Factors Associated with the Antibiotic Treatment of Children Hospitalized for COVID-19 during the Lockdown in Serbia
Abstract
:1. Introduction
2. Materials and Methods
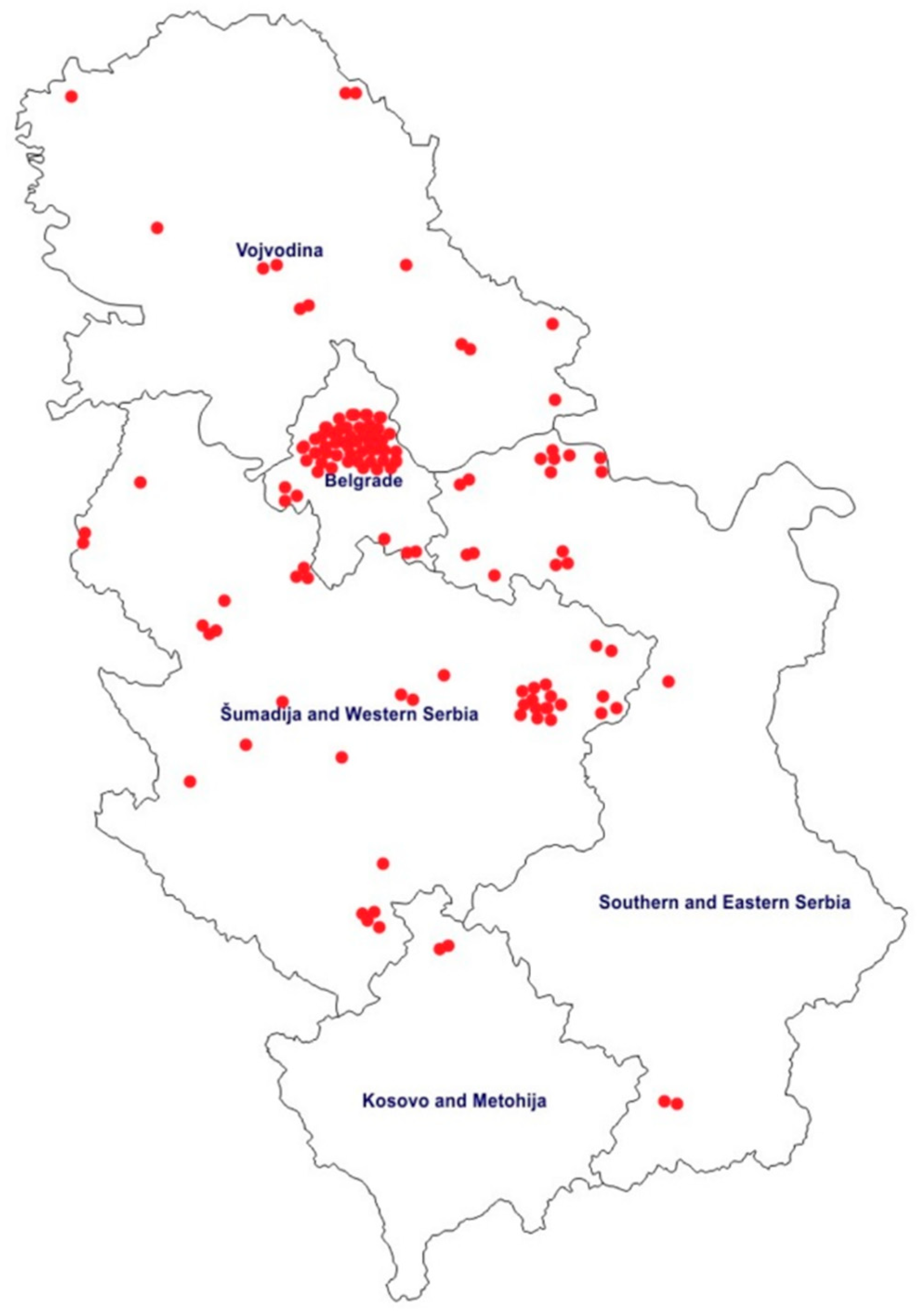
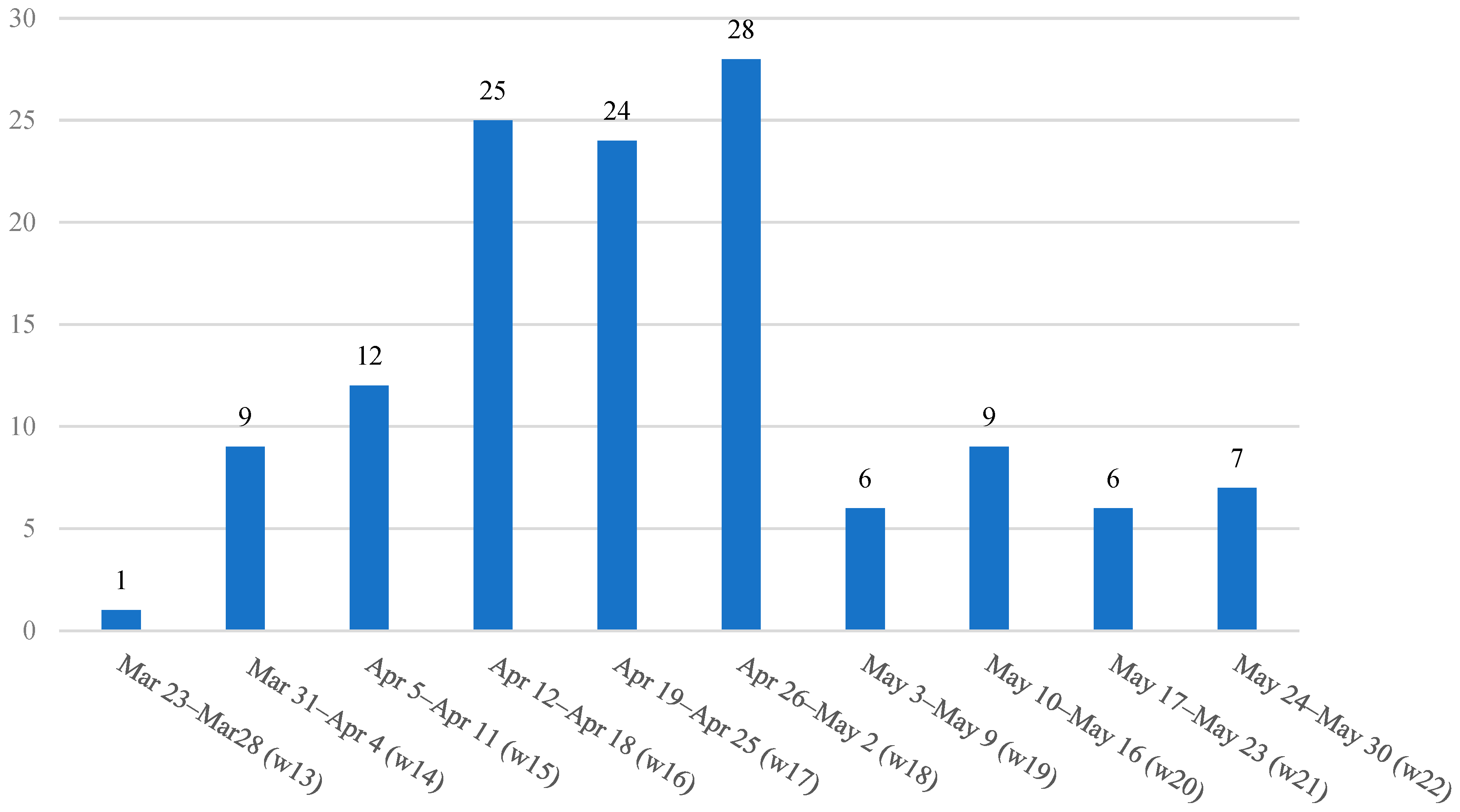
2.1. Study Population and Sample
2.2. Data Collection
2.3. Statistical Analyses
2.4. Ethical Approval
3. Results
3.1. Signs, Symptoms and Therapy of COVID-19
3.2. Linear Regression Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Procter, B.C.; Ross, C.; Pickard, V.; Smith, E.; Hanson, C.; McCullough, P.A. Clinical outcomes after early ambulatory multidrug therapy for high-risk SARS-CoV-2 (COVID-19) infection. Rev. Cardiovasc. Med. 2020, 21, 611–614. [Google Scholar] [PubMed]
- Popp, M.; Stegemann, M.; Riemer, M.; Metzendorf, M.-I.; Romero, C.S.; Mikolajewska, A.; Kranke, P.; Meybohm, P.; Skoetz, N.; Weibel, S. Antibiotics for the treatment of COVID-19. Cochrane Database Syst. Rev. 2021, 10, CD015025. [Google Scholar]
- Mussini, C.; Cozzi-Lepri, A. Another piece in the COVID-19 treatment puzzle. Lancet 2022, 399, 609–610. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Therapeutics and COVID-19 Living Guide. Available online: https://apps.who.int/iris/bitstream/handle/10665/345356/WHO-2019-nCoV-therapeutics-2021.3-eng.pdf (accessed on 5 November 2022).
