Cognitive Assessment Tools Recommended in Geriatric Oncology Guidelines: A Rapid Review
Abstract
1. Introduction
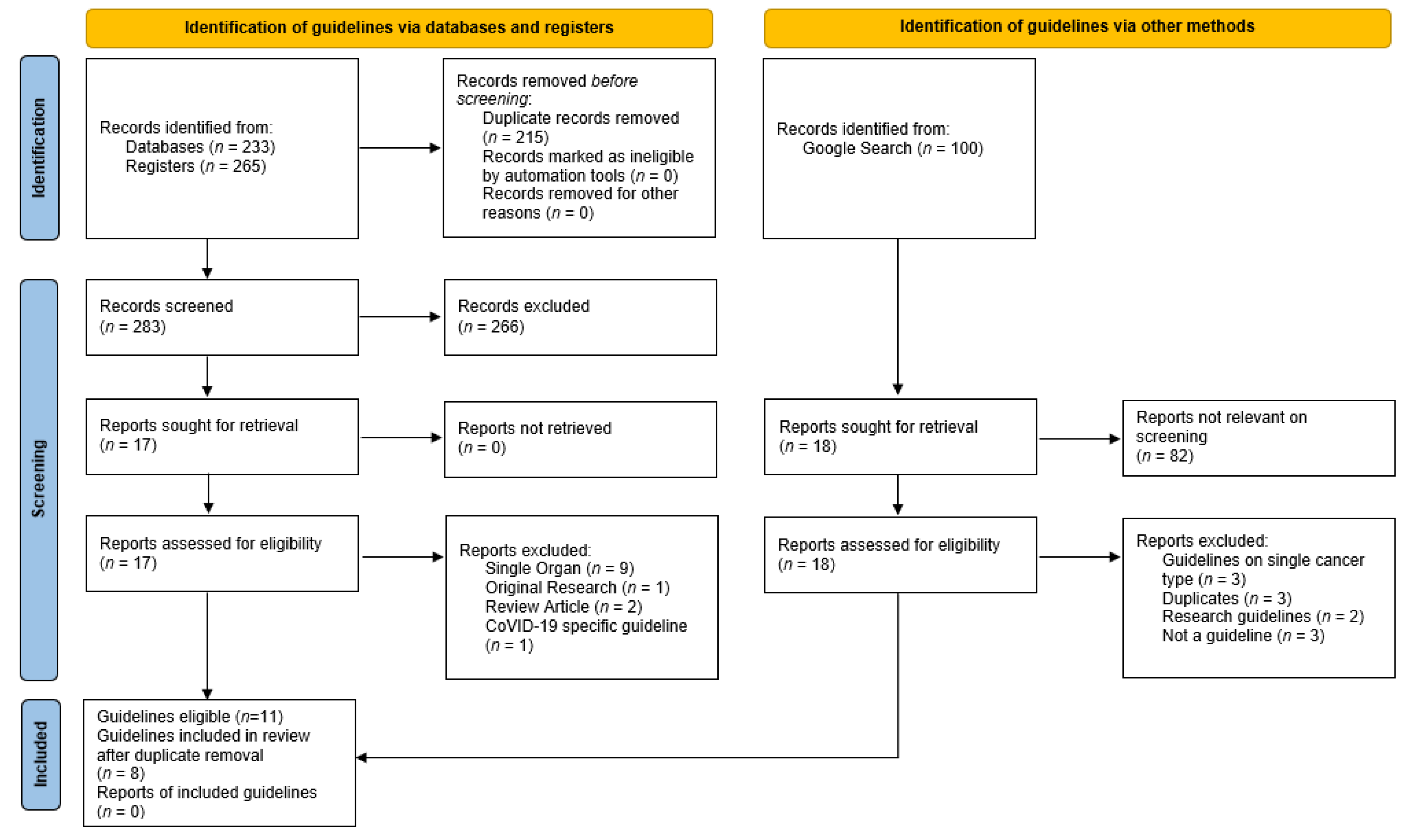
2. Materials and Methods
2.1. Part 1: Identification of Cognitive Assessment Tools
2.1.1. Published Resources
2.1.2. Web-Based Resources
2.1.3. Inclusion Criteria
- Published between 1 July 2016 and 1 July 2021;
- Available in English;
- Guidelines, position statements, consensus statements or recommendations;
- Primary focus is the assessment of older adults with cancer;
- Authored or published by a national or international Medical or Oncological Society or Organization; and
- Refer to specific cognitive assessment tools.
2.1.4. Exclusion Criteria
- Opinion pieces, research articles, and review articles;
- Authored or published by a single institution;
- Related to a single cancer type or groups of cancers of a single body system or organ; and
- Tools designed specifically for screening or assessing acute confusional states or delirium.
- Clinical practice guidelines: Evidence-based statements that include recommendations intended to optimize patient care and assist health care practitioners to make decisions on the appropriate health care for specific clinical circumstances [23].
- Cognitive assessment tool: Any instrument, tool or survey, developed or utilized to assess or screen cognitive function in adults.
