The Role of Denosumab for Surgical Outcomes in Patients with Giant Cell Tumour of Bone: A Systematic Review
Abstract
1. Introduction
2. Methods
2.1. Search for Existing Systematic Reviews
2.2. Study Selection Criteria
2.3. Data Extraction and Assessment of Risk of Bias
2.4. Synthesizing the Evidence
3. Results
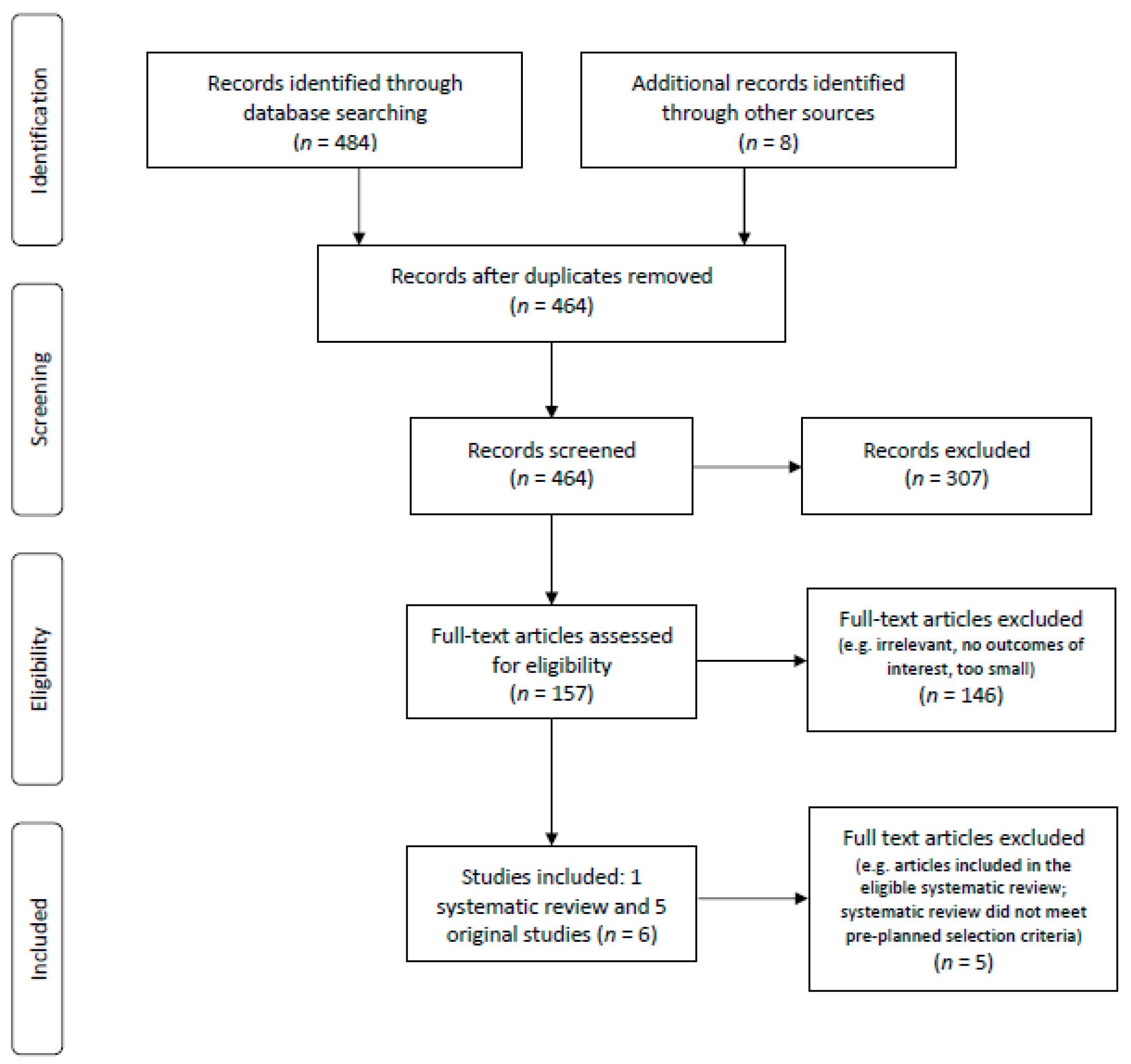
3.1. Search for Systematic Reviews
3.2. Search for Primary Studies
3.3. Outcomes
3.3.1. Facilitation of Surgery/Reduced Surgical Morbidity
3.3.2. Disease Recurrence
3.3.3. Pain Control
3.3.4. Disease Stability/Control
3.3.5. Adverse Effects
Malignant Transformation
Osteonecrosis of Jaw
Atypical Femur Fracture
3.4. Ongoing, Unpublished, or Incomplete Studies
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Literature Search Strategy
Appendix A.1. Giant Cell Tumour of Bone
- exp osteoclastoma/
- osteoclastoma.mp.
