Examining Treatment Patterns and Real-World Outcomes in Chronic Lymphocytic Leukemia Using Administrative Data in Ontario
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Patient Population
2.3. Data Sources
2.4. Statistical and Costing Analysis
3. Results
3.1. Baseline Characteristics
3.2. Treatment Patterns by Line of Therapy
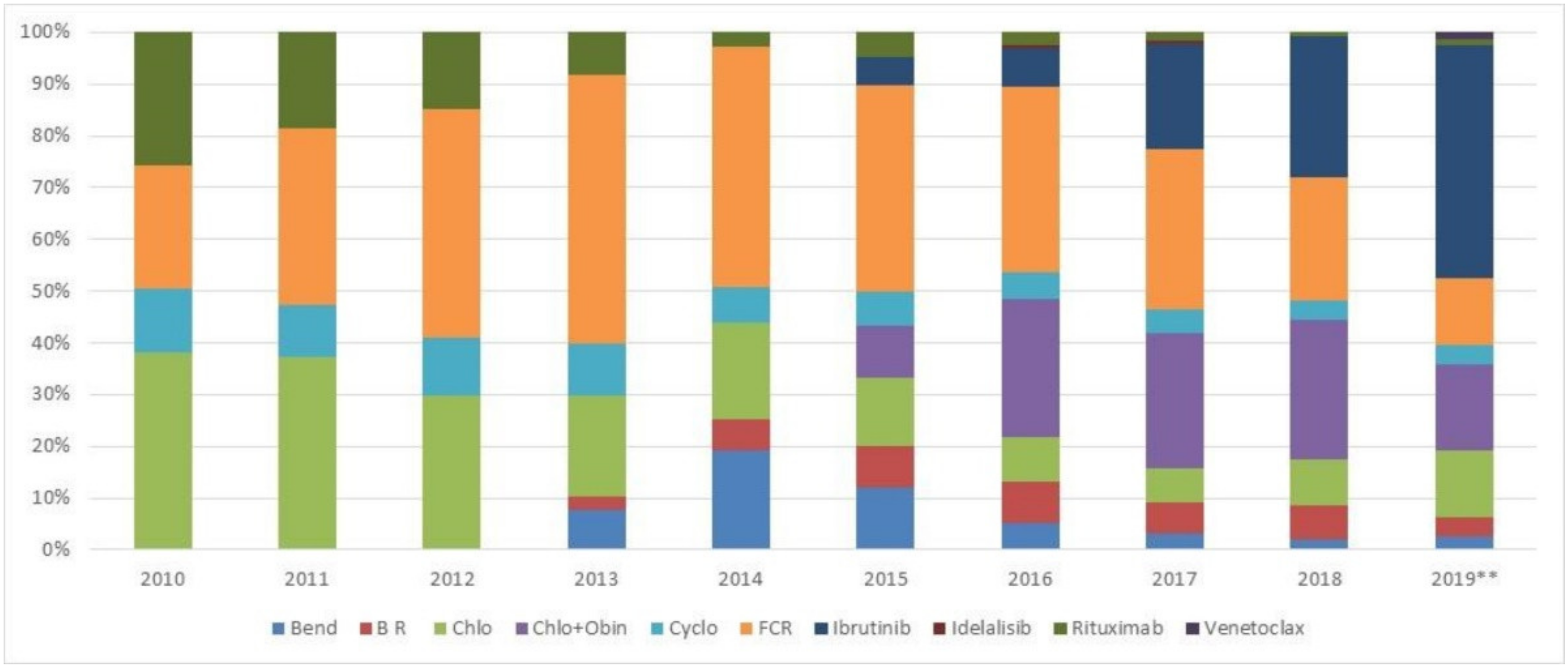
3.2.1. First Line Treatment
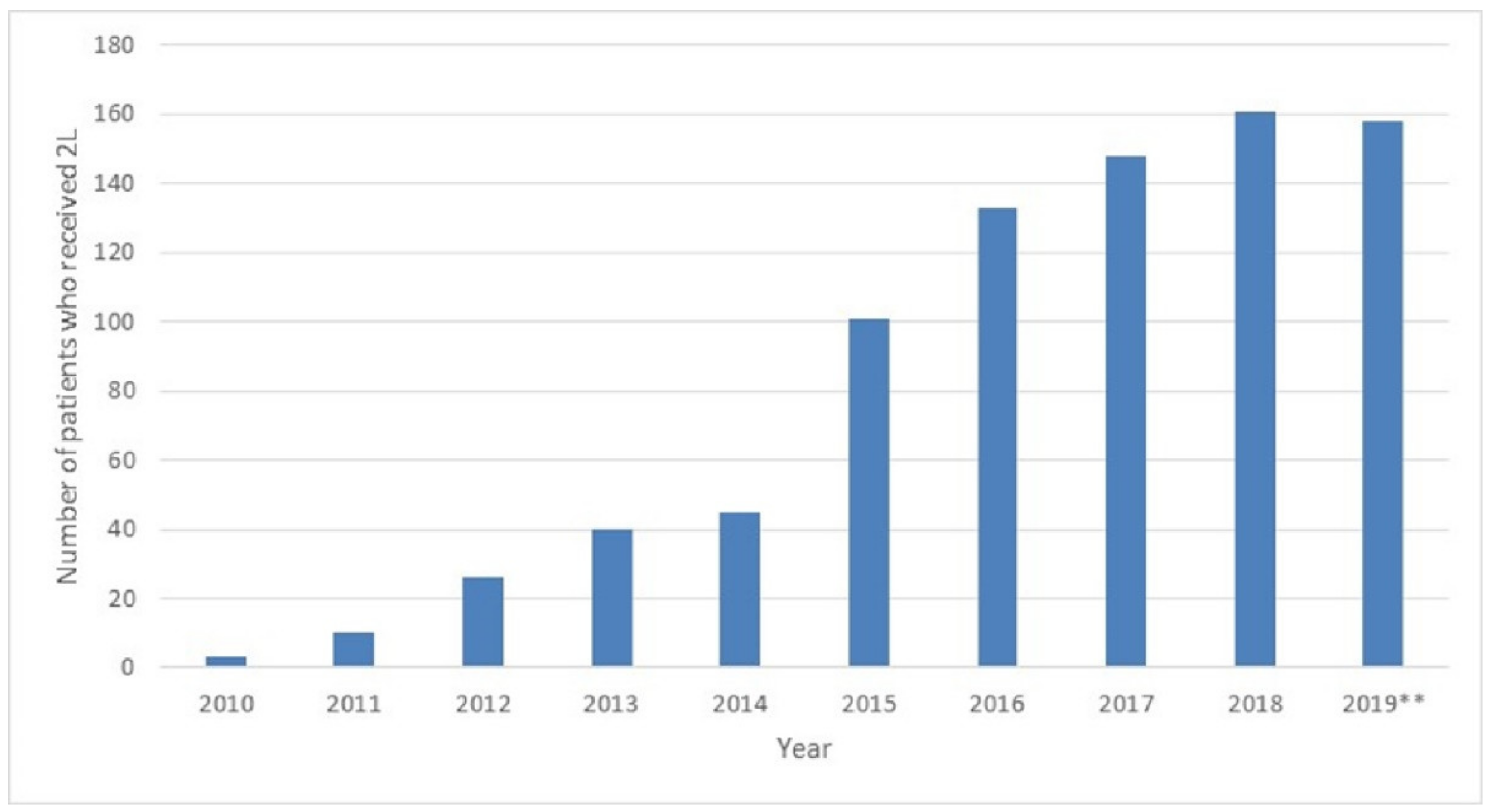
3.2.2. Second Line Treatment
3.2.3. Third Line Treatment
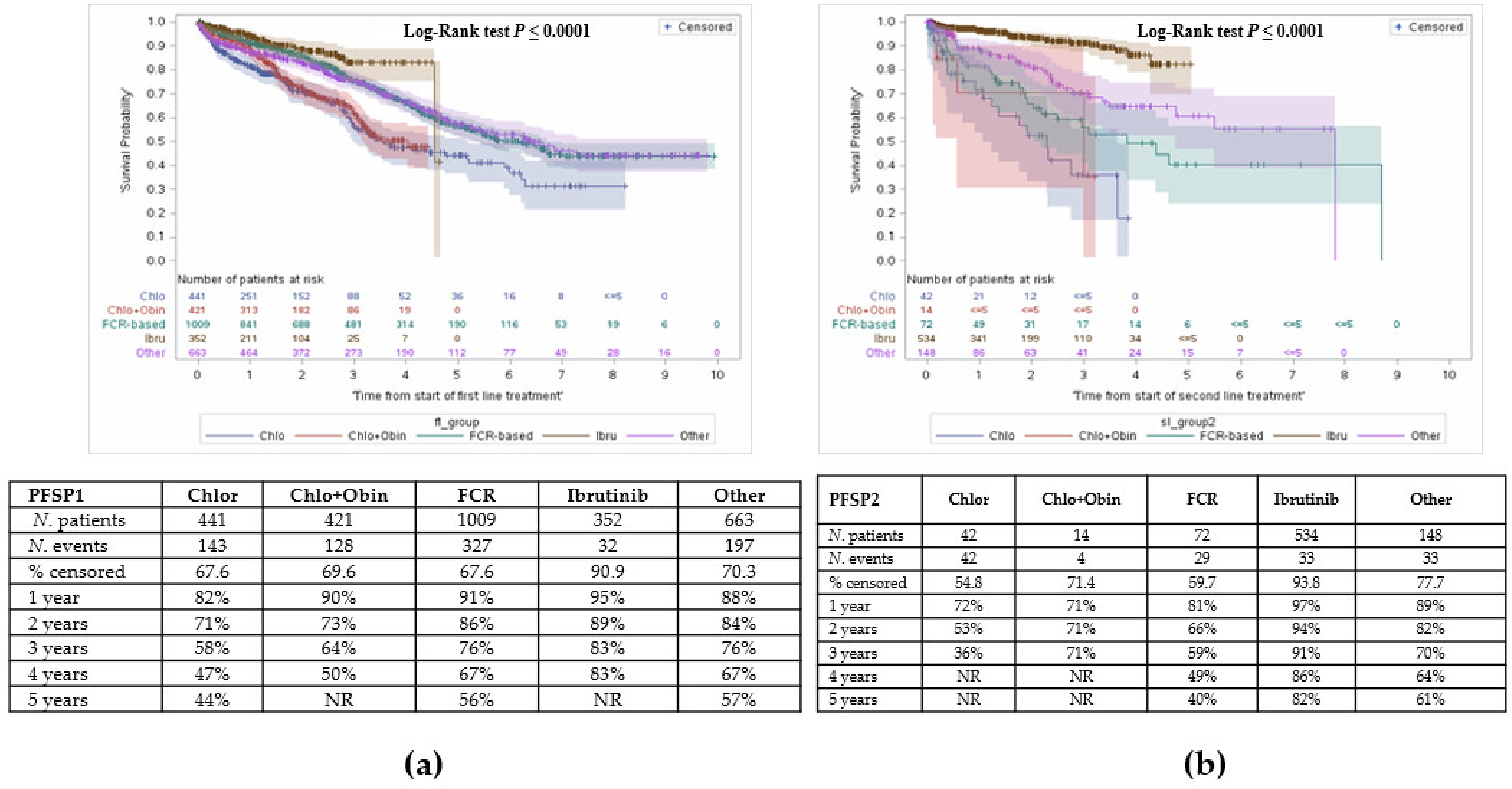
3.3. Survival
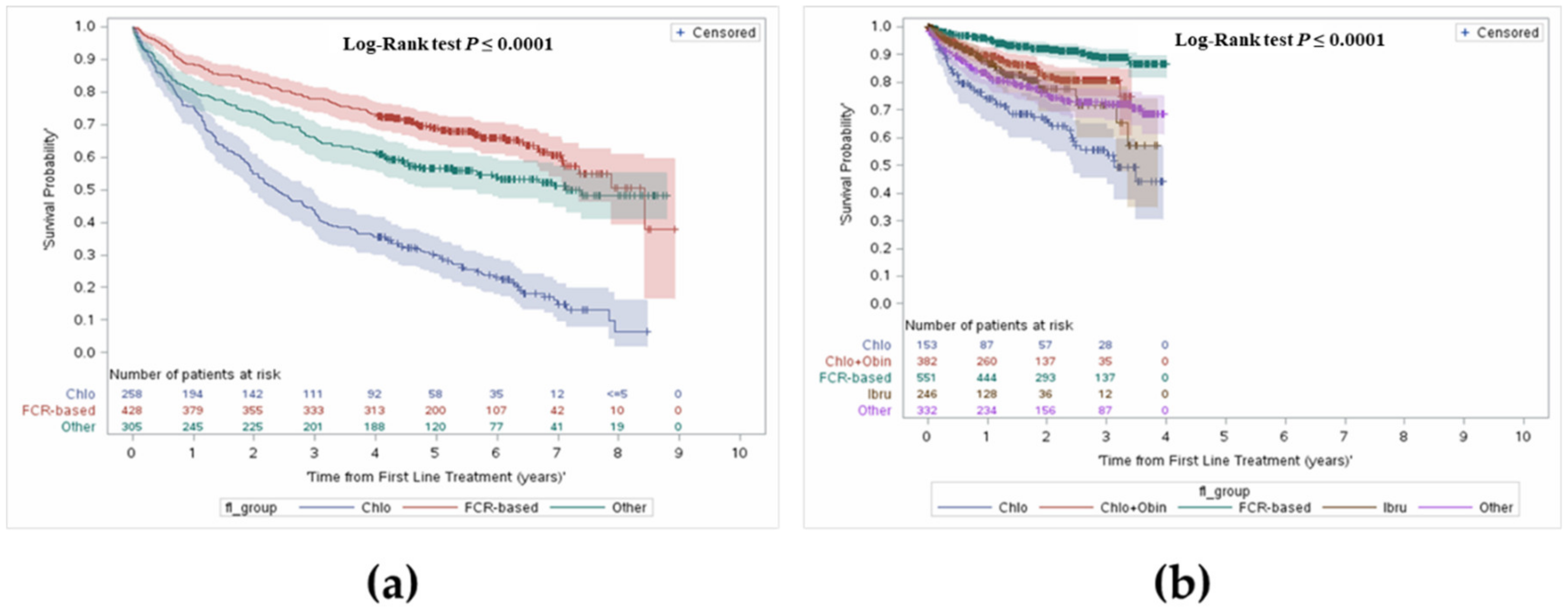
3.3.1. Overall Survival from First Line Treatment Initiation