2.2. Part 2: Focused Review of the Evidence Underpinning the Identified Tools
3. Results
3.1. Blessed Information Memory Concentration
3.2. Clock Draw Test
3.3. Mini-Cog
3.4. Mini-Mental Status Examination
3.5. Montreal Cognitive Assessment
3.6. Short Portable Mental Status Questionnaire
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mohile, S.G.; Dale, W.; Somerfield, M.R.; Schonberg, M.A.; Boyd, C.M.; Burhenn, P.S.; Canin, B.; Cohen, H.J.; Holmes, H.M.; Hopkins, J.O. Practical assessment and management of vulnerabilities in older patients receiving chemotherapy: ASCO guideline for geriatric oncology. J. Clin. Oncol. 2018, 36, 2326. [Google Scholar] [CrossRef]
- Magnuson, A.; Mohile, S.; Janelsins, M. Cognition and Cognitive Impairment in Older Adults with Cancer. Curr. Geriatr. Rep. 2016, 5, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Soo, W.K. Older adults with cancer and clinical decision-making: The importance of assessing cognition. Cancer Forum 2013, 37, 201–205. [Google Scholar]
- Pergolotti, M.; Battisti, N.M.L.; Padgett, L.; Sleight, A.G.; Abdallah, M.; Newman, R.; Van Dyk, K.; Covington, K.R.; Williams, G.R.; van den Bos, F.; et al. Embracing the complexity: Older adults with cancer-related cognitive decline-A Young International Society of Geriatric Oncology position paper. J. Geriatr. Oncol. 2020, 11, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Rostoft, S.; van den Bos, F.; Pedersen, R.; Hamaker, M.E. Shared decision-making in older patients with cancer—What does the patient want? J. Geriatr. Oncol. 2021, 12, 339–342. [Google Scholar] [CrossRef] [PubMed]
- Magnuson, A.; Ahles, T.; Chen, B.T.; Mandelblatt, J.; Janelsins, M.C. Cognitive Function in Older Adults with Cancer: Assessment, Management, and Research Opportunities. J. Clin. Oncol. 2021, 39, 2138–2149. [Google Scholar] [CrossRef]
- Chodosh, J.; Petitti, D.B.; Elliott, M.; Hays, R.D.; Crooks, V.C.; Reuben, D.B.; Galen Buckwalter, J.; Wenger, N. Physician recognition of cognitive impairment: Evaluating the need for improvement. J. Am. Geriatr. Soc. 2004, 52, 1051–1059. [Google Scholar] [CrossRef]
- Magnuson, A.; Sattar, S.; Nightingale, G.; Saracino, R.; Skonecki, E.; Trevino, K.M. A practical guide to geriatric syndromes in older adults with cancer: A focus on falls, cognition, polypharmacy, and depression. Am. Soc. Clin. Oncol. Educ. Book 2019, 39, e96–e109. [Google Scholar] [CrossRef] [PubMed]
- American Psychriatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Jung, P.; Puts, M.; Frankel, N.; Syed, A.T.; Alam, Z.; Yeung, L.; Malik, U.; Rosario, C.; Ayala, A.P.; Hudson, J.; et al. Delirium incidence, risk factors, and treatments in older adults receiving chemotherapy: A systematic review and meta-analysis. J. Geriatr. Oncol. 2021, 12, 352–360. [Google Scholar] [CrossRef]
- Ahles, T.A.; Root, J.C. Cognitive Effects of Cancer and Cancer Treatments. Ann. Rev. Clin. Psychol. 2018, 14, 425–451. [Google Scholar] [CrossRef]
- Wefel, J.S.; Vardy, J.; Ahles, T.; Schagen, S.B. International Cognition and Cancer Task Force recommendations to harmonise studies of cognitive function in patients with cancer. Lancet Oncol. 2011, 12, 703–708. [Google Scholar] [CrossRef]
- Extermann, M.; Hurria, A. Comprehensive Geriatric Assessment for Older Patients with Cancer. J. Clin. Oncol. 2007, 25, 1824–1831. [Google Scholar] [CrossRef]
- Hamaker, M.E.; van Huis-Tanja, L.H.; Rostoft, S. Optimizing the geriatrician’s contribution to cancer care for older patients. J. Geriatr. Oncol. 2020, 11, 389–394. [Google Scholar] [CrossRef]
- Hamaker, M.E.; Te Molder, M.; Thielen, N.; van Munster, B.C.; Schiphorst, A.H.; van Huis, L.H. The effect of a geriatric evaluation on treatment decisions and outcome for older cancer patients—A systematic review. J. Geriatr. Oncol. 2018, 9, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Mohile, S.G.; Mohamed, M.R.; Culakova, E.; Xu, H.; Loh, K.P.; Magnuson, A.; Flannery, M.A.; Ramsdale, E.E.; Dunne, R.F.; Gilmore, N.; et al. A geriatric assessment (GA) intervention to reduce treatment toxicity in older patients with advanced cancer: A University of Rochester Cancer Center NCI community oncology research program cluster randomized clinical trial (CRCT). J. Clin. Oncol. 2020, 38, 12009. [Google Scholar] [CrossRef]
- Li, D.; Sun, C.-L.; Kim, H.; Chung, V.; Koczywas, M.; Fakih, M.; Chao, J.; Chien, L.; Charles, K.; Hughes, S.F.D.S.; et al. Geriatric assessment-driven intervention (GAIN) on chemotherapy toxicity in older adults with cancer: A randomized controlled trial. J. Clin. Oncol. 2020, 38, 12010. [Google Scholar] [CrossRef]
- Soo, W.K.; King, M.; Pope, A.; Parente, P.; Darzins, P.; Davis, I.D. Integrated geriatric assessment and treatment (INTEGERATE) in older people with cancer planned for systemic anticancer therapy. J. Clin. Oncol. 2020, 38, 12011. [Google Scholar] [CrossRef]
- Lund, C.M.; Vistisen, K.K.; Olsen, A.P.; Bardal, P.; Schultz, M.; Dolin, T.G.; Rønholt, F.; Johansen, J.S.; Nielsen, D.L. The effect of geriatric intervention in frail older patients receiving chemotherapy for colorectal cancer: A randomised trial (GERICO). Br. J. Cancer 2021, 124, 1949–1958. [Google Scholar] [CrossRef]
- Burhenn, P.S.; McCarthy, A.L.; Begue, A.; Nightingale, G.; Cheng, K.; Kenis, C. Geriatric assessment in daily oncology practice for nurses and allied health care professionals: Opinion paper of the Nursing and Allied Health Interest Group of the International Society of Geriatric Oncology (SIOG). J. Geriatr. Oncol. 2016, 7, 315–324. [Google Scholar] [CrossRef]
- Loh, K.P.; Soto-Perez-de-Celis, E.; Hsu, T.; de Glas, N.A.; Battisti, N.M.L.; Baldini, C.; Rodrigues, M.; Lichtman, S.M.; Wildiers, H. What Every Oncologist Should Know About Geriatric Assessment for Older Patients With Cancer: Young International Society of Geriatric Oncology Position Paper. J. Oncol. Pr. 2018, 14, 85–94. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Older Adult Oncology Version 1.2019. Available online: http://www.gaca.org.cn/uploadfolder/files/201903/ny27_219665.pdf (accessed on 4 August 2021).
- National Health and Medical Research Council. Clinical Practice Guidelines Portal. Available online: https://www.clinicalguidelines.gov.au/portal (accessed on 1 August 2021).
