Resistance Training Improves Sleep and Anti-Inflammatory Parameters in Sarcopenic Older Adults: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
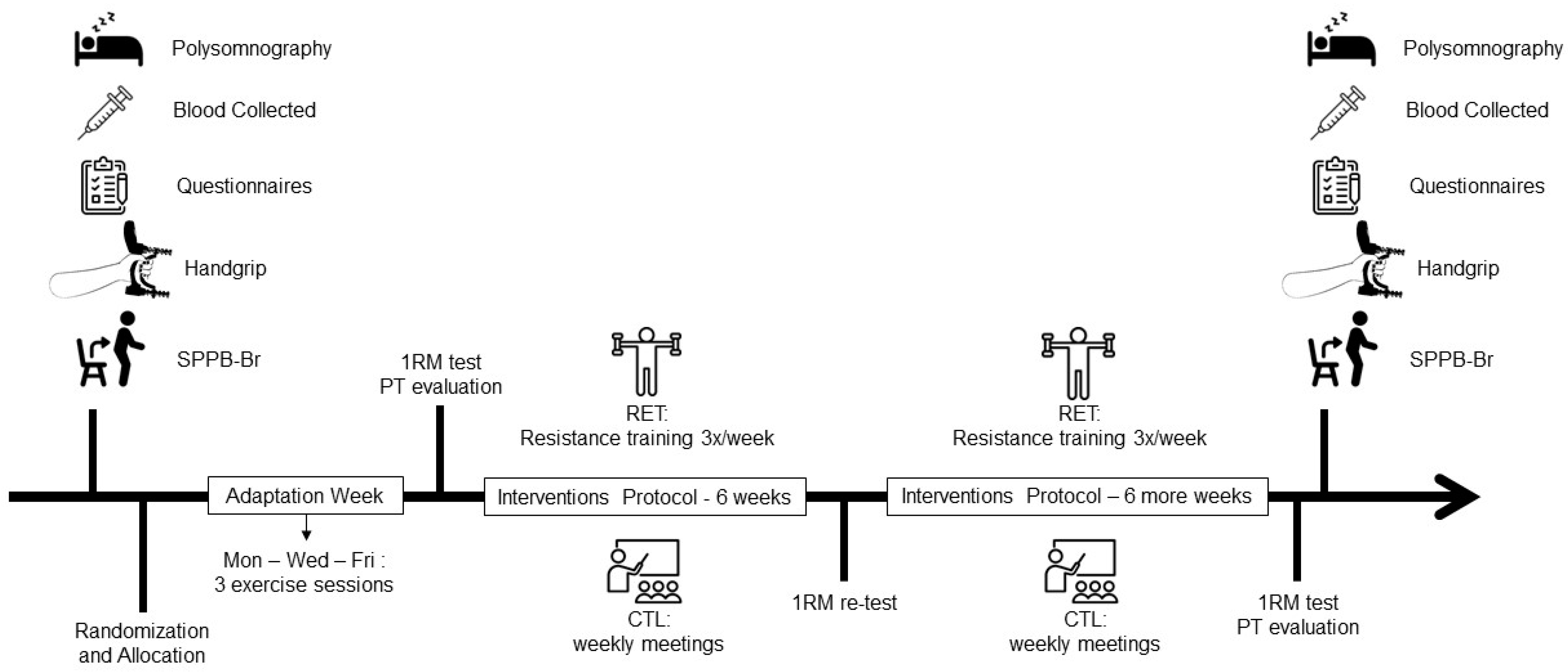
2.1. Study Design
2.2. Sarcopenia Identification
2.3. Sleep Measures and Definitions
2.4. Evaluation of Muscular Strength and Torque
2.5. Hormonal, Metabolic and Inflammatory Markers
2.6. Resistance Training Protocol
2.7. Calculation of Sample Size and Statistical Analysis
3. Results
3.1. Effects of RET on Sarcopenia Parameter Outcomes
3.2. Effects of RET on Objective and Subjective Sleep Parameter Outcomes
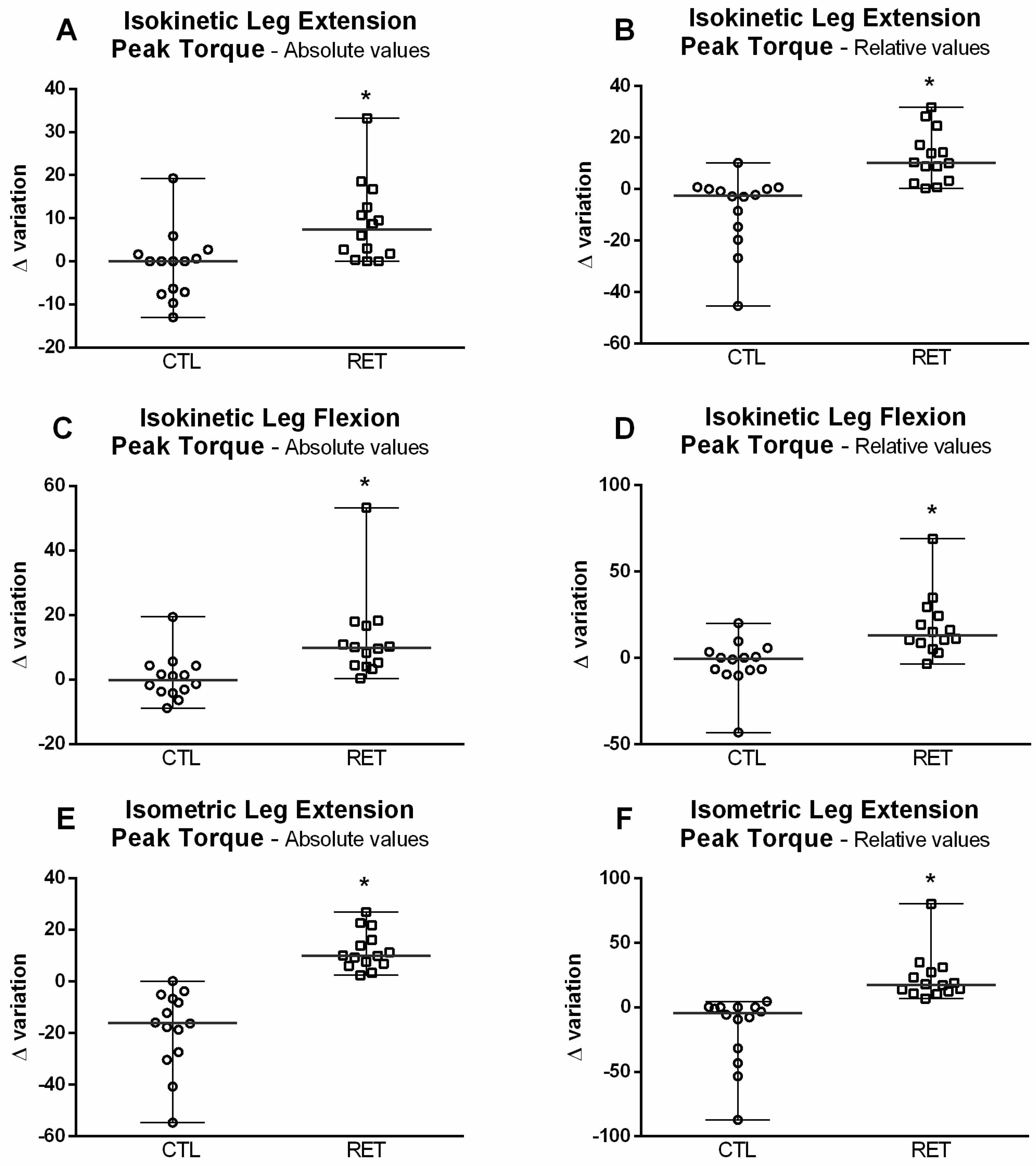
3.3. Effects of RET on Skeletal Muscle Strength
3.4. Changes in Hormonal, Metabolic and Inflammatory Marker Outcomes after the RET Intervention
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, J.M. Clues to the functions of mammalian sleep. Nature 2005, 437, 1264–1271. [Google Scholar] [CrossRef] [PubMed]
- Siegel, J.M. Sleep function: An evolutionary perspective. Lancet Neurol. 2022, 21, 937–946. [Google Scholar] [CrossRef] [PubMed]
- Dattilo, M.; Antunes, H.K.; Medeiros, A.; Monico Neto, M.; Souza, H.S.; Tufik, S.; de Mello, M.T. Sleep and muscle recovery: Endocrinological and molecular basis for a new and promising hypothesis. Med. Hypotheses 2011, 77, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Dattilo, M.; Antunes, H.K.; Medeiros, A.; Monico-Neto, M.; Souza Hde, S.; Lee, K.S.; Tufik, S.; de Mello, M.T. Paradoxical sleep deprivation induces muscle atrophy. Muscle Nerve 2012, 45, 431–433. [Google Scholar] [CrossRef]
- de Sa Souza, H.; Antunes, H.K.; Dattilo, M.; Lee, K.S.; Monico-Neto, M.; de Campos Giampa, S.Q.; Phillips, S.M.; Tufik, S.; de Mello, M.T. Leucine supplementation is anti-atrophic during paradoxical sleep deprivation in rats. Amino Acids 2016, 48, 949–957. [Google Scholar] [CrossRef] [PubMed]
- Monico-Neto, M.; Dattilo, M.; Ribeiro, D.A.; Lee, K.S.; de Mello, M.T.; Tufik, S.; Antunes, H.K.M. REM sleep deprivation impairs muscle regeneration in rats. Growth Factors 2017, 35, 12–18. [Google Scholar] [CrossRef]
- Dattilo, M.; Antunes, H.K.M.; Galbes, N.M.N.; Monico-Neto, M.; Souza, H.D.S.; Dos Santos Quaresma, M.V.L.; Lee, K.S.; Ugrinowitsch, C.; Tufik, S.; DE Mello, M.T. Effects of Sleep Deprivation on Acute Skeletal Muscle Recovery after Exercise. Med. Sci. Sports Exerc. 2020, 52, 507–514. [Google Scholar] [CrossRef]
- Piovezan, R.D.; Hirotsu, C.; Moizinho, R.; de Sa Souza, H.; D’Almeida, V.; Tufik, S.; Poyares, D. Associations between sleep conditions and body composition states: Results of the EPISONO study. J. Cachexia Sarcopenia Muscle 2019, 10, 962–973. [Google Scholar] [CrossRef]
- Piovezan, R.D.; Abucham, J.; Dos Santos, R.V.; Mello, M.T.; Tufik, S.; Poyares, D. The impact of sleep on age-related sarcopenia: Possible connections and clinical implications. Ageing Res. Rev. 2015, 23, 210–220. [Google Scholar] [CrossRef]
- Moraes, W.; Piovezan, R.; Poyares, D.; Bittencourt, L.R.; Santos-Silva, R.; Tufik, S. Effects of aging on sleep structure throughout adulthood: A population-based study. Sleep Med. 2014, 15, 401–409. [Google Scholar] [CrossRef]
