Deep Learning Algorithms with LIME and Similarity Distance Analysis on COVID-19 Chest X-ray Dataset
Abstract
1. Introduction
2. Related Work
3. Materials and Methods
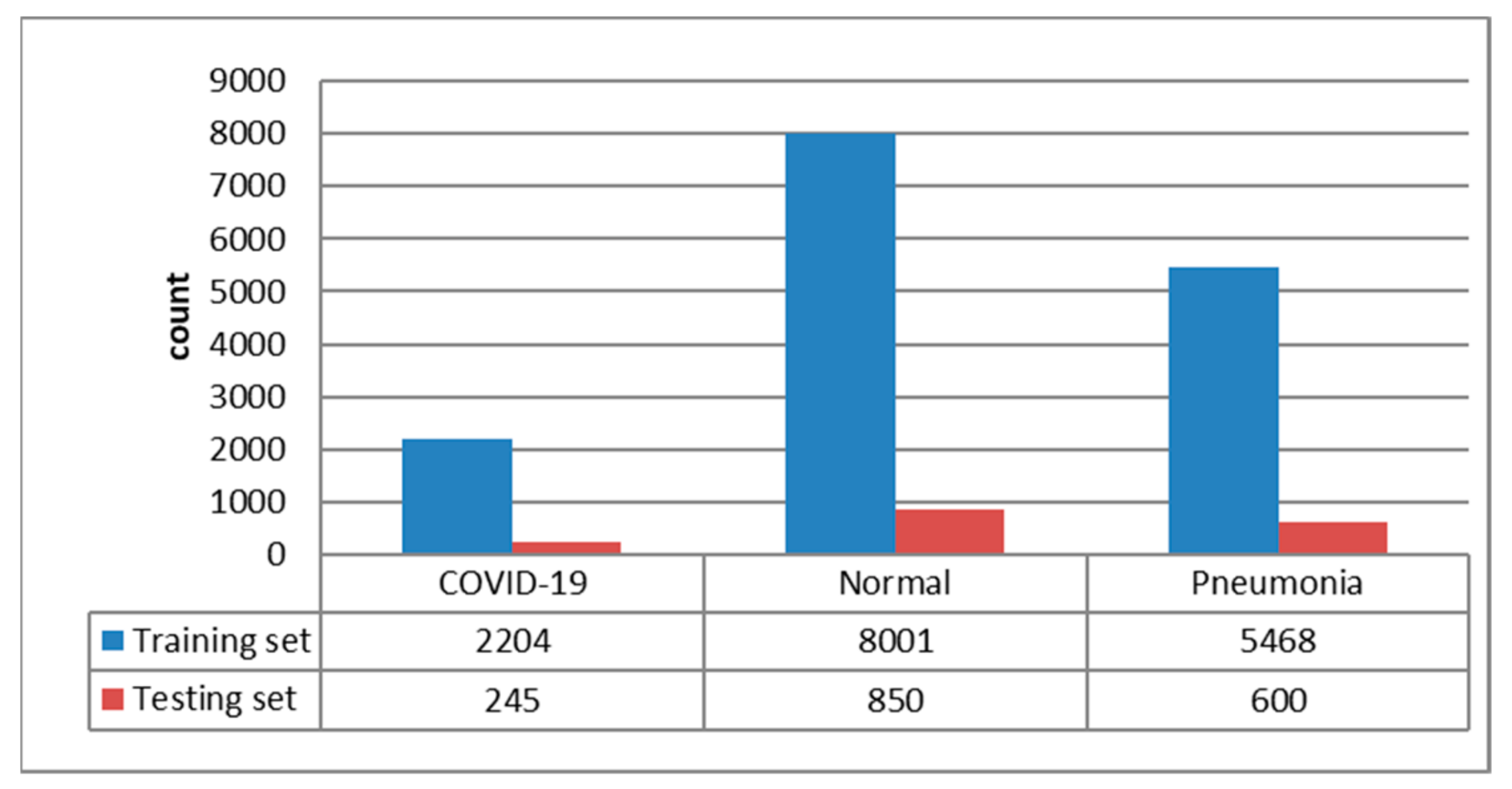
