Cadmium Body Burden and Inflammatory Arthritis: A Pilot Study in Patients from Lower Silesia, Poland
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Sample Collection
2.3. Determination of Blood Cadmium Content
2.4. Haematological and Biochemical Analysis
2.5. Statistical Analysis
3. Results
3.1. Basic Demographic Characteristics and Laboratory Parameters
3.2. Correlations between Blood Cadmium Concentrations and Basic Characteristics
3.3. Logistic Regression Model
3.4. Relationship between Disease Severity and Demographic Characteristics, Laboratory Parameters, and Cadmium Body Burden
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALT | alanine aminotransferase |
| AS | ankylosing spondylitis |
| AST | aspartate aminotransferase |
| Cd-B | blood cadmium concentration |
| CI | confidence interval |
| COX-2 | cyclooxygenase-2 |
| CRE | creatinine |
| ESR | erythrocyte sedimentation rate |
| HCT | haematocrit |
| hs-CRP | high-sensitivity C-reactive protein |
| IA | inflammatory arthritis |
| IL-10 | interleukin 10 |
| MCH | mean corpuscular haemoglobin |
| MCHC | mean corpuscular haemoglobin concentration |
| MCV | mean corpuscular volume |
| n.s. | not statistically significant |
| OR | odds ratio |
| PLT | platelets |
| PsA | psoriatic arthritis |
| RA | rheumatoid arthritis |
| RBC | red blood cells |
| WBC | white blood cells |
| 8-OHdG | 8-hydroxy-2’-deoxyguanosine |
References
- Lerner, A.; Jeremias, P.; Matthias, T. The world incidence and prevalence of autoimmune diseases is increasing. Int. J. Celiac Dis. 2015, 3, 151–155. [Google Scholar] [CrossRef] [Green Version]
- Rheumatic Diseases Declaration of the European Parliament on Rheumatic Diseases-Publications Office of the EU (europa.eu). Off. J. Eur. Union C 2009, 285 E, 67–68. Available online: https://op.europa.eu/en/publication-detail/-/publication/1f5aa9c8-285a-4b78-94f6-2437fdcafb7 (accessed on 23 January 2022).
- Alivernini, S.; Tolusso, B.; Petricca, L.; Ferraccioli, G.; Gremese, E. Chapter 46-Rheumatoid arthritis. In Mosaic of Autoimmunity: The Novel Factors of Autoimmune Diseases, 1st ed.; Perricone, C., Shoenfeld, Y., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; Section V; pp. 501–526. [Google Scholar] [CrossRef]
- Scotti, L.; Franchi, M.; Marchesoni, A.; Corrao, G. Prevalence and incidence of psoriatic arthritis: A systematic review and meta-analysis. Semin. Arthritis Rheum. 2018, 48, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Huang, C.; Huang, H.; Pan, J.-K.; Zeng, L.-F.; Luo, M.-H.; Liang, G.-H.; Yang, W.-Y.; Liu, J. Prevalence of ankylosing spondylitis in a Chinese population: A systematic review and meta-analysis. Rheumatol. Int. 2020, 40, 859–872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kosmaczewska, A.; Świerkot, J.; Ciszak, L.; Wiland, P. Rola subpopulacji limfocytów pomocniczych Th1, Th17 i Treg w patogenezie reumatoidalnego zapalenia stawów z uwzględnieniem przeciwzapalnego działania cytokin Th1. Postepy Hig. Med. Dosw. 2011, 65, 397–403. [Google Scholar] [CrossRef]
- McGonagle, D.G.; McInnes, I.B.; Kirkham, B.W.; Sherlock, J.; Moots, R. The role of IL-17A in axial spondyloarthritis and psoriatic arthritis: Recent advances and controversies. Ann. Rheum. Dis. 2019, 78, 1167–1178. [Google Scholar] [CrossRef] [Green Version]
- Simone, D.; Al Mossawi, M.H.; Bowness, P. Progress in our understanding of the pathogenesis of ankylosing spondylitis. Rheumatology 2018, 57 (Suppl. 6), vi4–vi9. [Google Scholar] [CrossRef] [Green Version]
- Shapira, Y.; Agmon-Levin, N.; Shoenfeld, Y. Geoepidemiology of autoimmune rheumatic diseases. Nat. Rev. Rheumatol. 2010, 6, 468–476. [Google Scholar] [CrossRef]
- Jiang, X.; Alfredsson, L. Modifiable environmental exposure and risk of rheumatoid arthritis—Current evidence from genetic studies. Arthritis Res. Ther. 2020, 22, 154. [Google Scholar] [CrossRef]
- Sankowski, A.J.; Łebkowska, U.M.; Ćwikła, J.; Walecka, I.; Walecki, J. Psoriatic arthritis. Pol. J. Radiol. 2013, 78, 7–17. [Google Scholar] [CrossRef] [Green Version]
- Zhu, W.; He, X.; Cheng, K.; Zhang, L.; Chen, D.; Wang, X.; Qiu, G.; Cao, X.; Weng, X. Ankylosing spondylitis: Etiol-ogy, pathogenesis, and treatments. Bone Res. 2019, 7, 22. [Google Scholar] [CrossRef] [Green Version]
- Chang, K.; Yang, S.M.; Kim, S.H.; Han, K.H.; Park, S.J.; Shin, J.I. Smoking and rheumatoid arthritis. Int. J. Mol. Sci. 2014, 15, 22279–22295. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nguyen, U.-S.D.T.; Zhang, Y.; Lu, N.; Louie-Gao, Q.; Niu, J.; Ogdie, A.; Gelfand, J.M.; LaValley, M.P.; Dubreuil, M.; Sparks, J.A.; et al. Smoking paradox in the development of psoriatic arthritis among patients with psoriasis: A population-based study. Ann. Rheum. Dis. 2018, 77, 119–123. [Google Scholar] [CrossRef]
- Zhang, S.; Li, Y.; Xu, X.; Feng, X.; Yang, D.; Lin, G. Effect of cigarette smoking and alcohol consumption on disease activity and physical functioning in ankylosing spondylitis: A cross-sectional study. Int. J. Clin. Exp. Med. 2015, 8, 13919–13927. [Google Scholar] [PubMed]
- Harel-Meir, M.; Sherer, Y.; Shoenfeld, Y. Tobacco smoking and autoimmune rheumatic diseases. Nat. Clin. Pract. Rheumatol. 2007, 3, 707–715. [Google Scholar] [CrossRef]
- Essouma, M.; Noubiap, J.J.N. Is air pollution a risk factor for rheumatoid arthritis? J. Inflamm. 2015, 12, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hart, J.E.; Laden, F.; Puett, R.C.; Costenbader, K.H.; Karlson, E.W. Exposure to traffic pollution and increased risk of rheumatoid arthritis. Environ. Health Perspect. 2009, 117, 1065–1069. [Google Scholar] [CrossRef] [Green Version]
- Shin, J.; Lee, J.; Lee, J.; Ha, E.-H. Association between exposure to ambient air pollution and rheumatoid arthritis in adults. Int. J. Environ. Res. Public Health 2019, 16, 1227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balali-Mood, M.; Naseri, K.; Tahergorabi, Z.; Khazdair, M.R.; Sadeghi, M. Toxic mechanisms of five heavy metals: Mercury, lead, chromium, cadmium, and arsenic. Front. Pharmacol. 2021, 12, 643972. [Google Scholar] [CrossRef]
- World Health Organization. Exposure to Cadmium: A Major Public Health Concern. Preventing Disease through Healthy Environments. 2019. Available online: https://apps.who.int/iris/handle/10665/329480 (accessed on 23 January 2022).