- Gulia, K.K.; Kumar, V.M. Sleep disorders in the elderly: A growing challenge. Psychogeriatrics 2018, 18, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Fulop, T.; Larbi, A.; Witkowski, J.M. Human Inflammaging. Gerontology 2019, 65, 495–504. [Google Scholar] [CrossRef] [PubMed]
- Fulop, T.; Larbi, A.; Pawelec, G.; Khalil, A.; Cohen, A.A.; Hirokawa, K.; Witkowski, J.M.; Franceschi, C. Immunology of Aging: The Birth of Inflammaging. Clin. Rev. Allergy Immunol. 2021, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Tessema, B.; Sack, U.; Konig, B.; Serebrovska, Z.; Egorov, E. Effects of Intermittent Hypoxia in Training Regimes and in Obstructive Sleep Apnea on Aging Biomarkers and Age-Related Diseases: A Systematic Review. Front. Aging Neurosci. 2022, 14, 878278. [Google Scholar] [CrossRef] [PubMed]
- Irwin, M.R.; Olmstead, R.; Carroll, J.E. Sleep Disturbance, Sleep Duration, and Inflammation: A Systematic Review and Meta-Analysis of Cohort Studies and Experimental Sleep Deprivation. Biol. Psychiatry 2016, 80, 40–52. [Google Scholar] [CrossRef]
- Tuttle, C.S.L.; Thang, L.A.N.; Maier, A.B. Markers of inflammation and their association with muscle strength and mass: A systematic review and meta-analysis. Ageing Res. Rev. 2020, 64, 101185. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Bano, G.; Trevisan, C.; Carraro, S.; Solmi, M.; Luchini, C.; Stubbs, B.; Manzato, E.; Sergi, G.; Veronese, N. Inflammation and sarcopenia: A systematic review and meta-analysis. Maturitas 2017, 96, 10–15. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef]
- Sharma, B.; Dabur, R. Role of Pro-inflammatory Cytokines in Regulation of Skeletal Muscle Metabolism: A Systematic Review. Curr. Med. Chem. 2020, 27, 2161–2188. [Google Scholar] [CrossRef] [PubMed]
- Pan, L.; Xie, W.; Fu, X.; Lu, W.; Jin, H.; Lai, J.; Zhang, A.; Yu, Y.; Li, Y.; Xiao, W. Inflammation and sarcopenia: A focus on circulating inflammatory cytokines. Exp. Gerontol. 2021, 154, 111544. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.Y.; Ho, K.H.; Chen, H.C.; Chien, M.Y. Exercise training improves sleep quality in middle-aged and older adults with sleep problems: A systematic review. J. Physiother. 2012, 58, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Walsh, N.P.; Gleeson, M.; Shephard, R.J.; Gleeson, M.; Woods, J.A.; Bishop, N.C.; Fleshner, M.; Green, C.; Pedersen, B.K.; Hoffman-Goetz, L.; et al. Position statement. Part one: Immune function and exercise. Exerc. Immunol. Rev. 2011, 17, 6–63. [Google Scholar] [PubMed]
- Syed-Abdul, M.M.; McClellan, C.L.; Parks, E.J.; Ball, S.D. Effects of a resistance training community programme in older adults. Ageing Soc. 2022, 42, 1863–1878. [Google Scholar] [CrossRef]
- Sardeli, A.V.; Tomeleri, C.M.; Cyrino, E.S.; Fernhall, B.; Cavaglieri, C.R.; Chacon-Mikahil, M.P.T. Effect of resistance training on inflammatory markers of older adults: A meta-analysis. Exp. Gerontol. 2018, 111, 188–196. [Google Scholar] [CrossRef]