3.1. Dataset Description
3.2. Methods
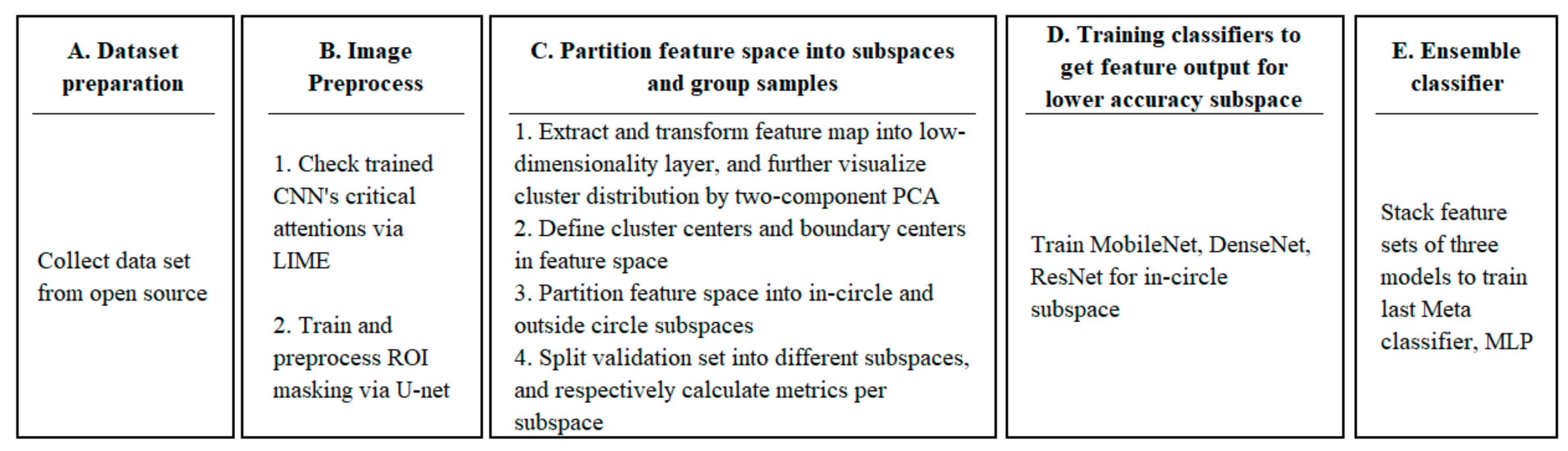
3.2.1. Model Training Phase
- Step A: Dataset preparation;
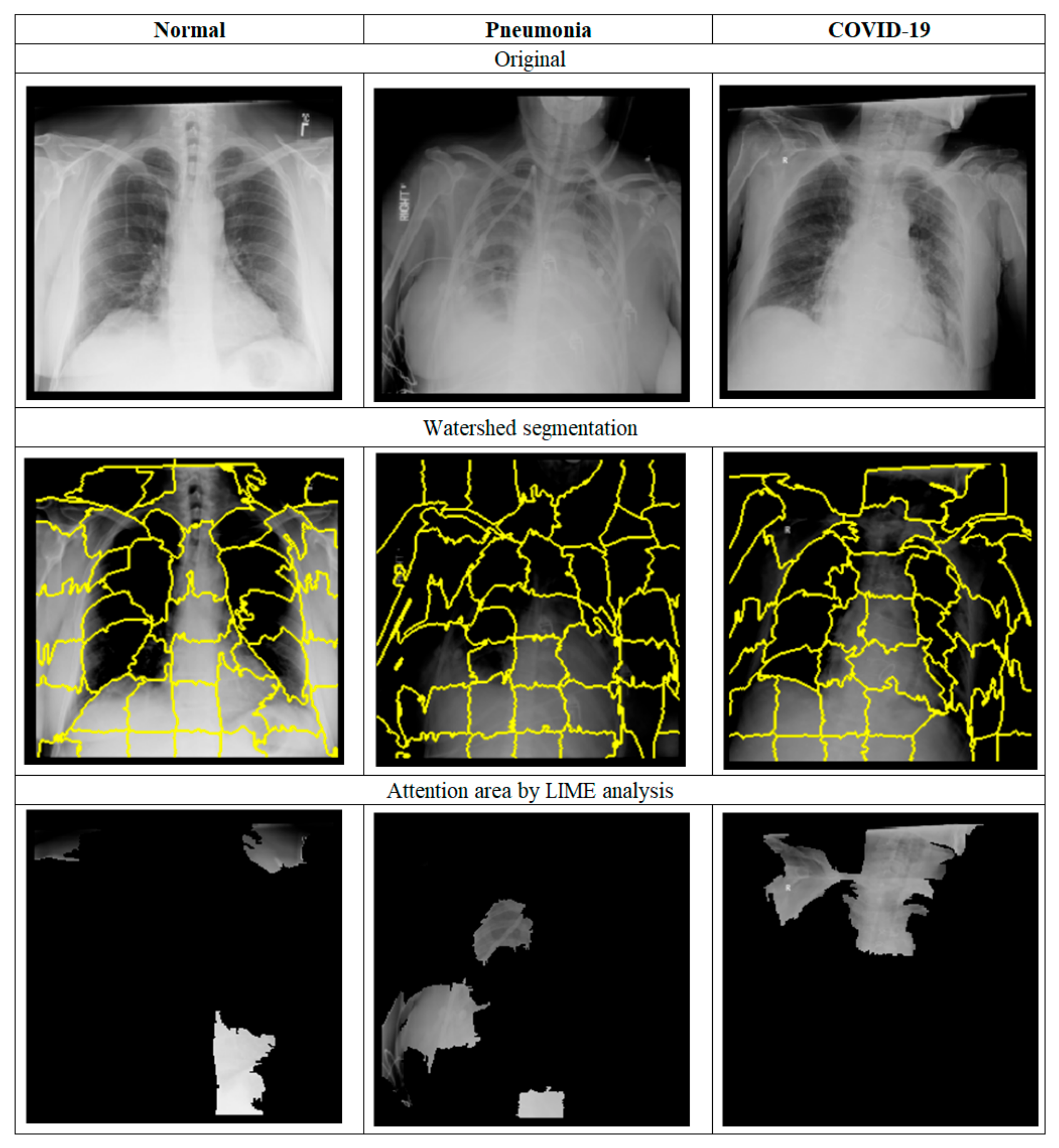
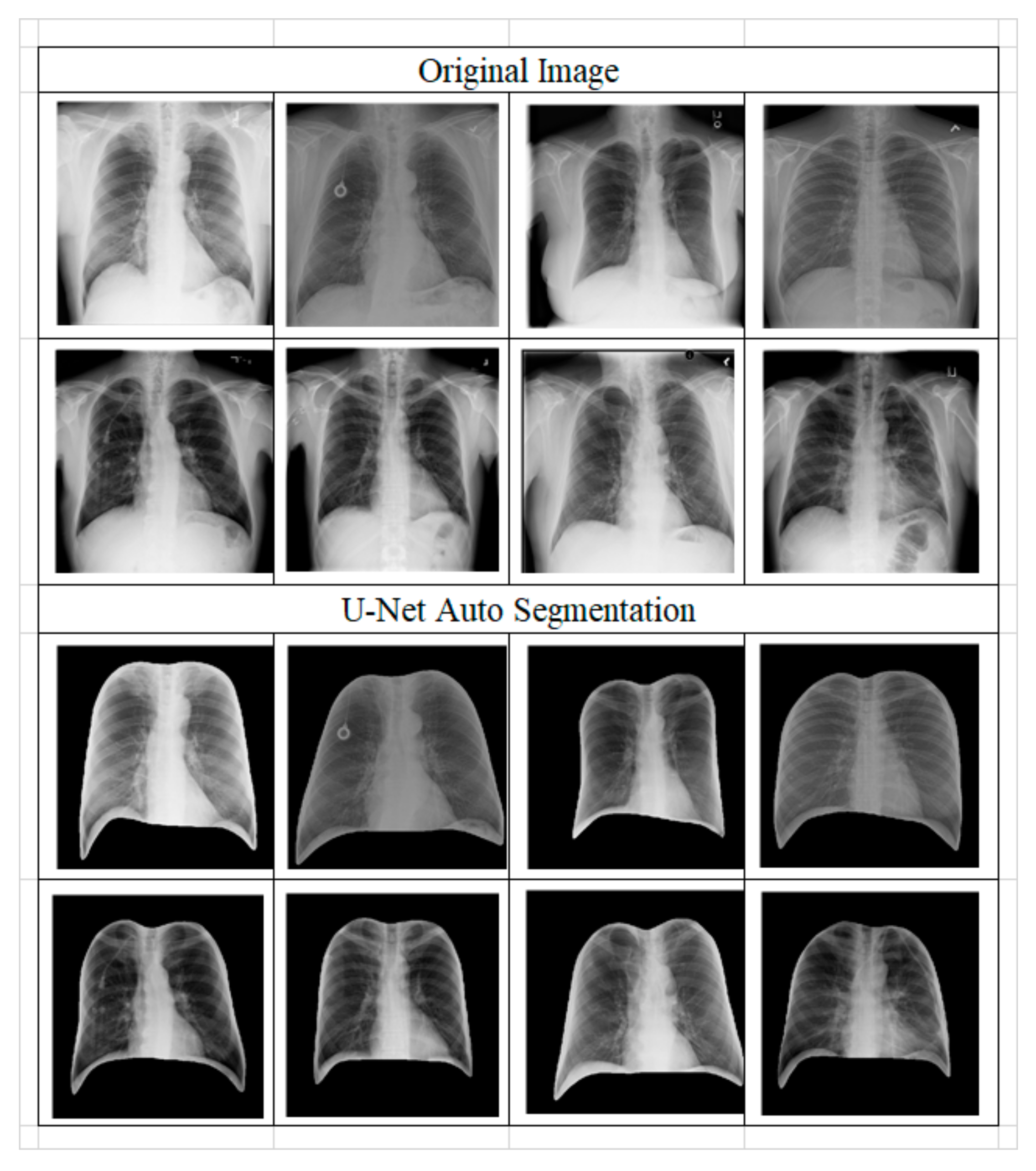
- Step B: Image preprocessing;
- Step C: Partition feature space into subspaces and group samples;
- Step D: Training classifiers to obtain feature output for lower accuracy subspace;
- Step E: Ensemble classifier.
