Safety Concerns Related to the Simultaneous Use of Prescription or Over-the-Counter Medications and Herbal Medicinal Products: Survey Results among Latvian Citizens
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Demographics
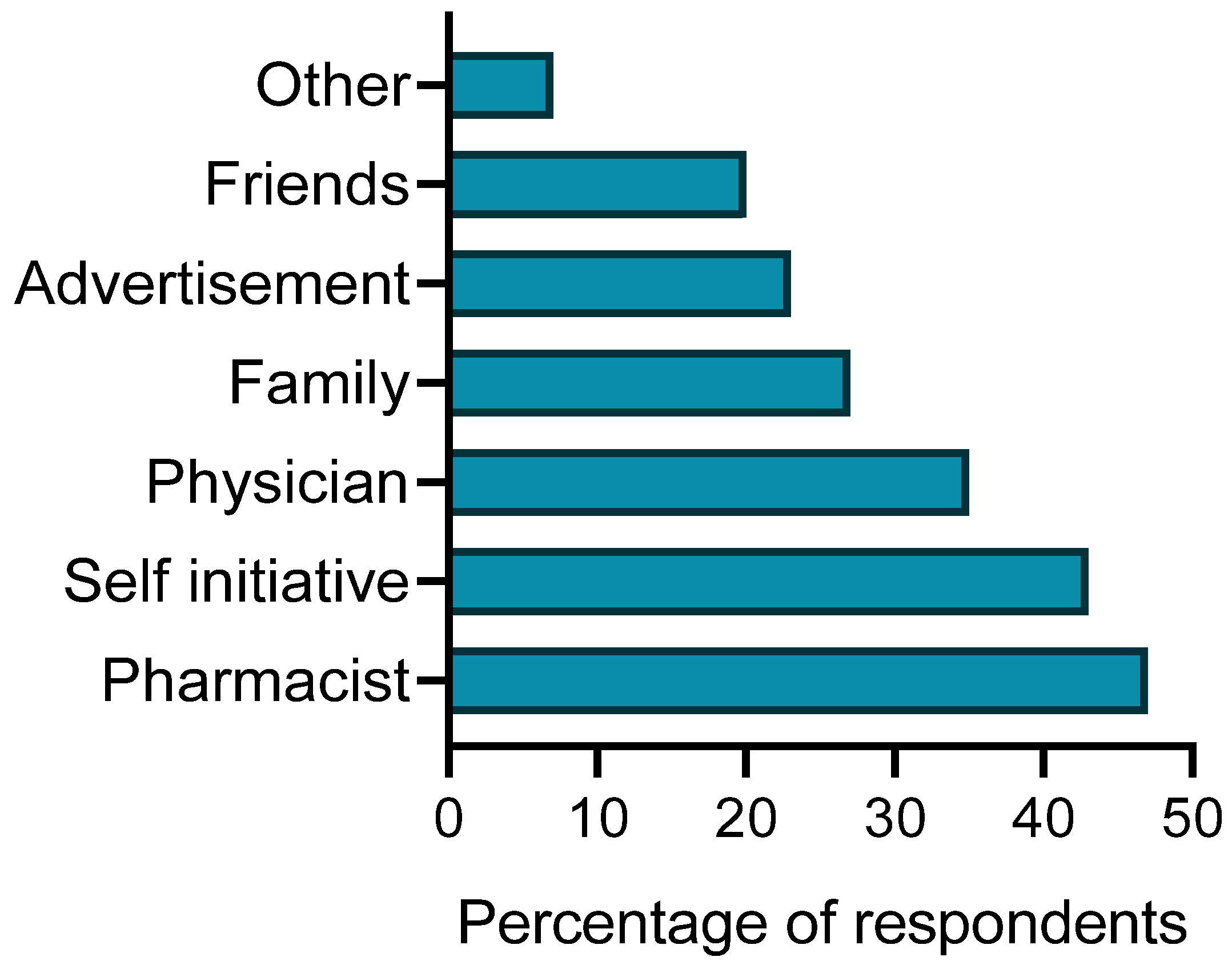
3.2. The Use of Herbal Medicines and Herbal Dietary Supplements and Their Recommended Sources
3.3. Attitudes towards the Safety of Herbal Medicines
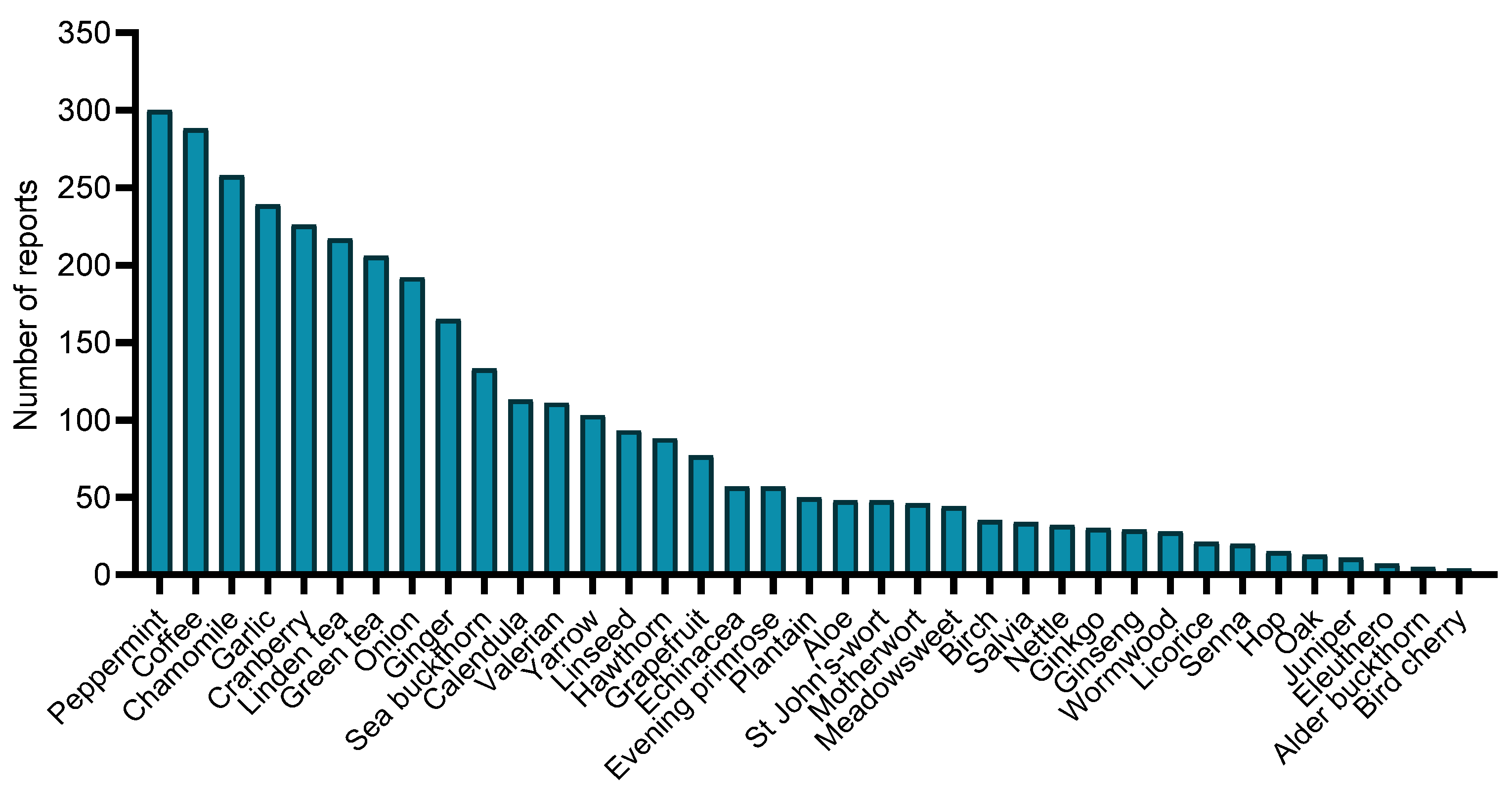
3.4. Medicinal Plants Used among Individuals Who Used Herbal Products
3.5. Respondents’ Health Concerns and Interest in Medication Side Effects
3.6. Potential Clinically Significant Interactions between Herbal Medicines and Conventional Drugs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Garcia-Alvarez, A.; Egan, B.; de Klein, S.; Dima, L.; Maggi, F.M.; Isoniemi, M.; Ribas-Barba, L.; Raats, M.M.; Meissner, E.M.; Badea, M.; et al. Usage of Plant Food Supplements across Six European Countries: Findings from the PlantLIBRA Consumer Survey. PLoS ONE 2014, 9, e92265. [Google Scholar] [CrossRef]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177. [Google Scholar] [CrossRef] [PubMed]
- Posadzki, P.; Watson, L.; Ernst, E. Herb-drug interactions: An overview of systematic reviews. Br. J. Clin. Pharmacol. 2013, 75, 603–618. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Klotz, U. Drug Interactions with Herbal Medicines. Clin. Pharmacokinet. 2012, 51, 77–104. [Google Scholar] [CrossRef]
- Fasinu, P.S.; Bouic, P.J.; Rosenkranz, B. An Overview of the Evidence and Mechanisms of Herb–Drug Interactions. Front. Pharmacol. 2012, 3, 69. [Google Scholar] [CrossRef]