- Monico-Neto, M.; Antunes, H.K.; Lee, K.S.; Phillips, S.M.; Giampa, S.Q.; Souza Hde, S.; Dattilo, M.; Medeiros, A.; de Moraes, W.M.; Tufik, S.; et al. Resistance training minimizes catabolic effects induced by sleep deprivation in rats. Appl. Physiol. Nutr. Metab.=Physiol. Appl. Nutr. Metab. 2015, 40, 1143–1150. [Google Scholar] [CrossRef]
- National Institute on Alcohol Abuse and Alcoholism (NIAAA). Helping Patients Who Drink Too Much: A Clinician’s Guide; National Institute on Alcohol Abuse and Alcoholism (NIAAA): Bethesda, MD, USA, 2005; pp. 1–33. [Google Scholar]
- Hurtig-Wennlof, A.; Hagstromer, M.; Olsson, L.A. The International Physical Activity Questionnaire modified for the elderly: Aspects of validity and feasibility. Public Health Nutr. 2010, 13, 1847–1854. [Google Scholar] [CrossRef]
- Iber, C.; Ancoli-Israel, S.; Chesson, A.L., Jr.; Quan, S.F. The AASM Manual for the Scoring of Sleep and Associated Events, 1st ed.; American Academy of Sleep Medicine: Downers Grove Township, IL, USA, 2007; p. 59. [Google Scholar]
- Berry, R.B.; Budhiraja, R.; Gottlieb, D.J.; Gozal, D.; Iber, C.; Kapur, V.K.; Marcus, C.L.; Mehra, R.; Parthasarathy, S.; Quan, S.F.; et al. Rules for scoring respiratory events in sleep: Update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J. Clin. Sleep Med. JCSM Off. Publ. Am. Acad. Sleep Med. 2012, 8, 597–619. [Google Scholar] [CrossRef]
- Tufik, S.; Santos-Silva, R.; Taddei, J.A.; Bittencourt, L.R. Obstructive sleep apnea syndrome in the Sao Paulo Epidemiologic Sleep Study. Sleep Med. 2010, 11, 441–446. [Google Scholar] [CrossRef]
- Johns, M.W. A new method for measuring daytime sleepiness: The Epworth sleepiness scale. Sleep 1991, 14, 540–545. [Google Scholar] [CrossRef]
- Buysse, D.J.; Reynolds, C.F., III; Monk, T.H.; Berman, S.R.; Kupfer, D.J. The Pittsburgh Sleep Quality Index: A new instrument for psychiatric practice and research. Psychiatry Res. 1989, 28, 193–213. [Google Scholar] [CrossRef] [PubMed]
- Bastien, C.H.; Vallieres, A.; Morin, C.M. Validation of the Insomnia Severity Index as an outcome measure for insomnia research. Sleep Med. 2001, 2, 297–307. [Google Scholar] [CrossRef]
- Vincent, K.R.; Braith, R.W.; Feldman, R.A.; Kallas, H.E.; Lowenthal, D.T. Improved cardiorespiratory endurance following 6 months of resistance exercise in elderly men and women. Arch. Intern. Med. 2002, 162, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Vincent, K.R.; Braith, R.W.; Feldman, R.A.; Magyari, P.M.; Cutler, R.B.; Persin, S.A.; Lennon, S.L.; Gabr, A.H.; Lowenthal, D.T. Resistance exercise and physical performance in adults aged 60 to 83. J. Am. Geriatr. Soc. 2002, 50, 1100–1107. [Google Scholar] [CrossRef] [PubMed]
- Bertani, R.F.; Bonardi, J.M.; de Oliveira Campos, G.; Goncalves Lima, L.; Ferriolli, E.; Moriguti, J.C.; da Costa Lima, N.K. One repetition maximum test in octogenarian: Evaluation of blood pressure. J. Sports Med. Phys. Fit. 2017, 57, 695–702. [Google Scholar] [CrossRef]
- Bottaro, M.; Russo, A.F.; de Oliveira, R.J. The effects of rest interval on quadriceps torque during an isokinetic testing protocol in elderly. J. Sports Sci. Med. 2005, 4, 285–290. [Google Scholar]