- Gironés Sarrió, R.; Antonio Rebollo, M.; Molina Garrido, M.J.; Guillén-Ponce, C.; Blanco, R.; Gonzalez Flores, E.; Saldaña, J. General recommendations paper on the management of older patients with cancer: The SEOM geriatric oncology task force’s position statement. Clin. Transl. Oncol. 2018, 20, 1246–1251. [Google Scholar] [CrossRef] [PubMed]
- National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines: Older Adult Oncology Version 2.2017. Available online: https://oncolife.com.ua/doc/nccn/Older_Adult_Oncology.pdf (accessed on 4 August 2021).
- Battisti, N.M.L.; Mislang, A.R.; Cooper, L.; O’Donovan, A.; Audisio, R.A.; Cheung, K.-L.; Sarrió, R.G.; Stauder, R.; Soto-Perez-de-Celis, E.; Jaklitsch, M.; et al. Adapting care for older cancer patients during the COVID-19 pandemic: Recommendations from the International Society of Geriatric Oncology (SIOG) COVID-19 Working Group. J. Geriatr. Oncol. 2020, 11, 1190–1198. [Google Scholar] [CrossRef] [PubMed]
- Fusco, D.; Ferrini, A.; Pasqualetti, G.; Giannotti, C.; Cesari, M.; Laudisio, A.; Ballestrero, A.; Scabini, S.; Odetti, P.R.; Colloca, G.F.; et al. Comprehensive geriatric assessment in older adults with cancer: Recommendations by the Italian Society of Geriatrics and Gerontology (SIGG). Eur. J. Clin. Investig. 2021, 51, e13347. [Google Scholar] [CrossRef] [PubMed]
- Katzman, R.; Brown, T.; Fuld, P.; Peck, A.; Schechter, R.; Schimmel, H. Validation of a short Orientation-Memory-Concentration Test of cognitive impairment. Am. J. Psychiatry 1983, 140, 734–739. [Google Scholar] [CrossRef] [PubMed]
- Upadhyaya, A.K.; Rajagopal, M.; Gale, T.M. The Six Item Cognitive Impairment Test (6-CIT) as a screening test for dementia: Comparison with Mini-Mental State Examination (MMSE). Curr. Aging Sci. 2010, 3, 138–142. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, D.; Brady, N.; Manning, E.; O’Shea, E.; O’Grady, S.; O’Regan, N.; Timmons, S. Validation of the 6-Item Cognitive Impairment Test and the 4AT test for combined delirium and dementia screening in older Emergency Department attendees. Age Ageing 2018, 47, 61–68. [Google Scholar] [CrossRef]
- Fillenbaum, G.G.; Heyman, A.; Wilkinson, W.E.; Haynes, C.S. Comparison of two screening tests in Alzheimer’s disease. The correlation and reliability of the Mini-Mental State Examination and the modified Blessed test. Arch. Neurol. 1987, 44, 924–927. [Google Scholar] [CrossRef] [PubMed]
- Yokomizo, J.E.; Simon, S.S.; Bottino, C.M. Cognitive screening for dementia in primary care: A systematic review. Int. Psychogeriatr 2014, 26, 1783–1804. [Google Scholar] [CrossRef]
- Tuijl, J.P.; Scholte, E.M.; de Craen, A.J.; van der Mast, R.C. Screening for cognitive impairment in older general hospital patients: Comparison of the Six-Item Cognitive Impairment Test with the Mini-Mental State Examination. Int. J. Geriatr. Psychiatry 2012, 27, 755–762. [Google Scholar] [CrossRef]
- Zama, I.N.; Maynard, W.K.; Davis, M.P. Clocking delirium: The value of the Clock Drawing Test with case illustrations. Am. J. Hosp. Palliat. Care 2008, 25, 385–388. [Google Scholar] [CrossRef]
- Shao, K.; Dong, F.M.; Guo, S.Z.; Wang, W.; Zhao, Z.M.; Yang, Y.M.; Wang, P.P.; Wang, J.H. Clock-drawing test: Normative data of three quantitative scoring methods for Chinese-speaking adults in Shijiazhuang City and clinical utility in patients with acute ischemic stroke. Brain Behav. 2020, 10, e01806. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Xu, S.; Jin, X.; Lu, X.; Liu, L.; Lou, Y.; Wang, Y.; Li, Y.; Jin, Y. A comparison of six clock-drawing test scoring methods in a nursing home. Aging Clin. Exp. Res. 2018, 30, 775–781. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Shao, K.; Guo, S.; Wang, W.; Yang, Y.; Zhao, Z.; Feng, R.; Wang, J. Clock-drawing test in vascular mild cognitive impairment: Validity of quantitative and qualitative analyses. J. Clin. Exp. Neuropsychol. 2020, 42, 622–633. [Google Scholar] [CrossRef] [PubMed]
- Carnero-Pardo, C.; Rego-García, I.; Barrios-López, J.M.; Blanco-Madera, S.; Calle-Calle, R.; López-Alcalde, S.; Vílchez-Carrillo, R.M. Assessment of the diagnostic accuracy and discriminative validity of the Clock Drawing and Mini-Cog tests in detecting cognitive impairment. Neurología 2019. [Google Scholar] [CrossRef]
- Lycke, M.; Ketelaars, L.; Boterberg, T.; Pottel, L.; Pottel, H.; Vergauwe, P.; Goethals, L.; Van Eygen, K.; Werbrouck, P.; Debruyne, D.; et al. Validation of the Freund Clock Drawing Test as a screening tool to detect cognitive dysfunction in elderly cancer patients undergoing comprehensive geriatric assessment. Psycho-Oncology 2014, 23, 1172–1177. [Google Scholar] [CrossRef]
- Shulman, K.I. Clock-drawing: Is it the ideal cognitive screening test? Int. J. Geriatr. Psychiatry 2000, 15, 548–561. [Google Scholar] [CrossRef]
- Lourenço, R.A.; Ribeiro-Filho, S.T.; Moreira Ide, F.; Paradela, E.M.; Miranda, A.S. The Clock Drawing Test: Performance among elderly with low educational level. Braz. J. Psychiatry 2008, 30, 309–315. [Google Scholar] [CrossRef]
- Borson, S.; Scanlan, J.; Brush, M.; Vitaliano, P.; Dokmak, A. The mini-cog: A cognitive ‘vital signs’ measure for dementia screening in multi-lingual elderly. Int. J. Geriatr. Psychiatry 2000, 15, 1021–1027. [Google Scholar] [CrossRef]
- Rezaei, M.; Rashedi, V.; Lotfi, G.; Shirinbayan, P.; Foroughan, M. Psychometric Properties of the Persian Adaptation of Mini-Cog Test in Iranian Older Adults. Int. J. Aging Hum. Dev. 2018, 86, 266–280. [Google Scholar] [CrossRef]