3.2.2. Inferring Phase
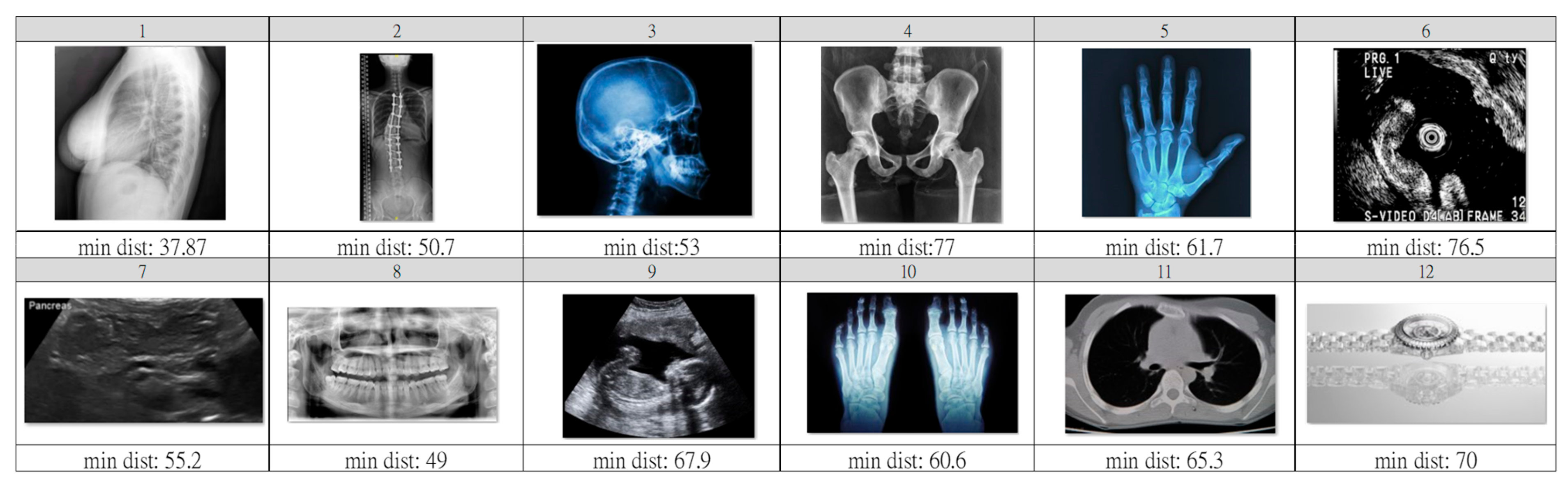
- Step A: Outlier pre-screening;
- Step B: Image Preprocess;
- Step C: Feature vector extraction and classification by a dedicated classifier.
4. Results
4.1. Model Training
4.2. Inferring
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Taiwan Centers for Disease Control. COVID-19 Introduction. Available online: https://www.cdc.gov.tw/En (accessed on 16 October 2022).