- Charlier, R.; Mertens, E.; Lefevre, J.; Thomis, M. Muscle mass and muscle function over the adult life span: A cross-sectional study in Flemish adults. Arch. Gerontol. Geriatr. 2015, 61, 161–167. [Google Scholar] [CrossRef]
- American College of Sports, M.; Chodzko-Zajko, W.J.; Proctor, D.N.; Fiatarone Singh, M.A.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S. American College of Sports Medicine position stand. Exercise and physical activity for older adults. Med. Sci. Sports Exerc. 2009, 41, 1510–1530. [Google Scholar] [CrossRef]
- Csapo, R.; Alegre, L.M. Effects of resistance training with moderate vs heavy loads on muscle mass and strength in the elderly: A meta-analysis. Scand. J. Med. Sci. Sports 2015, 26, 995–1006. [Google Scholar] [CrossRef]
- Garber, C.E.; Blissmer, B.; Deschenes, M.R.; Franklin, B.A.; Lamonte, M.J.; Lee, I.M.; Nieman, D.C.; Swain, D.P.; American College of Sports Medicine. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: Guidance for prescribing exercise. Med. Sci. Sports Exerc. 2011, 43, 1334–1359. [Google Scholar] [CrossRef]
- Jensen, S.M.; Bechshoft, C.J.L.; Heisterberg, M.F.; Schjerling, P.; Andersen, J.L.; Kjaer, M.; Mackey, A.L. Macrophage Subpopulations and the Acute Inflammatory Response of Elderly Human Skeletal Muscle to Physiological Resistance Exercise. Front. Physiol. 2020, 11, 811. [Google Scholar] [CrossRef]
- Abd El-Kader, S.M.; Al-Jiffri, O.H. Aerobic exercise modulates cytokine profile and sleep quality in elderly. Afr. Health Sci. 2019, 19, 2198–2207. [Google Scholar] [CrossRef] [PubMed]
- Phu, S.; Boersma, D.; Duque, G. Exercise and Sarcopenia. J. Clin. Densitom. Off. J. Int. Soc. Clin. Densitom. 2015, 18, 488–492. [Google Scholar] [CrossRef] [PubMed]
- Marzetti, E.; Calvani, R.; Tosato, M.; Cesari, M.; Di Bari, M.; Cherubini, A.; Broccatelli, M.; Savera, G.; D’Elia, M.; Pahor, M.; et al. Physical activity and exercise as countermeasures to physical frailty and sarcopenia. Aging Clin. Exp. Res. 2017, 29, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.Z.; No, M.H.; Heo, J.W.; Park, D.H.; Kang, J.H.; Kim, S.H.; Kwak, H.B. Role of exercise in age-related sarcopenia. J. Exerc. Rehabil. 2018, 14, 551–558. [Google Scholar] [CrossRef]
- Lohne-Seiler, H.; Torstveit, M.K.; Anderssen, S.A. Traditional versus functional strength training: Effects on muscle strength and power in the elderly. J. Aging Phys. Act. 2013, 21, 51–70. [Google Scholar] [CrossRef]
- Martin, H.J.; Yule, V.; Syddall, H.E.; Dennison, E.M.; Cooper, C.; Aihie Sayer, A. Is hand-held dynamometry useful for the measurement of quadriceps strength in older people? A comparison with the gold standard Bodex dynamometry. Gerontology 2006, 52, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Zapparoli, F.Y.; Riberto, M. Isokinetic Evaluation of the Hip Flexor and Extensor Muscles: A Systematic Review. J. Sport Rehabil. 2017, 26, 556–566. [Google Scholar] [CrossRef]
- Rantanen, T.; Avlund, K.; Suominen, H.; Schroll, M.; Frandin, K.; Pertti, E. Muscle strength as a predictor of onset of ADL dependence in people aged 75 years. Aging Clin. Exp. Res. 2002, 14, 10–15. [Google Scholar]