- Scanlan, J.; Borson, S. The Mini-Cog: Receiver operating characteristics with expert and naïve raters. Int. J. Geriatr. Psychiatry 2001, 16, 216–222. [Google Scholar] [CrossRef]
- Seitz, D.P.; Chan, C.C.; Newton, H.T.; Gill, S.S.; Herrmann, N.; Smailagic, N.; Nikolaou, V.; Fage, B.A. Mini-Cog for the diagnosis of Alzheimer’s disease dementia and other dementias within a primary care setting. Cochrane Database Syst. Rev. 2018, 2, Cd011415. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro Filho, S.T.; Lourenço, R.A. The performance of the Mini-Cog in a sample of low educational level elderly. Dement. Neuropsychol. 2009, 3, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”: A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Boban, M.; Malojčić, B.; Mimica, N.; Vuković, S.; Zrilić, I.; Hof, P.R.; Simić, G. The reliability and validity of the mini-mental state examination in the elderly Croatian population. Dement. Geriatr. Cogn. Disord. 2012, 33, 385–392. [Google Scholar] [CrossRef]
- Elhan, A.H.; Kutlay, S.; Küçükdeveci, A.A.; Cotuk, C.; Oztürk, G.; Tesio, L.; Tennant, A. Psychometric properties of the Mini-Mental State Examination in patients with acquired brain injury in Turkey. J. Rehabil. Med. 2005, 37, 306–311. [Google Scholar] [CrossRef]
- El-Hayeck, R.; Baddoura, R.; Wehbé, A.; Bassil, N.; Koussa, S.; Abou Khaled, K.; Richa, S.; Khoury, R.; Alameddine, A.; Sellal, F. An Arabic Version of the Mini-Mental State Examination for the Lebanese Population: Reliability, Validity, and Normative Data. J. Alzheimers Dis. 2019, 71, 525–540. [Google Scholar] [CrossRef]
- Ong, H.L.; Subramaniam, M.; Abdin, E.; Wang, P.; Vaingankar, J.A.; Lee, S.P.; Shafie, S.; Seow, E.; Chong, S.A. Performance of Mini-Mental State Examination (MMSE) in long-stay patients with schizophrenia or schizoaffective disorders in a psychiatric institute. Psychiatry Res. 2016, 241, 256–262. [Google Scholar] [CrossRef]
- Ansari, N.N.; Naghdi, S.; Hasson, S.; Valizadeh, L.; Jalaie, S. Validation of a Mini-Mental State Examination (MMSE) for the Persian population: A pilot study. Appl. Neuropsychol. 2010, 17, 190–195. [Google Scholar] [CrossRef]
- Matuoka, J.Y.; Kurita, G.P.; Nordly, M.; Sjøgren, P.; de Mattos-Pimenta, C.A. Validation of a Battery of Neuropsychological Tests for Patients With Metastatic Cancer. Clin. Nurs. Res. 2019, 29, 607–615. [Google Scholar] [CrossRef]
- Marioni, R.E.; Chatfield, M.; Brayne, C.; Matthews, F.E.; Medical Research Council Cognitive, F.; Ageing, S. The reliability of assigning individuals to cognitive states using the Mini Mental-State Examination: A population-based prospective cohort study. BMC Med. Res. Methodol. 2011, 11, 127. [Google Scholar] [CrossRef]
- Bowie, P.; Branton, T.; Holmes, J. Should the Mini Mental State Examination be used to monitor dementia treatments? Lancet 1999, 354, 1527–1528. [Google Scholar] [CrossRef]
- Creavin, S.T.; Wisniewski, S.; Noel-Storr, A.H.; Trevelyan, C.M.; Hampton, T.; Rayment, D.; Thom, V.M.; Nash, K.J.; Elhamoui, H.; Milligan, R.; et al. Mini-Mental State Examination (MMSE) for the detection of dementia in clinically unevaluated people aged 65 and over in community and primary care populations. Cochrane Database Syst. Rev. 2016, Cd011145. [Google Scholar] [CrossRef]
- Nasreddine, Z.S.; Phillips, N.A.; Bédirian, V.; Charbonneau, S.; Whitehead, V.; Collin, I.; Cummings, J.L.; Chertkow, H. The Montreal Cognitive Assessment, MoCA: A Brief Screening Tool for Mild Cognitive Impairment. J. Am. Geriatr. Soc. 2005, 53, 695–699. [Google Scholar] [CrossRef]
- Iiboshi, K.; Yoshida, K.; Yamaoka, Y.; Eguchi, Y.; Sato, D.; Kishimoto, M.; Funaki, K.; Mimura, M.; Kishimoto, T. A Validation Study of the Remotely Administered Montreal Cognitive Assessment Tool in the Elderly Japanese Population. Telemed. J. e-Health 2020, 26, 920–928. [Google Scholar] [CrossRef]
- Freud, T.; Vostrikov, A.; Dwolatzky, T.; Punchik, B.; Press, Y. Validation of the Russian Version of the MoCA Test as a Cognitive Screening Instrument in Cognitively Asymptomatic Older Individuals and Those With Mild Cognitive Impairment. Front. Med. 2020, 7, 447. [Google Scholar] [CrossRef] [PubMed]
- Gil, L.; Ruiz de Sánchez, C.; Gil, F.; Romero, S.J.; Pretelt Burgos, F. Validation of the Montreal Cognitive Assessment (MoCA) in Spanish as a screening tool for mild cognitive impairment and mild dementia in patients over 65 years old in Bogotá, Colombia. Int. J. Geriatr. Psychiatry 2015, 30, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Bartos, A.; Fayette, D. Validation of the Czech Montreal Cognitive Assessment for Mild Cognitive Impairment due to Alzheimer Disease and Czech Norms in 1,552 Elderly Persons. Dement. Geriatr. Cogn. Disord. 2018, 46, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Lifshitz, M.; Dwolatzky, T.; Press, Y. Validation of the Hebrew version of the MoCA test as a screening instrument for the early detection of mild cognitive impairment in elderly individuals. J. Geriatr. Psychiatry Neurol. 2012, 25, 155–161. [Google Scholar] [CrossRef]
- Tu, Q.Y.; Jin, H.; Ding, B.R.; Yang, X.; Lei, Z.H.; Bai, S.; Zhang, Y.D.; Tang, X.Q. Reliability, validity, and optimal cutoff score of the montreal cognitive assessment (changsha version) in ischemic cerebrovascular disease patients of hunan province, china. Dement. Geriatr. Cogn. Dis. Extra. 2013, 3, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Dupuis, K.; Pichora-Fuller, M.K.; Chasteen, A.L.; Marchuk, V.; Singh, G.; Smith, S.L. Effects of hearing and vision impairments on the Montreal Cognitive Assessment. Aging Neuropsychol. Cogn. 2015, 22, 413–437. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, E. A short portable mental status questionnaire for the assessment of organic brain deficit in elderly patients. J. Am. Geriatr. Soc. 1975, 23, 433–441. [Google Scholar] [CrossRef]