- Giacomelli, A.; Ridolfo, A.L.; Oreni, L.; Vimercati, S.; Albrecht, M.; Cattaneo, D.; Rimoldi, S.G.; Rizzardini, G.; Galli, M.; Antinori, S. Consumption of antibiotics at an Italian university hospital during the early months of the COVID-19 pandemic: Were all antibiotic prescriptions appropriate? Pharmacol. Res. 2021, 164, 105403. [Google Scholar] [CrossRef] [PubMed]
- Abelenda-Alonso, G.; Padullés, A.; Rombauts, A.; Gudiol, C.; Pujol, M.; Alvarez-Pouso, C.; Jodar, R.; Carratalà, J. Antibiotic prescription during the COVID-19 pandemic: A biphasic pattern. Infect. Control. Hosp. Epidemiol. 2020, 41, 1371–1372. [Google Scholar] [CrossRef]
- Silva, A.R.O.; Salgado, D.R.; Lopes, L.P.N.; Castanheira, D.; Emmerick, I.C.M.; Lima, E.C. Increased Use of Antibiotics in the Intensive Care Unit During Coronavirus Disease (COVID-19) Pandemic in a Brazilian Hospital. Front. Pharmacol. 2021, 12, 778386. [Google Scholar] [CrossRef]
- Lai, C.-C.; Chen, S.-Y.; Ko, W.-C.; Hsueh, P.-R. Increased antimicrobial resistance during the COVID-19 pandemic. Int. J. Antimicrob. Agents 2021, 57, 106324. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Bornman, C.; Zafer, M.M. Antimicrobial Resistance Threats in the emerging COVID-19 pandemic: Where do we stand? J. Infect Public Health 2021, 14, 555–560. [Google Scholar] [CrossRef]
- Gajdács, M.; Urbán, E.; Stájer, A.; Baráth, Z. Antimicrobial Resistance in the Context of the Sustainable Development Goals: A Brief Review. Eur. J. Investig. Health Psychol. Educ. 2021, 11, 6. [Google Scholar] [CrossRef] [PubMed]
- Capalbo, C.; Aceti, A.; Simmaco, M.; Bonfini, R.; Rocco, M.; Ricci, A.; Napoli, C.; Rocco, M.; Alfonsi, V.; Teggi, A.; et al. The Exponential Phase of the COVID-19 Pandemic in Central Italy: An Integrated Care Pathway. Int. J. Environ. Res. Public Health. 2020, 17, 3792. [Google Scholar] [CrossRef]
- Lucien, M.A.B.; Canarie, M.F.; Kilgore, P.E.; Jean-Denis, G.; Fénélon, N.; Pierre, M.; Cerpa, M.; Joseph, G.A.; Maki, G.; Zervos, M.J.; et al. Antibiotics and antimicrobial resistance in the COVID-19 era: Perspective from resource-limited settings. Int. J. Infect. Dis. 2021, 104, 250–254. [Google Scholar] [CrossRef]
- Chitungo, I.; Dzinamarira, T.; Nyazika, T.K.; Herrera, H.; Musuka, G.; Murewanhema, G. Inappropriate Antibiotic Use in Zimbabwe in the COVID-19 Era: A Perfect Recipe for Antimicrobial Resistance. Antibiotics 2022, 11, 244. [Google Scholar] [CrossRef] [PubMed]
- Mania, A.; Pokorska-Śpiewak, M.; Figlerowicz, M.; Pawłowska, M.; Mazur-Melewska, K.; Faltin, K.; Talarek, E.; Zawadka, K.; Dobrzeniecka, A.; Ciechanowski, P.; et al. Pneumonia, gastrointestinal symptoms, comorbidities, and coinfections as factors related to a lengthier hospital stay in children with COVID-19-analysis of a paediatric part of Polish register SARSTer. Infect. Dis. 2022, 54, 196–204. [Google Scholar] [CrossRef]
- Yock-Corrales, A.; Lenzi, J.; Ulloa-Gutiérrez, R.; Gómez-Vargas, J.; Antúnez-Montes, O.Y.; Aida, J.A.R.; del Aguila, O.; Arteaga-Menchaca, E.; Campos, F.; Uribe, F.; et al. High rates of antibiotic prescriptions in children with COVID-19 or multisystem inflammatory syndrome: A multinational experience in 990 cases from Latin America. Acta Paediatr. 2021, 110, 1902–1910. [Google Scholar] [CrossRef] [PubMed]
- Aguilera-Alonso, D.; Epalza, C.; Sanz-Santaeufemia, F.J.; Grasa, C.; Villanueva-Medina, S.; Pérez, S.M.; Hernández, E.C.; Urretavizcaya-Martínez, M.; Pino, R.; Gómez, M.N.; et al. Antibiotic Prescribing in Children Hospitalized With COVID-19 and Multisystem Inflammatory Syndrome in Spain: Prevalence, Trends, and Associated Factors. J. Pediatr. Infect. Dis. Soc. 2022, 11, 225–228. [Google Scholar] [CrossRef] [PubMed]
- Despotović, A.; Barać, A.; Cucanić, T.; Cucanić, K.; Stevanović, G. Antibiotic (Mis) Use in COVID-19 Patients before and after Admission to a Tertiary Hospital in Serbia. Antibiotics 2022, 11, 847. [Google Scholar] [CrossRef]
- Statistical Office of the Republic of Serbia. Estimates of Population by Age and Gender. 2020. Available online: https://data.stat.gov.rs/Home/Result/18010403?languageCode=sr-Latn (accessed on 29 May 2020).