- Ramirez-Campillo, R.; Castillo, A.; de la Fuente, C.I.; Campos-Jara, C.; Andrade, D.C.; Alvarez, C.; Martinez, C.; Castro-Sepulveda, M.; Pereira, A.; Marques, M.C.; et al. High-speed resistance training is more effective than low-speed resistance training to increase functional capacity and muscle performance in older women. Exp. Gerontol. 2014, 58, 51–57. [Google Scholar] [CrossRef]
- Ramirez-Campillo, R.; Alvarez, C.; Garcia-Hermoso, A.; Celis-Morales, C.; Ramirez-Velez, R.; Gentil, P.; Izquierdo, M. High-speed resistance training in elderly women: Effects of cluster training sets on functional performance and quality of life. Exp. Gerontol. 2018, 110, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Viana, V.A.; Esteves, A.M.; Boscolo, R.A.; Grassmann, V.; Santana, M.G.; Tufik, S.; de Mello, M.T. The effects of a session of resistance training on sleep patterns in the elderly. Eur. J. Appl. Physiol. 2012, 112, 2403–2408. [Google Scholar] [CrossRef] [PubMed]
- Uchida, S.; Shioda, K.; Morita, Y.; Kubota, C.; Ganeko, M.; Takeda, N. Exercise effects on sleep physiology. Front. Neurol. 2012, 3, 48. [Google Scholar] [CrossRef]
- Monico-Neto, M.; Antunes, H.K.; Dattilo, M.; Medeiros, A.; Souza, H.S.; Lee, K.S.; de Melo, C.M.; Tufik, S.; de Mello, M.T. Resistance exercise: A non-pharmacological strategy to minimize or reverse sleep deprivation-induced muscle atrophy. Med. Hypotheses 2013, 80, 701–705. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, E.T.; Villanueva, M.; West, D.D.; Phillips, S.M. Are acute post-resistance exercise increases in testosterone, growth hormone, and IGF-1 necessary to stimulate skeletal muscle anabolism and hypertrophy? Med. Sci. Sports Exerc. 2013, 45, 2044–2051. [Google Scholar] [CrossRef]
- Santos, R.V.; Tufik, S.; De Mello, M.T. Exercise, sleep and cytokines: Is there a relation? Sleep Med. Rev. 2007, 11, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Abedelmalek, S.; Souissi, N.; Chtourou, H.; Denguezli, M.; Aouichaoui, C.; Ajina, M.; Aloui, A.; Dogui, M.; Haddouk, S.; Tabka, Z. Effects of partial sleep deprivation on proinflammatory cytokines, growth hormone, and steroid hormone concentrations during repeated brief sprint interval exercise. Chronobiol. Int. 2013, 30, 502–509. [Google Scholar] [CrossRef]
- Nollet, M.; Wisden, W.; Franks, N.P. Sleep deprivation and stress: A reciprocal relationship. Interface Focus 2020, 10, 20190092. [Google Scholar] [CrossRef]
- Medzhitov, R. Inflammation 2010: New adventures of an old flame. Cell 2010, 140, 771–776. [Google Scholar] [CrossRef]
- Mullington, J.M.; Simpson, N.S.; Meier-Ewert, H.K.; Haack, M. Sleep loss and inflammation. Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 775–784. [Google Scholar] [CrossRef]
- Krueger, J.M.; Fang, J.; Taishi, P.; Chen, Z.; Kushikata, T.; Gardi, J. Sleep: A physiologic role for IL-1 β and TNF-α. Ann. N. Y. Acad. Sci. 1998, 856, 148–159. [Google Scholar] [CrossRef]
- Vgontzas, A.N.; Papanicolaou, D.A.; Bixler, E.O.; Hopper, K.; Lotsikas, A.; Lin, H.M.; Kales, A.; Chrousos, G.P. Sleep apnea and daytime sleepiness and fatigue: Relation to visceral obesity, insulin resistance, and hypercytokinemia. J. Clin. Endocrinol. Metab. 2000, 85, 1151–1158. [Google Scholar] [CrossRef] [PubMed]
