Adherence to Cervical Cancer Screening Programs in Migrant Populations: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
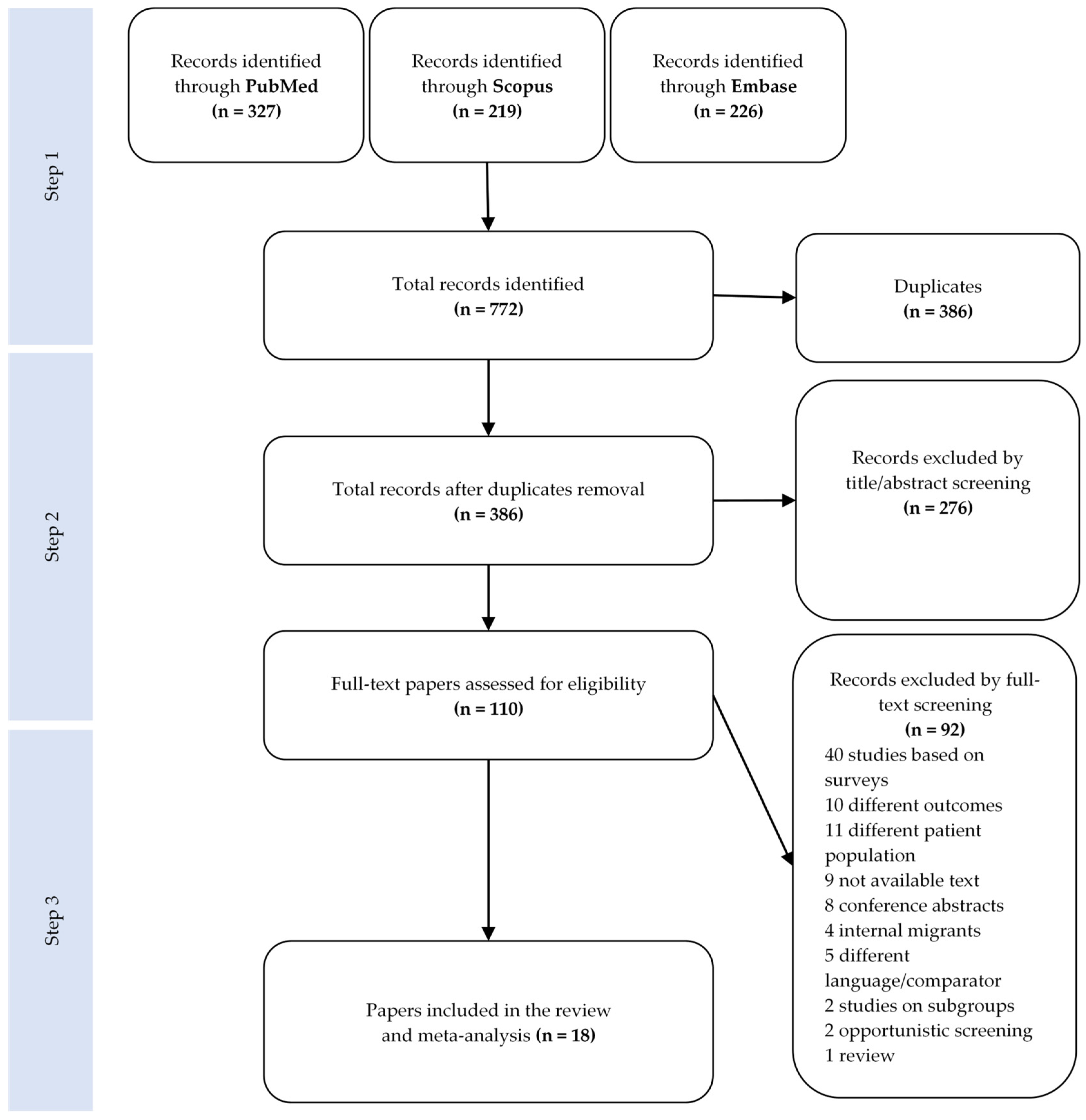
2. Materials and Methods
2.1. Literature Search
2.2. Data Extraction and Statistical Analysis
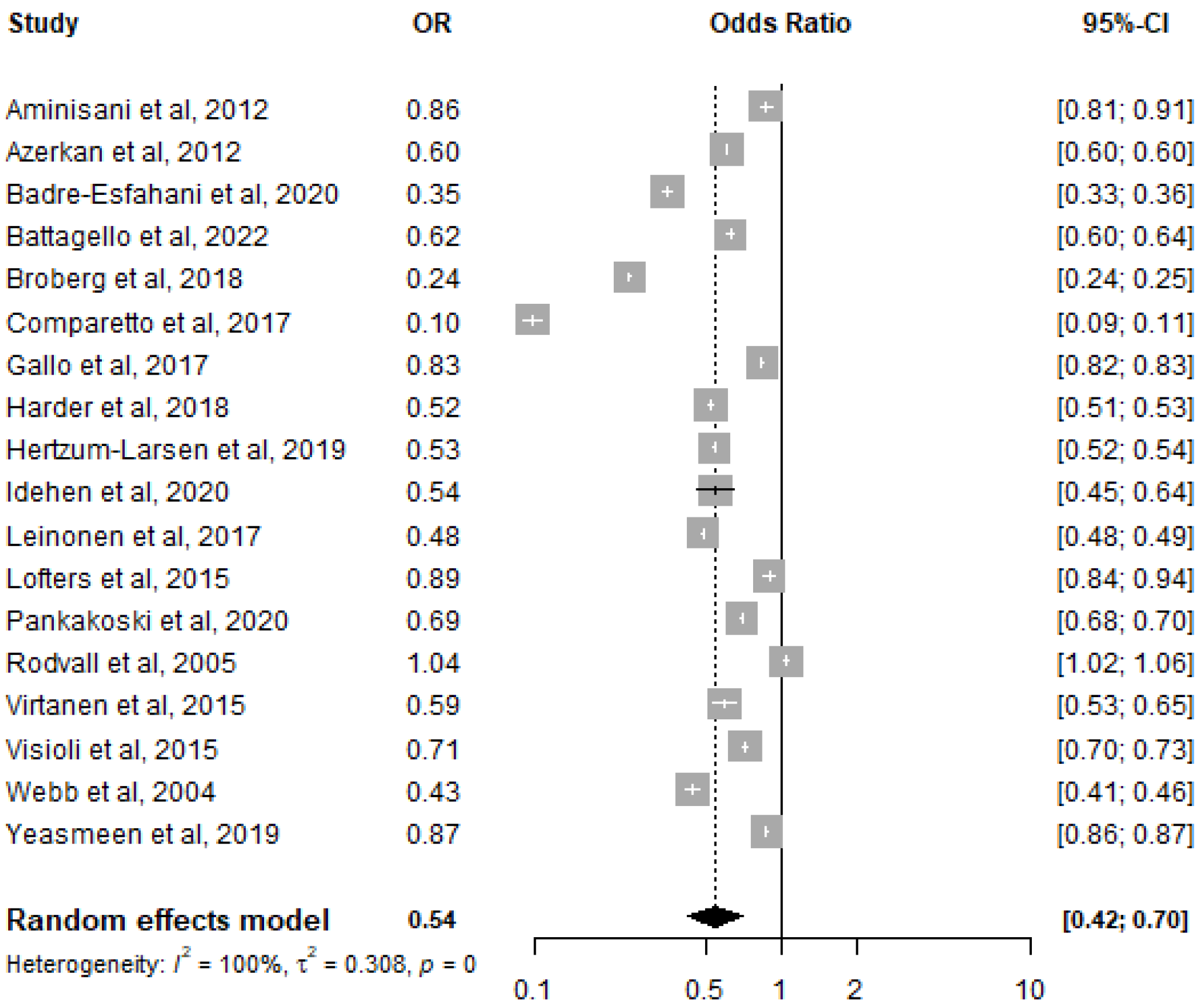
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Buskwofie, A.; David-West, G.; Clare, C.A. A Review of Cervical Cancer: Incidence and Disparities. J. Natl. Med. Assoc. 2020, 112, 229–232. [Google Scholar] [CrossRef] [PubMed]
- Olusola, P.; Banerjee, H.N.; Philley, J.V.; Dasgupta, S. Human Papilloma Virus-Associated Cervical Cancer and Health Disparities. Cells 2019, 8, 622. [Google Scholar] [CrossRef] [PubMed]
- Wardak, S. Human Papillomavirus (HPV) and Cervical Cancer. Med. Dosw. Mikrobiol. 2016, 68, 73–84. [Google Scholar] [PubMed]
- Kessler, T.A. Cervical Cancer: Prevention and Early Detection. Semin. Oncol. Nurs. 2017, 33, 172–183. [Google Scholar] [CrossRef]
- Kisling, L.A.; Das, J.M. Prevention Strategies. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Fowler, J.R.; Maani, E.V.; Jack, B.W. Cervical Cancer. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- WHO. Guideline for Screening and Treatment of Cervical Pre-Cancer Lesions for Cervical Cancer Prevention. Available online: https://www.who.int/publications/i/item/9789240030824 (accessed on 28 December 2022).