- Udugama, B.N.; Kadhiresan, P.; Kozlowski, H.N.; Malekjahani, A.; Osborne, M.; Li, V.Y.; Chen, H.; Mubareka, S.; Gubbay, J.B.; Chan, W.C. Diagnosing COVID-19: The Disease and Tools for Detection. ACS Nano 2020, 14, 3822–3835. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Zhang, H.; Xie, J.; Lin, M.; Ying, L.; Pang, P.; Ji, W. Sensitivity of Chest CT for COVID-19: Comparison to RT-PCR. Radiology 2020, 296, E115–E117. [Google Scholar] [CrossRef] [PubMed]
- Erickson, B.J.; Korfiatis, P.; Akkus, Z.; Kline, T.L. Machine Learning for Medical Imaging. Radiographics 2017, 37, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Litjens, G.J.; Kooi, T.; Bejnordi, B.E.; Setio, A.A.; Ciompi, F.; Ghafoorian, M.; Laak, J.V.; Ginneken, B.V.; Sánchez, C.I. A survey on deep learning in medical image analysis. Med. Image Anal. 2017, 42, 60–88. [Google Scholar] [CrossRef] [PubMed]
- Tajbakhsh, N.; Shin, J.Y.; Gurudu, S.R.; Hurst, R.T.; Kendall, C.B.; Gotway, M.B.; Liang, J. Convolutional Neural Networks for Medical Image Analysis: Full Training or Fine Tuning? IEEE Trans. Med. Imaging 2016, 35, 1299–1312. [Google Scholar] [CrossRef] [PubMed]
- Chouhan, V.; Singh, S.K.; Khamparia, A.; Gupta, D.; Tiwari, P.; Moreira, C.; Damaševičius, R.; Albuquerque, V.H. A Novel Transfer Learning Based Approach for Pneumonia Detection in Chest X-ray Images. Appl. Sci. 2020, 10, 559. [Google Scholar] [CrossRef]
- Gozes, O.; Frid-Adar, M.; Greenspan, H.; Browning, P.D.; Zhang, H.; Ji, W.; Bernheim, A.; Siegel, E. Rapid AI Development Cycle for the Coronavirus (COVID-19) Pandemic: Initial Results for Automated Detection & Patient Monitoring using Deep Learning CT Image Analysis. arXiv 2020, arXiv:abs/2003.05037. [Google Scholar]
- Chowdhury, M.E.; Rahman, T.; Khandakar, A.; Mazhar, R.; Kadir, M.A.; Mahbub, Z.B.; Islam, K.R.; Khan, M.S.; Iqbal, A.; Al-Emadi, N.A.; et al. Can AI Help in Screening Viral and COVID-19 Pneumonia? IEEE Access 2020, 8, 132665–132676. [Google Scholar] [CrossRef]
- Hemdan, E.E.; Shouman, M.A.; Karar, M. COVIDX-Net: A Framework of Deep Learning Classifiers to Diagnose COVID-19 in X-Ray Images. arXiv 2020, arXiv:abs/2003.11055. [Google Scholar]
