline carcinoma as a primary lung tumor treated with anlotinib combined with palliative radiotherapy: A case report. Diagn. Pathol. 2022, 17, 4. [Google Scholar] [CrossRef] [PubMed]
- Karakuş, E.; Poyraz, A.; Oğuz Erdogan, A.S.; Emir, S.; Özyörük, D. NUT Midline Carcinoma of the Lung in a Six-Year-Old Child. Fetal Pediatr. Pathol. 2017, 36, 472–474. [Google Scholar] [CrossRef]
- Keymel, S.; Krüger, S. Lung Cancer with Prominent Mediastinal Lymphadenopathy—Who Thinks of NUT Carcinoma? Pneumologie 2020, 74, 35–38. [Google Scholar]
- Kuroda, S.; Suzuki, S.; Kurita, A.; Muraki, M.; Aoshima, Y.; Tanioka, F.; Sugimura, H. Cytological Features of a Variant NUT Midline Carcinoma of the Lung Harboring the NSD3-NUT Fusion Gene: A Case Report and Literature Review. Case Rep. Pathol. 2015, 2015, 572951. [Google Scholar] [PubMed]
- Lee, T.; Choi, S.; Han, J.; Choi, Y.L.; Lee, K. Abrupt Dyskeratotic and Squamoid Cells in Poorly Differentiated Carcinoma: Case Study of Two Thoracic NUT Midline Carcinomas with Cytohistologic Correlation. J. Pathol. Transl. Med. 2018, 52, 349–353. [Google Scholar] [CrossRef]
- Liu, S.; Ferzli, G. NUT carcinoma: A rare and devastating neoplasm. BMJ Case Rep. 2018, 2018, bcr2018226526. [Google Scholar] [CrossRef]
- Liu, Y.; Li, Y.Y.; Ke, X.X.; Lu, Y. The primary pulmonary NUT carcinomas and some uncommon somatic mutations identified by next-generation sequencing: A case report. AME Case Rep. 2020, 4, 24. [Google Scholar] [CrossRef]
- Mao, N.; Liao, Z.; Wu, J.; Liang, K.; Wang, S.; Qin, S.; Dou, Y.; Lin, H.; Dong, X. Diagnosis of NUT carcinoma of lung origin by next-generation sequencing: Case report and review of the literature. Cancer Biol. Ther. 2019, 20, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Numakura, S.; Saito, K.; Motoi, N.; Mori, T.; Saito, Y.; Yokote, F.; Kanamoto, Y.; Asami, M.; Sakai, T.; Yamauchi, Y.; et al. P63-negative pulmonary NUT carcinoma arising in the elderly: A case report. Diagn. Pathol. 2020, 15, 134. [Google Scholar] [CrossRef] [PubMed]
- Pezzuto, F.; Fortarezza, F.; Mammana, M.; Pasello, G.; Pelosi, G.; Rea, F.; Calabrese, F. Immunohistochemical neuroendocrine marker expression in primary pulmonary NUT carcinoma: A diagnostic pitfall. Histopathology 2020, 77, 508–510. [Google Scholar] [CrossRef]
- Policarpio-Nicolas, M.L.; de Leon, E.M.; Jagirdar, J. Cytologic findings of NUT midline carcinoma in the hilum of the lung. Diagn Cytopathol. 2015, 43, 739–742. [Google Scholar] [CrossRef]
- Raza, A.; Cao, H.; Conrad, R.; Cobb, C.; Castelino-Prabhu, S.; Mirshahidi, S.; Shiraz, P.; Mirshahidi, H.R. Nuclear protein in testis midline carcinoma with unusual elevation of α-fetoprotein and synaptophysin positivity: A case report and review of the literature. Expert Rev. Anticancer Ther. 2015, 15, 1199–1213. [Google Scholar] [CrossRef]
- Riess, J.W.; Rahman, S.; Kian, W.; Edgerly, C.; Heilmann, A.M.; Madison, R.; Ramkissoon, S.H.; Klaitman, S.S.; Chung, J.H.; Trabucco, S.E.; et al. Genomic profiling of solid tumors harboring BRD4-NUT and response to immune checkpoint inhibitors. Transl. Oncol. 2021, 14, 101184. [Google Scholar] [CrossRef]