- Kojaie-Bidgoli, A.; Fadayevatan, R.; Sharifi, F.; Alizadeh-Khoei, M.; Vahabi, Z.; Aminalroaya, R. Applicability of SPMSQ in illiterate outpatients in clinics: The validity and reliability of the Short Portable Mental Status Questionnaire. Appl. Neuropsychol. Adult 2020, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Foreman, M.D. Reliability and validity of mental status questionnaires in elderly hospitalized patients. Nurs. Res. 1987, 36, 216–220. [Google Scholar] [CrossRef] [PubMed]
- De Roeck, E.E.; De Deyn, P.P.; Dierckx, E.; Engelborghs, S. Brief cognitive screening instruments for early detection of Alzheimer’s disease: A systematic review. Alzheimers Res. 2019, 11, 21. [Google Scholar] [CrossRef]
- Apóstolo, J.L.A.; Paiva, D.D.S.; Silva, R.; Santos, E.; Schultz, T.J. Adaptation and validation into Portuguese language of the six-item cognitive impairment test (6CIT). Aging Ment. Health 2018, 22, 1184–1189. [Google Scholar] [CrossRef] [PubMed]
- Taussig, I.M.; Mack, W.J.; Henderson, V.W. Concurrent validity of Spanish-language versions of the Mini-Mental State Examination, Mental Status Questionnaire, Information-Memory-Concentration test, and Orientation-Memory-Concentration test: Alzheimer’s disease patients and nondemented elderly comparison subjects. J. Int. Neuropsychol. Soc. 1996, 2, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Borson, S.; Brush, M.; Gil, E.; Scanlan, J.; Vitaliano, P.; Chen, J.; Cashman, J.; Sta Maria, M.M.; Barnhart, R.; Roques, J. The Clock Drawing Test: Utility for dementia detection in multiethnic elders. J. Gerontol. A Biol. Sci. Med. Sci. 1999, 54, M534–M540. [Google Scholar] [CrossRef]
- Palsetia, D.; Rao, G.P.; Tiwari, S.C.; Lodha, P.; De Sousa, A. The Clock Drawing Test versus Mini-mental Status Examination as a Screening Tool for Dementia: A Clinical Comparison. Indian J. Psychol. Med. 2018, 40, 1–10. [Google Scholar] [CrossRef]
- Borson, S. Mini-Cog: Screening for Cognitive Impairment in Older Adults. Available online: https://mini-cog.com/ (accessed on 12 August 2021).
- PAR, Inc. MMSE Mini Mental State Examination. Available online: https://www.parinc.com/Products/Pkey/237 (accessed on 8 August 2021).
- Molloy, D.W.; Standish, T.I. A guide to the standardized Mini-Mental State Examination. Int. Psychogeriatr. 1997, 9 (Suppl. 1), 87–94; discussion 143–150. [Google Scholar] [CrossRef]
- Teng, E.L.; Chui, H.C. The Modified Mini-Mental State (3MS) examination. J. Clin. Psychiatry 1987, 48, 314–318. [Google Scholar]
- Kennedy, R.E.; Williams, C.P.; Sawyer, P.; Allman, R.M.; Crowe, M. Comparison of in-person and telephone administration of the Mini-Mental State Examination in the University of Alabama at Birmingham Study of Aging. J. Am. Geriatr. Soc. 2014, 62, 1928–1932. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, Z. MoCA Cognitive Assessment. Available online: https://www.mocatest.org/ (accessed on 8 August 2021).
- Erkinjuntti, T.; Sulkava, R.; Wikström, J.; Autio, L. Short Portable Mental Status Questionnaire as a screening test for dementia and delirium among the elderly. J. Am. Geriatr. Soc. 1987, 35, 412–416. [Google Scholar] [CrossRef] [PubMed]
- Lorentz, W.J.; Scanlan, J.M.; Borson, S. Brief Screening Tests for Dementia. Can. J. Psychiatry 2002, 47, 723–733. [Google Scholar] [CrossRef] [PubMed]
- Yeh, S.-C.J.; Liu, Y.-Y. Influence of social support on cognitive function in the elderly. BMC Health Serv. Res. 2003, 3, 9. [Google Scholar] [CrossRef]
- Martínez de la Iglesia, J.; Dueñas Herrero, R.; Onís Vilches, M.C.; Aguado Taberné, C.; Albert Colomer, C.; Luque Luque, R. Spanish language adaptation and validation of the Pfeiffer’s questionnaire (SPMSQ) to detect cognitive deterioration in people over 65 years of age. Med. Clin. 2001, 117, 129–134. [Google Scholar] [CrossRef]
- Dion, C.; Arias, F.; Amini, S.; Davis, R.; Penney, D.; Libon, D.J.; Price, C.C. Cognitive Correlates of Digital Clock Drawing Metrics in Older Adults with and without Mild Cognitive Impairment. J. Alzheimer’s Dis. JAD 2020, 75, 73–83. [Google Scholar] [CrossRef]
- Petrazzuoli, F.; Vestberg, S.; Midlöv, P.; Thulesius, H.; Stomrud, E.; Palmqvist, S. Brief Cognitive Tests Used in Primary Care Cannot Accurately Differentiate Mild Cognitive Impairment from Subjective Cognitive Decline. J. Alzheimer’s Dis. JAD 2020, 75, 1191–1201. [Google Scholar] [CrossRef]
- Rakusa, M.; Jensterle, J.; Mlakar, J. Clock Drawing Test: A Simple Scoring System for the Accurate Screening of Cognitive Impairment in Patients with Mild Cognitive Impairment and Dementia. Dement. Geriatr. Cogn. Disord. 2018, 45, 326–334. [Google Scholar] [CrossRef]
- Buckley, R.A.; Atkins, K.J.; Fortunato, E.; Silbert, B.; Scott, D.A.; Evered, L. A novel digital clock drawing test as a screening tool for perioperative neurocognitive disorders: A feasibility study. Acta Anaesthesiol. Scand. 2021, 65, 473–480. [Google Scholar] [CrossRef]
- Trongsakul, S.; Lambert, R.; Clark, A.; Wongpakaran, N.; Cross, J. Development of the Thai version of Mini-Cog, a brief cognitive screening test. Geriatr. Gerontol. Int. 2015, 15, 594–600. [Google Scholar] [CrossRef]