- Chunhabundit, R. Cadmium exposure and potential health risk from foods in contaminated area, Thailand. Toxicol. Res. 2016, 32, 65–72. [Google Scholar] [CrossRef]
- Genchi, G.; Sinicropi, M.S.; Lauria, G.; Carocci, A.; Catalano, A. The effects of cadmium toxicity. Int. J. Environ. Res. Public Health 2020, 17, 3782. [Google Scholar] [CrossRef]
- Hayat, M.T.; Nauman, M.; Nazir, N.; Ali, S.; Bangash, N. Chapter 7-Environmental Hazards of Cadmium: Past, Present, and Future. In Cadmium Toxicity and Tolerance in Plants from Physiology to Remediation, 1st ed.; Hasanuzzaman, M., Prasad, M.N.V., Fujita, M., Eds.; Elsevier: London, UK, 2019; pp. 163–183. [Google Scholar]
- U.S. Food & Drug Administration. FDA’s Testing of Cosmetics for Arsenic, Cadmium, Chromium, Cobalt, Lead, Mercury, and Nickel Content. 2020. Available online: https://www.fda.gov/cosmetics/potential-contaminants-cosmetics/fdas-testing-cosmetics-arsenic-cadmium-chromium-cobalt-lead-mercury-and-nickel-content (accessed on 23 January 2022).
- Joo, S.H.; Lee, J.; Hutchinson, D.; Song, Y.W. Prevalence of rheumatoid arthritis in relation to serum cadmium concentrations: Cross-sectional study using Korean National Health and Nutrition Examination Survey (KNHANES) data. BMJ Open 2019, 9, e023233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cappelletti, R.; Ceppi, M.; Claudatus, J.; Gennaro, V. Health status of male steel workers at an electric arc furnace (EAF) in Trentino, Italy. J. Occup. Med. Toxicol. 2016, 11, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hutchinson, D. Cadmium, one of the villains behind the curtain: Has exposure to cadmium helped to pull the strings of seropositive rheumatoid arthritis pathogenesis all along? Int. J. Rheum. Dis. 2015, 18, 570–573. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murphy, D.; Hutchinson, D. Is male rheumatoid arthritis an occupational disease? A review. Open Rheumatol. J. 2017, 11, 88–105. [Google Scholar] [CrossRef] [Green Version]
- Murphy, D.; James, B.; Hutchinson, D. Could the significantly increased risk of rheumatoid arthritis reported in Italian male steel workers be explained by occupational exposure to cadmium? J. Occup. Med. Toxicol. 2016, 11, 21. [Google Scholar] [CrossRef] [Green Version]
- Bjørklund, G.; Dadar, M.; Chirumbolo, S.; Aaseth, J.; Peana, M. Metals, autoimmunity, and neuroendocrinology: Is there a connection? Environ. Res. 2020, 187, 109541. [Google Scholar] [CrossRef]
- Hemdan, N.Y.A.; Emmrich, F.; Faber, S.; Lehmann, J.; Sack, U. Alterations of TH1/TH2 reactivity by heavy metals: Possible consequences include induction of autoimmune diseases. Ann. N. Y. Acad. Sci. 2007, 1109, 129–137. [Google Scholar] [CrossRef]
- Fatima, G.; Raza, A.M.; Hadi, N.; Nigam, N.; Mahdi, A.A. Cadmium in human diseases: It’s more than just a mere metal. Indian J. Clin. Biochem. 2019, 34, 371–378. [Google Scholar] [CrossRef]
- Min, K.-S.; Iwata, N.; Tetsutikawahara, N.; Onosaka, S.; Tanaka, K. Effect of hemolytic and iron-deficiency anemia on intestinal absorption and tissue accumulation of cadmium. Toxicol. Lett. 2008, 179, 48–52. [Google Scholar] [CrossRef]
- Jackowska, T.; Wójtowicz, J. Niedokrwistość chorób przewlekłych. Anemia of chronic diseases. Post. Nauk Med. 2014, 27, 26–33. [Google Scholar]
- Wiciński, M.; Liczner, G.; Cadelski, K.; Kołnierzak, T.; Nowaczewska, M.; Malinowski, B. Anemia of Chronic Diseases: Wider Diagnostics-Better Treatment? Nutrients 2020, 12, 1784. [Google Scholar] [CrossRef] [PubMed]
- Smyrnova, G. The relationship between hemoglobin level and disease activity in patients with rheumatoid arthritis. Rev. Bras. Reumatol. 2014, 54, 437–440. [Google Scholar] [CrossRef] [Green Version]
- Frangos, T.; Maret, W. Zinc and cadmium in the aetiology and pathogenesis of osteoarthritis and rheumatoid arthritis. Nutrients 2020, 13, 53. [Google Scholar] [CrossRef] [PubMed]
- Rentschler, G.; Kippler, M.; Axmon, A.; Raqib, R.; Skerfving, S.; Vahter, M.; Broberg, K. Cadmium concentrations in human blood and urine are associated with polymorphisms in zinc transporter genes. Metallomics 2014, 6, 885–891. [Google Scholar] [CrossRef] [Green Version]