- Zimmermann, P.; Curtis, N. Coronavirus infections in children including COVID-19: An overview of the epidemiology, clinical features, diagnosis, treatment and prevention options in children. Pediatr. Infect. Dis. J. 2020, 39, 355. [Google Scholar] [CrossRef] [PubMed]
- Nava-Muñoz, Á.; Gómez-Peña, S.; Fuentes-Ferrer, M.; Cabeza, B.; Victoria, A.; Bustos, A. COVID-19 pneumonia: Relationship between initial chest X-rays and laboratory findings. Radiol. (Engl. Ed.) 2021, 63, 484–494. [Google Scholar] [CrossRef] [PubMed]
- D-Maps. Terms and Conditions of Use. Available online: https://d-maps.com/conditions.php?lang=en (accessed on 20 May 2022).
- Wang, J.; Tang, Y.; Ma, Y.; Zhou, Q.; Li, W.; Baskota, M.; Yang, Y.; Wang, X.; Li, Q.; Luo, X.; et al. Efficacy and safety of antibiotic agents in children with COVID-19: A rapid review. Ann. Transl. Med. 2020, 8, 619. [Google Scholar] [CrossRef]
- Allen, H.; Vusirikala, A.; Flannagan, J.; Twohig, K.A.; Zaidi, A.; Chudasama, D.; Lamagni, T.; Groves, N.; Turner, C.; Rawlinson, C.; et al. Household transmission of COVID-19 cases associated with SARS-CoV-2 delta variant (B.1.617.2): National case-control study. Lancet Reg. Health-Eur. 2022, 12, 100252. [Google Scholar] [CrossRef]
- Dhochak, N.; Singhal, T.; Kabra, S.K.; Lodha, R. Pathophysiology of COVID-19: Why Children Fare Better than Adults? Indian J. Pediatr. 2020, 87, 537–546. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, N.; Dixit, D. Human and novel coronavirus infections in children: A review. Paediatr. Int. Child Health 2021, 41, 36–55. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Han, H.; Liu, F.; Lv, Z.; Wu, K.; Liu, Y. Positive rate of RT-PCR detection of SARS-CoV-2 infection in 4880 cases from one hospital in Wuhan, China, from Jan to Feb 2020. Clin. Chim. Acta 2020, 505, 172–175. [Google Scholar] [CrossRef] [PubMed]
- McCormick, D.W.; Richardson, L.C.; Young, P.R.; Viens, L.J.; Gould, C.V.; Kimball, A.; Pindyck, T.; Rosenblum, H.G.; Siegel, D.A.; Vu, Q.M.; et al. Deaths in Children and Adolescents Associated With COVID-19 and MIS-C in the United States. Pediatrics 2021, 148, e2021052273. [Google Scholar] [CrossRef]
- De Jacobis, I.T.; Vona, R.; Cittadini, C.; Marchesi, A.; Cursi, L.; Gambardella, L.; Villani, A.; Straface, E. Clinical characteristics of children infected with SARS-CoV-2 in Italy. Ital. J. Pediatr. 2021, 47, 90. [Google Scholar] [CrossRef] [PubMed]
- Fortunato, F.; Martinelli, D.; Caputo, S.L.; Santantonio, T.; Dattoli, V.; Lopalco, P.L.; Prato, R. Sex and gender differences in COVID-19: An Italian local register-based study. BMJ Open 2021, 11, e051506. [Google Scholar] [CrossRef]
- Ortolan, A.; Lorenzin, M.; Felicetti, M.; Doria, A.; Ramonda, R. Does gender influence clinical expression and disease outcomes in COVID-19? A systematic review and meta-analysis. Int. J. Infect. Dis. 2020, 99, 496–504. [Google Scholar] [CrossRef]
- Wolfe, J.; Safdar, B.; Madsen, T.E.; Sethuraman, K.N.; Becker, B.; Greenberg, M.R.; McGregor, A.J. Sex- or Gender-specific Differences in the Clinical Presentation, Outcome, and Treatment of SARS-CoV-2. Clin. Ther. 2021, 43, 557–571. [Google Scholar] [CrossRef]
- Schwartz, R.A.; Suskind, R.M. Azithromycin and COVID-19: Prompt early use at first signs of this infection in adults and children, an approach worthy of consideration. Dermatol. Ther. 2020, 33, e13785. [Google Scholar] [CrossRef]
- Venturini, E.; Montagnani, C.; Garazzino, S.; Donà, D.; Pierantoni, L.; Vecchio, A.L.; Nicolini, G.; Bianchini, S.; Krzysztofiak, A.; Galli, L.; et al. Treatment of children with COVID-19: Position paper of the Italian Society of Pediatric Infectious Disease. Ital. J. Pediatr. 2020, 46, 139. [Google Scholar] [CrossRef] [PubMed]
- Perikleous, E.; Tsalkidis, A.; Bush, A.; Paraskakis, E. Coronavirus global pandemic: An overview of current findings among pediatric patients. Pediatr. Pulmonol. 2020, 55, 3252–3267. [Google Scholar] [CrossRef] [PubMed]
- National Institutes of Health. Therapeutic Management of Hospitalized Children with COVID-19. Available online: https://www.covid19treatmentguidelines.nih.gov/management/clinical-management-of-children/hospitalized-children-therapeutic-management/ (accessed on 1 November 2022).