3.3.2. Overall Survival from Initiation of Second or Third Line Treatment
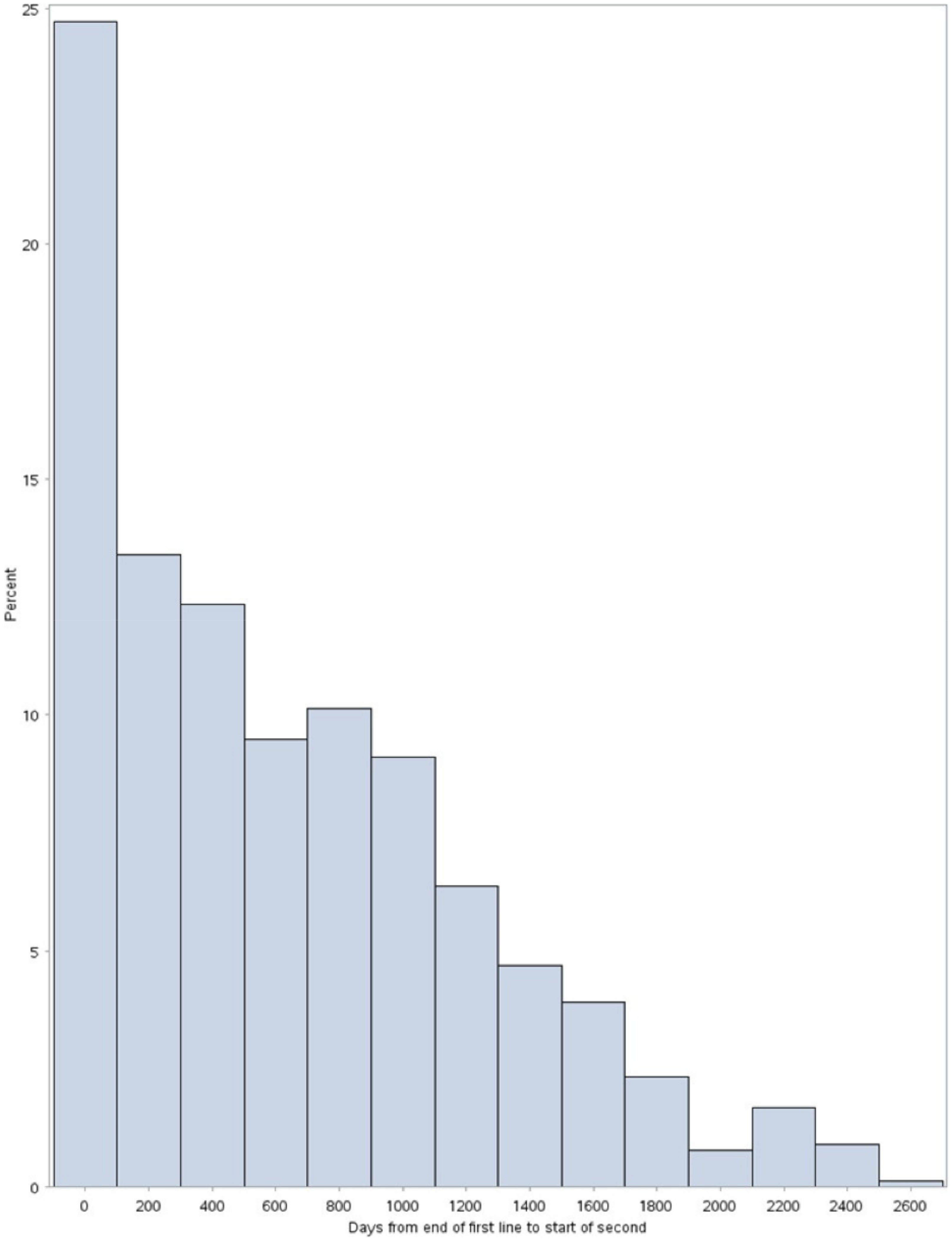
3.4. Other Outcomes: Time to Subsequent Treatments
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Leukemia & Lymphoma Society of Canada. Blood Cancer in Canada: Facts and Stats 2016. Available online: https://www.llscanada.org/sites/default/files/National/CANADA/Pdf/InfoBooklets/Blood_Cancer_in_Canada_Facts_%26_Stats_2016.pdf (accessed on 4 May 2020).
- Canadian Cancer Society. What Is Chronic Lymphocytic Leukemia? Available online: https://cancer.ca/en/cancer-information/cancer-types/chronic-lymphocytic-leukemia-cll/what-is-chronic-lymphocytic-leukemia (accessed on 4 May 2020).
