Comparison of Perioperative Chemotherapy versus Postoperative Chemoradiotherapy for Operable Stomach Cancer: A Western Canadian Province Experience
Abstract
1. Introduction
2. Methods
Analysis of Primary and Secondary Endpoints
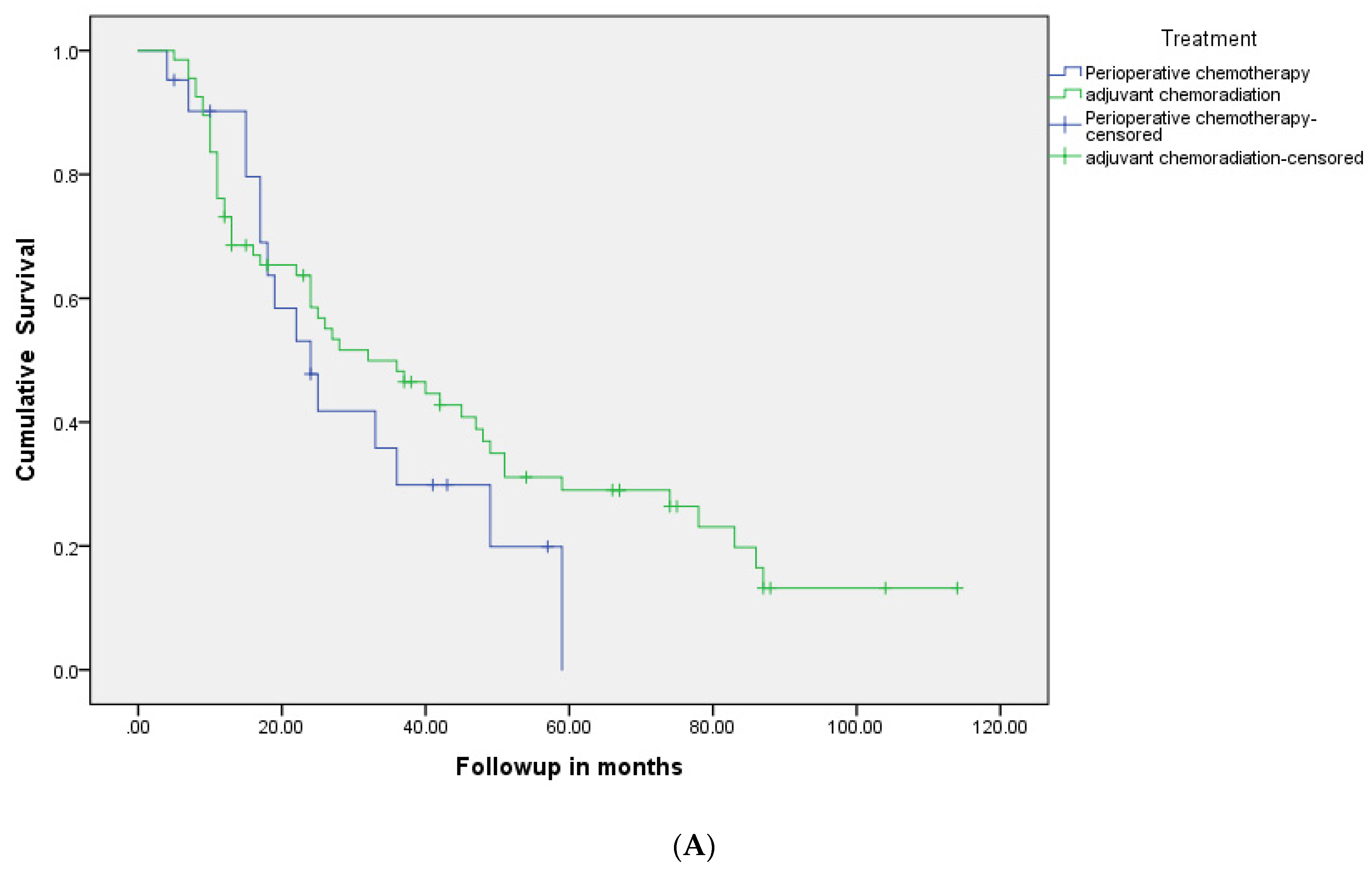
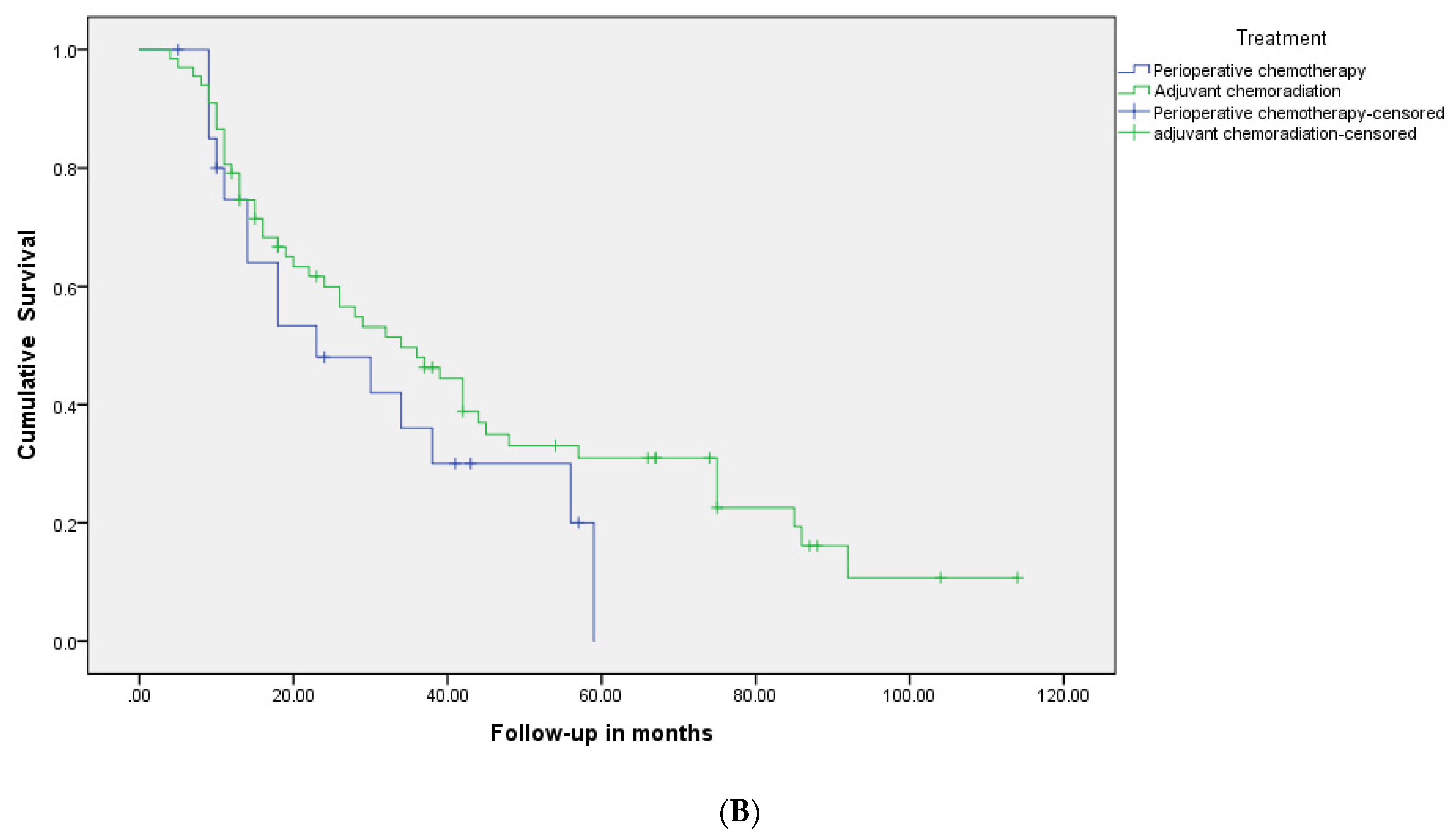
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics, 2013. CA Cancer J. Clin. 2013, 63, 11–30. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Shin, H.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D. GLOBOCAN 2008 v2.0, Cancer Incidence and Mortality Worldwide: IARC Cancer Base No. 10; International Agency Research Cancer: Lyon, France, 2010. [Google Scholar]
- Macdonald, J.S.; Smalley, S.R.; Benedetti, J.; Hundahl, S.A.; Estes, N.C.; Stemmermann, G.N.; Haller, D.G.; Ajani, A.J.; Gunderson, L.L.; Jessup, J.M.; et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N. Engl. J. Med. 2001, 345, 725–730. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, D.; Allum, W.H.; Stenning, S.P.; Thompson, J.N.; Van de Velde, C.J.H.; Nicolson, M.; Scarffe, J.H.; Lofts, F.J.; Falk, S.J.; Iveson, T.J.; et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N. Engl. J. Med. 2006, 355, 11–20. [Google Scholar] [CrossRef]
- Boige, V.; Pignon, J.-P.; Saint-Aubert, B.; Lasser, P.; Conroy, T.; Bouché, O.; Segol, P.; Bedenne, L.; Rougier, P.; Ychou, M. Final results of a randomized trial comparing preoperative 5-fluorouracil/cisplatin to surgery alone in adenocarcinoma of stomach and lower esophagus (ASLE): FNLCC ACCORD07-FFCD 9703 trial (abstract). J. Clin. Oncol. 2007, 25, 200s. [Google Scholar] [CrossRef]
- Nio, Y.; Koike, M.; Omori, H.; Hashimoto, K.; Itakura, M.; Yano, S.; Higami, T.; Maruyama, R. A randomized consent design trial of neoadjuvant chemotherapy with tegafur plus uracil (UFT) for gastric cancer—A single institute study. Anticancer Res. 2004, 24, 1879–1887. [Google Scholar]