- Pierce, J.; Stevens, M.P. COVID-19 and antimicrobial stewardship: Lessons learned, best practices, and future implications. Int. J. Infect. Dis. 2021, 113, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Langford, B.J.; So, M.; Raybardhan, S.; Leung, V.; Soucy, J.R.; Westwood, D.; Daneman, N.; MacFadden, D.R. Antibiotic prescribing in patients with COVID-19: Rapid review and meta-analysis. Clin. Microbiol. Infect. 2021, 27, 520–531. [Google Scholar] [CrossRef]
- Kalam, A.; Shano, S.; Khan, M.A.; Islam, A.; Warren, N.; Hassan, M.M.; Davis, M. Understanding the social drivers of antibiotic use during COVID-19 in Bangladesh: Implications for reduction of antimicrobial resistance. PLoS ONE. 2021, 16, e0261368. [Google Scholar] [CrossRef] [PubMed]
- Papst, L.; Luzzati, R.; Carević, B.; Tascini, C.; Miksić, N.G.; Palčevski, V.V.; Djordjevic, Z.M.; Simonetti, O.; Sozio, E.; Lukić, M.; et al. Antimicrobial Use in Hospitalised Patients with COVID-19: An International Multicentre Point-Prevalence Study. Antibiotics 2022, 11, 176. [Google Scholar] [CrossRef]
- Pourbagheri-Sigaroodi, A.; Bashash, D.; Fateh, F.; Abolghasemi, H. Laboratory findings in COVID-19 diagnosis and prognosis. Clin. Chim. Acta 2020, 510, 475–482. [Google Scholar] [CrossRef]
- Molteni, E.; Sudre, C.H.; Canas, L.S.; Bhopal, S.S.; Hughes, R.C.; Antonelli, M.; Murray, B.; Kläser, K.; Kerfoot, E.; Chen, L.; et al. Illness duration and symptom profile in symptomatic UK school-aged children tested for SARS-CoV-2. Lancet Child Adolesc. Health 2021, 5, 708–718. [Google Scholar] [CrossRef]
- Molteni, E.; Sudre, C.H.; Canas, L.D.S.; Bhopal, S.S.; Hughes, R.C.; Chen, L.; Deng, J.; Murray, B.; Kerfoot, E.; Antonelli, M.; et al. Illness Characteristics of COVID-19 in Children Infected with the SARS-CoV-2 Delta Variant. Children 2022, 9, 652. [Google Scholar] [CrossRef]
- Lazova, S.; Alexandrova, T.; Gorelyova-Stefanova, N.; Atanasov, K.; Tzotcheva, I.; Velikova, T. Liver Involvement in Children with COVID-19 and Multisystem Inflammatory Syndrome: A Single-Center Bulgarian Observational Study. Microorganisms 2021, 9, 1958. [Google Scholar] [CrossRef]
- de Munain, A.I.L.; Veintemilla, C.J.; Aguirre, M.H.; Sánchez, N.V.; Ramos-Lacuey, B.; Urretavizcaya-Martínez, M.; Esandi, L.E.; López, M.D.M.P.; Howard, M.G.; Fernández-Montero, A.; et al. Chest radiograph in hospitalized children with COVID-19. A review of findings and indications. Eur. J. Radiol. Open 2021, 8, 100337. [Google Scholar] [CrossRef] [PubMed]
- Biko, D.M.; Ramirez-Suarez, K.I.; Barrera, C.A.; Banerjee, A.; Matsubara, D.; Kaplan, S.L.; Cohn, K.A.; Rapp, J.B. Imaging of children with COVID-19: Experience from a tertiary children’s hospital in the United States. Pediatr. Radiol. 2021, 51, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.; Meena, J.; Yadav, A.; Yadav, J. Radiological Findings of COVID-19 in Children: A Systematic Review and Meta-Analysis. J. Trop. Pediatr. 2021, 67, fmaa045. [Google Scholar] [CrossRef] [PubMed]