- Canadian Cancer Statistics Advisory Committee. Canadian Cancer Statistics 2019; Canadian Cancer Society: Toronto, ON, Canada, 2019; Available online: https://cdn.cancer.ca/-/media/files/research/cancer-statistics/2019-statistics/canadian-cancer-statistics-2019-en.pdf (accessed on 4 May 2020).
- Sagatys, E.M.; Zhang, L. Clinical and Laboratory Prognostic Indicators in Chronic Lymphocytic Leukemia. Cancer Control 2012, 19, 18–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Bockstaele, F.; Verhasselt, B.; Philippé, J. Prognostic Markers in Chronic Lymphocytic Leukemia: A Comprehensive Review. Blood Rev. 2009, 23, 25–47. [Google Scholar] [CrossRef] [PubMed]
- Shanafelt, T. Treatment of Older Patients with Chronic Lymphocytic Leukemia: Key Questions and Current Answers. Hematol. Am. Soc. Hematol. Educ. Program 2013, 2013, 158–167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The International CLL-IPI Working Group. An International Prognostic Index for Patients with Chronic Lymphocytic Leukaemia (CLL-IPI): A Meta-Analysis of Individual Patient Data. Lancet Oncol. 2016, 17, 779–790. [Google Scholar] [CrossRef]
- Owen, C.; Gerrie, A.S.; Banerji, V.; Assouline, S.; Chen, C.; Robinson, K.S.; Lye, E.; Fraser, G. Canadian Evidence-Based Guideline for the First-Line Treatment of Chronic Lymphocytic Leukemia. Curr. Oncol. 2018, 25, e461–e474. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shanafelt, T.D.; Wang, X.V.; Kay, N.E.; Hanson, C.A.; O’Brien, S.; Barrientos, J.; Jelinek, D.F.; Braggio, E.; Leis, J.F.; Zhang, C.C.; et al. Ibrutinib–Rituximab or Chemoimmunotherapy for Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2019, 381, 432–443. [Google Scholar] [CrossRef] [PubMed]
- Pan-Canadian Oncology Drug Review. Provincial Funding Summary: Ibrutinib (Imbruvica) for Chronic Lymphocytic Leukemia/Small Lymphocytic Leukemia (Previously Untreated) (pCODR 10085). Available online: https://www.cadth.ca/sites/default/files/pcodr/pcodr_ibrutinib_imbruvica_cll-sll_in_egr.pdf (accessed on 4 May 2020).