- Sachin; Bhat, A. Automated Detection of COVID-19 from X-ray Images using Deep Convolutional Neural Networks. In Proceedings of the 2021 3rd International Conference on Advances in Computing, Communication Control and Networking (ICAC3N), Greater Noida, India, 17–18 December 2021; IEEE: Piscataway, NJ, USA, 2021; pp. 2076–2081. [Google Scholar]
- Wang, L.; Lin, Z.Q.; Wong, A. COVID-Net: A tailored deep convolutional neural network design for detection of COVID-19 cases from chest X-ray images. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Wang, C.; Nie, J.; Kumar, N.; Zhang, Y.; Xiong, Z.; Barnawi, A. EDL-COVID: Ensemble Deep Learning for COVID-19 Case Detection From Chest X-Ray Images. IEEE Trans. Ind. Inform. 2021, 17, 6539–6549. [Google Scholar] [CrossRef]
- Rincy, T.N.; Gupta, R. Ensemble Learning Techniques and its Efficiency in Machine Learning: A Survey. In Proceedings of the 2nd International Conference on Data, Engineering and Applications (IDEA), Bhopal, India, 28–29 February 2020; IEEE: Piscataway, NJ, USA, 2020; pp. 1–6. [Google Scholar]
- Ganaie, M.A.; Hu, M.; Tanveer, M.; Suganthan, P.N. Ensemble deep learning: A review. arXiv 2021, arXiv:abs/2104.02395. [Google Scholar] [CrossRef]
- Lin, T.; Lee, H. COVID-19 Chest Radiography Images Analysis Based on Integration of Image Preprocess, Guided Grad-CAM, Machine Learning, and Risk Management. In Proceedings of the 4th International Conference on Medical and Health Informatics, Kamakura City, Japan, 14–16 August 2020; Association for Computing Machinery: New York, NY, USA, 2020. [Google Scholar]
- Lundberg, S.M.; Lee, S. A Unified Approach to Interpreting Model Predictions. Adv. Neural Inf. Process. Syst. 2017, 30, 4765–4774. [Google Scholar]
- Zhou, B.; Khosla, A.; Lapedriza, À.; Oliva, A.; Torralba, A. Learning Deep Features for Discriminative Localization. In Proceedings of the 2016 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Las Vegas, NV, USA, 27–30 June 2016; pp. 2921–2929. [Google Scholar]
- Ribeiro, M.; Singh, S.; Guestrin, C. “Why Should I Trust You?”: Explaining the Predictions of Any Classifier. In Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining, San Francisco, CA, USA, 13–17 August 2016; Association for Computing Machinery: New York, NY, USA, 2016. [Google Scholar]