- Ferroni, E.; Camilloni, L.; Jimenez, B.; Furnari, G.; Borgia, P.; Guasticchi, G.; Giorgi Rossi, P. Methods to increase participation Working Group How to Increase Uptake in Oncologic Screening: A Systematic Review of Studies Comparing Population-Based Screening Programs and Spontaneous Access. Prev. Med. 2012, 55, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Dillner, J. Primary Human Papillomavirus Testing in Organized Cervical Screening. Curr. Opin. Obstet. Gynecol. 2013, 25, 11. [Google Scholar] [CrossRef]
- Maver, P.J.; Poljak, M. Primary HPV-Based Cervical Cancer Screening in Europe: Implementation Status, Challenges, and Future Plans. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2020, 26, 579–583. [Google Scholar] [CrossRef]
- Bruni, L.; Serrano, B.; Roura, E.; Alemany, L.; Cowan, M.; Herrero, R.; Poljak, M.; Murillo, R.; Broutet, N.; Riley, L.M.; et al. Cervical Cancer Screening Programmes and Age-Specific Coverage Estimates for 202 Countries and Territories Worldwide: A Review and Synthetic Analysis. Lancet Glob. Health 2022, 10, e1115–e1127. [Google Scholar] [CrossRef]
- Bedell, S.L.; Goldstein, L.S.; Goldstein, A.R.; Goldstein, A.T. Cervical Cancer Screening: Past, Present, and Future. Sex. Med. Rev. 2020, 8, 28–37. [Google Scholar] [CrossRef]
- Reyes-Ortiz, C.A.; Velez, L.F.; Camacho, M.E.; Ottenbacher, K.J.; Markides, K.S. Health Insurance and Cervical Cancer Screening among Older Women in Latin American and Caribbean Cities. Int. J. Epidemiol. 2008, 37, 870–878. [Google Scholar] [CrossRef][Green Version]
- Williams, J.H.; Carter, S.M.; Rychetnik, L. ‘Organised’ Cervical Screening 45 Years on: How Consistent Are Organised Screening Practices? Eur. J. Cancer 2014, 50, 3029–3038. [Google Scholar] [CrossRef] [PubMed]
- Idehen, E.E.; Koponen, P.; Härkänen, T.; Kangasniemi, M.; Pietilä, A.-M.; Korhonen, T. Disparities in Cervical Screening Participation: A Comparison of Russian, Somali and Kurdish Immigrants with the General Finnish Population. Int. J. Equity Health 2018, 17, 56. [Google Scholar] [CrossRef] [PubMed]
- Aminisani, N.; Armstrong, B.K.; Canfell, K. Participation in Cervical Screening by Older Asian and Middle Eastern Migrants in New South Wales, Australia. Health Promot. Perspect. 2012, 2, 274–286. [Google Scholar] [CrossRef] [PubMed]
- Marques, P.; Nunes, M.; da Luz Antunes, M.; Heleno, B.; Dias, S. Factors Associated with Cervical Cancer Screening Participation among Migrant Women in Europe: A Scoping Review. Int. J. Equity Health 2020, 19, 160. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Bhargava, S.; Moen, K.; Qureshi, S.A.; Hofvind, S. Mammographic Screening Attendance among Immigrant and Minority Women: A Systematic Review and Meta-Analysis. Acta Radiol. 2018, 59, 1285–1291. [Google Scholar] [CrossRef]
- Bradby, H.; Humphris, R.; Newall, D.; Phillimore, J. Definitions of Refugees, Asylum Seekers and Migrants in the Literature; WHO Regional Office for Europe: Copenhagen, Denmark, 2015. [Google Scholar]
- Reeves, M.; de Wildt, G.; Murshali, H.; Williams, P.; Gill, P.; Kralj, L.; Rushby, M. Access to Health Care for People Seeking Asylum in the UK. Br. J. Gen. Pract. J. R. Coll. Gen. Pract. 2006, 56, 306–308. [Google Scholar]
- Bradby, H.; Humphris, R.; Newall, D.; Phillimore, J. Public Health Aspects of Migrant Health: A Review of the Evidence on Health Status for Refugees and Asylum Seekers in the European Region; WHO Regional Office for Europe: Copenhagen, Denmark, 2015. [Google Scholar]
- Kindermann, D.; Zeyher, V.; Nagy, E.; Friederich, H.-C.; Bozorgmehr, K.; Nikendei, C. Predictors of Asylum Seekers’ Health Care Utilization in the Early Phase of Resettlement. Front. Psychiatry 2020, 11, 475. [Google Scholar] [CrossRef]
- Lee, S.K.; Sulaiman-Hill, C.M.R.; Thompson, S.C. Providing Health Information for Culturally and Linguistically Diverse Women: Priorities and Preferences of New Migrants and Refugees. Health Promot. J. Austr. 2013, 24, 98–103. [Google Scholar] [CrossRef]
- Lofters, A.; Vahabi, M.; Glazier, R.H. The Validity of Self-Reported Cancer Screening History and the Role of Social Disadvantage in Ontario, Canada. BMC Public Health 2015, 15, 28. [Google Scholar] [CrossRef]
- Yeh, P.T.; Kennedy, C.E.; de Vuyst, H.; Narasimhan, M. Self-Sampling for Human Papillomavirus (HPV) Testing: A Systematic Review and Meta-Analysis. BMJ Glob. Health 2019, 4, e001351. [Google Scholar] [CrossRef] [PubMed]
- Bennett, K.F.; Waller, J.; Chorley, A.J.; Ferrer, R.A.; Haddrell, J.B.; Marlow, L.A. Barriers to Cervical Screening and Interest in Self-Sampling among Women Who Actively Decline Screening. J. Med. Screen. 2018, 25, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Rebolj, M.; Sargent, A.; Njor, S.H.; Cuschieri, K. Widening the Offer of Human Papillomavirus Self-Sampling to All Women Eligible for Cervical Screening: Make Haste Slowly. Int. J. Cancer, 2022; Epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.T.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring Inconsistency in Meta-Analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef]