- Schuhmacher, C.; Gretschel, S.; Lordick, F.; Reichardt, P.; Hohenberger, W.; Eisenberger, C.F.; Haag, C.; Mauer, M.E.; Hasan, B.; Welch, J.; et al. Neoadjuvant chemotherapy compared with surgery alone for locally advanced cancer of the stomach and cardia: European Organisation for Research and Treatment of Cancer randomized trial 40954. J. Clin. Oncol. 2010, 28, 5210–5218. [Google Scholar] [CrossRef]
- Xiong, B.H.; Cheng, Y.; Ma, L.; Zhang, C.Q. An updated meta-analysis of randomized controlled trial assessing the effect of neoadjuvant chemotherapy in advanced gastric cancer. Cancer Investig. 2014, 32, 272–284. [Google Scholar] [CrossRef]
- Bamias, A.; Karina, M.; Papakostas, P.; Kostopoulos, I.; Bobos, M.; Vourli, G.; Samantas, E.; Christodoulou, C.; Pentheroudakis, G.; Pectasides, D.; et al. A randomized phase III study of adjuvant platinum/docetaxel chemotherapy with or without radiation therapy in patients with gastric cancer. Cancer Chemother. Pharmacol. 2010, 65, 1009–1021. [Google Scholar] [CrossRef]
- Bang, Y.J.; Kim, Y.W.; Yang, H.K.; Chung, H.C.; Park, Y.-K.; Lee, K.H.; Lee, K.-W.; Kim, Y.H.; Noh, S.-I.; Cho, J.Y.; et al. Adjuvant capecitabine and oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): A phase 3 open-label, randomised controlled trial. Lancet 2012, 379, 315–321. [Google Scholar] [CrossRef]
- Kim, T.H.; Park, S.R.; Ryu, K.W.; Lim, D.H.; Hong, M.E.; Kim, K.-M.; Sohn, I.; Jung, S.H.; Choi, M.G.; Lee, J.H.; et al. Phase 3 trial of postoperative chemotherapy alone versus chemoradiation therapy in stage III-IV gastric cancer treated with R0 gastrectomy and D2 lymph node dissection. Int. J. Radiat. Oncol. Biol. Phys. 2012, 84, e585. [Google Scholar] [CrossRef]
- Park, S.H.; Sohn, T.S.; Lee, J.; Lim, D.H.; Hong, M.E.; Kim, K.-M.; Sohn, I.; Jung, S.H.; Choi, M.G.; Lee, J.H.; et al. Phase III trial to compare adjuvant chemotherapy with capecitabine and cisplatin versus concurrent chemoradiotherapy in gastric cancer: Final report of the adjuvant chemoradiotherapy in stomach tumors trial, including survival and subset analyses. J. Clin. Oncol. 2015, 33, 3130–3136. [Google Scholar] [CrossRef]