- Kepenekli, E.; Yakut, N.; Ergenc, Z.; Aydıner, Ö.; Sarınoğlu, R.C.; Karahasan, A.; Karakoc-Aydıner, E.; Memişoğlu, A.; Gökdemir, Y.; Eralp, E.E.; et al. COVID-19 disease characteristics in different pediatric age groups. J. Infect. Dev. Ctries. 2022, 16, 16–24. [Google Scholar] [CrossRef] [PubMed]
| Variable | Neonates | Infants | Children 1–5 y | Children Aged 5–17 y |
|---|---|---|---|---|
| N = 6 | N = 10 | N = 30 | N = 81 | |
| n (%) | n (%) | n (%) | n (%) | |
| Gender | ||||
| Male | 2 (33.3) | 8 (80.0) | 17 (56.7) | 47 (58.0) |
| Female | 4 (66.7) | 2 (20.0) | 13 (43.3) | 34 (42.0) |
| Chronic diseases | ||||
| Yes | 0 (0) | 0 (0) | 3 (10.0) | 10 (12.3) |
| No | 6 (100.0) | 10 (100.0) | 27 (90.0) | 71 (87.7) |
| Confirmed COVID-19 in family | ||||
| Yes | 6 (100.0) | 9 (90.0) | 22 (73.3) | 77 (95.1) |
| No | 0 (0) | 1 (10.0) | 8 (26.7) | 4 (4.9) |
| Variable | Neonates | Infants | Children 1–5 y | Children Aged 5–17 y |
|---|---|---|---|---|
| N = 6 | N = 10 | N = 30 | N = 81 | |
| n (%) | n (%) | n (%) | n (%) | |
| Cough | ||||
| Yes | 6 (100.0) | 7 (70.0) | 20 (66.7) | 60 (74.1) |
| No | 0 (0) | 3 (30.0) | 10 (33.3) | 21 (25.9) |
| Pharyngeal erythema | ||||
| Yes | 0 (0) | 1 (10.0) | 0 (0) | 5 (6.2) |
| No | 6 (100.0) | 9 (90.0) | 30 (100.0) | 76 (93.8) |
| Fever | ||||
| Yes | 2 (33.3) | 7 (70.0) | 12 (40.0) | 37 (45.7) |
| No | 4 (66.7) | 3 (30.0) | 18 (60.0) | 44 (54.3) |
| Body temperature when having fever (range) | 37.7 °C (37.3–38.2) 99.8 °F (99.1–100.7) | 38.3 °C (37.3–40.6) 100.9 °F (99.1–105.1) | 38.2 °C (37.2–39.4) 100.7 °F (99.0–102.9) | 38.0 °C (37.1–39.5) 100.4 °F (98.8–103.1) |
| Duration of fever in days (range) | 1 * | 2.7 (1–6) | 3.8 (1–8) | 3.3 (0–10) |
| Diarrhea | ||||
| Yes | 0 (0) | 2 (20.0) | 0 (0) | 3 (3.7) |
| No | 6 (100.0) | 8 (80.0) | 30 (100.0) | 78 (96.3) |
| Vomiting | ||||
| Yes | 1 (16.7) | 2 (20.0) | 0 (0) | 1 (1.2) |
| No | 5 (83.3) | 8 (80.0) | 30 (100.0) | 80 (98.8) |
| Rhinorrhea | ||||
| Yes | 0 (0) | 1 (10.0) | 0 (0) | 2 (2.5) |
| No | 6 (100.0) | 9 (90.0) | 30 (100.0) | 79 (97.5) |
| Nasal congestion | ||||
| Yes | 1 (16.7) | 4 (40.0) | 0 (0) | 1 (1.2) |
| No | 5 (83.3) | 6 (60.0) | 30 (100.0) | 80 (98.8) |
| Tachypnea | ||||
| Yes | 0 (0) | 0 (0) | 0 (0) | 0 (0) |
| No | 6 (100.0) | 10 (100.0) | 30 (100.0) | 81 (100.0) |
| Tachycardia | ||||
| Yes | 1 (16.7) | 0 (0) | 0 (0) | 0 (0) |
| No | 5 (83.3) | 10 (100.0) | 30 (100.0) | 81 (100.0) |
| Oxygen saturation % (range) | 98 (96–99) | 97 (96–99) | 98 (95–99) | 98 (96–99) |
| C-reactive protein (range) | 3.8 (1.0–5.30) | 5.8 (1.0–9.50) | 4.1 (1.0–25.4) | 4.3 (1.0–58.0) |
| Leukocytes (×109/L) | 11.1 (8.9–12.5) | 8.3 (5.0–15.0) | 7.4 (4.3–13.9) | 6.8 (2.8–14.9) |
| Thrombocytes (×109/L) | 277.4 (212.0–372.0) | 308.1 (186.0–432.0) | 309.0 (119–779) | 237.8 (57.0–410.0) |
| Pneumonia seen on chest X-ray | ||||
| Yes | 0 (0) | 1 (10.0) | 8 (26.7) | 21 (28.0) |
| No | 6 (100.0) | 9 (90.0) | 22 (73.3) | 60 (72.0) |
| Variable | Neonates | Infants | Children 1–5 y | Children Aged 5–17 y |