- Stoeppler, M.; Brandt, K. Contributions to automated trace analysis. Z. Anal. Chem. 1980, 300, 372–380. [Google Scholar] [CrossRef]
- Adams, S.V.; Newcomb, P.A. Cadmium blood and urine concentrations as measures of exposure: NHANES 1999–2010. J. Expo. Sci. Environ. Epidemiol. 2014, 24, 163–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lamkarkach, F.; Ougier, E.; Garnier, R.; Viau, C.; Kolossa-Gehring, M.; Lange, R.; Apel, P. Human biomonitoring initiative (HBM4EU): Human biomonitoring guidance values (HBM-GVs) derived for cadmium and its compounds. Environ. Int. 2021, 147, 106337. [Google Scholar] [CrossRef]
- Nordberg, G.F.; Nogawa, K.; Nordberg, M. Cadmium. In Handbook on the Toxicology of Metals, 4th ed.; Nordberg, G.F., Fowler, B.A., Nordberg, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 667–716. [Google Scholar]
- Nordberg, G.F.; Bernard, A.; Diamond, G.L.; Duffus, J.H.; Illing, P.; Nordberg, M.; Bergdahl, I.A.; Jin, T.; Skerfving, S. Risk assessment of effects of cadmium on human health (IUPAC Technical Report). Pure Appl. Chem. 2018, 90, 755–808. [Google Scholar] [CrossRef] [Green Version]
- Barregard, L.; Sallsten, G.; Fagerberg, B.; Borné, Y.; Persson, M.; Hedblad, B.; Engström, G. Blood cadmium levels and incident cardiovascular events during follow-up in a population-based cohort of Swedish adults: The Malmö Diet and Cancer Study. Environ. Health Perspect. 2016, 124, 594–600. [Google Scholar] [CrossRef]
- Garner, R.E.; Levallois, P. Associations between cadmium levels in blood and urine, blood pressure and hypertension among Canadian adults. Environ. Res. 2017, 155, 64–72. [Google Scholar] [CrossRef]
- Järup, L. Hazards of heavy metal contamination. Br. Med. Bull. 2003, 68, 167–182. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Repić, A.; Bulat, P.; Antonijević, B.; Antunović, M.; Džudović, J.; Buha, A.; Bulat, Z. The influence of smoking habits on cadmium and lead blood levels in the Serbian adult people. Environ. Sci. Pollut. Res. Int. 2020, 27, 751–760. [Google Scholar] [CrossRef] [PubMed]
- Goyal, T.; Mitra, P.; Singh, P.; Sharma, P.; Sharma, S. Evaluation of oxidative stress and pro-inflammatory cytokines in occupationally cadmium exposed workers. Work 2021, 69, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Moulis, J.-M. Cellular mechanisms of cadmium toxicity related to the homeostasis of essential metals. Biometals 2010, 23, 877–896. [Google Scholar] [CrossRef] [PubMed]
- Rani, A.; Kumar, A.; Lal, A.; Pant, M. Cellular mechanisms of cadmium-induced toxicity: A review. Int. J. Environ. Health Res. 2014, 24, 378–399. [Google Scholar] [CrossRef]
- Domingo-Relloso, A.; Riffo-Campos, A.L.; Haack, K.; Rentero-Garrido, P.; Ladd-Acosta, C.; Fallin, D.M.; Tang, W.Y.; Herreros-Martinez, M.; Gonzalez, J.R.; Bozack, A.K.; et al. Cadmium, smoking, and human blood DNA methylation profiles in adults from the Strong Heart Study. Environ. Health Perspect. 2020, 128, 67005. [Google Scholar] [CrossRef]
- Aljazzar, A.; El-Ghareeb, W.R.; Darwish, W.S.; Abdel-Raheem, S.M.; Ibrahim, A.M. Content of total aflatoxin, lead, and cadmium in the bovine meat and edible offal: Study of their human dietary intake, health risk assessment, and molecular biomarkers. Environ. Sci. Pollut. Res. Int. 2021, 28, 61225–61234. [Google Scholar] [CrossRef]
- Mojska, H.; Kłosiewicz-Latoszek, L.; Jasińska-Melon, E.; Gielecińska, I. Kwasy omega-3. In Normy Żywienia Dla Populacji Polski i Ich Zastosowanie; Jarosz, M., Rychlik, E., Stoś, K., Charzewska, J., Eds.; PZH, Narodowy Instytut Zdrowia Publicznego–Państwowy Zakład Higieny: Warsaw, Poland, 2020; pp. 98–121. Available online: https://ncez.pzh.gov.pl/wp-content/uploads/2021/03/normy_zywienia_2020web.pdf (accessed on 23 January 2022).
- Bernatowicz, A. Podroby–Wartości Odżywcze, Potrawy Z Podrobów–Czy Są Zdrowe I Bezpieczne? 2019. Available online: https://wylecz.to/zywnosc/podroby-wartosci-odzywcze-potrawy-z-podrobow-czy-sa-zdrowe-i-bezpieczne/ (accessed on 23 January 2022).