- Smalley, S.R.; Benedetti, J.K.; Haller, D.G.; Hundahl, S.A.; Estes, N.C.; Ajani, J.A.; Gunderson, L.L.; Goldman, B.; Martenson, J.A.; Jessup, J.M.; et al. Updated analysis of SWOG-directed intergroup study 0116: A phase III trial of adjuvant radiochemotherapy versus observation after curative gastric cancer resection. J. Clin. Oncol. 2012, 30, 2327–2333. [Google Scholar] [CrossRef]
- GASTRIC Group; Paoletti, X.; Oba, K.; Burzykowski, T.; Michiels, S.; Ohashi, Y.; Pignon, J.-P.; Rougier, P.; Sakamoto, J.; Sargent, D.; et al. Benefit of adjuvant chemotherapy for resectable gastric cancer: A meta-analysis. JAMA 2010, 303, 1729–1737. [Google Scholar] [PubMed]
- Dai, Q.; Jiang, L.; Lin, R.J.; Wei, K.K.; Gan, L.L.; Deng, C.H.; Guan, Q.L. Adjuvant chemoradiotherapy versus chemotherapy for gastric cancer: A meta-analysis of randomized controlled trials. J. Surg. Oncol. 2015, 111, 277–284. [Google Scholar] [CrossRef]
- Fitzgerald, T.L.; Efird, J.T.; Bellamy, N.; Russo, S.M.; Jindal, C.; Mosquera, C.; Holliday, E.G.; Biswas, T. Perioperative chemotherapy versus postoperative chemoradiotherapy in patients with resectable gastric/gastroesophageal junction adenocarcinomas: A survival analysis of 5058 patients. Cancer 2017, 123, 2909–2917. [Google Scholar] [CrossRef]
- Al-Batran, S.-E.; Homann, N.; Pauligk, C.; O’Goetze, T.; Meiler, J.; Kasper, S.; Kopp, H.-G.; Mayer, F.; Haag, G.M.; Luley, K.; et al. Perioperative chemotherapy with fluorouracil plus leucovorin, oxaliplatin, and docetaxel versus fluorouracil or capecitabine plus cisplatin and epirubicin for locally advanced, resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4): A randomised, phase 2/3 trial. Lancet 2019, 393, 1948–1957. [Google Scholar] [CrossRef]
- Jayanathan, M.; Ryan, P.; Molacek, E.N.; Fluck, M.; Hunsinger, M.; Wild, J.; Arora, T.K.; Shabahang, M.M.; Franko, J.; Blansfield, J.A.; et al. MAGIC versus MacDonald treatment regimens for gastric cancer: Trends and predictors of multimodal therapy for gastric cancer using the National Cancer Database. Am. J. Surg. 2020, 219, 129–135, ISSN 0002-9610. [Google Scholar] [CrossRef]