- Garreau, D.; Mardaoui, D. What does LIME really see in images? In Proceedings of the 38th International Conference on Machine Learning, Virtual Event, 18–24 July 2021. [Google Scholar]
- Ronneberger, O.; Fischer, P.; Brox, T. U-Net: Convolutional Networks for Biomedical Image Segmentation. arXiv 2015, arXiv:abs/1505.04597. [Google Scholar]
- Wolpert, D.H. Stacked generalization. Neural Netw. 1992, 5, 241–259. [Google Scholar] [CrossRef]
- Aggarwal, C.C. Outlier Analysis; Springer: New York, NY, USA, 2013. [Google Scholar]
- Cohen, J.P.; Morrison, P.; Dao, L. COVID-19 image data collection. arXiv 2020, arXiv:2003.11597. [Google Scholar]
- Chung, A. Figure 1 COVID-19 Chest X-ray Data Initiative. 2022. Available online: https://github.com/agchung/Figure1-COVID-chestxray-dataset (accessed on 29 October 2022).
- Chung, A. Actualmed COVID-19 Chest X-ray Data Initiative. 2022. Available online: https://github.com/agchung/Actualmed-COVID-chestxray-dataset (accessed on 29 October 2022).
- Qatar University. COVID-19 Radiography Database. 2022. Available online: https://www.kaggle.com/tawsifurrahman/covid19-radiography-database (accessed on 29 October 2022).
- Radiological Society of North America. RSNA Pneumonia Detection Challenge. 2022. Available online: https://www.kaggle.com/c/rsna-pneumonia-detection-challenge/data (accessed on 29 October 2022).
- Howard, A.G.; Zhu, M.; Chen, B.; Kalenichenko, D.; Wang, W.; Weyand, T.; Andreetto, M.; Adam, H. MobileNets: Efficient Convolutional Neural Networks for Mobile Vision Applications. arXiv 2017, arXiv:abs/1704.04861. [Google Scholar]
- Diamantaras, K.I.; Kung, S.Y. Principal Component Neural Networks: Theory and Applications; John Wiley: New York, NY, USA, 1996. [Google Scholar]
- He, K.; Zhang, X.; Ren, S.; Sun, J. Deep Residual Learning for Image Recognition. In 2016 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Las Vegas, NV, USA, 27–30 June 2016; pp. 770–778. [Google Scholar]
- He, K.; Zhang, X.; Ren, S.; Sun, J. Identity Mappings in Deep Residual Networks. arXiv 2016, arXiv:abs/1603.05027. [Google Scholar]