- Al-Ramahi, R.; Jaradat, N.; Shalalfeh, R.; Nasir, S.; Manasra, Y.; Shalalfeh, I.; Esam, Y. Evaluation of potential drug- herb interactions among a group of Palestinian patients with chronic diseases. BMC Complement. Altern. Med. 2015, 15, 221. [Google Scholar] [CrossRef]
- Awortwe, C.; Makiwane, M.; Reuter, H.; Muller, C.; Louw, J.; Rosenkranz, B. Critical evaluation of causality assessment of herb-drug interactions in patients. Br. J. Clin. Pharmacol. 2018, 84, 679–693. [Google Scholar] [CrossRef]
- Gavronski, M.; Volmer, D. Safety concerns in simultaneous use of prescription and ‘over-the-counter’ medicines- results of patient survey in Estonia. Springerplus 2014, 3, 143. [Google Scholar] [CrossRef]
- Ara, T.; Viqar, M.; Arshad, J. Use of Herbal Products and Potential Interactions in Patients With Cardiovascular Diseases. J. Am. Coll. Cardiol. 2010, 55, 515–525. [Google Scholar]
- De Smet, P.A.G.M. Clinical risk management of herb-drug interactions. Br. J. Clin. Pharmacol. 2007, 63, 258–267. [Google Scholar] [CrossRef]
- Strauch, K.; Lutz, U.; Bittner, N.; Lutz, W.K. Dose–response relationship for the pharmacokinetic interaction of grapefruit juice with dextromethorphan investigated by human urinary metabolite profiles. Food Chem. Toxicol. 2009, 47, 1928–1935. [Google Scholar] [CrossRef]
- Markowitz, J.S.; Donovan, J.L.; Lindsay Devane, C.; Taylor, R.M.; Ruan, Y.; Wang, J.-S.; Chavin, K.D. Effect of St John’s Wort on Drug Metabolism by Induction of Cytochrome P450 3A4 Enzyme. JAMA 2003, 290, 1500–1504. [Google Scholar] [CrossRef] [PubMed]
- Izzo, A.A. Drug Interactions with St. John’s Wort (Hypericum Perforatum): A Review of the Clinical Evidence. Int. J. Clin. Pharmacol. Ther. 2004, 42, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Borrelli, F.; Izzo, A.A. Herb–Drug Interactions with St John’s Wort (Hypericum perforatum): An Update on Clinical Observations. AAPS J. 2009, 11, 710–727. [Google Scholar] [CrossRef]