- Burger, J.A.; Barr, P.M.; Robak, T.; Owen, C.; Ghia, P.; Tedeschi, A.; Bairey, O.; Hillmen, P.; Coutre, S.E.; Devereux, S.; et al. Long-Term Efficacy and Safety of First-Line Ibrutinib Treatment for Patients with CLL/SLL: 5 Years of Follow-up from the Phase 3 RESONATE-2 Study. Leukemia 2020, 34, 787–798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moreno, C.; Greil, R.; Demirkan, F.; Tedeschi, A.; Anz, B.; Larratt, L.; Simkovic, M.; Samoilova, O.; Novak, J.; Ben-Yehuda, D.; et al. Ibrutinib plus Obinutuzumab versus Chlorambucil plus Obinutuzumab in First-Line Treatment of Chronic Lymphocytic Leukaemia (ILLU-MINATE): A Multicentre, Randomised, Open-Label, Phase 3 Trial. Lancet Oncol. 2019, 20, 43–56. [Google Scholar] [CrossRef]
- Woyach, J.A.; Ruppert, A.S.; Heerema, N.A.; Zhao, W.; Booth, A.M.; Ding, W.; Bartlett, N.L.; Brander, D.M.; Barr, P.M.; Rogers, K.A.; et al. Ibrutinib Regimens versus Chemoimmunotherapy in Older Patients with Untreated CLL. N. Engl. J. Med. 2018, 379, 2517–2528. [Google Scholar] [CrossRef] [PubMed]
- Munir, T.; Brown, J.R.; O’Brien, S.; Barrientos, J.C.; Barr, P.M.; Reddy, N.M.; Coutre, S.; Tam, C.S.; Mulligan, S.P.; Jaeger, U.; et al. Final Analysis from RESONATE: Up to Six Years of Follow-up on Ibrutinib in Patients with Previously Treated Chronic Lymphocytic Leukemia or Small Lymphocytic Lymphoma. Am. J. Hematol. 2019, 94, 1353–1363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Janssen Inc. Imbruvica Product Monograph. 2019. Available online: https://pdf.hres.ca/dpd_pm/00050262.PDF (accessed on 4 May 2020).
- AstraZeneca Canada Inc. Calquence Product Monograph. 2019. Available online: https://www.astrazeneca.ca/content/dam/az-ca/downloads/productinformation/calquence-product-monograph-en.pdf (accessed on 4 May 2020).
- Pan-Canadian Oncology Drug Review. Provincial Funding Summary: Obinutuzumab (Gazyva) for Chronic Lymphocytic Leukemia (pCODR 10041). Available online: https://www.cadth.ca/sites/default/files/pcodr/pcodr-provfund_gazyva-cll.pdf (accessed on 4 May 2020).
- Pan-Canadian Oncology Drug Review. Provincial Funding Summary: Ibrutinib (Imbruvica) for Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma (pCODR 10043). Available online: https://www.cadth.ca/sites/default/files/pcodr/pcodr-ibrutinib-cll-sll-fn-cgr.pdf (accessed on 4 May 2020).
- Huang, S.J.; Gerrie, A.S.; Young, S.; Tucker, T.; Bruyere, H.; Hrynchak, M.; Galbraith, P.; Al Tourah, A.J.; Dueck, G.; Noble, M.C.; et al. Comparison of Real-World Treatment Patterns in Chronic Lymphocytic Leukemia Management before and after Availability of Ibrutinib in the Province of British Columbia, Canada. Leuk. Res. 2020, 91, 106335. [Google Scholar] [CrossRef] [PubMed]