- Villa, M.P.; Ianniello, F.; Tocci, G.; Evangelisti, M.; Miano, S.; Ferrucci, A.; Ciavarella, G.M.; Volpe, M. Early cardiac abnormalities and increased C-reactive protein levels in a cohort of children with sleep disordered breathing. Sleep Breath.=Schlaf Atm. 2012, 16, 101–110. [Google Scholar] [CrossRef]
- Wu, H.; Yuan, X.; Wang, L.; Sun, J.; Liu, J.; Wei, Y. The Relationship between Obstructive Sleep Apnea Hypopnea Syndrome and Inflammatory Markers and Quality of Life in Subjects with Acute Coronary Syndrome. Respir. Care 2016, 61, 1207–1216. [Google Scholar] [CrossRef] [PubMed]
- Levy, P.; Kohler, M.; McNicholas, W.T.; Barbe, F.; McEvoy, R.D.; Somers, V.K.; Lavie, L.; Pepin, J.L. Obstructive sleep apnoea syndrome. Nat. Rev. Dis. Prim. 2015, 1, 15015. [Google Scholar] [CrossRef] [PubMed]
- Bonaldo, P.; Sandri, M. Cellular and molecular mechanisms of muscle atrophy. Dis. Model Mech. 2013, 6, 25–39. [Google Scholar] [CrossRef]
- Moore, K.W.; O’Garra, A.; de Waal Malefyt, R.; Vieira, P.; Mosmann, T.R. Interleukin-10. Annu. Rev. Immunol. 1993, 11, 165–190. [Google Scholar] [CrossRef] [PubMed]
- Matthys, P.; Mitera, T.; Heremans, H.; Van Damme, J.; Billiau, A. Anti-gamma interferon and anti-interleukin-6 antibodies affect staphylococcal enterotoxin B-induced weight loss, hypoglycemia, and cytokine release in D-galactosamine-sensitized and unsensitized mice. Infect. Immun. 1995, 63, 1158–1164. [Google Scholar] [CrossRef]
- Harrell, C.R.; Markovic, B.S.; Fellabaum, C.; Arsenijevic, N.; Djonov, V.; Volarevic, V. The role of Interleukin 1 receptor antagonist in mesenchymal stem cell-based tissue repair and regeneration. Biofactors 2020, 46, 263–275. [Google Scholar] [CrossRef]
- Wilson, D.; Jackson, T.; Sapey, E.; Lord, J.M. Frailty and sarcopenia: The potential role of an aged immune system. Ageing Res. Rev. 2017, 36, 1–10. [Google Scholar] [CrossRef]
- D’Aurea, C.V.R.; Poyares, D.; Passos, G.S.; Santana, M.G.; Youngstedt, S.D.; Souza, A.A.; Bicudo, J.; Tufik, S.; de Mello, M.T. Effects of resistance exercise training and stretching on chronic insomnia. Braz. J. Psychiatry 2019, 41, 51–57. [Google Scholar] [CrossRef]
- Rico-Rosillo, M.G.; Vega-Robledo, G.B. Sleep and immune system. Rev. Alerg. Mex. 2018, 65, 160–170. [Google Scholar] [CrossRef]
- Youngstedt, S.D.; O’Connor, P.J.; Dishman, R.K. The effects of acute exercise on sleep: A quantitative synthesis. Sleep 1997, 20, 203–214. [Google Scholar] [CrossRef] [PubMed]


| Variables/Groups | CTL | RET |
|---|---|---|
| Age (years) | 74.64 ± 7.13 | 77.42 ± 6.25 |
| Height (cm) | 155.17 ± 11.45 | 162.04 ± 12.81 |
| Body Mass (kg) | 64.93 ± 14.94 | 67.63 ± 12.77 |
| Body Fat (%) | 42.10 ± 6.85 | 35.62 ± 10.07 |
| AMI (kg/m2) | 6.58 ± 0.88 | 7.20 ± 2.54 |
| Body Mass Index (kg/m2) | 26.78 ± 4.44 | 25.54 ± 2.04 |
| Hemoglobin (g/dL) | 14.04 ± 1.73 | 14.50 ± 1.15 |
| Hematocrit (%) | 41.56 ± 5.50 | 43.77 ± 3.63 |
| Platelets (thousands/mm3) | 192.20 ± 30.84 | 228.00 ± 62.07 |