- Albanna, M.; Yehya, A.; Khairi, A.; Dafeeah, E.; Elhadi, A.; Rezgui, L.; Al Kahlout, S.; Yousif, A.; Uthman, B.; Al-Amin, H. Validation and cultural adaptation of the Arabic versions of the Mini-Mental Status Examination—2 and Mini-Cog test. Neuropsychiatr. Dis. Treat. 2017, 13, 793–801. [Google Scholar] [CrossRef] [PubMed]
- Milian, M.; Leiherr, A.-M.; Straten, G.; Müller, S.; Leyhe, T.; Eschweiler, G.W. The Mini-Cog, Clock Drawing Test, and the Mini-Mental State Examination in a German Memory Clinic: Specificity of separation dementia from depression. Int. Psychogeriatr. 2013, 25, 96–104. [Google Scholar] [CrossRef] [PubMed]
- O’Bryant, S.E.; Humphreys, J.D.; Smith, G.E.; Ivnik, R.J.; Graff-Radford, N.R.; Petersen, R.C.; Lucas, J.A. Detecting dementia with the mini-mental state examination in highly educated individuals. Arch. Neurol. 2008, 65, 963–967. [Google Scholar] [CrossRef] [PubMed]
- Franco-Marina, F.; García-González, J.J.; Wagner-Echeagaray, F.; Gallo, J.; Ugalde, O.; Sánchez-García, S.; Espinel-Bermúdez, C.; Juárez-Cedillo, T.; Rodríguez, M.A.; García-Peña, C. The Mini-mental State Examination revisited: Ceiling and floor effects after score adjustment for educational level in an aging Mexican population. Int. Psychogeriatr. 2010, 22, 72–81. [Google Scholar] [CrossRef] [PubMed]
- Hoops, S.; Nazem, S.; Siderowf, A.D.; Duda, J.E.; Xie, S.X.; Stern, M.B.; Weintraub, D. Validity of the MoCA and MMSE in the detection of MCI and dementia in Parkinson disease. Neurology 2009, 73, 1738–1745. [Google Scholar] [CrossRef] [PubMed]
- Burton, L.; Tyson, S.F. Screening for cognitive impairment after stroke: A systematic review of psychometric properties and clinical utility. J. Rehabil. Med. 2015, 47, 193–203. [Google Scholar] [CrossRef]
- Freitas, S.; Simões, M.R.; Alves, L.; Duro, D.; Santana, I. Montreal Cognitive Assessment (MoCA): Validation study for frontotemporal dementia. J. Geriatr. Psychiatry Neurol. 2012, 25, 146–154. [Google Scholar] [CrossRef]
- Freitas, S.; Simões, M.R.; Alves, L.; Vicente, M.; Santana, I. Montreal Cognitive Assessment (MoCA): Validation study for vascular dementia. J. Int. Neuropsychol. Soc. 2012, 18, 1031–1040. [Google Scholar] [CrossRef]
- Roccaforte, W.H.; Burke, W.J.; Bayer, B.L.; Wengel, S.P. Reliability and validity of the Short Portable Mental Status Questionnaire administered by telephone. J. Geriatr. Psychiatry Neurol. 1994, 7, 33–38. [Google Scholar] [CrossRef]
- Borson, S.; Scanlan, J.M.; Chen, P.; Ganguli, M. The Mini-Cog as a screen for dementia: Validation in a population-based sample. J. Am. Geriatr. Soc. 2003, 51, 1451–1454. [Google Scholar] [CrossRef]
- Borson, S.; Scanlan, J.M.; Watanabe, J.; Tu, S.P.; Lessig, M. Simplifying detection of cognitive impairment: Comparison of the Mini-Cog and Mini-Mental State Examination in a multiethnic sample. J. Am. Geriatr. Soc. 2005, 53, 871–874. [Google Scholar] [CrossRef] [PubMed]
- Bravo, G.; Hébert, R. Age- and education-specific reference values for the Mini-Mental and modified Mini-Mental State Examinations derived from a non-demented elderly population. Int. J. Geriatr. Psychiatry 1997, 12, 1008–1018. [Google Scholar] [CrossRef]
- Scazufca, M.; Almeida, O.P.; Vallada, H.P.; Tasse, W.A.; Menezes, P.R. Limitations of the Mini-Mental State Examination for screening dementia in a community with low socioeconomic status: Results from the Sao Paulo Ageing & Health Study. Eur. Arch. Psychiatry Clin. Neurosci. 2009, 259, 8–15. [Google Scholar] [CrossRef]
- Lange, M.; Binarelli, G.; Joly, F. Geriatric phone follow-up including validated cognitive assessment in older patients treated for cancer. J. Geriatr. Oncol. 2021, 12, 838–839. [Google Scholar] [CrossRef] [PubMed]
- Tsoi, K.K.; Chan, J.Y.; Hirai, H.W.; Wong, S.Y.; Kwok, T.C. Cognitive Tests to Detect Dementia: A Systematic Review and Meta-analysis. JAMA Intern. Med. 2015, 175, 1450–1458. [Google Scholar] [CrossRef]
- Lezak, M.D.; Howieson, D.B.; Loring, D.W.; Hannay, H.J.; Fischer, J.S. Neuropsychological Assessment, 4th ed.; Oxford University Press: New York, NY, USA, 2004; pp. xiv, 1016–xiv, 1016. [Google Scholar]
- Calamia, M.; Markon, K.; Tranel, D. Scoring higher the second time around: Meta-analyses of practice effects in neuropsychological assessment. Clin. Neuropsychol. 2012, 26, 543–570. [Google Scholar] [CrossRef]
- Galvin, J.E.; Roe, C.M.; Xiong, C.; Morris, J.C. Validity and reliability of the AD8 informant interview in dementia. Neurology 2006, 67, 1942–1948. [Google Scholar] [CrossRef]
- Galvin, J.E.; Roe, C.M.; Powlishta, K.K.; Coats, M.A.; Muich, S.J.; Grant, E.; Miller, J.P.; Storandt, M.; Morris, J.C. The AD8: A brief informant interview to detect dementia. Neurology 2005, 65, 559–564. [Google Scholar] [CrossRef]
- Jorm, A.F. The Informant Questionnaire on cognitive decline in the elderly (IQCODE): A review. Int. Psychogeriatr. 2004, 16, 275–293. [Google Scholar] [CrossRef]
- Jorm, A.F.; Jacomb, P.A. The Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE): Socio-demographic correlates, reliability, validity and some norms. Psychol. Med. 1989, 19, 1015–1022. [Google Scholar] [CrossRef] [PubMed]
- Nicholas, L.H.; Langa, K.M.; Bynum, J.P.W.; Hsu, J.W. Financial Presentation of Alzheimer Disease and Related Dementias. JAMA Intern. Med. 2021, 181, 220–227. [Google Scholar] [CrossRef]
- Ott, B.R.; Daiello, L.A. How does dementia affect driving in older patients? Aging Health 2010, 6, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Dementia Australia. Tests Used in Diagnosing Dementia. Available online: https://www.dementia.org.au/national/about-dementia/how-can-i-find-out-more/tests-used-in-diagnosing-dementia (accessed on 14 August 2021).