- Pacelli, B.; Zengarini, N.; Broccoli, S.; Caranci, N.; Spadea, T.; Di Girolamo, C.; Cacciani, L.; Petrelli, A.; Ballotari, P.; Cestari, L.; et al. Differences in Mortality by Immigrant Status in Italy. Results of the Italian Network of Longitudinal Metropolitan Studies. Eur. J. Epidemiol. 2016, 31, 691–701. [Google Scholar] [CrossRef] [PubMed]
- Leinonen, M.K.; Campbell, S.; Klungsøyr, O.; Lönnberg, S.; Hansen, B.T.; Nygård, M. Personal and Provider Level Factors Influence Participation to Cervical Cancer Screening: A Retrospective Register-Based Study of 1.3 Million Women in Norway. Prev. Med. 2017, 94, 31–39. [Google Scholar] [CrossRef]
- Badre-Esfahani, S.; Larsen, M.B.; Seibæk, L.; Petersen, L.K.; Blaakær, J.; Støvring, H.; Andersen, B. Non-Adherence To Childhood HPV Vaccination Is Associated With Non-Participation In Cervical Cancer Screening—A Nationwide Danish Register-Based Cohort Study. Clin. Epidemiol. 2019, 11, 969–980. [Google Scholar] [CrossRef]
- Azerkan, F.; Sparén, P.; Sandin, S.; Tillgren, P.; Faxelid, E.; Zendehdel, K. Cervical Screening Participation and Risk among Swedish-Born and Immigrant Women in Sweden. Int. J. Cancer 2012, 130, 937–947. [Google Scholar] [CrossRef]
- Badre-Esfahani, S.; Larsen, M.B.; Seibæk, L.; Petersen, L.K.; Blaakær, J.; Andersen, B. Low Attendance by Non-Native Women to Human Papillomavirus Vaccination and Cervical Cancer Screening—A Danish Nationwide Register-Based Cohort Study. Prev. Med. Rep. 2020, 19, 101106. [Google Scholar] [CrossRef]
- Battagello, J.; Monetti, D.; Rizzato, S.; Rosano, A.; Stocco, C.F.; Zamberlan, S.; Rugge, M.; Zorzi, M. Young Immigrant Women and Cervical Cancer Screening: Participation and Lesions Detected at the First Screening Round. Epidemiol. Prev. 2022, 46, 173–180. [Google Scholar] [CrossRef]
- Broberg, G.; Wang, J.; Östberg, A.-L.; Adolfsson, A.; Nemes, S.; Sparén, P.; Strander, B. Socio-Economic and Demographic Determinants Affecting Participation in the Swedish Cervical Screening Program: A Population-Based Case-Control Study. PLoS ONE 2018, 13, e0190171. [Google Scholar] [CrossRef] [PubMed]
- Comparetto, C.; Epifani, C.; Manca, M.C.; Lachheb, A.; Bravi, S.; Cipriani, F.; Bellomo, F.; Olivieri, S.; Fiaschi, C.; Di Marco, L.; et al. Uptake of Cervical Cancer Screening among the Migrant Population of Prato Province, Italy. Int. J. Gynaecol. Obstet. Off. Organ Int. Fed. Gynaecol. Obstet. 2017, 136, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Gallo, F.; Caprioglio, A.; Castagno, R.; Ronco, G.; Segnan, N.; Giordano, L. Inequalities in Cervical Cancer Screening Utilisation and Results: A Comparison between Italian Natives and Immigrants from Disadvantaged Countries. Health Policy 2017, 121, 1072–1078. [Google Scholar] [CrossRef] [PubMed]
- Harder, E.; Juul, K.E.; Jensen, S.M.; Thomsen, L.T.; Frederiksen, K.; Kjaer, S.K. Factors Associated with Non-Participation in Cervical Cancer Screening—A Nationwide Study of Nearly Half a Million Women in Denmark. Prev. Med. 2018, 111, 94–100. [Google Scholar] [CrossRef]
- Hertzum-Larsen, R.; Kjær, S.K.; Frederiksen, K.; Thomsen, L.T. Participation in Cervical Cancer Screening among Immigrants and Danish-Born Women in Denmark. Prev. Med. 2019, 123, 55–64. [Google Scholar] [CrossRef]
- Idehen, E.E.; Virtanen, A.; Lilja, E.; Tuomainen, T.-P.; Korhonen, T.; Koponen, P. Cervical Cancer Screening Participation among Women of Russian, Somali, and Kurdish Origin Compared with the General Finnish Population: A Register-Based Study. Int. J. Environ. Res. Public. Health 2020, 17, 7899. [Google Scholar] [CrossRef]
- Leinonen, M.K.; Campbell, S.; Ursin, G.; Tropé, A.; Nygård, M. Barriers to Cervical Cancer Screening Faced by Immigrants: A Registry-Based Study of 1.4 Million Women in Norway. Eur. J. Public Health 2017, 27, 873–879. [Google Scholar] [CrossRef]
- Pankakoski, M.; Heinävaara, S.; Anttila, A.; Sarkeala, T. Differences in Cervical Test Coverage by Age, Socioeconomic Status, Ethnic Origin and Municipality Type—A Nationwide Register-Based Study. Prev. Med. 2020, 139, 106219. [Google Scholar] [CrossRef]
- Rodvall, Y.; Kemetli, L.; Tishelman, C.; Törnberg, S. Factors Related to Participation in a Cervical Cancer Screening Programme in Urban Sweden. Eur. J. Cancer Prev. Off. J. Eur. Cancer Prev. Organ. 2005, 14, 459–466. [Google Scholar] [CrossRef]
- Virtanen, A.; Anttila, A.; Luostarinen, T.; Malila, N.; Nieminen, P. Improving Cervical Cancer Screening Attendance in Finland. Int. J. Cancer 2015, 136, E677–E684. [Google Scholar] [CrossRef]