| Leukocytes (thousands/mm3) | 5.14 ± 0.92 | 6.46 ± 1.78 |
| Neutrophils (thousands/mm3) | 3.14 ± 0.60 | 3.84 ± 1.43 |
| Eosinophils (thousands/mm3) | 0.08 ± 0.07 | 0.25 ± 0.042 |
| Basophils (thousands/mm3) | 0.03 ± 0.02 | 0.03 ± 0.01 |
| Typical lymphocytes (thousands/mm3) | 1.57 ± 0.44 | 1.90 ± 0.60 |
| Total lymphocytes (thousands/mm3) | 1.57 ± 0.44 | 1.90 ± 0.60 |
| Monocytes (thousands/mm3) | 0.30 ± 0.06 | 0.42 ± 0.18 |
| Cholesterol (mg/dL) | 171.80 ± 28.98 | 187.09 ± 42.42 |
| HDL (mg/dL) | 63.40 ± 18.18 | 54.63 ± 10.82 |
| NO HDL (mg/dL) | 108.40 ± 15.10 | 132.45 ± 41.12 |
| LDL (mg/dL) | 88.20 ± 19.13 | 109.72 ± 36.33 |
| VLDL (mg/dL) | 20.20 ± 5.40 | 22.72 ± 8.05 |
| Triglycerides (mg/dL) | 101.20 ± 27.72 | 114.27 ± 40.48 |
| Glucose (mg/dL) | 99.60 ± 6.61 | 94.63 ± 12.17 |
| Urea (mg/dL) | 32.20 ± 3.11 | 40.90 ± 11.86 |
| Creatinine (mg/dL) | 0.83 ± 0.08 | 0.83 ± 0.18 |
| Albumin (g/dL) | 4.34 ± 0.24 | 4.24 ± 0.29 |
| Variables/Groups | CTL | RET | ||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | After 12 Weeks | ∆ Variation | Baseline | After 12 Weeks | ∆ Variation | |||
| Median | 95%CI | Median | 95%CI | |||||
| Sleep Latency (min) | 21.59 ± 15.67 | 29.98 ± 16.09 | 5.25 | 9.84–21.87 | 25.86 ± 20.16 | 16.09 ± 15.21 * | −6.25 # | 8.00–17.78 |
| REM sleep latency (min) | 86.21 ± 44.34 | 103.00 ± 45.42 | 9.00 | 29.32–65.15 | 108.53 ± 73.17 | 117.10 ± 70.87 | 0.25 | 55.69–123.76 |
| Total Sleep Time (min) | 342.00 ± 90.03 | 325.14 ± 66.56 | −28.00 | 43.34–96.31 | 310.00 ± 61.17 | 298.86 ± 84.93 | 8.50 | 46.72–103.84 |
| Sleep Efficiency (%) | 72.32 ± 18.74 | 69.49 ± 13.31 | −3.80 | 10.60–23.57 | 67.05 ± 13.52 | 67.92 ± 20.07 | 1.70 | 12.26–27.26 |
| N1 (%) | 10.27 ± 4.39 | 10.23 ± 3.85 | 0.80 | 3.79–8.42 | 20.62 ± 15.79 | 22.31 ± 21.34 * | −0.15 | 12.27–27.28 |
| N2 (%) | 39.57 ± 11.79 | 42.60 ± 10.63 | 3.20 | 4.73–10.52 | 37.34 ± 11.95 | 37.37 ± 8.66 | −1.80 | 9.70–21.57 |
| N3 (%) | 32.18 ± 11.57 | 27.80 ± 9.08 | −4.90 | 7.06–15.70 | 25.55 ± 9.79 | 30.25 ± 11.23 | 0.70 # | 7.27–16.16 |
| WASO (min) | 80.21 ± 46.55 | 98.63 ± 40.77 | 17.30 | 36.04–80.10 | 128.42 ± 58.21 | 131.21 ± 94.95 | 9.05 | 61.99–137.76 |
| Apnea/hour (n°/h) | 9.68 ± 4.52 | 13.29 ± 7.66 | 1.25 | 5.56–12.37 | 16.82 ± 14.11 | 7.37 ± 7.55 † | −9.45 | 6.93–15.41 |
| AHI (n°/h) | 15.20 ± 13.09 | 15.13 ± 15.89 | 12.35 | 4.91–10.93 | 14.08 ± 15.59 | 9.37 ± 13.70 * | 9.90 | 5.34–11.88 |
| SPO2 < 90% (min) | 7.02 ± 10.67 | 6.55 ± 11.03 | 0.00 | 4.74–10.54 | 6.84 ± 10.29 | 5.00 ± 8.67 | −0.85 | 2.04–4.53 |
| Variables/Groups | CTL | RET | ||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | After 12 Weeks | ∆ Variation | Baseline | After 12 Weeks | ∆ Variation | |||
| Median | 95%CI | Median | 95%CI | |||||
| Epworth (score) | 5.21 ± 3.09 | 5.57 ± 3.50 | 0.00 | 2.85–6.33 | 6.78 ± 4.13 | 6.50 ± 5.18 | 0.00 | 2.63–5.84 |
| Insomnia Severity Index (score) | 5.71 ± 3.89 | 5.78 ± 3.78 | −11.00 | 5.24–11.64 | 8.71 ± 4.37 | 5.14 ± 3.73 * | −14.00 # | 5.08–11.30 |
| Pittsburgh (score) | 7.75 ± 5.12 | 4.50 ± 3.87 | 0.00 | 1.67–3.84 | 5.33 ± 3.21 | 4.33 ± 3.78 | −1.50 # | 2.76–6.14 |