- Huang, G.; Liu, Z.; Weinberger, K.Q. Densely Connected Convolutional Networks. In 2017 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), Honolulu, HI, USA, 21–26 July 2017; pp. 2261–2269. [Google Scholar]

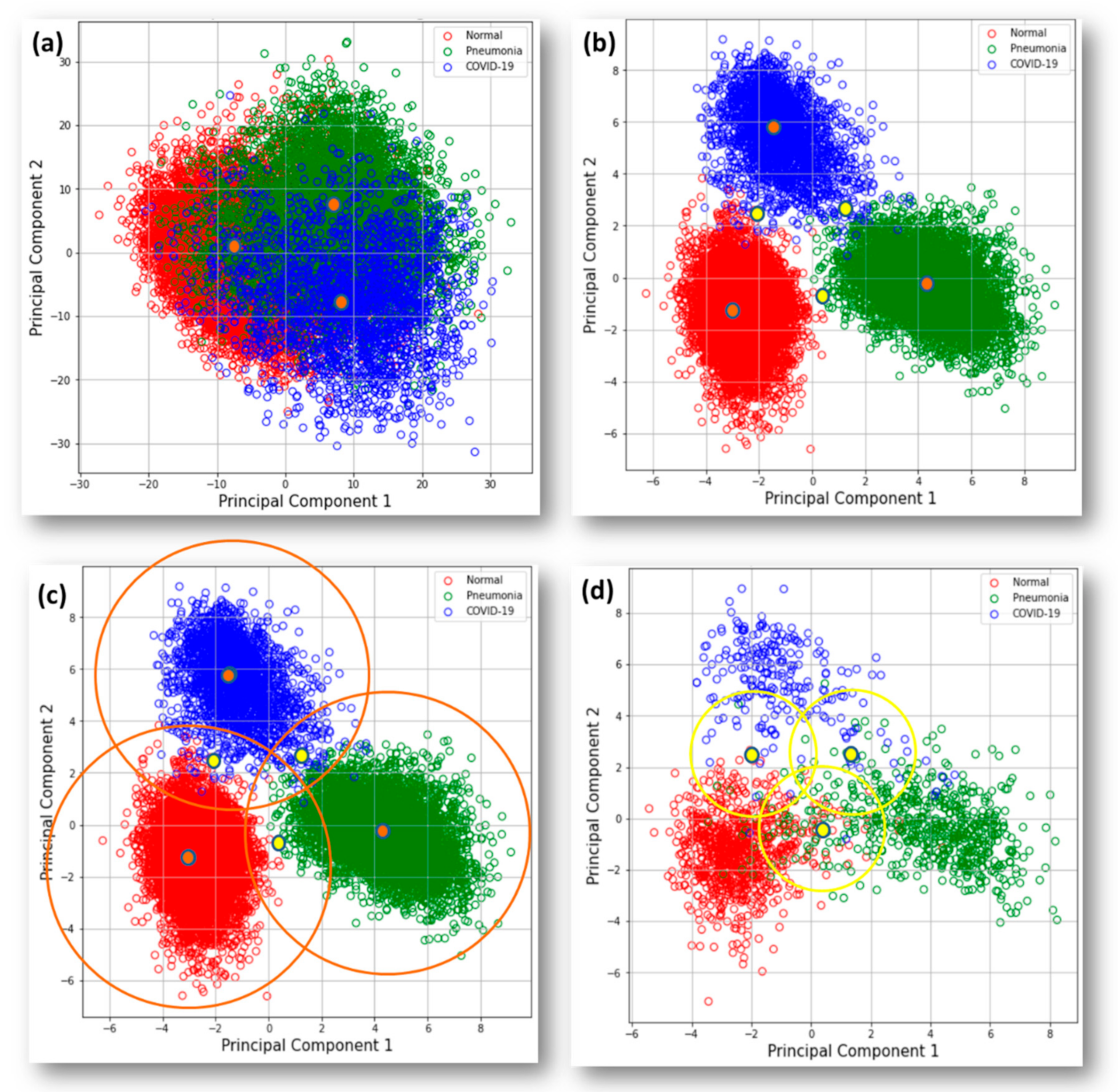
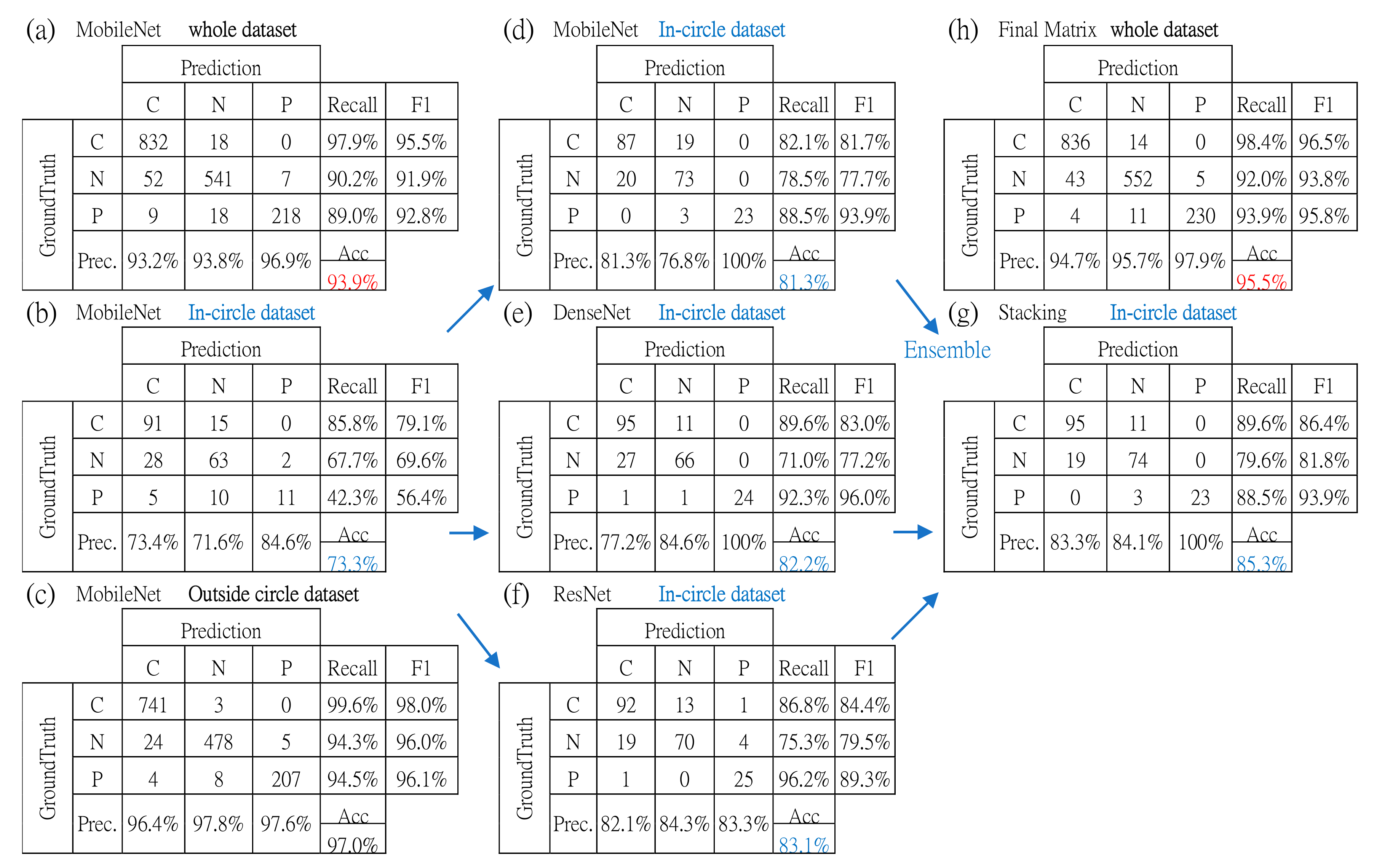
| In-Circle Testing Dataset | Image Size | Overall Accuracy | Avg. Sensitivity of All | Avg. Precision of All | Avg. F-Score of All | Sensitivity of COVID-19 |
|---|---|---|---|---|---|---|
| Individual CNN Stage | ||||||
| MobileNet | 224 × 224 | 81.3% | 83.0% | 86.1% | 84.4% | 82.1% |
| DenseNet | 224 × 224 | 82.2% | 84.3% | 87.3% | 85.4% | 89.6% |
| ResNet | 224 × 224 | 83.1% | 86.1% | 83.3% | 84.4% | 86.8% |
| Ensemble stage | ||||||
| Stacking with MLP | NA | 85.3% | 85.9% | 89.1% | 87.3% | 89.6% |
| Method | Accuracy | Sensitivity | Precision |
|---|---|---|---|
| DarkCovidNet | 88.6% | 89.0% | 94.6% |
| COVID-Net | 93.3% | 91.0% | 98.9% |
| EDL-COVID | 95.0% | 96.0% | 94.1% |
| MobileNet50V1 | 93.9% | 97.9% | 93.2% |
| Proposed-model | 95.9% | 98.4% | 94.7% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, K.-Y.; Lee, H.-C.; Lin, T.-C.; Lee, C.-Y.; Ho, Z.-P. Deep Learning Algorithms with LIME and Similarity Distance Analysis on COVID-19 Chest X-ray Dataset. Int. J. Environ. Res. Public Health 2023, 20, 4330. https://doi.org/10.3390/ijerph20054330
Chen K-Y, Lee H-C, Lin T-C, Lee C-Y, Ho Z-P. Deep Learning Algorithms with LIME and Similarity Distance Analysis on COVID-19 Chest X-ray Dataset. International Journal of Environmental Research and Public Health. 2023; 20(5):4330. https://doi.org/10.3390/ijerph20054330
Chicago/Turabian StyleChen, Kuan-Yung, Hsi-Chieh Lee, Tsung-Chieh Lin, Chih-Ying Lee, and Zih-Ping Ho. 2023. "Deep Learning Algorithms with LIME and Similarity Distance Analysis on COVID-19 Chest X-ray Dataset" International Journal of Environmental Research and Public Health 20, no. 5: 4330. https://doi.org/10.3390/ijerph20054330
APA StyleChen, K.-Y., Lee, H.-C., Lin, T.-C., Lee, C.-Y., & Ho, Z.-P. (2023). Deep Learning Algorithms with LIME and Similarity Distance Analysis on COVID-19 Chest X-ray Dataset. International Journal of Environmental Research and Public Health, 20(5), 4330. https://doi.org/10.3390/ijerph20054330