- Moore, L.B.; Goodwin, B.; Jones, S.A.; Wisely, G.B.; Serabjit-Singh, C.J.; Willson, T.M.; Collins, J.L.; Kliewer, S.A.S. John’s wort induces hepatic drug metabolism through activation of the pregnane X receptor. Proc. Natl. Acad. Sci. USA 2000, 97, 7500–7502. [Google Scholar] [CrossRef]
- Ioannides, C. Topics in Xenobiochemistry Pharmacokinetic Interactions between Herbal Remedies and Medicinal Drugs. Xenobiotica 2002, 32, 451–478. [Google Scholar] [CrossRef]
- Mannel, M. Drug Interactions with St John’s Wort Mechanisms and Clinical Implications. Drug Saf. 2004, 27, 773–797. [Google Scholar]
- Yue, Q.-Y.; Bergquist, C.; Gerdén, B. Safety of St John’s wort (Hypericum perforatum). Lancet 2000, 355, 576–577. [Google Scholar] [CrossRef]
- Schwarz, U.I.; Büschel, B.; Kirch, W. Unwanted Pregnancy on Self-Medication with St John’s Wort despite Hormonal Contraception. Br. J. Clin. Pharmacol. 2003, 55, 112–113. [Google Scholar] [PubMed]
- Miller, L.G. Herbal Medicinals Selected Clinical Considerations Focusing on Known or Potential Drug-Herb Interactions. Arch. Intern. Med. 1998, 158, 2200–2211. [Google Scholar] [CrossRef]
- Carrasco, M.C.; Ramón Vallejo, J.; Pardo-De-Santayana, M.; Peral, D.; Martín, M.Á.; Altimiras, J. Interactions of Valeriana officinalis L. and Passiflora incarnata L. in a Patient Treated with Lorazepam. Phytother. Res. 2009, 23, 1795–1796. [Google Scholar] [CrossRef] [PubMed]
- Committee on Herbal Medicinal Products (HMPC). Assessment Report on Rhamnus frangula L. Cortex. European Medicines Agency; 2019; EMA/HMPC/483550/2018. Available online: https://www.ema.europa.eu/ (accessed on 11 June 2023).