- Visioli, C.B.; Crocetti, E.; Zappa, M.; Iossa, A.; Andersson, K.L.; Bulgaresi, P.; Alfieri, A.; Amunni, G. Participation and Risk of High Grade Cytological Lesions among Immigrants and Italian-Born Women in an Organized Cervical Cancer Screening Program in Central Italy. J. Immigr. Minor. Health 2015, 17, 670–678. [Google Scholar] [CrossRef] [PubMed]
- Webb, R.; Richardson, J.; Esmail, A.; Pickles, A. Uptake for Cervical Screening by Ethnicity and Place-of-Birth: A Population-Based Cross-Sectional Study. J. Public Health Oxf. Engl. 2004, 26, 293–296. [Google Scholar] [CrossRef]
- Yeasmeen, T.; Kelaher, M.; Brotherton, J.M.L.; Malloy, M.J. Understanding the Participation in Cervical Screening of Muslim Women in Victoria, Australia from Record-Linkage Data. J. Cancer Policy 2019, 22, 100201. [Google Scholar] [CrossRef]
- Ferdous, M.; Lee, S.; Goopy, S.; Yang, H.; Rumana, N.; Abedin, T.; Turin, T.C. Barriers to Cervical Cancer Screening Faced by Immigrant Women in Canada: A Systematic Scoping Review. BMC Womens Health 2018, 18, 165. [Google Scholar] [CrossRef] [PubMed]
- Access To Primary Care And Preventative Health Services Of Migrants—Rosano Aldo (Curatore). Libro Springer 05/2018—HOEPLI.it. Available online: https://www.hoepli.it/libro/access-to-primary-care-and-preventative-health-services-of-migrants/9783319736297.html (accessed on 28 December 2022).
- Waller, J.; McCaffery, K.; Wardle, J. Beliefs about the Risk Factors for Cervical Cancer in a British Population Sample. Prev. Med. 2004, 38, 745–753. [Google Scholar] [CrossRef]
- Damiani, G.; Federico, B.; Basso, D.; Ronconi, A.; Bianchi, C.B.N.A.; Anzellotti, G.M.; Nasi, G.; Sassi, F.; Ricciardi, W. Socioeconomic Disparities in the Uptake of Breast and Cervical Cancer Screening in Italy: A Cross Sectional Study. BMC Public Health 2012, 12, 99. [Google Scholar] [CrossRef]
- Zhang, W.; Gao, K.; Fowkes, F.J.I.; Adeloye, D.; Rudan, I.; Song, P.; Jin, M.; Chen, K. Associated Factors and Global Adherence of Cervical Cancer Screening in 2019: A Systematic Analysis and Modelling Study. Glob. Health 2022, 18, 101. [Google Scholar] [CrossRef]
- Walsh, B.; Silles, M.; O’Neill, C. The Importance of Socio-Economic Variables in Cancer Screening Participation: A Comparison between Population-Based and Opportunistic Screening in the EU-15. Health Policy 2011, 101, 269–276. [Google Scholar] [CrossRef]
- Palència, L.; Espelt, A.; Rodríguez-Sanz, M.; Puigpinós, R.; Pons-Vigués, M.; Pasarín, M.I.; Spadea, T.; Kunst, A.E.; Borrell, C. Socio-Economic Inequalities in Breast and Cervical Cancer Screening Practices in Europe: Influence of the Type of Screening Program. Int. J. Epidemiol. 2010, 39, 757–765. [Google Scholar] [CrossRef]
- Marlow, L.A.V.; Waller, J.; Wardle, J. Barriers to Cervical Cancer Screening among Ethnic Minority Women: A Qualitative Study. J. Fam. Plann. Reprod. Health Care 2015, 41, 248–254. [Google Scholar] [CrossRef]
- Moser, K.; Patnick, J.; Beral, V. Inequalities in Reported Use of Breast and Cervical Screening in Great Britain: Analysis of Cross Sectional Survey Data. BMJ 2009, 338, b2025. [Google Scholar] [CrossRef] [PubMed]
- Morrison, T.B.; Flynn, P.M.; Weaver, A.L.; Wieland, M.L. Cervical Cancer Screening Adherence among Somali Immigrants and Refugees to the United States. Health Care Women Int. 2013, 34, 980–988. [Google Scholar] [CrossRef] [PubMed]
- Rechel, B.; Mladovsky, P.; Ingleby, D.; Mackenbach, J.P.; McKee, M. Migration and Health in an Increasingly Diverse Europe. Lancet Lond. Engl. 2013, 381, 1235–1245. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.; Kiss, N.; Łaszewska, A.; Mayer, S. Public Health Aspects of Migrant Health: A Review of the Evidence on Health Status for Labour Migrants in the European Region; WHO Health Evidence Network Synthesis Reports; WHO Regional Office for Europe: Copenhagen, Denmark, 2015; ISBN 9789289051095. [Google Scholar]
- Dunn, S.F.; Lofters, A.K.; Ginsburg, O.M.; Meaney, C.A.; Ahmad, F.; Moravac, M.C.; Nguyen, C.T.J.; Arisz, A.M. Cervical and Breast Cancer Screening After CARES: A Community Program for Immigrant and Marginalized Women. Am. J. Prev. Med. 2017, 52, 589–597. [Google Scholar] [CrossRef]
- Johnson, C.E.; Mues, K.E.; Mayne, S.L.; Kiblawi, A.N. Cervical Cancer Screening Among Immigrants and Ethnic Minorities: A Systematic Review Using the Health Belief Model. J. Low. Genit. Tract Dis. 2008, 12, 232. [Google Scholar] [CrossRef]
- Giorgi Rossi, P.; Baldacchini, F.; Ronco, G. The Possible Effects on Socio-Economic Inequalities of Introducing HPV Testing as Primary Test in Cervical Cancer Screening Programs. Front. Oncol. 2014, 4, 20. [Google Scholar] [CrossRef]
- Vahabi, M.; Lofters, A. Muslim Immigrant Women’s Views on Cervical Cancer Screening and HPV Self-Sampling in Ontario, Canada. BMC Public Health 2016, 16, 868. [Google Scholar] [CrossRef]