- Findlay, J.M.; Antonowicz, S.; Segaran, A.; El Kafsi, J.; Zhang, A.; Bradley, K.M.; Gillies, R.S.; Maynard, N.D.; Middleton, M.R. Routinely staging gastric cancer with 18F-FDG PET-CT detects additional metastases and predicts early recurrence and death after surgery. Eur. Radiol. 2019, 29, 2490–2498. [Google Scholar] [CrossRef] [PubMed]
- Jabo, B.; Selleck, M.J.; Morgan, J.W.; Lum, S.S.; Bahjri, K.A.; Aljehani, M.; Garberoglio, C.A.; Reeves, M.E.; Namm, J.P.; Solomon, N.L.; et al. Comparison of perioperative chemotherapy with adjuvant chemoradiotherapy for resectable gastric cancer: Findings from a population-based study. J. Gastrointest. Oncol. 2018, 9, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Orman, S.; Cayci, H.M. Gastric cancer: Factors affecting survival. Acta Chir. Belg. 2019, 119, 24–30. [Google Scholar] [CrossRef]
- Cordero-García, E.; Ramos-Esquivel, A.; Alpízar-Alpízar, W. Predictors of overall survival after surgery in gastric cancer patients from a Latin-American country. J. Gastrointest. Oncol. 2018, 9, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lu, J.J.; Du, Y.P.; Feng, C.X.; Wang, L.Q.; Chen, M.B. Prognostic value of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio in gastric cancer. Medicine 2018, 97, e0144. [Google Scholar] [CrossRef] [PubMed]
- Smyth, E.C.; Verheij, M.; Allum, W.; Cunningham, D.; Cervantes, A.; Arnold, D. Gastric cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2016, 27, v38–v49. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf (accessed on 12 March 2021).
- Bang, Y.-J.; Van Cutsem, E.; Fuchs, C.S.; Ohtsu, A.; Tabernero, J.; Ilson, D.H.; Hyung, W.J.; Strong, V.E.; Goetze, T.O.; Yoshikawa, T.; et al. KEYNOTE-585: Phase 3 study of chemotherapy (chemo) + pembrolizumab (pembro) vs chemo + placebo as neoadjuvant/adjuvant treatment for patients (pts) with gastric or gastroesophageal junction (G/GEJ) cancer. J. Clin. Oncol. 2018, 36, TPS4136. [Google Scholar] [CrossRef]

| Variables | All Patients N = 88 (%) | Adjuvant Chemoradiotherapy Group N = 67 (%) | Peri-Operative Chemotherapy Group N = 21 (%) | p Value |