- (giant cell tumo$r adj4 bone).mp.
- exp denosumab/
- (denosumab or amgiva or prolia or xgeva or amg_162 or amg162).mp.
- (1 or 2 or 3) and (4 or 5)
- exp “giant cell tumor of bone”/
- (giant cell tumo$r adj4 bone).mp.
- exp denosumab/
- (denosumab or amgiva or prolia or xgeva or amg_162 or amg162).mp.
- (1 or 2) and (3 or 4)
- remove duplicates from 5
Appendix A.2. Benign Fibrous Histiocytoma of Bone and Secondary Aneurysmal Bone Cyst
- exp osteoclastoma/
- osteoclastoma.mp.
- (giant cell tumo$r adj4 bone).mp.
- (benign fibrous histiocytoma).mp.
- exp bone cysts, aneurysmal/
- (aneurysmal bone cystS).mp.
- exp denosumab/
- (denosumab or amgiva or prolia or xgeva or amg_162 or amg162).mp.
- (4 or 5 or 6) not (1 or 2 or 3)
- 9 and (7 or 8)
- Remove duplicates from 10
- exp “giant cell tumor of bone”/
- (giant cell tumo$r adj4 bone).mp.
- exp Histiocytoma, Benign Fibrous/
- Exp Bone Cysts, Aneurysmal/
- (aneurysmal bone cyst$).mp.
- exp denosumab/
- (denosumab or amgiva or prolia or xgeva or amg_162 or amg162).mp.
- (3 or 4 or 5) not (1 or 2)
- 8 and (6 or 7)
- Remove duplicates from 9
References
- van der Heijden, L.; Dijkstra, P.D.S.; Blay, J.Y.; Gelderblom, H. Giant cell tumour of bone in the denosumab era. Eur. J. Cancer 2017, 77, 75–83. [Google Scholar] [CrossRef]
- Xu, S.F.; Adams, B.; Yu, X.C.; Xu, M. Denosumab and giant cell tumour of bone-a review and future management considerations. Curr. Oncol. 2013, 20, e442–e447. [Google Scholar] [CrossRef]
- Sobti, A.; Agrawal, P.; Agarwala, S.; Agarwal, M. Giant Cell Tumor of Bone—An Overview. Arch. Bone. Jt. Surg. 2016, 4, 2–9. [Google Scholar]
- Chawla, S.; Henshaw, R.; Seeger, L.; Choy, E.; Blay, J.Y.; Ferrari, S.; Kroep, J.; Grimer, R.; Reichardt, P.; Rutkowski, P.; et al. Safety and efficacy of denosumab for adults and skeletally mature adolescents with giant cell tumour of bone: Interim analysis of an open-label, parallel-group, phase 2 study. Lancet Oncol. 2013, 14, 901–908. [Google Scholar] [CrossRef]
- Traub, F.; Singh, J.; Dickson, B.C.; Leung, S.; Mohankumar, R.; Blackstein, M.E.; Razak, A.B.; Griffin, A.M.; Ferguson, P.C.; Wunder, J.S. Efficacy of denosumab in joint preservation for patients with giant cell tumour of the bone. Eur. J. Cancer 2016, 59, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rutkowski, P.; Gaston, L.; Borkowska, A.; Stacchiotti, S.; Gelderblom, H.; Baldi, G.G.; Palmerini, E.; Casali, P.; Gronchi, A.; Parry, M.; et al. Denosumab treatment of inoperable or locally advanced giant cell tumor of bone—Multicenter analysis outside clinical trial. Eur. J. Surg. Oncol. 2018, 44, 1384–1390. [Google Scholar] [CrossRef]
- Thomas, D.; Henshaw, R.; Skubitz, K.; Chawla, S.; Staddon, A.; Blay, J.Y.; Roudier, M.; Smith, J.; Ye, Z.; Sohn, W.; et al. Denosumab in patients with giant-cell tumour of bone: An open-label, phase 2 study. Lancet Oncol. 2010, 11, 275–280. [Google Scholar] [CrossRef]
- Errani, C.; Tsukamoto, S.; Mavrogenis, A.F. How safe and effective is denosumab for bone giant cell tumour? Int. Orthop. 2017, 41, 2397–2400. [Google Scholar] [CrossRef] [PubMed]
- Chawla, S.; Blay, J.Y.; Rutkowski, P.; Le Cesne, A.; Reichardt, P.; Gelderblom, H.; Grimer, R.J.; Choy, E.; Skubitz, K.; Seeger, L.; et al. Denosumab in patients with giant-cell tumour of bone: A multicentre, open-label, phase 2 study. Lancet Oncol. 2019, 20, 1719–1729. [Google Scholar] [CrossRef]