- Dementia Australia. Diagnosing Dementia. Available online: https://www.dementia.org.au/information/diagnosing-dementia (accessed on 14 August 2021).
- Hamaker, M.E.; Schiphorst, A.H.; ten Bokkel Huinink, D.; Schaar, C.; van Munster, B.C. The effect of a geriatric evaluation on treatment decisions for older cancer patients–a systematic review. Acta Oncol. 2014, 53, 289–296. [Google Scholar] [CrossRef]
- Blackwood, J.; Rybicki, K.; Huang, M. Cognitive measures in older cancer survivors: An examination of validity, reliability, and minimal detectable change. J. Geriatr. Oncol. 2021, 12, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Karuturi, M.; Wong, M.L.; Hsu, T.; Kimmick, G.G.; Lichtman, S.M.; Holmes, H.M.; Inouye, S.K.; Dale, W.; Loh, K.P.; Whitehead, M.I.; et al. Understanding cognition in older patients with cancer. J. Geriatr. Oncol. 2016, 7, 258–269. [Google Scholar] [CrossRef] [PubMed]
- Malik, U.; Alam, Z.; Loucks, A.; Jin, R.; Yokom, D.; Watt, S.; Berger, A.; Romanovsky, L.; Puts, M.; Alibhai, S.M.H. Downstream consequences of abnormal cognitive screening in older adults seen pretreatment in a geriatric oncology clinic. J. Geriatr. Oncol. 2020, 11, 784–789. [Google Scholar] [CrossRef]
| Cancer*[ti/ab] OR Neoplasm*[ti/ab] OR Oncolog*[ti/ab] | |
| AND | geriatric*[ti/ab] OR elderly*[ti/ab] OR “older adult*”[ti/ab] OR “senior adult*”[ti/ab] OR “older patient*”[ti/ab] |
| AND | assessment*[ti/ab] OR “evaluation*[ti/ab] |
| AND | guideline*[ti/ab] OR “position statement”[ti/ab] OR “consensus statement” [ti/ab] or recommendation*”[ti/ab] |
| Psychometric Considerations for Clinical Use |
| Validated populations |
| Validation in older adults with cancer |
| Reliability |
| Sensitivity and specificity for detecting cognitive impairment |
| Effect of literacy or education |
| Effect of visual impairment |
| Practical Considerations |
| Cognitive domains assessed |
| Completion time |
| Training recommendations and frequency |
| Cost |
| Accessibility |
| Available in languages other than English |
| Available in alternate versions for repeat testing |
| Suitability for telehealth consultations |
| Guidelines | Organization | Year of Publication | Recommended Tools |
|---|---|---|---|
| Geriatric assessment in daily oncology practice for nurses and allied health care professionals: Opinion paper of the Nursing and Allied Health Interest Group of the International Society of Geriatric Oncology (SIOG) [20] | Nursing and Allied Health Interest Group of SIOG (International Society for Geriatric Oncology) | 2016 | Mini-Cog, MMSE, MoCA |
| NCCN Clinical Practice Guidelines in Oncology: Older adult oncology, version 2.2017 [25] | NCCN (National Comprehensive Cancer Network) | 2017 | Mini-Cog, MMSE, MoCA |
| Practical assessment and management of vulnerabilities in older patients receiving chemotherapy: ASCO guideline for geriatric oncology [1] | ASCO (American Society of Clinical Oncology) | 2018 | Mini-Cog, BOMC, MMSE, MoCA |
| General recommendations paper on the management of older patients with cancer: The SEOM geriatric oncology task forces position statement [24] | SEOM (Spanish Society for Medical Oncology) | 2018 | SPMSQ |
| What every oncologist should know about geriatric assessment for older patients with cancer: Young International Society of Geriatric Oncology position paper [21] | Young SIOG (Young International Society for Geriatric Oncology) | 2018 | Mini-Cog, MMSE, MoCA, BOMC, Clock-drawing |
| NCCN Clinical Practice Guidelines in Oncology: Older adult oncology, version 1.2019 [22] | NCCN (National Comprehensive Cancer Network) | 2019 | Mini-Cog, MMSE, MoCA |
| Adapting care for older cancer patients during the COVID-19 pandemic: Recommendations for the International Society for Geriatric Oncology COVID-19 Working Group [26] | SIOG (International Society for Geriatric Oncology) | 2020 | BOMC |
| Comprehensive geriatric assessment in older adults with cancer: Recommendations by the Italian Society of Geriatrics and Gerontology [27] | SIGG (Italian Society of Geriatrics and Gerontology) | 2021 | MoCA, MMSE |
| Tool Name | Year Published | Validated Population | Validated in Older Adults with Cancer? | Reliability | Sensitivity and Specificity at Standard Cut-Points | Effects of Literacy or Education | Effects of Visual Impairment |
|---|---|---|---|---|---|---|---|
| BOMC [28] | 1983 | Validated in a variety of clinical settings [29,30] | No | Test-retest reliability correlation coefficient: 0.77 [31] | Sensitivity 78.5–83%, specificity 77–100% to detect dementia [32] | Not considered sensitive to educational level [33] | No visual elements |
| CDT [34] | 1963 | Widely validated in multiple countries and various neurological conditions [35,36,37,38] | Comparable with MMSE results in cancer population [39] | High inter-rater and test-retest reliability [40] | Mean sensitivity 85% and specificity 85% to detect dementia [40] | Not valid in those with ≤4 years of formal schooling [41] | Requires adequate vision |