- Masna, M. Podroby: Rodzaje, Charakterystyka i Wartości Odżywcze. 2019. Available online: https://www.poradnikzdrowie.pl/diety-i-zywienie/co-jesz/podroby-rodzaje-charakterystyka-wartosci-odzywcze-aa-6wSB-4jxv-49uS.html (accessed on 23 January 2022).
- Abbas, M.; Chand, N.; Khan, R.U.; Ahmad, N.; Pervez, U.; Naz, S. Public health risk of heavy metal residues in meat and edible organs of broiler in an intensive production system of a region in Pakistan. Environ. Sci. Pollut. Res. Int. 2019, 26, 23002–23009. [Google Scholar] [CrossRef]
- Adetunji, V.O.; Famakin, I.O.; Chen, J. Lead and cadmium levels in cattle muscle and edible tissues collected from a slaughter slab in Nigeria. Food Addit. Contam. Part B Surveill. 2014, 7, 79–83. [Google Scholar] [CrossRef]
- Giżejewska, A.; Szkoda, J.; Nawrocka, A.; Żmudzki, J.; Giżejewski, Z. Can red deer antlers be used as an indicator of environmental and edible tissues’ trace element contamination? Environ. Sci. Pollut. Res. Int. 2017, 24, 11630–11638. [Google Scholar] [CrossRef] [Green Version]
- Gallagher, C.M.; Chen, J.J.; Kovach, J.S. The relationship between body iron stores and blood and urine cadmium concentrations in US never-smoking, non-pregnant women aged 20–49 years. Environ. Res. 2011, 111, 702–707. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.; Chu, K.-A.; Lin, M.-C.; Chu, Y.-H.; Hung, Y.-M.; Wei, J.C.-C. Newly diagnosed iron deficiency anemia and subsequent autoimmune disease: A matched cohort study in Taiwan. Curr. Med. Res. Opin. 2020, 36, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Aleksandrov, A.P.; Mirkov, I.; Tucovic, D.; Kulas, J.; Zeljkovic, M.; Popovic, D.; Ninkov, M.; Jankovic, S.; Kataranovski, M. Immunomodulation by heavy metals as a contributing factor to inflammatory diseases and autoimmune reactions: Cadmium as an example. Immunol. Lett. 2021, 240, 106–122. [Google Scholar] [CrossRef] [PubMed]
- Dessein, P.H.; Joffe, B.I.; Stanwix, A.E. High sensitivity C-reactive protein as a disease activity marker in rheumatoid arthritis. J. Rheumatol. 2004, 31, 1095–1097. [Google Scholar]
- Abramson, S.B.; Attur, M. Developments in the scientific understanding of osteoarthritis. Arthritis Res. Ther. 2009, 11, 227. [Google Scholar] [CrossRef] [Green Version]
- Prame Kumar, K.; Nicholls, A.J.; Wong, C.H.Y. Partners in crime: Neutrophils and monocytes/macrophages in inflammation and disease. Cell Tissue Res. 2018, 371, 551–565. [Google Scholar] [CrossRef] [Green Version]
- Das, S.C.; Al-Naemi, H.A. Cadmium toxicity: Oxidative stress, inflammation and tissue injury. Occup. Dis. Environ. Med. 2019, 7, 144–163. [Google Scholar] [CrossRef] [Green Version]
- Hossein-Khannazer, N.; Azizi, G.; Eslami, S.; Mohammed, H.A.; Fayyaz, F.; Hosseinzadeh, R.; Usman, A.B.; Kamali, A.N.; Mohammadi, H.; Jadidi-Niaragh, F.; et al. The effects of cadmium exposure in the induction of inflammation. Immunopharmacol. Immunotoxicol. 2020, 42, 1–8. [Google Scholar] [CrossRef]
- Xiao, L.; Zhou, Y.; Ma, J.; Cao, L.; Zhu, C.; Li, W.; Wang, D.; Fan, L.; Ye, Z.; Chen, W. Roles of C-reactive protein on the association between urinary cadmium and type 2 diabetes. Environ. Pollut. 2019, 255 Pt 2, 113341. [Google Scholar] [CrossRef]