- Gorski, J.; Huang, S.; Pinto, A.; Hamman, M.A.; Hilligoss, J.K.; Zaheer, N.A.; Desai, M.; Miller, M.; Hall, S.D. The effect of echinacea (Echinacea purpurea root) on cytochrome P450 activity in vivo. Clin. Pharmacol. Ther. 2004, 75, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, P.K.; Ponnusankar, S.; Pandit, S.; Hazam, P.K.; Ahmmed, M.; Mukherjee, K. Botanicals as medicinal food and their effects on drug metabolizing enzymes. Food Chem. Toxicol. 2011, 49, 3142–3153. [Google Scholar] [CrossRef]
- Chen, X.-W.; Serag, E.S.; Sneed, K.B.; Liang, J.; Chew, H.; Pan, S.-Y.; Zhou, S.-F. Clinical Herbal Interactions with Conventional Drugs: From Molecules to Maladies. Curr. Med. Chem. 2011, 18, 4836–4850. [Google Scholar] [CrossRef]
- Abebe, W. Herbal Medication: Potential for Adverse Interactions with Analgesic Drugs. J. Clin. Pharm. Ther. 2002, 27, 391–401. [Google Scholar] [CrossRef] [PubMed]
- Ali, B.H.; Blunden, G.; Tanira, M.O.; Nemmar, A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): A review of recent research. Food Chem. Toxicol. 2008, 46, 409–420. [Google Scholar] [CrossRef]
- Marx, W.; McKavanagh, D.; McCarthy, A.L.; Bird, R.; Ried, K.; Chan, A.; Isenring, L. The Effect of Ginger (Zingiber officinale) on Platelet Aggregation: A Systematic Literature Review. PLoS ONE 2015, 10, e0141119. [Google Scholar] [CrossRef]
- Izzo, A.A.; Di Carlo, G.; Borrelli, F.; Ernst, E. Cardiovascular pharmacotherapy and herbal medicines: The risk of drug interaction. Int. J. Cardiol. 2005, 98, 1–14. [Google Scholar] [CrossRef]
- Williamson, E.M. Drug Interactions Between Herbal and Prescription Medicines. Drug Saf. 2003, 26, 1075–1092. [Google Scholar] [CrossRef]
- Kashihara, Y.; Ieiri, I.; Yoshikado, T.; Maeda, K.; Fukae, M.; Kimura, M.; Hirota, T.; Matsuki, S.; Irie, S.; Izumi, N.; et al. Small-Dosing Clinical Study: Pharmacokinetic, Pharmacogenomic (SLCO2B1 and ABCG2), and Interaction (Atorvastatin and Grapefruit Juice) Profiles of 5 Probes for OATP2B1 and BCRP. J. Pharm. Sci. 2017, 106, 2688–2694. [Google Scholar] [CrossRef]
- Holtzman, C.W.; Wiggins, B.S.; Spinler, S.A. Role of P-glycoprotein in Statin Drug Interactions. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2006, 26, 1601–1607. [Google Scholar] [CrossRef]
- Nugala, B.; Namasi, A.; Emmadi, P.; Krishna, P.M. Role of green tea as an antioxidant in periodontal disease: The Asian paradox. J. Indian Soc. Periodontol. 2012, 16, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Varma, R.; Ross, C.N. Liquorice: A root cause of secondary hypertension. JRSM Open 2017, 8, 205427041668520. [Google Scholar] [CrossRef]
- Atkinson, H.C.; Potts, A.L.; Anderson, B.J. Potential cardiovascular adverse events when phenylephrine is combined with paracetamol: Simulation and narrative review. Eur. J. Clin. Pharmacol. 2015, 71, 931–938. [Google Scholar] [CrossRef]
- Ritter, M.; Melichar, K.; Strahler, S.; Kuchta, K.; Schulte, J.; Sartiani, L.; Cerbai, E.; Mugelli, A.; Mohr, F.-W.; Rauwald, H.W.; et al. Cardiac and Electrophysiological Effects of Primary and Refined Extracts from Leonurus cardiaca L. (Ph. Eur.). Planta Medica 2010, 76, 572–582. [Google Scholar] [CrossRef] [PubMed]
- Agbabiaka, T.B.; Spencer, N.H.; Khanom, S.; Goodman, C. Prevalence of Drug—Herb and Drug—Supplement Interactions in Older Adults: A cross-sectional survey. Br. J. Gen. Pract. 2018, 68, e711–e717. [Google Scholar] [PubMed]
- Tsai, H.-H.; Lin, H.-W.; Lu, Y.-H.; Chen, Y.-L.; Mahady, G.B. A Review of Potential Harmful Interactions between Anticoagulant/Antiplatelet Agents and Chinese Herbal Medicines. PLoS ONE 2013, 8, e64255. [Google Scholar] [CrossRef]
- Donovan, J.L.; DeVane, C.L.; Chavin, K.D.; Taylor, R.M.; Markowitz, J.S. Siberian ginseng (Eleutheroccus senticosus) effects on cyp2d6 and cyp3a4 activity in normal volunteers. Drug Metab. Dispos. 2003, 31, 519–522. [Google Scholar] [CrossRef]
- Lippert, A.; Renner, B. Herb–Drug Interaction in Inflammatory Diseases: Review of Phytomedicine and Herbal Supplements. J. Clin. Med. 2022, 11, 1567. [Google Scholar] [CrossRef] [PubMed]