|---|---|---|---|---|
| N = 6 | N = 10 | N = 30 | N = 81 | |
| n (%) | n (%) | n (%) | n (%) | |
| Azithromycin | ||||
| Yes | 1 (16.7) | 2 (20.0) | 13 (43.3) | 32 (39.5) |
| No | 5 (83.3) | 8 (80.0) | 17 (56.7) | 49 (60.5) |
| Cephalosporin (ceftriaxone) | ||||
| Yes | 1 (16.7) | 2 (20.0) | 4 (13.3) | 22 (27.2) |
| No | 5 (83.3) | 8 (80.0) | 26 (86.7) | 59 (72.8) |
| Ampicillin-amikacin | ||||
| Yes | 1 (16.7) | 1 (10.0) | 0 (0) | 0 (0) |
| No | 5 (83.3) | 9 (90.0) | 30 (100.0) | 81 (100.0) |
| Chloroquine | ||||
| Yes | 0 (0) | 0 (0) | 0 (0) | 5 (6.2) |
| No | 6 (100.0) | 10 (100.0) | 30 (100.0) | 76 (93.8) |
| Number of medications used | ||||
| 0 | 0 (0) | 5 (50.0) | 17 (56.7) | 45 (55.6) |
| 1 | 5 (83.3) | 5 (50.0) | 9 (30.0) | 18 (22.2) |
| 2 | 1 (16.7) | 0 (0) | 4 (13.3) | 12 (16.0) |
| 3 | 0 (0) | 0 (0) | 0 (0) | 5 (6.2) |
| Maximum duration of therapy in days for children who received medications (range) | 7.0 * | 5.6 (5–7) | 6.4 (5–11) | 6.4 (5–10) |
| Supplemental oxygen | ||||
| Yes | 0 (0) | 0 (0) | 1 (3.3) | 1 (1.2) |
| No | 6 (100.0) | 10 (100.0) | 29 (96.7) | 80 (98.8) |
| Mechanical ventilation | ||||
| Yes | 0 (0) | 0 (0) | 1 (3.3) | 0 (0) |
| No | 6 (100.0) | 10 (100.0) | 29 (96.7) | 81 (100.0) |
| Variable | Total Number of Medications Used | Duration of Medication Therapy | ||||
|---|---|---|---|---|---|---|
| B | 95%CI | p | B | 95%CI | p | |
| Socio-epidemiologic model | ||||||
| Gender Male vs. female | −0.29 | −0.78, 0.20 | 0.232 | −2.13 | −4.48, 0.23 | 0.075 |
| Having chronic diseases Yes vs. no | 0.04 | −0.81, 0.88 | 0.925 | 0.96 | −3.02, 4.95 | 0.623 |
| Confirmed positive family members Yes vs. no | −1.38 | −2.43, −0.34 | 0.011 | −5.16 | −10.08, −0.23 | 0.041 |
| Model summary | R2 = 0.325, adj R2 = 0.248, p = 0.015 | R2 = 0.340, adj R2 = 0.261, p = 0.014 | ||||
| Clinical symptoms model | ||||||
| Gender Male vs. female | −0.14 | −0.60, 0.32 | 0.532 | −1.76 | −3.61, 0.08 | 0.060 |
| Cough Yes vs. no | 0.29 | −0.26, 0.84 | 0.292 | 0.25 | −2.12, 2.63 | 0.826 |
| Fever Yes vs. no | 0.44 | −0.07, 0.85 | 0.086 | 3.20 | 1.03, 5.37 | 0.006 |
| Pneumonia on X-ray Yes vs. no | 0.81 | 0.34, 1.29 | 0.002 | 4.02 | 2.07, 5.97 | 0.001 |
| Model summary | R2 = 0.546, adj R2 = 0.470, p = 0.001 | R2 = 0.684, adj R2 = 0.629, p = 0.001 | ||||
| Laboratory findings model | ||||||
| Gender Male vs. female | −0.65 | −1.17, −0.13 | 0.018 | −3.48 | −6.14, −0.82 | 0.013 |
| Oxygen saturation | −0.12 | −0.35, 0.10 | 0.253 | −0.82 | −1.95, 0.32 | 0.147 |
| C-reactive protein | 0.12 | 0.07, 0.17 | 0.001 | 0.37 | 0.10, 0.64 | 0.011 |
| Leukocytes | −0.07 | −0.20, 0.05 | 0.236 | −0.04 | −0.71, 0.62 | 0.893 |
| Thrombocytes | 0.01 | −0.01, 0.01 | 0.210 | 0.01 | −0.01, 0.01 | 0.266 |
| Model summary | R2 = 0.679, adj R2 = 0.594, p = 0.001 | R2 = 0.639, adj R2 = 0.539, p = 0.001 | ||||
| Variable | Total Number of Medications Used | Duration of Medication Therapy | ||||
|---|---|---|---|---|---|---|
| B | 95%CI | p | B | 95%CI | p | |
| Socio-epidemiologic model | ||||||