- Ma, S.; Zhang, J.; Xu, C.; Da, M.; Xu, Y.; Chen, Y.; Mo, X. Increased serum levels of cadmium are associated with an elevated risk of cardiovascular disease in adults. Environ. Sci. Pollut. Res. 2022, 29, 1836–1844. [Google Scholar] [CrossRef]
- Gong, Z.; Liu, G.; Liu, W.; Zou, H.; Song, R.; Zhao, H.; Yuan, Y.; Gu, J.; Bian, J.; Zhu, J.; et al. The epigenetic regulator BRD4 is involved in cadmium-triggered inflammatory response in rat kidney. Ecotoxicol. Environ. Saf. 2021, 224, 112620. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhu, Y.; Lu, Z.; Guo, W.; Tumen, B.; He, Y.; Chen, C.; Hu, S.; Xu, K.; Wang, Y.; et al. Cadmium induces acute liver injury by inhibiting Nrf2 and the role of NF-κB, NLRP3, and MAPKs signaling pathway. Int. J. Environ. Res. Public Health 2019, 17, 138. [Google Scholar] [CrossRef] [Green Version]
- Larson-Casey, J.L.; Gu, L.; Fiehn, O.; Carter, A.B. Cadmium-mediated lung injury is exacerbated by the persistence of classically activated macrophages. J. Biol. Chem. 2020, 295, 15754–15766. [Google Scholar] [CrossRef] [PubMed]
- Salama, S.A.; Arab, H.H.; Hassan, M.H.; Al Robaian, M.M.; Maghrabi, I.A. Cadmium-induced hepatocellular injury: Modulatory effects of γ-glutamyl cysteine on the biomarkers of inflammation, DNA damage, and apoptotic cell death. J. Trace Elem. Med. Biol. 2019, 52, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Beebe, A.M.; Cua, D.J.; de Waal Malefyt, R. The role of interleukin-10 in autoimmune disease: Systemic lupus erythematosus (SLE) and multiple sclerosis (MS). Cytokine Growth Factor Rev. 2002, 13, 403–412. [Google Scholar] [CrossRef]
- Saraiva, M.; Vieira, P.; O’Garra, A. Biology and therapeutic potential of interleukin-10. J. Exp. Med. 2020, 217, e20190418. [Google Scholar] [CrossRef] [Green Version]
- Howes, A.; Stimpson, P.; Redford, P.; Gabrysova, L.; O’Garra, A. Interleukin-10: Cytokines in anti-inflammation and tolerance. In Cytokine Frontiers: Regulation of Immune Responses in Health and Disease; Yoshimoto, T., Yoshimoto, T., Eds.; Springer: Tokyo, Japan, 2014; pp. 327–352. [Google Scholar]
- Carter, N.A.; Rosser, E.C.; Mauri, C. Interleukin-10 produced by B cells is crucial for the suppression of Th17/Th1 responses, induction of T regulatory type 1 cells and reduction of collagen-induced arthritis. Arthritis Res. Ther. 2012, 14, R32. [Google Scholar] [CrossRef] [Green Version]
- Greenhill, C.J.; Jones, G.W.; Nowell, M.A.; Newton, Z.; Harvey, A.K.; Moideen, A.N.; Collins, F.L.; Bloom, A.C.; Coll, R.C.; Robertson, A.A.B.; et al. Interleukin-10 regulates the inflammasome-driven augmentation of inflammatory arthritis and joint destruction. Arthritis Res. Ther. 2014, 16, 419. [Google Scholar] [CrossRef] [Green Version]
- Akinyemi, A.J.; Adeniyi, P.A. Effect of essential oils from ginger (Zingiber officinale) and turmeric (Curcuma longa) rhizomes on some inflammatory biomarkers in cadmium induced neurotoxicity in rats. J. Toxicol. 2018, 4109491. [Google Scholar] [CrossRef] [Green Version]