- Landau, D.A.; Tausch, E.; Taylor-Weiner, A.N.; Stewart, C.; Reiter, J.G.; Bahlo, J.; Kluth, S.; Bozic, I.; Lawrence, M.; Böttcher, S.; et al. Mutations Driving CLL and Their Evolution in Progression and Relapse. Nature 2015, 526, 525–530. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Döhner, H.; Stilgenbauer, S.; Benner, A.; Leupolt, E.; Kröber, A.; Bullinger, L.; Döhner, K.; Bentz, M.; Lichter, P. Genomic Aberrations and Survival in Chronic Lymphocytic Leukemia. N. Engl. J. Med. 2000, 343, 1910–1916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Edelmann, J.; Gribben, J.G. Managing Patients with TP53-Deficient Chronic Lymphocytic Leukemia. J. Oncol. Pract. 2017, 13, 371–377. [Google Scholar] [CrossRef] [PubMed]
- Kipps, T.J.; Fraser, G.; Coutre, S.E.; Brown, J.R.; Barrientos, J.C.; Barr, P.M.; Byrd, J.C.; O’Brien, S.M.; Dilhuydy, M.-S.; Hillmen, P.; et al. Long-Term Studies Assessing Outcomes of Ibrutinib Therapy in Patients with Del(11q) Chronic Lymphocytic Leukemia. Clin. Lymphoma Myeloma Leuk. 2019, 19, 715–722.e6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mato, A.R.; Nabhan, C.; Thompson, M.C.; Lamanna, N.; Brander, D.M.; Hill, B.; Howlett, C.; Skarbnik, A.; Cheson, B.D.; Zent, C.; et al. Toxicities and outcomes of 616 ibrutinib-treated patients in the United States: A real-world analysis. Haematologica 2018, 103, 874–879. [Google Scholar] [CrossRef] [PubMed]
- Aarup, K.; Rotbain, E.C.; Enggaard, L.; Pedersen, R.S.; Bergmann, O.J.; Thomsen, R.H.; Frederiksen, M.; Frederiksen, H.; Nielsen, T.; Christiansen, I.; et al. Real-world outcomes for 205 patients with chronic lymphocytic leukemia treated with ibrutinib. Eur. J. Haematol. 2020, 105, 646–654. [Google Scholar] [CrossRef] [PubMed]

| C + O N = 421 | FCR-Based N = 1009 | Ibrutinib N = 352 | Other * N = 1105 | Total N = 2887 | |
|---|---|---|---|---|---|
| CLL Diagnosis Year | |||||
| 2010 | 28 (6.7%) | 119 (11.8%) | 10 (2.8%) | 185 (16.7%) | 342 (11.8%) |
| 2011 | 27 (6.4%) | 137 (13.6%) | 22 (6.3%) | 172 (15.6%) | 358 (12.4%) |
| 2012 | 32 (7.6%) | 145 (14.4%) | 17 (4.8%) | 145 (13.1%) | 339 (11.7%) |
| 2013 | 45 (10.7%) | 151 (15.0%) | 22 (6.3%) | 170 (15.4%) | 388 (13.4%) |
| 2014 | 54 (12.8%) | 148 (14.7%) | 39 (11.1%) | 125 (11.3%) | 366 (12.7%) |
| 2015 | 91 (21.6%) | 125 (12.4%) | 68 (19.3%) | 120 (10.9%) | 404 (14.0%) |
| 2016 | 72 (17.1%) | 89 (8.8%) | 76 (21.6%) | 101 (9.1%) | 338 (11.7%) |
| 2017 | 72 (17.1%) | 95 (9.4%) | 98 (27.8%) | 87 (7.9%) | 352 (12.2%) |
| Age at Diagnosis | |||||
| Mean ± SD | 73.55 ± 6.84 | 61.09 ± 9.39 | 67.92 ± 10.67 | 73.09 ± 10.82 | 68.33 ± 11.28 |
| Median (IQR) | 74 (69–78) | 61 (55–67) | 69 (62–76) | 74 (66–82) | 69 (61–77) |
| Age at Treatment | |||||
| Mean ± SD | 76.09 ± 6.38 | 62.63 ± 9.17 | 70.60 ± 10.33 | 74.47 ± 10.77 | 70.10 ± 11.17 |
| Median (IQR) | 76 (72–81) | 63 (56–69) | 71 (65–77) | 76 (67–83) | 71 (63–78) |
| Sex | |||||
| Female | 131 (31.1%) | 299 (29.6%) | 105 (29.8%) | 430 (38.9%) | 965 (33.4%) |
| Male | 290 (68.9%) | 710 (70.4%) | 247 (70.2%) | 675 (61.1%) | 1922 (66.6%) |
| Income Quintile | |||||
| Missing | 1–5 ** | 1–5 ** | 1–5 ** | 1–5 ** | 9 (0.3%) |
| 1 (Lowest) | 80 (19.0%) | 155 (15.4%) | 47 (13.4%) | 204 (18.5%) | 486 (16.8%) |
| 2 | 76–80 ** | 202–206 ** | 70–74 ** | 245–249 ** | 604 (20.9%) |