- Whiting, P.; Savović, J.; Higgins, J.P.T.; Caldwell, D.M.; Reeves, B.C.; Shea, B.; Davies, P.; Kleijnen, J.; Churchill, R.; ROBIS Group. ROBIS: A new tool to assess risk of bias in systematic reviews was developed. J. Clin. Epidemiol. 2016, 69, 225–234. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.; Corbett, M.S.; Eldrige, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Hernan, M.A.; Reeves, B.C.; Savovic, J.; Berkman, N.D.; Viswanathan, M.; Herry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I. ROBINS-I: A Tool for Assessing Risk of Bias in Non-Randomized Studies of Interventions. BMJ 2016, 355. [Google Scholar] [CrossRef]
- StataCorp. Stata Statistical Software: Release 15.1 2017; StataCorp LP: College Station, TX, USA, 2017. [Google Scholar]
- Biermann, J.S.; Chow, W.; Boles, S.; Brigman, B.; Callan, A.K.; Cranmer, L.D.; Dianat, S.; Dommelly, E.; Fabbri, N.; Getty, P.; et al. National Clinical Practice Guidelines in Oncology. Bone Cancer. Version 1. 2021 [internet]. Pennsylvania: National Clinical Practice Guidelines. 2021. Available online: https://www.nccn.org/professionals/physician_gls/ (accessed on 19 March 2021).
- Charest-Morin, R.; Boriani, S.; Fisher, C.G.; Patel, S.R.; Kawahara, N.; Mendel, E.; Bettegowda, C.; Rhines, L.D. Benign tumors of the spine: Has new chemotherapy and interventional radiology changed the treatment paradigm? Spine 2016, 41, S178–S185. [Google Scholar] [CrossRef]
- Tsukamoto, S.; Tanaka, Y.; Mavrogenis, A.F.; Kido, A.; Kawaguchi, M.; Errani, C. Is Treatment with Denosumab Associated with Local Recurrence in Patients with Giant Cell Tumor of Bone Treated with Curettage? A Systematic Review. Clin. Orthop. Relat. Res. 2020, 478, 1076–1085. [Google Scholar] [CrossRef]
- Agarwal, M.G.; Gundavda, M.K.; Gupta, R.; Reddy, R. Does Denosumab Change the Giant Cell Tumor Treatment Strategy? Lessons Learned From Early Experience. Clin. Orthop. Relat. Res. 2018, 476, 1773–1782. [Google Scholar] [CrossRef]
- Lim, C.Y.; Liu, X.; He., F.; Liang, H.; Yang, Y.; Ji, T.; Yang, R.; Guo, W. Retrospective cohort study of 68 sacral giant cell tumours treated with nerve-sparing surgery and evaluation on therapeutic benefits of denosumab therapy. Bone Jt. J. 2020, 102–B, 177–185. [Google Scholar] [CrossRef]
- Medellin, M.R.; Fujiwara, T.; Tillman, R.M.; Jeys, L.M.; Gregory, J.; Stevenson, J.D.; Parry, M.; Abudu, A. Prognostic factors for local recurrence in extremity-located giant cell tumours of bone with pathological fracture. Bone Jt. J. 2018, 100–B, 1626–1632. [Google Scholar] [CrossRef]
- Scoccianti, G.; Totti, F.; Scorianz, M.; Baldi, G.; Roselli, G.; Beltrami, G.; Franchi, A.; Capanna, R.; Campanacci, D.A. Preoperative denosumab with curettage and cryotherapy in giant cell tumor of bone: Is there an increased risk of local recurrence? Clin. Orthop. Relat. Res. 2018, 476, 1783–1790. [Google Scholar] [CrossRef]
- Tsukamoto, S.; Mavrogenis, A.F.; Leone, G.; Righi, A.; Akahane, M.; Tanzi, P.; Kido, A.; Honoki, K.; Tanaka, Y.; Donati, D.M.; et al. Denosumab does not decrease the risk of lung metastases from bone giant cell tumour. Int. Orthop. 2019, 43, 483–489. [Google Scholar] [CrossRef]