- Häggqvist, B.; Hultman, P. Interleukin-10 in murine metal-induced systemic autoimmunity. Clin. Exp. Immunol. 2005, 141, 422–431. [Google Scholar] [CrossRef]
- Odewumi, C.; Latinwo, L.M.; Sinclair, A.; Badisa, V.L.D.; Abdullah, A.; Badisa, R.B. Effect of cadmium on the expression levels of interleukin-1α and interleukin-10 cytokines in human lung cells. Mol. Med. Rep. 2015, 12, 6422–6426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castrogiovanni, P.; Di Rosa, M.; Ravalli, S.; Castorina, A.; Guglielmino, C.; Imbesi, R.; Vecchio, M.; Drago, F.; Szychlinska, M.A.; Musumeci, G. Moderate physical activity as a prevention method for knee osteoarthritis and the role of synoviocytes as biological key. Int. J. Mol. Sci. 2019, 20, 511. [Google Scholar] [CrossRef] [Green Version]
- El-Ali, Z.; El-Kassas, G.; Ziade, F.M.; Shivappa, N.; Hébert, J.R.; Zmerly, H.; Bissar, N. Evaluation of circulating levels of interleukin-10 and interleukin-16 and dietary inflammatory index in Lebanese knee osteoarthritis patients. Heliyon 2021, 7, e07551. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A.; Khosraviani, S.; Noel, S.; Mohan, D.; Donner, T.; Hamad, A.R.A. Interleukin-10 paradox: A potent immunoregulatory cytokine that has been difficult to harness for immunotherapy. Cytokine 2015, 74, 27–34. [Google Scholar] [CrossRef] [Green Version]
- Hajialilo, M.; Ghorbanihaghjo, A.; Maddahi, S.; Khabbazi, A.; Mahdavi, A.M.; Rashtchizadeh, N. Association between serum toll-like receptor 4 and 8-hydroxy-2’-deoxyguanosine levels with disease activity in rheumatoid arthritis patients. Egypt. Rheumatol. 2020, 42, 95–99. [Google Scholar] [CrossRef]
- Sukkar, S.G.; Rossi, E. Oxidative stress and nutritional prevention in autoimmune rheumatic diseases. Autoimmun. Rev. 2014, 3, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Pawlas, N.; Olewińska, E.; Markiewicz-Górka, I.; Kozłowska, A.; Januszewska, L.; Lundh, T.; Januszewska, E.; Pawlas, K. Oxidative damage of DNA in subjects occupationally exposed to lead. Adv. Clin. Exp. Med. 2017, 26, 939–945. [Google Scholar] [CrossRef]
- Xu, P.; Mo, Z.; Wu, L.; Chen, W.; He, S.; Chen, Y.; Xu, D.; Xiang, J.; Chen, Z.; Lou, X.; et al. Elevated cadmium and 8-hydroxy-2’-deoxyguanosine (8-OHdG) levels in residents living near electroplating industries. Environ. Sci. Pollut. Res. Int. 2021, 28, 34427–34435. [Google Scholar] [CrossRef]
| Characteristics | IA Patients (RA, PsA, AS) n = 51 | Controls n = 46 | Overall n = 97 | p1 |
|---|---|---|---|---|
| Age (years), mean ± SD | 48.1 ± 13.8 | 47.5 ± 13.7 | 47.8 ± 13.6 | n.s. |
| Gender male, n (%) | 30 (58.8%) | 28 (60.9%) | 58 (59.8%) | n.s. |
| Place of residence | ||||
| Big city, n (%) | 24 (47.1%) | 29 (64.4%) | 53 (55.2%) | n.s. |
| Education level | ||||
| Higher, n (%) | 26 (51%) | 23 (51%) | 49 (51%) | n.s. |
| Potential sources of cadmium | ||||
| Exposure to low-pollutant emissions (burning coal and rubbish in domestic furnaces, living/working in a heavy-traffic zone), n (%) | 45 (88.2%) | 33 (71.7%) | 78 (80.4%) | n.s. |
| Diet | ||||
| Frequent consumption of offal (≥once a week), | ||||