|---|---|---|---|---|
| Median Age | 63 (IQR: 56–71) | 63 (IQR: 56–73) | 61 (IQR: 52–70) | 0.41 |
| Men | 61 (69) | 48 (72) | 13 (62) | 0.42 |
| Comorbid illness | 51 (58) | 40 (60) | 11 (52) | 0.61 |
| WHO Performance Status | ||||
| 0 | 27 (31) | 20 (30) | 7 (33) | 0.79 |
| 1 | 59 (67) | 45 (67) | 14 (67) | 1.0 |
| 2 | 2 (2) | 2 (3) | 0 | 1.0 |
| Current Smoker | 18 (21) | 13 (19) | 5 (24) | 0.75 |
| Past Smoker | 42 (48) | 34 (51) | 8 (38) | 0.33 |
| Secondary Cancer | 12 (14) | 10 (15) | 2 (10) | 0.72 |
| Clinical Stage | ||||
| I | 33 (38) | 26 (38) | 7 (33) | 0.80 |
| II | 47 (53) | 36 (54) | 11 (52) | 1.0 |
| III | 6 (7) | 3 (5) | 3 (15) | 0.14 |
| Not known | 2 (2) | 2 (3) | 0 | |
| Site | ||||
| Gastroesophageal (GE) Junction/cardia | 41 (47) | 33 (49) | 8 (38) | 0.45 |
| Body | 8 (9) | 4 (6) | 4 (19) | 0.08 |
| Antrum/Pylorus | 39 (44) | 30 (45) | 9 (43) | 1.0 |
| Clinical node positive | 36 (41) | 25 (37) | 11 (52) | 0.20 |
| Pathological node positive | 73 (83) | 58 (87) | 15 (71) | 0.18 |
| Pathological Stage | ||||
| 0 | 1 (1) | 0 | 1 (5) | 0.23 |
| I | 5 (6) | 2 (3) | 3 (14) | 0.08 |
| II | 36 (41) | 28 (42) | 8 (38) | 0.80 |
| III | 46 (52) | 37 (55) | 9 (43) | 0.45 |
| Positive margin | 21 (24) | 17 (25) | 4 (19) | 0.77 |
| Grade ≥ 3 | 56 (64) | 42 (63) | 14 (67) | 0.80 |
| Mean white blood cell (WBC) | 7.7 ± 2.5 | 7.7 ± 2.7 | 7.9 ± 2.1 | 0.68 |
| Mean Hemoglobin | 123 ± 19 | 123 ± 17 | 125 ± 26 | 0.52 |
| Mean Platelets | 300 ± 102 | 310 ± 102 | 271 ± 97 | 0.12 |
| Mean Creatinine | 75 ± 21 | 76 ± 23 | 75 ± 17 | 0.91 |
| Mean Albumin | 35 ± 5 | 35 ± 5 | 35 ± 5 | 0.81 |
| Mean Bilirubin | 9.1 ± 5.6 | 9.4 ± 5.9 | 8.5 ± 4.5 | 0.54 |
| Mean Alkaline Phosphatase | 84 ± 36 | 81 ± 23 | 93 ± 65 | 0.22 |
| Mean Alanine aminotransferase (ALT) | 23 ± 35 | 23 ± 39 | 23 ± 14 | 0.98 |
| Mean carcinoembryonic antigen (CEA) | 1.8 ± 1.1 | 1.8 ± 1.1 | 1.6 ± 1.2 | 0.63 |
| Variables | All Patients N = 88 (%) | Adjuvant Chemoradiotherapy Group N = 67 (%) | Peri-Operative Chemotherapy Group N = 21 (%) | p Value |