| 3 | 89 (21.1%) | 187 (18.5%) | 73 (20.7%) | 198 (17.9%) | 547 (18.9%) |
| 4 | 87 (20.7%) | 221 (21.9%) | 75 (21.3%) | 231 (20.9%) | 614 (21.3%) |
| 5 (Highest) | 84 (20.0%) | 239 (23.7%) | 82 (23.3%) | 222 (20.1%) | 627 (21.7%) |
| Charlson Comorbidity Index | |||||
| Mean ± SD | 1.06 ± 1.65 | 1.30 ± 1.59 | 0.95 ± 1.47 | 1.92 ± 2.03 | 1.46 ± 1.81 |
| Median (IQR) | 0 (0–2) | 0 (0–2) | 0 (0–2) | 2 (0–3) | 1 (0–2) |
| Comorbidities | |||||
| Chronic obstructive pulmonary disease (COPD) | 85 (20.2%) | 131 (13.0%) | 65 (18.5%) | 250 (22.6%) | 531 (18.4%) |
| Diabetes | 128 (30.4%) | 188 (18.6%) | 90 (25.6%) | 294 (26.6%) | 700 (24.2%) |
| Myocardial infarction (MI) | 18 (4.3%) | 34 (3.4%) | 14 (4.0%) | 48 (4.3%) | 114 (3.9%) |
| Congestive heart failure (CHF) | 38 (9.0%) | 31 (3.1%) | 20 (5.7%) | 100 (9.0%) | 189 (6.5%) |
| Rheumatoid arthritis (RA) | 9–13 ** | 12 (1.2%) | 1–5 ** | 20 (1.8%) | 46 (1.6%) |
| Prior cancer | 82 (19.5%) | 96 (9.5%) | 66 (18.8%) | 232 (21.0%) | 476 (16.5%) |
| Before 2015 | ||||||
|---|---|---|---|---|---|---|
| Overall | FCR | Chlo + Obin | Ibrutinib | Chlorambucil | Other | |
| Median (Years) | 6.2 | 8.6 | 2.4 | 7.3 | ||
| Landmark Survival by Year | ||||||
| 1 | 89% | NA | NA | 76% | 81% | |
| 2 | 83% | 56% | 74% | |||
| 3 | 78% | 45% | 66% | |||
| 4 | 73% | 36% | 62% | |||
| 5 | 69% | 30% | 56% | |||
| After 2015 | ||||||
| Overall | FCR | Chlo + Obin | Ibrutinib | Chlorambucil | Other | |
| Median (Years) | NR | NR | NR | NR | 3.3 | NR |
| Landmark Survival by Year | ||||||
| 1 | 96% | 90% | 87% | 75% | 84% | |
| 2 | 92% | 82% | 78% | 66% | 76% | |
| 3 | 89% | 81% | 72% | 56% | 73% | |
| 4 | 87% | NR | NR | NR | 69% | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seung, S.J.; Hurry, M.; Hassan, S.; Elnoursi, A.; Scheider, K.A.B.; Wagner, D.; Edwin, J.J.; Aw, A.T.W. Examining Treatment Patterns and Real-World Outcomes in Chronic Lymphocytic Leukemia Using Administrative Data in Ontario. Curr. Oncol. 2021, 28, 4832-4844. https://doi.org/10.3390/curroncol28060408
Seung SJ, Hurry M, Hassan S, Elnoursi A, Scheider KAB, Wagner D, Edwin JJ, Aw ATW. Examining Treatment Patterns and Real-World Outcomes in Chronic Lymphocytic Leukemia Using Administrative Data in Ontario. Current Oncology. 2021; 28(6):4832-4844. https://doi.org/10.3390/curroncol28060408
Chicago/Turabian StyleSeung, Soo Jin, Manjusha Hurry, Shazia Hassan, Ashlie Elnoursi, Krystin A. B. Scheider, Dennis Wagner, Jonathan J. Edwin, and Andrew T. W. Aw. 2021. "Examining Treatment Patterns and Real-World Outcomes in Chronic Lymphocytic Leukemia Using Administrative Data in Ontario" Current Oncology 28, no. 6: 4832-4844. https://doi.org/10.3390/curroncol28060408
APA StyleSeung, S. J., Hurry, M., Hassan, S., Elnoursi, A., Scheider, K. A. B., Wagner, D., Edwin, J. J., & Aw, A. T. W. (2021). Examining Treatment Patterns and Real-World Outcomes in Chronic Lymphocytic Leukemia Using Administrative Data in Ontario. Current Oncology, 28(6), 4832-4844. https://doi.org/10.3390/curroncol28060408