| Gender Male vs. female | 0.31 | −0.12, 0.74 | 0.158 | 0.99 | −0.55, 2.52 | 0.204 |
| Having chronic diseases Yes vs. no | 0.23 | −0.39, 0.85 | 0.460 | −0.01 | −2.24, 2.21 | 0.991 |
| Confirmed positive family members Yes vs. no | −0.86 | −1.83, 0.12 | 0.086 | −4.05 | −7.55, −0.55 | 0.024 |
| Model summary | R2 = 0.092, adj R2 = 0.057, p = 0.057 | R2 = 0.107, adj R2 = 0.072, p = 0.032 | ||||
| Clinical symptoms model | ||||||
| Gender Male vs. female | 0.27 | −0.08, 0.61 | 0.129 | 1.04 | −0.16, 2.24 | 0.089 |
| Cough Yes vs. no | 0.07 | −0.33, 0.47 | 0.737 | −0.58 | −1.97, 0.82 | 0.413 |
| Pharyngeal erythema Yes vs. no | 1.37 | 0.61, 2.13 | 0.001 | 3.08 | 0.44, 5.72 | 0.023 |
| Fever Yes vs. no | 0.43 | 0.07, 0.79 | 0.018 | 2.29 | 1.07, 3.52 | 0.001 |
| Diarrhea Yes vs. no | 0.92 | −0.04, 1.88 | 0.059 | 2.05 | −1.26, 5.36 | 0.221 |
| Rhinorrhea Yes vs. no | −1.27 | −2.47, −0.08 | 0.037 | −3.80 | −7.92, 0.32 | 0.070 |
| Pneumonia on X-ray Yes vs. no | 0.91 | 0.53, 1.29 | 0.001 | 3.88 | 2.57–5.18 | 0.001 |
| Model summary | R2 = 0.502, adj R2 = 0.450, p = 0.001 | R2 = 0.536, adj R2 = 0.487, p = 0.001 | ||||
| Laboratory findings model | ||||||
| Gender Male vs. female | 0.52 | 0.08, 0.97 | 0.021 | 1.68 | 0.06, 3.30 | 0.043 |
| Oxygen saturation | −0.18 | −0.47, 0.10 | 0.206 | −0.49 | −1.53, 0.55 | 0.351 |
| C-reactive protein | 0.04 | 0.01, 0.06 | 0.006 | 0.14 | 0.05, 0.24 | 0.004 |
| Leukocytes | −0.02 | −0.12, 0.07 | 0.650 | −0.09 | −0.44, 0.25 | 0.584 |
| Thrombocytes | −0.01 | −0.01, 0.01 | 0.478 | 0.01 | −0.01, 0.01 | 0.744 |
| Model summary | R2 = 0.224, adj R2 = 0.164, p = 0.005 | R2 = 0.199, adj R2 = 0.137, p = 0.012 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prijić, A.; Gazibara, T.; Prijić, S.; Mandić-Rajčević, S.; Maksimović, N. Factors Associated with the Antibiotic Treatment of Children Hospitalized for COVID-19 during the Lockdown in Serbia. Int. J. Environ. Res. Public Health 2022, 19, 15590. https://doi.org/10.3390/ijerph192315590
Prijić A, Gazibara T, Prijić S, Mandić-Rajčević S, Maksimović N. Factors Associated with the Antibiotic Treatment of Children Hospitalized for COVID-19 during the Lockdown in Serbia. International Journal of Environmental Research and Public Health. 2022; 19(23):15590. https://doi.org/10.3390/ijerph192315590
Chicago/Turabian StylePrijić, Andreja, Tatjana Gazibara, Sergej Prijić, Stefan Mandić-Rajčević, and Nataša Maksimović. 2022. "Factors Associated with the Antibiotic Treatment of Children Hospitalized for COVID-19 during the Lockdown in Serbia" International Journal of Environmental Research and Public Health 19, no. 23: 15590. https://doi.org/10.3390/ijerph192315590
APA StylePrijić, A., Gazibara, T., Prijić, S., Mandić-Rajčević, S., & Maksimović, N. (2022). Factors Associated with the Antibiotic Treatment of Children Hospitalized for COVID-19 during the Lockdown in Serbia. International Journal of Environmental Research and Public Health, 19(23), 15590. https://doi.org/10.3390/ijerph192315590