| Mini-Cog [42] | 2000 | Validated in a variety of community and clinical settings and populations [43] | No | High inter-tester reliability [44] | Sensitivity 76% and specificity 73% to detect dementia [45] | Not recommended in those with ≤5 years of formal education [46] | Requires adequate vision |
| MMSE [47] | 1975 | Extensively validated in many countries, populations, and in a range of different neurological and neurocognitive conditions [48,49,50,51,52] | Validated in cancer populations [53] | Variability reported in the test-retest reliability [47,54,55] | Sensitivity 0.85, specificity 0.90 for detecting dementia [56] | Scores decrease with advancing age and less education | Includes writing, drawing, and reading tasks |
| MoCA [57] | 2005 | Extensively validated in many countries, populations, and in a range of neurological and neurocognitive conditions [58,59,60,61,62] | No | High test-retest reliability [63] | MCI sensitivity 90%, specificity 87% *AD sensitivity 100%, specificity 87% [57] | Add 1 point if ≤12 years of education [57] | Hearing and visual impairment affect the MoCA performance [64] |
| SPMSQ [65] | 1975 | Assessed in hospital inpatients, nursing homes, Finish, Singaporean, Iranian populations [66] | No | Test-retest reliability: 0.8–0.83 Interrater reliability: 0.62–0.87 [67] | Sensitivity 55–85.7% and specificity 78.9–96% for detecting dementia [32] | Subtract 1 from error score if grade school education, add 1 to error score if education beyond high school [65] | No visual elements |
| Tool Name | Cognitive Domains | Score Interpretation | Completion Time | Training Requirements | Fee | Copyright, Open Access, and Permission to Reuse | Languages | Alternate Versions | Telehealth Version | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Memory | Visuospatial | Orientation | Attention | Language | Praxis | Executive | |||||||||
| BOMC [28] | 🗸 | 🗸 | 🗸 | ≤7 = Normal ≥8 = Abnormal | 2–3 min [68] | No training recommended | Free usage for healthcare professionals | Copyrighted | Three languages: English, Portuguese, and Spanish [69,70] | No | Suitable for telephone consultation | ||||
| CDT [34] | 🗸 | 🗸 | Multiple different scoring methods [36,40] | 1–5 min [71] | Can be administered by non-trained professionals [72] | Freely available | Open access | Does not require translation | No | Suitable for video consultation | |||||
| Mini-Cog [42] | 🗸 | 🗸 | 🗸 | ≥3 = Lower likelihood of dementia [73] | 3 min [69] | Training not required, 90% concordance between “expert” and regular raters [44] | No cost | Copyrighted, but may be used without permission in clinical and educational settings [73] | Six languages: English, Spanish, Portuguese, Chinese, Malay, and Arabic [73] | No | Suitable for video consultation | ||||
| MMSE [47] | 🗸 | 🗸 | 🗸 | 🗸 | 🗸 | 🗸 | ≥24 = Normal [47] | 10–15 min [47] | No specific training is recommended | There is a cost to purchasing tests [74] | Copyrighted | 75 languages [74] | Many variations are published [75,76] | Validated telephone versions are available [77] | |
| MoCA [57] | 🗸 | 🗸 | 🗸 | 🗸 | 🗸 | 🗸 | >26 = normal ≤26 = Abnormal [57] | 10 min [54] | Mandatory training and certification program. Retraining recommended twice yearly [78] | USD 150 for training. No cost for using the test for clinical or teaching purposes [78] | Copyrighted, available for use without permission for clinical and teaching purposes [78] | Paper version: Nearly 100 languages App version: Five languages [78] | Three versions are available for repeat testing, if retesting within 3 months [78] | Video consultation with modified instructions. Abbreviated telephone version available [78] | |
| SPMSQ [65] | 🗸 | 🗸 | 🗸 | 0–3 errors = normal [79] | 5–10 min [80] | Can be applied without formal training [81] | Freely available | Open access | Three languages: English, Spanish, and Iranian [66,82] | No | Suitable for telephone consultation | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tuch, G.; Soo, W.K.; Luo, K.-Y.; Frearson, K.; Oh, E.L.; Phillips, J.L.; Agar, M.; Lane, H. Cognitive Assessment Tools Recommended in Geriatric Oncology Guidelines: A Rapid Review. Curr. Oncol. 2021, 28, 3987-4003. https://doi.org/10.3390/curroncol28050339
Tuch G, Soo WK, Luo K-Y, Frearson K, Oh EL, Phillips JL, Agar M, Lane H. Cognitive Assessment Tools Recommended in Geriatric Oncology Guidelines: A Rapid Review. Current Oncology. 2021; 28(5):3987-4003. https://doi.org/10.3390/curroncol28050339
Chicago/Turabian StyleTuch, Gina, Wee Kheng Soo, Ki-Yung Luo, Kinglsey Frearson, Ek Leone Oh, Jane L. Phillips, Meera Agar, and Heather Lane. 2021. "Cognitive Assessment Tools Recommended in Geriatric Oncology Guidelines: A Rapid Review" Current Oncology 28, no. 5: 3987-4003. https://doi.org/10.3390/curroncol28050339
APA StyleTuch, G., Soo, W. K., Luo, K.-Y., Frearson, K., Oh, E. L., Phillips, J. L., Agar, M., & Lane, H. (2021). Cognitive Assessment Tools Recommended in Geriatric Oncology Guidelines: A Rapid Review. Current Oncology, 28(5), 3987-4003. https://doi.org/10.3390/curroncol28050339