- Errani, C.; Tsukamoto, S.; Leone, G.; Righi, A.; Akahane, M.; Tanaka, Y.; Donati, D.M. Denosumab may increase the risk of local recurrence in patients with giant-cell tumor of bone treated with curettage. J. Bone Jt. Surg. Am. 2018, 100, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Urakawa, H.; Yonemoto, T.; Matsumoto, S.; Takagi, T.; Asanuma, K.; Watanuki, M.; Takemoto, A.; Naka, N.; Matsumoto, Y.; Kawai, A.; et al. Clinical outcome of primary giant cell tumor of bone after curettage with or without perioperative denosumab in Japan: From a questionnaire for JCOG 1610 study. World J. Surg. Oncol. 2018, 16, 160. [Google Scholar] [CrossRef]
- Fedenko, A.A.; Tararykova, A. Neoadjuvant denosumab for the treatment of resectable giant cell tumor of bone: First results of Russian multicenter study. J. Clin. Oncol. Conf. 2018, 36, 11521. [Google Scholar] [CrossRef]
| Study | Domain 1: Study Eligibility Criteria | Domain 2: Identification and Selection of studies | Domain 3: Data Collection and Study Appraisal | Domain 4: Synthesis and Findings | Overall Risk of Bias |
|---|---|---|---|---|---|
| Charest-Morin 2016 [15] | Unclear | Low | Unclear | Unclear | Unclear |
| Tsukamoto 2019a [16] | Low | Low | Low | Low | Low |
| Study | Bias Due to Confounding | Bias Due to Selection of Participants | Bias in Measurement of Interventions | Bias Due to Deviations of Interventions | Bias Due to Missing Data | Bias in Measurement of Outcomes | Bias in Selection of Reported Results | Overall Risk of Bias Judgement |
|---|---|---|---|---|---|---|---|---|
| Agarwal 2018 [17] | Serious | Serious | Moderate | Moderate | Moderate | Moderate | Moderate | Serious |
| Lim 2020 [18] | Serious | Moderate | Moderate | Serious | Moderate | Moderate | Moderate | Serious |
| Medellin 2018 [19] | Moderate | Serious | Moderate | Serious | Moderate | Serious | Moderate | Serious |
| Scoccianti 2018 [20] | Moderate | Moderate | Moderate | Moderate | Moderate | Moderate | Moderate | Moderate |
| Tsukamoto 2019b [21] | Moderate | Serious | Moderate | Serious | Moderate | Serious | Moderate | Serious |
| Author and Study Type | Patient Population; Mean/ Median Age; Median Follow-Up Time | Arms or Comparisons | Number of Pts Analyzed | Facilitation of Surgery/Reduced Morbidity after Surgery | Disease Recurrence | Pain Control | Disease Stability/ Control | Adverse Effects |
|---|---|---|---|---|---|---|---|---|
| Agarwal 2018, Case-matched control [17] | 54 pts with primary or recurrent GCTB located in the axial skeleton, appendicular skeleton, or distal tibia and sacrum; 32 (17–67) yr; 27–60 mths | Group 1: Neoadjuvant DENO 120 mg every month for 4 mths with additional doses of 120 mg on d8, d15 during 1st mth only | 25 | DENO aided surgical resection by hardening the tumour and the bony shell | Group 1: 11 (44%) in curettage, Group 2: 7 (21%), OR = 3.03 (95% CI 0.96 to 9.54), p = 0.085 | NR | NR | No osteonecrosis of jaw |
| Group 2: Surgery alone | 34 | NR | ||||||
| Lim 2020, Retro cohort [18] | 64 pts with sacral GCTB; 34 (11–65) yr; 48 (12–91) mths | Group 1: Surgery alone | 36 | Mean operating time (mins (SD)): 199.4 (49.5) vs. 200.6 (69.8) vs. 181.2 (38.6), p = NS. Blood loss during surgery (ml (SD)): 1715 vs. 1600 vs. 1418, p = 0.008 | Local recurrence (n): 12 vs. 2 vs. 3. 1 yr RFS (%): 86.1 vs. 100 vs. 94.1, p = NS. 2 yr RFS (%) 72.2 vs. 100 vs. 86.3, p = NS. 3 yr RFS (%) 69.4 vs. 75.0 vs. 69.0, p = NS | NR | Local control rate: 66.7% vs. 77.8% vs. 87.5%, p = NS | NR |
| Group 2: Adjuvant DENO 120 mg mthly. Continuation based on progress. | 9 | No osteonecrosis of jaw | ||||||