- Raudonė, L.; Raudonis, R.; Janulis, V.; Viškelis, P. Quality evaluation of different preparations of dry extracts of birch (Betula pendula Roth) leaves. Nat. Prod. Res. 2014, 28, 1645–1648. [Google Scholar] [CrossRef]
- Agbabiaka, T.B.; Wider, B.; Watson, L.K.; Goodman, C. Concurrent Use of Prescription Drugs and Herbal Medicinal Products in Older Adults: A Systematic Review. Drugs Aging 2017, 34, 891–905. [Google Scholar] [CrossRef]
- Chan, W.J.J.; McLachlan, A.J.; Luca, E.J.; Harnett, J.E. Garlic (Allium sativum L.) in the Management of Hypertension and Dyslipidemia—A Systematic Review. J. Herb. Med. 2020, 19, 100292. [Google Scholar] [CrossRef]
- Mouly, S.; Lloret-Linares, C.; Sellier, P.-O.; Sene, D.; Bergmann, J.-F. Is the clinical relevance of drug-food and drug-herb interactions limited to grapefruit juice and Saint-John’s Wort? Pharmacol. Res. 2017, 118, 82–92. [Google Scholar] [CrossRef]
- Werba, J.P.; Misaka, S.; Giroli, M.G.; Shimomura, K.; Amato, M.; Simonelli, N.; Vigo, L.; Tremoli, E. Update of green tea interactions with cardiovascular drugs and putative mechanisms. J. Food Drug Anal. 2018, 26, S72–S77. [Google Scholar] [CrossRef] [PubMed]
- Sá, C.M.; Ramos, A.A.; Azevedo, M.F.; Lima, C.F.; Fernandes-Ferreira, M.; Pereira-Wilson, C. Sage Tea Drinking Improves Lipid Profile and Antioxidant Defences in Humans. Int. J. Mol. Sci. 2009, 10, 3937–3950. [Google Scholar] [CrossRef] [PubMed]
- Sile, I.; Romane, E.; Reinsone, S.; Maurina, B.; Tirzite, D.; Dambrova, M. Medicinal plants and their uses recorded in the Archives of Latvian Folklore from the 19th century. J. Ethnopharmacol. 2020, 249, 112378. [Google Scholar] [CrossRef]
- Yilmaz, M.B.; Yontar, O.C.; Turgut, O.O.; Yilmaz, A.; Yalta, K.; Gul, M.; Tandogan, I. Herbals in cardiovascular practice: Are physicians neglecting anything? Int. J. Cardiol. 2007, 122, 48–51. [Google Scholar] [CrossRef]
- Kennedy, J. Herb and supplement use in the US adult population. Clin. Ther. 2005, 27, 1847–1858. [Google Scholar] [CrossRef]
- Picking, D.; Younger, N.; Mitchell, S.; Delgoda, R. The prevalence of herbal medicine home use and concomitant use with pharmaceutical medicines in Jamaica. J. Ethnopharmacol. 2011, 137, 305–311. [Google Scholar] [CrossRef]
- Samojlik, I.; Mijatović, V.; Gavarić, N.; Krstin, S.; Božin, B. Consumers’ attitude towards the use and safety of herbal medicines and herbal dietary supplements in Serbia. Int. J. Clin. Pharm. 2013, 35, 835–840. [Google Scholar] [CrossRef]
- Sánchez, M.; González-Burgos, E.; Iglesias, I.; Lozano, R.; Gómez-Serranillos, M.P. Current uses and knowledge of medicinal plants in the Autonomous Community of Madrid (Spain): A descriptive cross-sectional study. BMC Complement. Med. Ther. 2020, 20, 306. [Google Scholar] [CrossRef]
- Kennedy, D.A.; Lupattelli, A.; Koren, G.; Nordeng, H. Safety classification of herbal medicines used in pregnancy in a multinational study. BMC Complement. Altern. Med. 2016, 16, 102. [Google Scholar] [CrossRef]
- Welz, A.N.; Emberger-Klein, A.; Menrad, K. The importance of herbal medicine use in the German health-care system: Prevalence, usage pattern, and influencing factors. BMC Health Serv. Res. 2019, 19, 952. [Google Scholar] [CrossRef]
- Qato, D.M.; Alexander, G.C.; Conti, R.M.; Johnson, M.; Schumm, P.; Lindau, S.T. Use of Prescription and Over-the-counter Medications and Dietary Supplements Among Older Adults in the United States. JAMA 2008, 300, 2867–2878. [Google Scholar] [CrossRef]
- González-Stuart, A. Herbal product use by older adults. Maturitas 2011, 68, 52–55. [Google Scholar] [CrossRef]
- Rahmawati, R.; Bajorek, B.V. Self-medication among people living with hypertension: A review. Fam. Pract. 2017, 34, 147–153. [Google Scholar] [CrossRef]
- Hassen, G.; Belete, G.; Carrera, K.G.; Iriowen, R.O.; Araya, H.; Alemu, T.; Solomon, N.; Bam, D.S.; Nicola, S.M.; Araya, M.E.; et al. Clinical Implications of Herbal Supplements in Conventional Medical Practice: A US Perspective. Cureus 2022, 14, e26893. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Onakpoya, I.J.; Posadzki, P.; Eddouks, M. The Safety of Herbal Medicine: From Prejudice to Evidence. Evid. -Based Complement. Altern. Med. 2015, 2015, 316706. [Google Scholar]