- Petrelli, A.; Di Napoli, A.; Agabiti, N.; Barbieri, G.; Bardin, A.; Bargagli, A.M.; Biggeri, A.; Bonvicini, L.; Broccoli, S.; Cacciani, L.; et al. Immigrants’ health and socioeconomic inequalities of overall population residing in Italy evaluated through the Italian network of Longitudinal Metropolitan Studies. Epidemiol. Prev. 2019, 43, 1–80. [Google Scholar] [CrossRef]
- Shewamene, Z.; Dune, T.; Smith, C.A. Acculturation and Use of Traditional Medicine among African Migrant Women in Sydney: A Mixed Method Study. BMC Complement. Med. Ther. 2021, 21, 249. [Google Scholar] [CrossRef]
- Moxham, R.; Moylan, P.; Duniec, L.; Fisher, T.; Furestad, E.; Manolas, P.; Scott, N.; Oam, D.K.; Finlay, S. Knowledge, Attitudes, Beliefs, Intentions and Behaviours of Australian Indigenous Women from NSW in Response to the National Cervical Screening Program Changes: A Qualitative Study. Lancet Reg. Health – West. Pac. 2021, 13, 100195. [Google Scholar] [CrossRef]
- Møen, K.A.; Kumar, B.; Qureshi, S.; Diaz, E. Differences in Cervical Cancer Screening between Immigrants and Nonimmigrants in Norway: A Primary Healthcare Register-Based Study. Eur. J. Cancer Prev. Off. J. Eur. Cancer Prev. Organ. 2017, 26, 521–527. [Google Scholar] [CrossRef] [PubMed]
- Harcourt, N.; Ghebre, R.G.; Whembolua, G.-L.; Zhang, Y.; Osman, S.W.; Okuyemi, K.S. Factors Associated with Breast and Cervical Cancer Screening Behavior among African Immigrant Women in Minnesota. J. Immigr. Minor. Health Cent. Minor. Public Health 2014, 16, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Lofters, A.K.; Moineddin, R.; Hwang, S.W.; Glazier, R.H. Predictors of Low Cervical Cancer Screening among Immigrant Women in Ontario, Canada. BMC Womens Health 2011, 11, 20. [Google Scholar] [CrossRef] [PubMed]
- Malmusi, D.; Borrell, C.; Benach, J. Migration-Related Health Inequalities: Showing the Complex Interactions between Gender, Social Class and Place of Origin. Soc. Sci. Med. 1982 2010, 71, 1610–1619. [Google Scholar] [CrossRef] [PubMed]

| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Original papers published up to July 2022 English language | Reviews, conference abstracts, commentaries, editorials, letters to Editor, and pilot studies Languages different from English |
| Subjects: female migrants aged 18+ and native controls (age range for screening invitation may vary in different countries) | Subjects: selected groups of participants (patients at high risk of cervical cancer, asylum seekers/refugees and internal migrants) |
| Outcome: measure of participation in organized cervical cancer screening (both Pap smear and HPV DNA test are included) Data source: population-based data (national registries and databases) | Outcome: participation in cervical cancer screening through self-sampling strategies or opportunistic screening Data source: surveys and questionnaires (self-reported measures) |
| Reference | Location | Screening Program Characteristics | Data Sources | Definition of Adherent Subject | Exclusion Criteria | Definition of Migrants | Characteristics of the Sample |
|---|---|---|---|---|---|---|---|
| Aminisani et al., 2012 [16] | Australia | Age: 18–69 years Time interval: 2 years | NSW Pap Test Register, NSW Midwives Data Collection | Pap test register screening record in the calendar/fiscal year 2001–2002, and screening performed within 2–3 years of giving birth | Women died after giving birth/during the follow-up period, abnormal cervical test, and unsatisfactory result in the 5 years preceding the study period | Country of birth | Migrants: women aged 20–54 years giving birth between 1 January and 31 December 2000; natives: women matched by birth in the same period, 5-year age group and residence area |
| Azerkan et al., 2012 [33] | Sweden | Age: 23–50 years Time interval: 3 years Age: 51–60 Time interval: 5 years | National Cancer Screening Register, and Swedish Total Population Register | Women aged 23–50 years were considered participants for 3 years from the last test, and women aged 51–60 years for 5 years after the last test | Missing information, emigration out of Sweden, death, and carcinoma in situ/invasive cancer before entry to the cohort | Country of birth | Migrants and native women with data from 1993 to 2005 |
| Badre-Esfahani et al., 2020 [34] | Denmark | Age: 23–49 years Time interval: 3 years Age: 50–64 years Time interval: 5 years | Danish Civil Registration System, and Danish Pathology Register | Women with at least one registered cytology sample between the age of 22.5 and 24 years | Women with cervical cytology obtained before the age of 22.5 years, history of surgical removal of cervix, and diagnosis of cervical cancer | Country of origin | Migrants: women born during the period of 1985–1993 outside Denmark or with two immigrant parents. Natives: women born in Denmark in the same period |