| n (%) | 25 (49%) | 11 (24%) | 36 (37.5%) | 0.0085 |
| Frequent consumption of legumes | ||||
| (≥once a week) n (%) | 25 (49%) | 19 (41.3%) | 44 (45.4%) | n.s. |
| Using colour cosmetics (lipstick, powder, eye shadows), n (% of females) | 16 (76.2%) | 9 (50%) | 25 (64.1%) | n.s. |
| Smoking status | n.s. | |||
| Never smoked, n (%) | 33 (64.7%) | 24(53.3%) | 57 (59.4%) | |
| Former smoker, n (%) | 11 (21.6%) | 14 (31.1%) | 25 (26.0%) | |
| Current smoker, n (%) | 7 (13.7%) | 7 (15.6%) | 14 (14.6%) | |
| Passive smoker, n (%) | 15 (29.4%) | 12 (26.7%) | 27 (28.1%) | n.s. |
| Occupational exposure to Cd, n (%) | 8 (15.7%) | 4 (8.7%) | 12 (12.4%) | n.s. |
| Disease duration (years), mean ± SD | 4.5 ± 6.6 | |||
| Disease, n (%) | ||||
| RA | 21 (41.2%) | |||
| AS | 17 (33.3%) | |||
| PsA | 13 (25.5%) | |||
| Disease activity | ||||
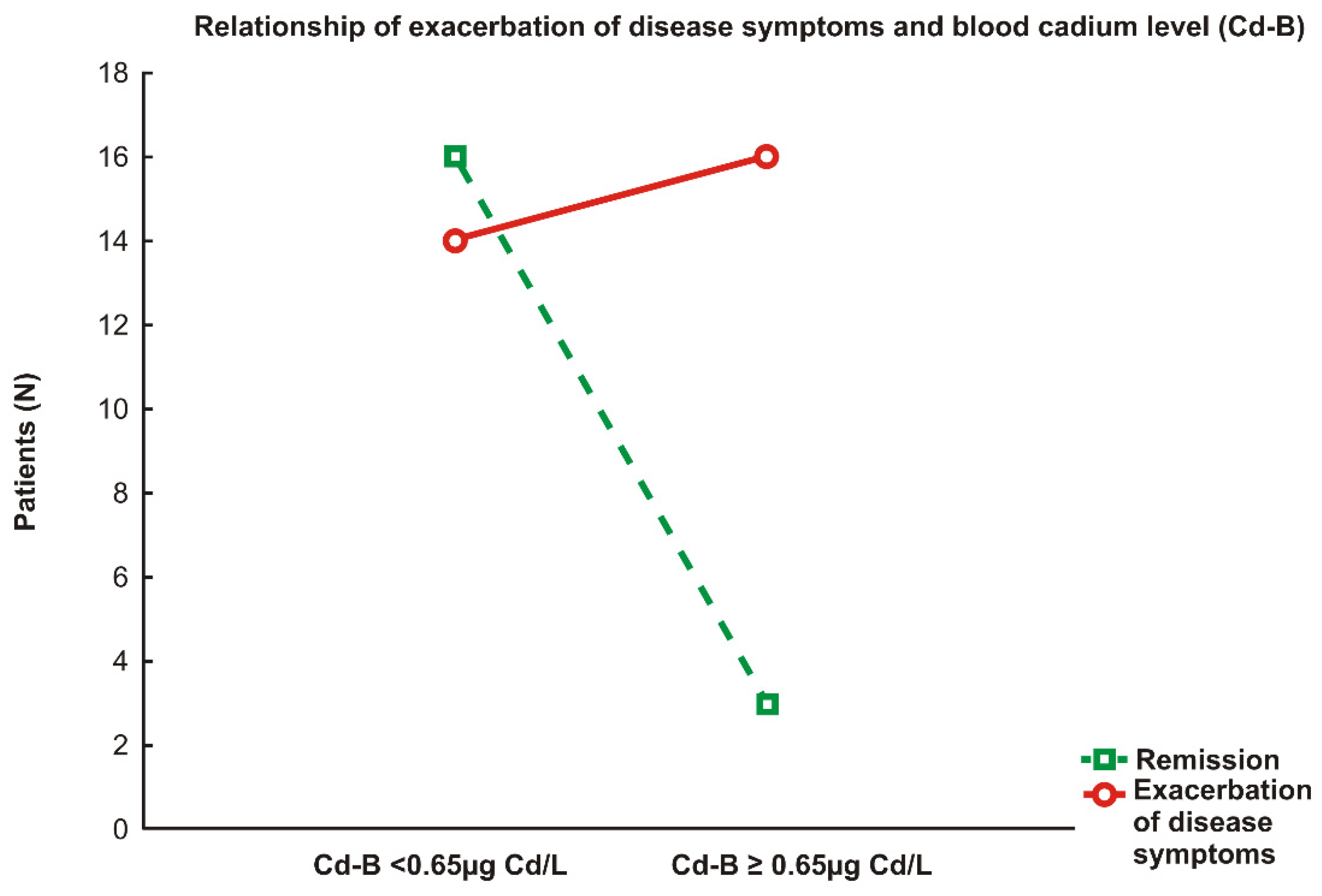
| Remission, n (%) | 20 (39.2%) | |||
| Exacerbation of disease symptoms, n (%) | 31 (68.0%) | |||
| Low | 13 (25.5%) | |||
| Moderate | 5 (5.0%) | |||
| High | 13 (25.5%) | |||
| Blood cadmium concentration | ||||
| Mean ± SD (μg/L) | 0.67 ± 0.51 | 0.40 ± 0.47 | 0.543 ± 0.48 | 0.00003 |
| Geometric mean (μg/L) | 0.532 | 0.258 | 0.373 | |
| Range (min–max) (μg/L) | (0.13–2.16) | (0.00–2.15) | (0.00–2.16) | |
| Parameters | IA Patients (RA, PsA, AS) n = 51 | Controls n = 46 | Overall n = 97 | p1 |
|---|---|---|---|---|
| WBC (×103/μL) | 7.74 ± 2.28 | 6.19 ± 1.52 | 7.01 ± 2.1 | 0.0003 |
| Lymphocytes (×103/μL) | 1.93 ± 0.69 | 2.01 ± 0.63 | 1.97 ± 0.66 | n.s. |
| Neutrophils (×103/μL) | 4.90 ± 2.22 | 3.39 ± 1.11 | 4.19 ± 1.93 | 0.0001 |
| Monocytes (×103/μL) | 0.67 ± 0.22 | 0.59 ± 0.49 | 0.63 ± 0.37 | 0.001 |
| Haemoglobin (g/dL) | 13.75 ± 1.68 | 15.02 ± 1.45 | 14.34 ± 1.69 | 0.0001 |
| RBC (×103/μL) | 4.67 ± 0.49 | 4.92 ± 0.48 | 4.79 ± 0.50 | 0.01 |
| HCT (%) | 40.37 ± 7.45 | 43.95 ± 3.92 | 41.73 ± 6.55 | 0.0006 |
| MCV (fL) | 89.29 ± 4.95 | 90.32 ± 4.69 | 89.98 ± 4.83 | n.s. |
| MCH (pg) | 29.56 ± 2.26 | 30.50 ± 1.54 | 30.00 ± 2.00 | 0.02 |
| MCHC (g/dL) | 32.70 ± 1.25 | 33.74 ± 0.93 | 33.10 ± 1.24 | 0.0004 |
| PLT (×103/μL) | 287.8 ± 98.3 | 247.2 ± 63.7 | 273.2 ± 95.4 | n.s. |
| AST (U/L) | 26.75 ± 11.24 | 25.59 ± 7.61 | 26.31 ± 9.97 | n.s. |
| ALT (U/L) | 29.45 ± 22.73 | 27.81 ± 14.63 | 28.83 ± 19.94 | n.s |
| CRE (mg/dL) | 0.93 ± 0.19 | 0.98 ± 0.15 | 0.95 ± 0.17 | 0.03 |
| ESR (mm/h) | 16.8 ± 19.6 | 8.07 ± 7.7 | 12.7 ± 15.8 | 0.005 |
| hs-CRP (mg/L) | 11.03 ± 26.8 | 2.30 ± 3.6 | 7.00 ± 20.2 | 0.005 |
| COX-2 (ng/mL) | 18.46 ± 11.8 | 9.90 ± 11.91 | 14.36 ± 12.57 | 0.00005 |
| 8-OHdG (ng/mL) | 12.02 ± 12.6 | 7.03 ± 3.2 | 9.8 ± 9.8 | n.s. |
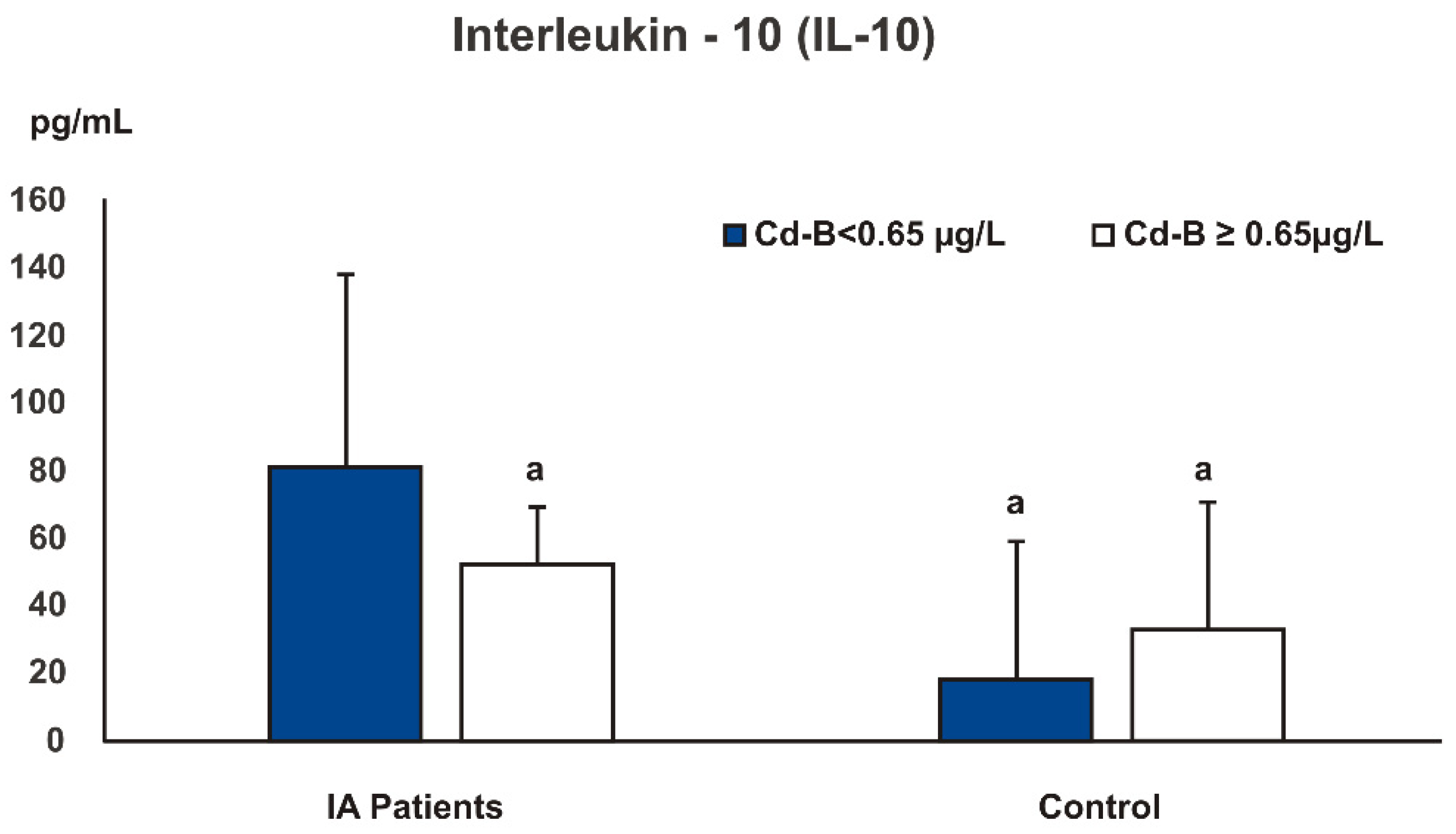
| IL-10 (pg/mL) | 69.87 ± 52.90 | 19.49 ± 20.4 | 45.72 ± 47.76 | 0.000001 |
| Age (Years) | Level of Education | Current Smoking | Smoking in the Past | Passive Smoking | Living in a Heavy-Traffic Zone | |
|---|---|---|---|---|---|---|
| Cd-B | r = 0.25 * | r = –0.20 * | r = 0.46 ** | r = 0.36 ** | r = 0.29 ** | r = 0.32 ** |
| ESR | hs-CRP | MCHC | COX-2 | |
|---|---|---|---|---|
| Cd-B | r = 0.27 ** | r = 0.24 * | r = –0.34 ** | r = 0.30 ** |