|---|---|---|---|---|
| Median RFS months (95% CI) | 32 (21.3–42.7) | 34 (20.7–47.3) | 23 (6.7–39.3) | NS |
| Median OS months (95% CI) | 34 (23–45) | 38 (24.6–51.4) | 30 (14.3–45.7) | NS |
| Three year-RFS | 49% | 55% | 43% | NS |
| Five year-RFS | 31% | 33% | 29% | NS |
| Three year-OS | 52% | 55% | 48% | NS |
| Five year-OS | 34% | 39% | 29% | NS |
| Variables | HR (95% CI) | |||
|---|---|---|---|---|
| Univariate | Multivariate | p | ||
| Age ≥ 70 years | 1.01 (0.58–1.73) | 0.97 | 1.10 (0.64–1.88) | 0.75 |
| Men | 1.11 (0.62–1.97) | 0.72 | ------ | |
| Comorbid illness | 1.16 (0.70–1.92) | 0.56 | 1.10 (0.63–1.78) | 0.83 |
| WHO performance status > 0 | 1.45 (0.83–2.53) | 0.19 | 1.48 (0.80–2.71) | 0.21 |
| Secondary cancer | 0.79 (0.40–1.57) | 0.51 | ------ | |
| Current smoking | 1.15 (0.62–2.12) | 0.66 | ------ | |
| Children | 0.71 (0.39–1.27) | 0.25 | ------ | |
| Married | 0.63 (0.32–1.25) | 0.18 | ------ | |
| Urban City | 0.97 (0.58–1.62) | 0.92 | ------ | |
| Neutrophil to lymphocyte ratio (NLR) > 2.80 | 1.66 (1.01–2.75) | 0.048 | 1.85 (1.05–3.25) | 0.034 |
| Node positive | 1.47 (0.74–2.89) | 0.27 | 1.31 (0.63–2.71) | 0.48 |
| T ≥ 3 | 3.57 (1.52–8.40) | 0.003 | 3.45 (1.39–8.56) | 0.008 |
| Positive margin | 2.24 (1.32–3.80) | 0.003 | 1.89 (1.06–3.37) | 0.03 |
| Grade 3 | 1.10 (0.64–1.89) | 0.72 | ------ | |
| Perioperative therapy | 1.38 (0.76–2.50) | 0.29 | 1.39 (0.71–2.68) | 0.33 |
| Variables | All Patients N = 88 (%) | Adjuvant Chemoradiotherapy Group N = 67 (%) | Peri-Operative Chemotherapy Group N = 21 (%) | p Value |
|---|---|---|---|---|
| Hospital Admission | 30 (34) | 19 (29) | 11 (52) | 0.06 |
| Febrile Neutropenia | 4 (5) | 4 (6) | 0 | 0.56 |
| Neutropenia | 11 (13) | 9 (13) | 2 (10) | 1.0 |
| Other infection | 6 (7) | 6 (9) | 0 | 0.32 |
| Thrombocytopenia | 1 (1) | 1 (1) | 1 (5) | 0.42 |
| Anemia | 4 (5) | 3 (5) | 1 (5) | 1.0 |
| Nausea | 33 (37) | 26 (39) | 7 (33) | 0.79 |
| Vomiting | 22 (25) | 18 (27) | 4 (20) | 0.57 |
| Diarrhea | 28 (32) | 27 (40) | 1 (5) | 0.002 |