| Pittsburgh Sleep Efficiency (%) | 84.50 ± 11.37 | 81.91 ± 11.25 | 0.00 | 8.04–17.87 | 75.57 ± 15.37 | 83.57 ± 14.56 * | 9.50 # | 9.87–21.94 |
| Variables/Groups | CTL | RET | ||||||
|---|---|---|---|---|---|---|---|---|
| Baseline | After 12 Weeks | ∆ Variation | Baseline | After 12 Weeks | ∆ Variation | |||
| Median | 95%CI | Median | 95%CI | |||||
| Testosterone (ng/dL) | 127.00 ± 211.60 | 130.65 ± 226.45 | −2.20 | 10.89–27.35 | 329.02 ± 298.30 | 322.16 ± 317.70 | −4.70 | 64.71–148.97 |
| GH (ng/mL) | 1.95 ± 2.42 | 1.11 ± 1.32 | −0.29 | 1.26–2.80 | 1.21 ± 1.26 | 1.26 ± 1.23 | −0.52 | 1.30–3.01 |
| IGF 1 (ng/mL) | 12.27 ± 28.20 | 14.60 ± 28.25 | −0.01 | 6.00–13.34 | 7.76 ± 20.47 | 7.33 ± 20.46 | −0.04 | 1.25–2.78 |
| Cortisol (ug/dL) | 12.29 ± 3.07 | 13.40 ± 2.10 | 1.10 | 2.39–5.32 | 10.05 ± 3.23 | 11.29 ± 2.65 | 0.20 | 2.87–6.38 |
| TNF-α (pg/mL) | 4.30 ± 0.36 | 4.17 ± 0.32 | −0.07 | 0.25–0.56 | 4.40 ± 0.32 | 4.41 ± 0.31 | −0.06 | 0.23–0.51 |
| IL-6 (pg/mL) | 2.88 ± 0.75 | 2.56 ± 0.64 | −0.25 | 0.77–1.71 | 2.92 ± 1.08 | 3.29 ± 1.71 | 0.16 | 0.88–1.97 |
| IL-10 (pg/mL) | 1.96 ± 0.58 | 2.26 ± 0.88 | 0.26 | 0.53–1.18 | 2.13 ± 0.80 | 2.51 ± 0.99 * | 0.27 | 0.31–0.69 |
| IL-1 RA (ng/mL) | 0.95 ± 0.06 | 0.94 ± 0.05 | −0.01 | 0.04–0.08 | 0.93 ± 0.03 | 0.99 ± 0.10 * | 0.04 # | 0.06–0.15 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Sá Souza, H.; de Melo, C.M.; Piovezan, R.D.; Miranda, R.E.E.P.C.; Carneiro-Junior, M.A.; Silva, B.M.; Thomatieli-Santos, R.V.; Tufik, S.; Poyares, D.; D’Almeida, V. Resistance Training Improves Sleep and Anti-Inflammatory Parameters in Sarcopenic Older Adults: A Randomized Controlled Trial. Int. J. Environ. Res. Public Health 2022, 19, 16322. https://doi.org/10.3390/ijerph192316322
de Sá Souza H, de Melo CM, Piovezan RD, Miranda REEPC, Carneiro-Junior MA, Silva BM, Thomatieli-Santos RV, Tufik S, Poyares D, D’Almeida V. Resistance Training Improves Sleep and Anti-Inflammatory Parameters in Sarcopenic Older Adults: A Randomized Controlled Trial. International Journal of Environmental Research and Public Health. 2022; 19(23):16322. https://doi.org/10.3390/ijerph192316322
Chicago/Turabian Stylede Sá Souza, Helton, Camila Maria de Melo, Ronaldo Delmonte Piovezan, Rafael Eduardo Eustórgio Pinheiro Chagas Miranda, Miguel Araujo Carneiro-Junior, Bruno Moreira Silva, Ronaldo Vagner Thomatieli-Santos, Sergio Tufik, Dalva Poyares, and Vânia D’Almeida. 2022. "Resistance Training Improves Sleep and Anti-Inflammatory Parameters in Sarcopenic Older Adults: A Randomized Controlled Trial" International Journal of Environmental Research and Public Health 19, no. 23: 16322. https://doi.org/10.3390/ijerph192316322
APA Stylede Sá Souza, H., de Melo, C. M., Piovezan, R. D., Miranda, R. E. E. P. C., Carneiro-Junior, M. A., Silva, B. M., Thomatieli-Santos, R. V., Tufik, S., Poyares, D., & D’Almeida, V. (2022). Resistance Training Improves Sleep and Anti-Inflammatory Parameters in Sarcopenic Older Adults: A Randomized Controlled Trial. International Journal of Environmental Research and Public Health, 19(23), 16322. https://doi.org/10.3390/ijerph192316322