| Battagello et al., 2022 [35] | Italy | Age: 25–29 years Time interval: 3 years Age: 30–64 years Time interval: 5 years | Cervical cancer screening databases from Local Health Units | Women screened after invitation | Women with spontaneous HPV vaccination and Western country citizenship, and undelivered invitations | Country of origin/ citizenship | Foreign and Italian women residing in the study area born between 1986 and 1992 and invited for the 1st time between 2011 and 2017 |
| Broberg et al., 2018 [36] | Sweden | Age: 23–50 years Time interval: 3 years Age: 51–60 Time interval: 5 years | Swedish Total Population Register, Swedish National Cervical Screening Registry, and Statistics Sweden | Women who attended a screening program within 90 days of invitation between 1 January 2012 and 31 December 2012 | Women migrated during the study period, with total hysterectomy, without regular invitation in 2012, and with regular invitation in 2012 but not participating within 90 days | Country of birth | Immigrant and Swedish women between 30–60 years of age on 31 December 2012 |
| Comparetto et al., 2017 [37] | Italy | Age: 25–64 years Time interval: 3 years | Archives of LHU Serviceable Registry of Prato Province, and Cancer Registry of Tuscany | Woman respondents to the invitation | Women who did not receive the invitation letter, underwent a hysterectomy since the last test, and had a cervical test in the 12 months before the invitation | Citizenship | Residents of Prato with at least 1 invitation between 1 July 2004 and 30 June 2007 |
| Gallo et al., 2017 [38] | Italy | Age: 25–64 years Time interval: 3 years | Screening program archives | Attendees at first appointment or at recall 1 month later | Not available | Country of birth/ citizenship | Women resident in Piedmont who received at least one invitation during the period of 2001–2013 |
| Harder et al., 2018 [39] | Denmark | Age: 23–49 years Time interval: 3 years Age: 50–64 years Time interval: 5 years | Pathology Databank, National Patient Register, Cancer Register, Medical Birth Register, Prescription and Psychiatric Research Register, and Statistics Denmark | Women with a cervical cytology registered within the 4-year follow-up period | Women with missing information, with hysterectomy registered before baseline or in follow-up, and emigrated or died during follow-up | Country of origin | Migrant and native women invited for routine cervical cancer screening in 2008–2009 |
| Hertzum-Larsen et al., 2019 [40] | Denmark | Age: 23–49 years Time interval: 3 years Age: 50–64 years Time interval: 5 years | Civil Registration System, Pathology Databank, National Patient Register, National Health Service Register, Medical Birth Register, Psychiatric Central Register, and Employment Register | Cervical cytology registered in the Pathology Databank during follow-up | Women born in Denmark to immigrant parents, were not residing in Denmark, were unsubscribed or fully hysterectomized, were pregnant, had missing data, and had not resided continuously in Denmark during the study period | Country of origin | Migrant and native women invited to the screening program between 2008 and 2009 |
| Idehen et al., 2020 [41] | Finland | Age: 30–60 years Time interval: 5 years | Finnish National Population Registry, Mass Screening Registry, Care Register, Medical Birth Register, Register of Induced Abortions, Statistics Finland, and Social Insurance Institution of Finland | Positive response to invitation in 2008–2012 | Males, women aged < 30 years, and not invited women | Country of origin | Migrant and native women invited to the organized cervical screening program during the period of 2008–2012 |
| Leinonen et al., 2017 [42] | Norway | Age: 25–69 years Time interval: 3 years | Norwegian cervical cancer screening program system | Screening test recorded in 2008–2012 | Incomplete screening history, women with missing data, women who had opted out from the program, women with previous diagnosis of gynecological cancer, and women under surveillance for cervical abnormalities | Country of origin | Migrant and native women alive and were a resident in Norway on 31 December 2012 |
| Lofters et al., 2015 [25] | Canada | Age: 25–69 years Time interval: 3 years | Cytobase (Pap test registry) | Record of screening in the 3 years before study period | Women not eligible for health insurance coverage, hysterectomized, and with previous diagnosis of cervical cancer | Country of birth | Migrant and native women eligible for cervical screening from 2000 to 2007 |
| Pankakoski et al., 2020 [43] | Finland | Age: 30–60 years (up to 25–65) | Mass Screening Registry, Population Registry, and Statistics Finland | Women who attended organized screening in the 5-year interval studied | Women with no information on socioeconomic status, mother tongue or home municipalities | Mother tongue | Migrant and native women born in 1950–1984 and residing in Finland in 2010–2014 |