- Delgoda, R.; Westlake, A.C.G. Herbal Interactions Involving Cytochrome P450 Enzymes: A Mini Review. Toxicol. Rev. 2004, 23, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Gurley, B.J.; Gardner, S.F.; Hubbard, M.A.; Williams, D.K.; Gentry, W.B.; Khan, I.A.; Shah, A. In vivo effects of goldenseal, kava kava, black cohosh, and valerian on human cytochrome P450 1A2, 2D6, 2E1, and 3A4/5 phenotypes. Clin. Pharmacol. Ther. 2005, 77, 415–426. [Google Scholar] [CrossRef]
- Izzo, A.A. Interactions between Herbs and Conventional Drugs: Overview of the Clinical Data. Med. Princ. Pract. 2012, 21, 404–428. [Google Scholar] [CrossRef] [PubMed]


| Characteristics | Frequency | Percentage, n = 504 | |
|---|---|---|---|
| Age | 18–29 | 65 | 12.9 |
| 30–39 | 103 | 20.4 | |
| 40–49 | 106 | 21.0 | |
| 50–59 | 91 | 18.1 | |
| 60–69 | 70 | 13.9 | |
| 70–79 | 51 | 10.1 | |
| 80–95 | 18 | 3.6 | |
| Sex | Female | 380 | 75.4 |
| Male | 124 | 24.6 | |
| Education | Basic education | 29 | 5.8 |
| Secondary education | 238 | 47.2 | |
| Higher education | 236 | 46.8 | |
| Doctoral degree | 1 | 0.2 | |
| Region | City | 297 | 58.9 |
| Town | 119 | 23.6 | |
| Countryside | 88 | 17.5 |
| Long-Term Health Condition | Frequency (%) |
|---|---|
| Back pain | 185 (69.5%) |
| High blood pressure | 157 (59.0%) |
| Allergies | 72 (27.1%) |
| Depression, anxiety | 56 (21.1%) |
| High blood cholesterol and fats | 56 (21.1%) |
| Gastric or peptic ulcer disease | 55 (20.7%) |
| Heart disease | 49 (18.4%) |
| Kidney and urinary tract diseases | 49 (18.4%) |
| Different types of arthritis | 41 (15.4%) |
| Lung diseases | 29 (10.9%) |
| Anaemia or any other blood condition | 17 (6.4%) |
| Osteoporosis (bone atrophy) | 15 (5.6%) |
| Diabetes | 15 (5.6%) |
| Cancer | 12 (4.5%) |
| Liver diseases | 5 (2.3%) |
| Other (thyroid problems, bone fractures, joint injuries, gynaecologic diseases, venous problems, and others) | 62 (23.3%) |
| Herbal Medicine | Conventional Drug | Clinical Result of Interaction | Classification of Drug Interaction * | Comments | References |
|---|---|---|---|---|---|
| Grapefruit Citrus paradisi | Dextromethorphan | Grapefruit increases the level or effect of dextromethorphan and ivabradine by affecting hepatic/intestinal enzyme CYP3A4 metabolism. | Serious | [11] | |
| St. John’s wort Hypericum perforatum | Amitriptyline, citalopram, clomipramine, dextromethorphan, escitalopram, mirtazapine, paroxetine, sertraline, trazodone, venlafaxine | Coadministration may potentiate the risk of serotonin syndrome, which is a rare but serious and potentially fatal condition thought to result from hyperstimulation of brainstem 5-HT1A and 2A receptors. | Serious | [12,13,14] | |
| Alprazolam, atorvastatin, carbamazepine, clopidogrel, diazepam, felodipine | St. John’s wort decreases the level or effect of the drug by affecting hepatic/intestinal enzyme CYP3A4 metabolism. | Serious | Special attention should be given when using any drug that is metabolized by CYP3A4, CYP2C9, CYP2C19, or P-glycoprotein in combination with another substance because it may lead to a reduction of plasma concentrations. | [15,16] | |
| Dabigatran | Coadministration with potent inducers of P-glycoprotein may significantly reduce the bioavailability of dabigatran following oral administration. | Serious | [17,18] | ||
| Drospirenone, oestradiol, ethinylestradiol, ivabradine, loratadine, nifedipine, prednisolone, sildenafil, rivaroxaban, trazodone | St. John’s wort decreases the level or effect of these drugs by affecting hepatic/intestinal enzyme CYP3A4 metabolism. | Serious | Coadministration with St. John’s wort may reduce the efficacy of contraceptive hormones. Nonhormonal contraception is recommended. | [17,19] | |
| Valerian Valeriana officinalis | Alprazolam, clonazepam, codeine, diazepam, gabapentin, phenobarbital | Coadministration of valerian and these drugs increases sedation effects. | Serious | [20,21] | |