- Reddy, R.; Woods, T.R.; Allan, R.W.; Malhotra, P.; Mehta, H.J.; Sarkar, P.K.; Boyce, B.J.; Asirvatham, J.R. NUT (Nuclear Protein in Testis) Carcinoma: A Report of Two Cases With Different Histopathologic Features. Int. J. Surg. Pathol. 2019, 27, 225–229. [Google Scholar] [CrossRef]
- Tanaka, M.; Kato, K.; Gomi, K.; Yoshida, M.; Niwa, T.; Aida, N.; Kigasawa, H.; Ohama, Y.; Tanaka, Y. NUT midline carcinoma: Report of 2 cases suggestive of pulmonary origin. Am. J. Surg. Pathol. 2012, 36, 381–388. [Google Scholar] [CrossRef]
- Zhang, Y.; Han, K.; Dong, X.; Hou, Q.; Li, T.; Li, L.; Zhou, G.; Liu, X.; Zhao, G.; Li, W. Case Report and Literature Review: Primary Pulmonary NUT-Midline Carcinoma. Front. Oncol. 2021, 11, 700781. [Google Scholar] [CrossRef]
- Zhou, L.; Yong, X.; Zhou, J.; Wang, C. Clinicopathological Analysis of Five Cases of NUT Midline Carcinoma, including One with the Gingiva. Biomed. Res. Int. 2020, 2020, 9791208. [Google Scholar] [CrossRef]
- Chen, J.J.; Gao, X.L. Primary Pulmonary NUT Midline Carcinoma:a case report and review of the literature. Int. J. Respir. 2018, 38, 820–825. [Google Scholar]
- Huang, Y.; Wu, W.; Hou, L.K. Nuclear protein in testis carcinoma of lung:a clinicopathologic study of three cases. Chin. J. Diagn. Pathol. 2017, 24, 350–353. [Google Scholar]
- Liu, X.; Li, Y.; Yu, M.; Zhou, L. A Case Report of Primary Pulmonary NUT Carcinoma and Literature Review. Chin. J. Lung Cancer 2021, 24, 63–68. [Google Scholar]
- Wang, L.; Wang, W.; Xin, Y. A case report of NUT midline carcinoma with multiple metastases in the elderly. Chin. J. Clin. Oncol. 2016, 43, 1067. [Google Scholar]
- Zhang, S.; Zhao, D. NUT Midline Carcinoma of the Lung in A Child: A case report. Acta Acad. Med. Nanjing 2020, 40, 1078–1080. [Google Scholar]
- Hung, Y.P.; Chen, A.L.; Taylor, M.S.; Huynh, T.G.; Kem, M.; Selig, M.K.; Nielsen, G.P.; Lennerz, J.K.; Azzoli, C.G.; Dagogo-Jack, I.; et al. Thoracic nuclear protein in testis (NUT) carcinoma: Expanded pathological spectrum with expression of thyroid transcription factor-1 and neuroendocrine markers. Histopathology 2021, 78, 896–904. [Google Scholar] [CrossRef]
- Xie, X.H.; Wang, L.Q.; Qin, Y.Y.; Lin, X.Q.; Xie, Z.H.; Liu, M.; Zhang, J.X.; Ouyang, M.; Liu, J.; Gu, Y.Y.; et al. Clinical features, treatment, and survival outcome of primary pulmonary NUT midline carcinoma. Orphanet J. Rare Dis. 2020, 15, 183. [Google Scholar] [CrossRef]
- Sholl, L.M.; Nishino, M.; Pokharel, S.; Mino-Kenudson, M.; French, C.A.; Janne, P.A.; Lathan, C. Primary Pulmonary NUT Midline Carcinoma: Clinical, Radiographic, and Pathologic Characterizations. J. Thorac. Oncol. 2015, 10, 951–959. [Google Scholar] [CrossRef]
- French, C.A.; Miyoshi, I.; Kubonishi, I.; Grier, H.E.; Perez-Atayde, A.R.; Fletcher, J.A. BRD4-NUT fusion oncogene: A novel mechanism in aggressive carcinoma. Cancer Res. 2003, 63, 304–307. [Google Scholar] [PubMed]