| Rodvall et al., 2005 [44] | Sweden | Age: 25–40 years Time interval: 3 years Age: 41–59 years Time interval: 4 years | Dataset of invited women, and National Longitudinal Population Database | Women having taken a smear within the program one year of receiving the invitation | Women not invited due to a recent smear, including those taken as an opportunistic screening test | Country of birth | Migrant and native women invited to the screening program between 1994 and 1996 |
| Virtanen et al., 2015 [45] | Finland | Age: 30–60 years (up to 25–65) | Mass Screening Registry, and Statistics Finland | Women who attended screening after receiving an invitation letter in 2011–2012 | Not invited women, women with missing information, emigrated, dead, and moved to other municipalities during the study | Mother tongue | Migrant and native women invited to screening between 2011 and 2012 |
| Visioli et al., 2015 [46] | Italy | Age: 25–69 years Time interval: 3 years | Archive of invitations to the screening and archive of the Research Institute laboratory | Pap test performed within one year from the date of invitation | Missing information on country of birth, and undelivered invitation letters | Country of birth | Migrants and Italians who were residents in the Florence district invited to screening between 2000 and 2008 |
| Webb et al., 2004 [47] | United Kingdom | Age: 25–49 years Time interval: 3 years Age: 50–64 Time interval: 5 years | Manchester Health Authority, and National Database of Primary Care Trusts | Women screened in the last 5 years | Absence of cervix | Country of birth | All eligible migrant and native women aged 30–64 years |
| Yeasmeen et al., 2019 [48] | Australia | Age: 25–74 years Time interval: 3 years | Victorian Cervical Cytology Registry, and Victorian Admitted Episodes Dataset | Women identified in both selected datasets was defined as a screening participant | Women who previously underwent hysterectomy or who died prior to the period of interest | Country of birth | Migrant and native women aged 15 years or over in the period from 1 January 2000 to 31 December 2013 |
| Study | N Migrant Women | N Screened Migrant Women (%) | N Natives | N Screened Native Women (%) |
|---|---|---|---|---|
| Aminisani et al., 2012 [16] | 11,477 | 6879 (59.9%) | 10,762 | 6834 (63.5%) |
| Azerkan et al., 2012 [33] | 445,547 | 220,247 (49.4%) | 2,176,255 | 1,349,278 (62.0%) |
| Badre-Esfahani et al., 2020 [34] | 18,273 | 4965 (27.2%) | 151,885 | 78,903 (51.9%) |
| Battagello et al., 2022 [35] | 27,958 | 10,443 (37.4%) | 96,105 | 47,069 (49.0%) |
| Broberg et al., 2018 [36] | 178,917 | 46,317 (25.9%) | 369,574 | 218,036 (59.0%) |
| Comparetto et al., 2017 [37] | 4992 | 555 (11.1%) | 40,688 | 22,728 (55.9%) |
| Gallo et al., 2017 [38] | 500,381 | 220,155 (44.0%) | 4,017,764 | 1,955,373 (48.7%) |
| Harder et al., 2018 [39] | 48,218 | 36,357 (75.4%) | 428,452 | 366,627 (85.6%) |
| Hertzum-Larsen et al., 2019 [40] | 57,329 | 34,844 (60.8%) | 553,578 | 411,898 (74.4%) |
| Idehen et al., 2020 [41] | 1790 | 926 (51.7%) | 789 | 525 (66.5%) |
| Leinonen et al., 2017 [42] | 208,626 | 106,399 (51.0%) | 1,157,223 | 791,228 (68.4%) |
| Lofters et al., 2015 [25] | 7737 | 5370 (69.4%) | 31,268 | 22,482 (71.9%) |
| Pankakoski et al., 2020 [43] | 129,049 | 78,658 (61.0%) | 1,098,410 | 762,296 (69.4%) |
| Rodvall et al., 2005 [44] | 67,581 | 38,071 (56.3%) | 239,971 | 132,872 (55.4%) |
| Virtanen et al., 2015 [45] | 1818 | 1283 (70.6%) | 29,009 | 23,317 (80.4%) |
| Visioli et al., 2015 [46] | 52,281 | 20,094 (38.4%) | 488,498 | 228,129 (46.7%) |
| Webb et al., 2004 [47] | 8921 | 5120 (57.4%) | 15,937 | 12,048 (75.6%) |
| Yeasmeen et al., 2019 [48] | 499,967 | 171,865 (34.4%) | 1,298,494 | 489,532 (37.7%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosato, I.; Dalla Zuanna, T.; Tricarico, V.; Barbiellini Amidei, C.; Canova, C. Adherence to Cervical Cancer Screening Programs in Migrant Populations: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2023, 20, 2200. https://doi.org/10.3390/ijerph20032200
Rosato I, Dalla Zuanna T, Tricarico V, Barbiellini Amidei C, Canova C. Adherence to Cervical Cancer Screening Programs in Migrant Populations: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health. 2023; 20(3):2200. https://doi.org/10.3390/ijerph20032200
Chicago/Turabian StyleRosato, Isabella, Teresa Dalla Zuanna, Valentina Tricarico, Claudio Barbiellini Amidei, and Cristina Canova. 2023. "Adherence to Cervical Cancer Screening Programs in Migrant Populations: A Systematic Review and Meta-Analysis" International Journal of Environmental Research and Public Health 20, no. 3: 2200. https://doi.org/10.3390/ijerph20032200
APA StyleRosato, I., Dalla Zuanna, T., Tricarico, V., Barbiellini Amidei, C., & Canova, C. (2023). Adherence to Cervical Cancer Screening Programs in Migrant Populations: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health, 20(3), 2200. https://doi.org/10.3390/ijerph20032200