| Group 3: Neoadjuvant DENO 120 mg d1, d8 and d15 with additional doses on d28 and every 4 wks, if required; Adjuvant: DENO 120 mg mthly, continuously based on progression. | 17 | No osteonecrosis of jaw, Malignant transformation: 3.2% | ||||||
| Medellin 2018, Retro Cohort [19] | 120 patients with GCTB located in the femur and other bones; 33 (14–86) yr; 75 (12–301) mths | Group 1: Neoadjuvant DENO: 120 mg wk 1,2,3,5 and mthly until surgery. Mean duration of denosumab treatment prior to surgery was 8.9 (3–19) mths. | 7 | Mean time interval until initial surgery (Group 1 vs. Group 2): 61 wks (13–134) vs. 4 wks (0–19), p < 0.001. After initial surgery n = 41 (41%) in Group 2 required further surgical intervention. No data in Group 1. | Multivariate analysis showed DENO associated with higher risk of local recurrence (HR 3.2, 95% CI 1.07–9.55, p = 0.037) | NR | NR | No significant adverse effects that warranted cessation of DENO |
| Group 2: Surgery alone | 100 | NR | ||||||
| Scoccianti 2018, Retro Cohort [20] | 97 pts with GCTB located at the distal femur, distal tibia, distal radius and sacrum, proximal humerus, distal humerus, finger phalanx, iliac wing, proximal tibia, patella; 42 (17–66) yr; 27–39 mths | Group 1: Neoadjuvant DENO: 120 mg weekly for 3 wks, then monthly for 3 mths, then surgery | 12 | All showed new bone formation around and partially inside the lesion. | 5 (42%) pts, Median 23 (7–54) mths post-surgery | NR | NR | No malignant transformation or osteonecrosis of jaw |
| Group 2: Surgery alone | 9 | Curettage was considered feasible already at presentation. | 1 (11%) pt, 14 mths post-surgery | NR | ||||
| Tsukamoto 2019b, Retro Cohort [21] | 411 pts with GCTB located in the distal radius and other sites such as the fibula, distal ulna, proximal radius, scapula, and patella; 29 (23–41) yr; 85 (IQR 54–124) | Group 1: Neoadjuvant DENO 120 mg once weekly for first mth and then once a mth for 6–9 mths, then surgery | 30 | NR | 15 (50%) pts vs. 58 (15.2%) pts, p <0.0001 | NR | Partial response: 22 (73.3%) pts, stable disease: 8 (26.7%) pts | 1 (3.3%) pt experienced lung metastases vs. 18 (4.7%) pts, p = 0.589 |
| Group 2: Surgery alone | 381 | Not applicable |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gupta, A.; Durocher-Allen, L.; Popovic, S.; Tozer, R.; Yao, X.; Ghert, M. The Role of Denosumab for Surgical Outcomes in Patients with Giant Cell Tumour of Bone: A Systematic Review. Curr. Oncol. 2021, 28, 1302-1313. https://doi.org/10.3390/curroncol28020124
Gupta A, Durocher-Allen L, Popovic S, Tozer R, Yao X, Ghert M. The Role of Denosumab for Surgical Outcomes in Patients with Giant Cell Tumour of Bone: A Systematic Review. Current Oncology. 2021; 28(2):1302-1313. https://doi.org/10.3390/curroncol28020124
Chicago/Turabian StyleGupta, Abha, Lisa Durocher-Allen, Snezana Popovic, Richard Tozer, Xiaomei Yao, and Michelle Ghert. 2021. "The Role of Denosumab for Surgical Outcomes in Patients with Giant Cell Tumour of Bone: A Systematic Review" Current Oncology 28, no. 2: 1302-1313. https://doi.org/10.3390/curroncol28020124
APA StyleGupta, A., Durocher-Allen, L., Popovic, S., Tozer, R., Yao, X., & Ghert, M. (2021). The Role of Denosumab for Surgical Outcomes in Patients with Giant Cell Tumour of Bone: A Systematic Review. Current Oncology, 28(2), 1302-1313. https://doi.org/10.3390/curroncol28020124