| Alder buckthorn Frangula alnus | Hydrochlorothiazide, indapamide | Prolonged or excessive use of laxatives can enhance the pharmacological impact of diuretics. Laxatives have the potential to lead to substantial depletion of fluids and essential electrolytes like sodium, potassium (resulting in hypokalemia), magnesium, and zinc. These effects could be cumulative when combined with the effects of diuretics. | Moderate | [22] | |
| Echinacea Echinacea purpurea | Acetaminophen | Coadministration with echinacea may increase the plasma concentrations and the risk of adverse effects of drugs that are substrates of CYP450 1A2. | Moderate | [23,24] | |
| Amlodipine, atorvastatin | Coadministration with echinacea may alter the plasma concentrations and therapeutic effects of drugs that are substrates of CYP450 3A4. | Moderate | [23,24] | ||
| Garlic Allium sativum | Aceclofenac, aspirin, diclofenac, ibuprofen | Garlic has the potential to enhance the effects of anticoagulants, platelet inhibitors, and thrombolytic agents, which may lead to an increased risk of bleeding. | Moderate | Garlic powder, aged garlic preparations, garlic oil, and fresh garlic all contain antiplatelet activity. Some isolated reports have linked chronic and high dietary intake of garlic to bleeding complications. | [25,26] |
| Ginger Zingiber officinale | Aceclofenac, aspirin, diclofenac, ibuprofen | Ginger has the potential to enhance the effects of anticoagulants, platelet inhibitors, and thrombolytic agents, which could potentially increase the risk of bleeding. | Moderate | [27,28] | |
| Ginkgo Ginkgo biloba | Aspirin, ibuprofen, diclofenac | Ginkgo may potentiate the risk of bleeding associated with anticoagulants, platelet inhibitors, nonsteroidal anti-inflammatory drugs (NSAIDs), and thrombolytic agents. | Moderate | Consumption of ginkgo should be avoided during the use of coagulation-modifying agents and at least two weeks prior to surgery. | [20,29] |
| Hydrochlorothiazide, indapamide | Blood pressure may increase following the addition of ginkgo while receiving a thiazide diuretic. | Moderate | [25] | ||
| Ginseng Panax ginseng | Aceclofenac, aspirin, diclofenac, ibuprofen | Ginseng may potentiate the effects of anticoagulants, platelet inhibitors, and thrombolytic agents, possibly increasing the risk of bleeding. | Moderate | [30] | |
| Grapefruit Citrus paradisi | Atorvastatin | Grapefruit juice can increase atorvastatin blood levels. | Moderate | The combination of certain medications, such as atorvastatin, with grapefruit juice can increase the risk of side effects like liver damage and a rare but serious condition known as rhabdomyolysis, which involves the breakdown of skeletal muscle tissue. Therefore, it is recommended to limit grapefruit juice consumption to no more than 1 quart per day while undergoing atorvastatin treatment to mitigate these risks. | [31,32] |
| Green tea Camellia sinensis | Aspirin, diclofenac, ibuprofen | Combination may increase the risk of bleeding. | Moderate | [33] | |
| Licorice Glycyrrhiza glabra | Amlodipine, bisoprolol, metoprolol, nebivolol, perindopril, ramipril | Licorice use has been associated with hypertension and may antagonize the effects of antihypertensive agents. | Moderate | The use of licorice should be avoided in conjunction with diuretics, cardiac glycosides, corticosteroids, stimulant laxatives, or other medications that may worsen electrolyte imbalance. | [20,34] |
| Indapamide, hydrochlorothiazide | Chronic use of licorice may potentiate the hypokalemic effects of some diuretics and other drugs that deplete potassium. | Moderate | Severe hypokalaemia can lead to muscle paralysis, rhabdomyolysis, metabolic alkalosis, cardiac arrhythmias, and respiratory arrest. | [20] | |
| Motherwort Leonurus cardiaca | Phenylephrine | Motherwort decreases both blood pressure and heart rate, while phenylephrine increases both blood pressure and heart rate. | Moderate | [35,36] | |
| Nettle Urtica dioica; U. urens | Aceclofenac, aspirin, diclofenac, ibuprofen | Nettle reduces the anticoagulant effect of aceclofenac, aspirin, diclofenac, and ibuprofen and lower plasma concentrations. | Moderate | [5] | |