| Neuropathy | 5 (6) | 3 (5) | 2 (10) | 0.58 |
| Thromboembolism | 3 (3) | 2 (3) | 1 (5) | 0.56 |
| Mucositis | 31 (35) | 27 (40) | 4 (19) | 0.11 |
| Fatigue | 17 (19) | 15 (22) | 2 (10) | 0.34 |
| Rash | 6 (7) | 6 (9) | 0 | 0.32 |
| Others | 37 (42) | 29 (43) | 8 (38) | 0.80 |
| Post-operative complications | 50 (57) | 38 (57) | 12 (57) | 1.0 |
| Wound infection | 7 (8) | 6 (9) | 1 (5) | 1.0 |
| Other infection | 14 (16) | 8 (12) | 6 (29) | 0.08 |
| Leakage | 14 (16) | 9 (13) | 5 (24) | 0.30 |
| Thromboembolism | 1 (1) | 0 | 1 (5) | 0.23 |
| Cardiovascular events | 6 (7) | 4 (6) | 2 (10) | 0.62 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaidi, A.; Khan, A.; Duval, C.; Haider, K.; Ahmed, O.; Dueck, D.-A.; Brunet, B.; Gardiner, D.; Ahmed, S. Comparison of Perioperative Chemotherapy versus Postoperative Chemoradiotherapy for Operable Stomach Cancer: A Western Canadian Province Experience. Curr. Oncol. 2021, 28, 1262-1273. https://doi.org/10.3390/curroncol28020120
Zaidi A, Khan A, Duval C, Haider K, Ahmed O, Dueck D-A, Brunet B, Gardiner D, Ahmed S. Comparison of Perioperative Chemotherapy versus Postoperative Chemoradiotherapy for Operable Stomach Cancer: A Western Canadian Province Experience. Current Oncology. 2021; 28(2):1262-1273. https://doi.org/10.3390/curroncol28020120
Chicago/Turabian StyleZaidi, Adnan, Amal Khan, Claire Duval, Kamal Haider, Osama Ahmed, Dorie-Anna Dueck, Bryan Brunet, Donald Gardiner, and Shahid Ahmed. 2021. "Comparison of Perioperative Chemotherapy versus Postoperative Chemoradiotherapy for Operable Stomach Cancer: A Western Canadian Province Experience" Current Oncology 28, no. 2: 1262-1273. https://doi.org/10.3390/curroncol28020120
APA StyleZaidi, A., Khan, A., Duval, C., Haider, K., Ahmed, O., Dueck, D.-A., Brunet, B., Gardiner, D., & Ahmed, S. (2021). Comparison of Perioperative Chemotherapy versus Postoperative Chemoradiotherapy for Operable Stomach Cancer: A Western Canadian Province Experience. Current Oncology, 28(2), 1262-1273. https://doi.org/10.3390/curroncol28020120