- Haack, H.; Johnson, L.A.; Fry, C.J.; Crosby, K.; Polakiewicz, R.D.; Stelow, E.B.; Hong, S.M.; Schwartz, B.E.; Cameron, M.J.; Rubin, M.A.; et al. Diagnosis of NUT midline carcinoma using a NUT-specific monoclonal antibody. Am. J. Surg. Pathol. 2009, 33, 984–991. [Google Scholar] [CrossRef]
- Mertz, J.A.; Conery, A.R.; Bryant, B.M.; Sandy, P.; Balasubramanian, S.; Mele, D.A.; Bergeron, L.; Sims, R.J., 3rd. Targeting MYC dependence in cancer by inhibiting BET bromodomains. Proc. Natl. Acad. Sci. USA 2011, 108, 16669–16674. [Google Scholar] [CrossRef]
- Sahai, V.; Redig, A.J.; Collier, K.A.; Eckerdt, F.D.; Munshi, H.G. Targeting BET bromodomain proteins in solid tumors. Oncotarget 2016, 7, 53997–54009. [Google Scholar] [CrossRef]
- Schwartz, B.E.; Hofer, M.D.; Lemieux, M.E.; Bauer, D.E.; Cameron, M.J.; West, N.H.; Agoston, E.S.; Reynoird, N.; Khochbin, S.; Ince, T.A.; et al. Differentiation of NUT midline carcinoma by epigenomic reprogramming. Cancer Res. 2011, 71, 2686–2696. [Google Scholar] [CrossRef]
| Sex | Male (n) | Female (n) | Male:Female | ||
| 36/55 | 19/55 | 1.89:1 | |||
| Age | 0–30 (n) | 30–60 (n) | >60 (n) | Mean (years old) | Median (years old) |
| 17/55 | 26/55 | 12/55 | 41.4 | 36 | |
| Smoking history | Yes (n) | No (n) | Yes:No | ||
| 14/42 | 28/42 | 1:2 | |||
| Symptom | Cough (n) | Dyspnea (n) | Chest pain (n) | Hemoptysis (n) | Wheezing (n) |
| 28/44 | 13/44 | 8/44 | 8/44 | 7/44 |
| Lesion Location | n < 3 | n ≥ 3, < 6 | n ≥ 6, < 9 | n ≥ 9 | NA | Total (n) | |
|---|---|---|---|---|---|---|---|
| Left Lung | LUL * | 1 | 0 | 1 | 1 | 2 | 5 |
| LLL | 0 | 5 | 3 | 0 | 2 | 10 | |
| LH | 0 | 1 | 0 | 1 | 0 | 2 | |
| NA | 0 | 0 | 1 | 1 | 3 | 5 | |
| Right Lung | RUL | 1 | 2 | 2 | 0 | 2 | 7 |
| RML | 0 | 1 | 1 | 0 | 0 | 2 | |
| RLL | 1 | 2 | 3 | 2 | 2 | 10 | |
| RH | 1 | 1 | 2 | 1 | 0 | 5 | |
| NA | 0 | 0 | 3 | 0 | 0 | 3 | |
| Trachea | 2 | 0 | 0 | 0 | 0 | 2 | |
| Total (n) | 6 | 12 | 16 | 6 | 11 | 51 | |
| Treatment | Recorded/Total (n) | Mean OS (Months) |
|---|---|---|
| Untreated | 5/5 | 1.3 |
| Surgery only | 11/11 | 3.54 |
| Surgery combined with adjuvant therapy | 1/4 | (8) * |
| Chemotherapy only | 13/16 | 5.58 |
| Chemoradiotherapy | 6/8 | 13.2 |
| Chemotherapy combined with immunotherapy | 2/3 | 5.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, J.; Xu, Z.; Guo, Y. Diagnosis, Treatment and Prognosis of Primary Pulmonary NUT Carcinoma: A Literature Review. Curr. Oncol. 2022, 29, 6807-6815. https://doi.org/10.3390/curroncol29100536
Yuan J, Xu Z, Guo Y. Diagnosis, Treatment and Prognosis of Primary Pulmonary NUT Carcinoma: A Literature Review. Current Oncology. 2022; 29(10):6807-6815. https://doi.org/10.3390/curroncol29100536
Chicago/Turabian StyleYuan, Jiaqian, Zhili Xu, and Yong Guo. 2022. "Diagnosis, Treatment and Prognosis of Primary Pulmonary NUT Carcinoma: A Literature Review" Current Oncology 29, no. 10: 6807-6815. https://doi.org/10.3390/curroncol29100536
APA StyleYuan, J., Xu, Z., & Guo, Y. (2022). Diagnosis, Treatment and Prognosis of Primary Pulmonary NUT Carcinoma: A Literature Review. Current Oncology, 29(10), 6807-6815. https://doi.org/10.3390/curroncol29100536