| Senna Cassia senna; C. angustifolia | Hydrochlorothiazide, indapamide | Prolonged or excessive use of laxatives can enhance the pharmacological impact of diuretics. Laxatives have the potential to cause considerable loss of fluids and essential electrolytes, such as sodium, potassium (resulting in hypokalemia), magnesium, and zinc. These effects may add up to the effects of diuretics. | Moderate | [37] | |
| Siberian ginseng Eleutherococcus senticosus | Aceclofenac, aspirin, diclofenac, ibuprofen | Coadministration increases anticoagulation. | Moderate | [38] | |
| Atorvastatin | St. John’s wort decreases the level or effect of atorvastatin by P-glycoprotein (MDR1) efflux transporter. | Moderate | [39] | ||
| Amlodipine, omeprazole | Coadministration with potent inducers of CYP450 2C19 and 3A4 may significantly decrease the plasma concentrations of the drug. | Moderate | The interaction is likely to also occur with esomeprazole, an enantiomer of omeprazole. | [40] | |
| Birch Betula | Hydrochlorothiazide, indapamide | Birch increases the effects of these drugs by pharmacodynamic synergism. | Minor | [41] | |
| Chamomile Matricaria chamomilla | Aspirin | In theory, consuming large amounts of chamomile could increase the risk of bleeding in patients undergoing treatment with drugs that impact the body’s ability to control bleeding, such as anticoagulants, platelet inhibitors, thrombin inhibitors, thrombolytic agents, or medications that often lead to low platelet counts (thrombocytopenia). | Minor | For patients who have extensively used chamomile before starting anticoagulation, antiplatelet, or thrombolytic therapy, it is crucial to consider the potential for interactions. | [20,42] |
| Evening primrose Oenothera biennis | Aspirin | Theoretically, the use of evening primrose oil with anticoagulants or antiplatelet aggregation drugs may increase the risk of bleeding in some patients. | Minor | [42] | |
| Garlic Allium sativum | Amlodipine, bisoprolol, hydrochlorothiazide, indapamide, metoprolol, nebivolol, perindopril, ramipril | Garlic has been found in some studies to lower blood pressure and may theoretically potentiate the effects of hypotensive agents. There have been no reports of clinical hypotension associated with the concomitant use of garlic and antihypertensive agents. | Minor | [43] | |
| Grapefruit Citrus paradisi | Amlodipine | Grapefruit increases the level or effect of amlodipine by affecting hepatic/intestinal enzyme CYP3A4 metabolism. | Minor | [11] | |
| Green tea Camellia sinensis | Acetaminophen | Combination may increase the risk of bleeding. | Minor | [44] | |
| Atorvastatin, rosuvastatin | Coadministration with green tea may decrease oral bioavailability. | Minor | [45] | ||
| Sage Salvia officinalis | Phenylephrine | Sage lowers blood pressure, while phenylephrine increases blood pressure | Minor | [35,46] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sile, I.; Teterovska, R.; Onzevs, O.; Ardava, E. Safety Concerns Related to the Simultaneous Use of Prescription or Over-the-Counter Medications and Herbal Medicinal Products: Survey Results among Latvian Citizens. Int. J. Environ. Res. Public Health 2023, 20, 6551. https://doi.org/10.3390/ijerph20166551
Sile I, Teterovska R, Onzevs O, Ardava E. Safety Concerns Related to the Simultaneous Use of Prescription or Over-the-Counter Medications and Herbal Medicinal Products: Survey Results among Latvian Citizens. International Journal of Environmental Research and Public Health. 2023; 20(16):6551. https://doi.org/10.3390/ijerph20166551
Chicago/Turabian StyleSile, Inga, Renate Teterovska, Oskars Onzevs, and Elita Ardava. 2023. "Safety Concerns Related to the Simultaneous Use of Prescription or Over-the-Counter Medications and Herbal Medicinal Products: Survey Results among Latvian Citizens" International Journal of Environmental Research and Public Health 20, no. 16: 6551. https://doi.org/10.3390/ijerph20166551
APA StyleSile, I., Teterovska, R., Onzevs, O., & Ardava, E. (2023). Safety Concerns Related to the Simultaneous Use of Prescription or Over-the-Counter Medications and Herbal Medicinal Products: Survey Results among Latvian Citizens. International Journal of Environmental Research and Public Health, 20(16), 6551. https://doi.org/10.3390/ijerph20166551