| Estimate (B) | p | Wald chi2 | Wald p | OR | 95% CI | |
|---|---|---|---|---|---|---|
| Cd-B (quartile intervals) | 0.646 | 0.001 | 11.17 | 0.0008 | 1.9 | 1.3–2.8 |
| Variables | Estimate (B) | p | Wald chi2 | Wald p | OR | 95% CI |
|---|---|---|---|---|---|---|
| Constant (B0) | 9.90 | 0.046 | 4.08 | 0.043 | 1.99 × 104 | 1.2–3.4 × 108 |
| Cd-B ≥ 0.65 μg/L (≥upper quartile) | 1.49 | 0.040 | 4.32 | 0.038 | 4.4 | 1.1–18.4 |
| 8-OHdG ≥ 7.66 ng/mL (≥median) | 2.36 | 0.001 | 12.05 | 0.001 | 10.6 | 2.7–40.7 |
| COX-2 ≥ 22.9 ng/mL (≥upper quartile) | 1.76 | 0.016 | 6.01 | 0.014 | 5.8 | 1.4–24.2 |
| MCH (pg) | –0.39 | 0.022 | 5.47 | 0.019 | 0.7 | 0.5–0.9 |
| Frequent consumption of offal (≥once a week) | 1.29 | 0.025 | 5.19 | 0.023 | 3.6 | 1.2–11.1 |
| Parameters | Disease Severity Rating Scale |
|---|---|
| Gender (female) | r = 0.19 *, p = 0.045 |
| Level of education (higher) | r = 0.27 **, p = 0.004 |
| ESR | r = 0.39 **, p = 0.00005 |
| ESR (norm–0, above the norm–1) | r = 0.27 **, p = 0.006 |
| hs-CRP | r = 0.30 **, p = 0.002 |
| hs-CRP (norm–0, above the norm–1) | r = 0.38 **, p = 0.00009 |
| HB | r = −0.19 *, p = 0.044 |
| HCT | r = −0.20 *, p = 0.036 |
| PLT | r = 0.23 *, p = 0.015 |
| MCV | r = −0.20 *, p = 0.039 |
| MCH | r = −0.22 *, p = 0.019 |
| MCHC | r = −0.30 **, p = 0.002 |
| Creatinine | r = −0.27 **, p = 0.005 |
| Disease duration (years) | r = −0.21 *, p = 0.044 |
| IL-10 | r = −0.51 **, p = 0.0000 |
| 8-OHdG ≥ 10.1 ng/mL (≥upper quartile–1) | r = 0.31 *, p = 0.015 |
| Cd-B ≥ 0.65 μg/L (≥upper quartile–1) | r = 0.23 *, p = 0.016 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Markiewicz-Górka, I.; Chowaniec, M.; Martynowicz, H.; Wojakowska, A.; Jaremków, A.; Mazur, G.; Wiland, P.; Pawlas, K.; Poręba, R.; Gać, P. Cadmium Body Burden and Inflammatory Arthritis: A Pilot Study in Patients from Lower Silesia, Poland. Int. J. Environ. Res. Public Health 2022, 19, 3099. https://doi.org/10.3390/ijerph19053099
Markiewicz-Górka I, Chowaniec M, Martynowicz H, Wojakowska A, Jaremków A, Mazur G, Wiland P, Pawlas K, Poręba R, Gać P. Cadmium Body Burden and Inflammatory Arthritis: A Pilot Study in Patients from Lower Silesia, Poland. International Journal of Environmental Research and Public Health. 2022; 19(5):3099. https://doi.org/10.3390/ijerph19053099
Chicago/Turabian StyleMarkiewicz-Górka, Iwona, Małgorzata Chowaniec, Helena Martynowicz, Anna Wojakowska, Aleksandra Jaremków, Grzegorz Mazur, Piotr Wiland, Krystyna Pawlas, Rafał Poręba, and Paweł Gać. 2022. "Cadmium Body Burden and Inflammatory Arthritis: A Pilot Study in Patients from Lower Silesia, Poland" International Journal of Environmental Research and Public Health 19, no. 5: 3099. https://doi.org/10.3390/ijerph19053099
APA StyleMarkiewicz-Górka, I., Chowaniec, M., Martynowicz, H., Wojakowska, A., Jaremków, A., Mazur, G., Wiland, P., Pawlas, K., Poręba, R., & Gać, P. (2022). Cadmium Body Burden and Inflammatory Arthritis: A Pilot Study in Patients from Lower Silesia, Poland. International Journal of Environmental Research and Public Health, 19(5), 3099. https://doi.org/10.3390/ijerph19053099








