Broccoli Sprouts Promote Sex-Dependent Cardiometabolic Health and Longevity in Long-Evans Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Treatments
2.2. Body Composition and Metabolic Parameters
2.3. Tail-Cuff Plethysmography
2.4. Echocardiography
2.5. Behavioral Tests
2.6. Lifespan Estimation
2.7. Statistical Analyses
3. Results
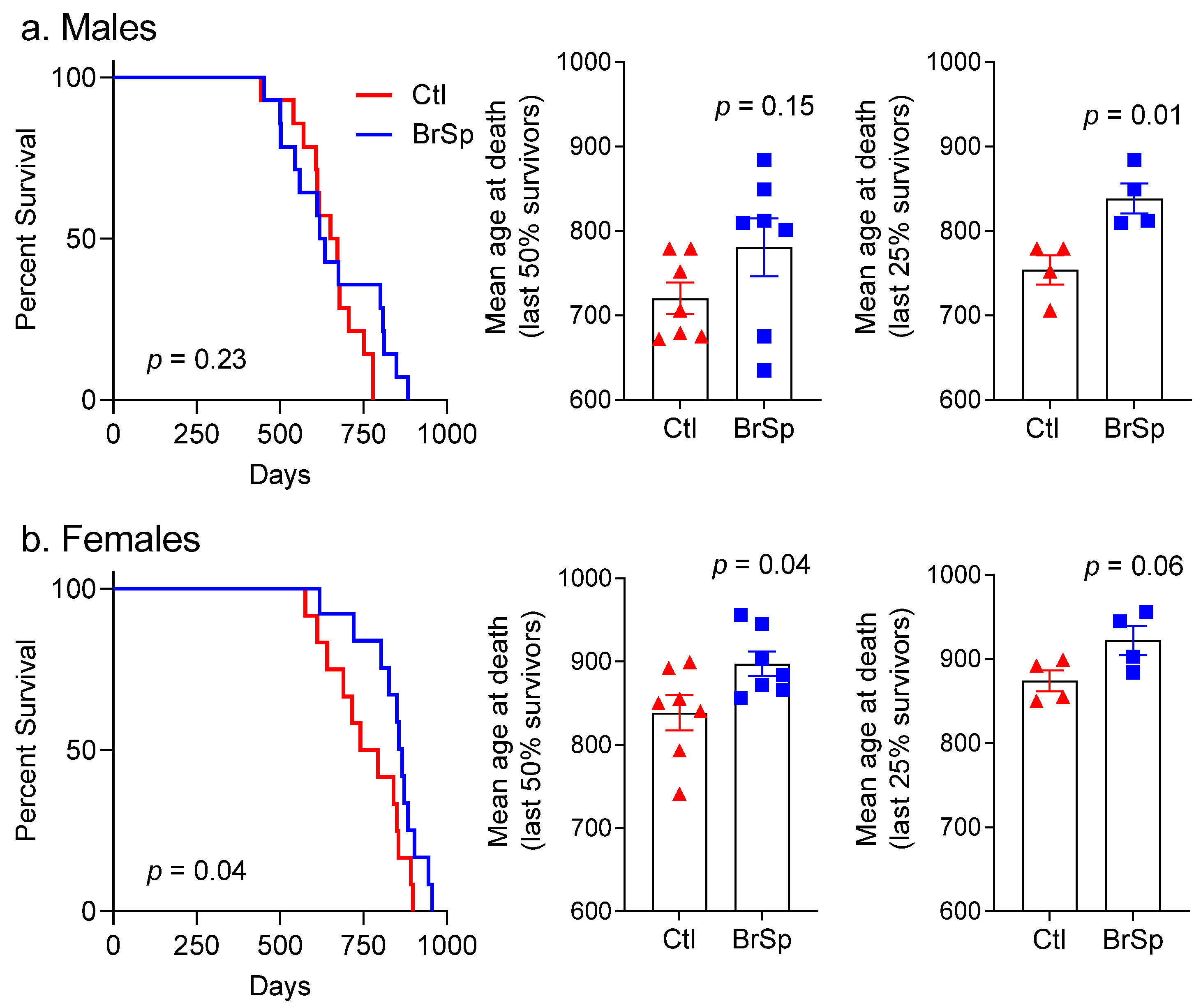
3.1. Survival and Mean Age at Death
3.2. Cause of Death
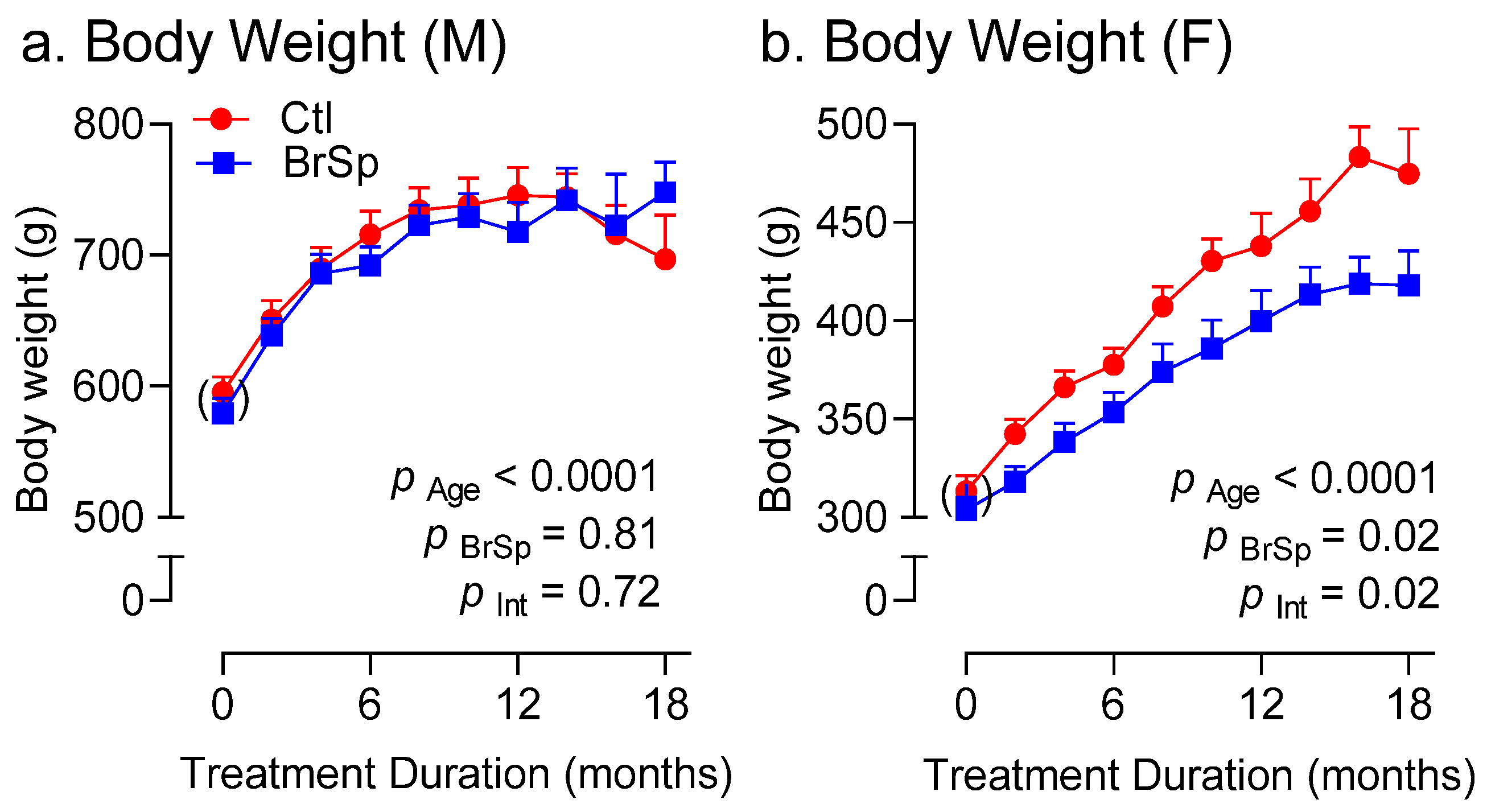
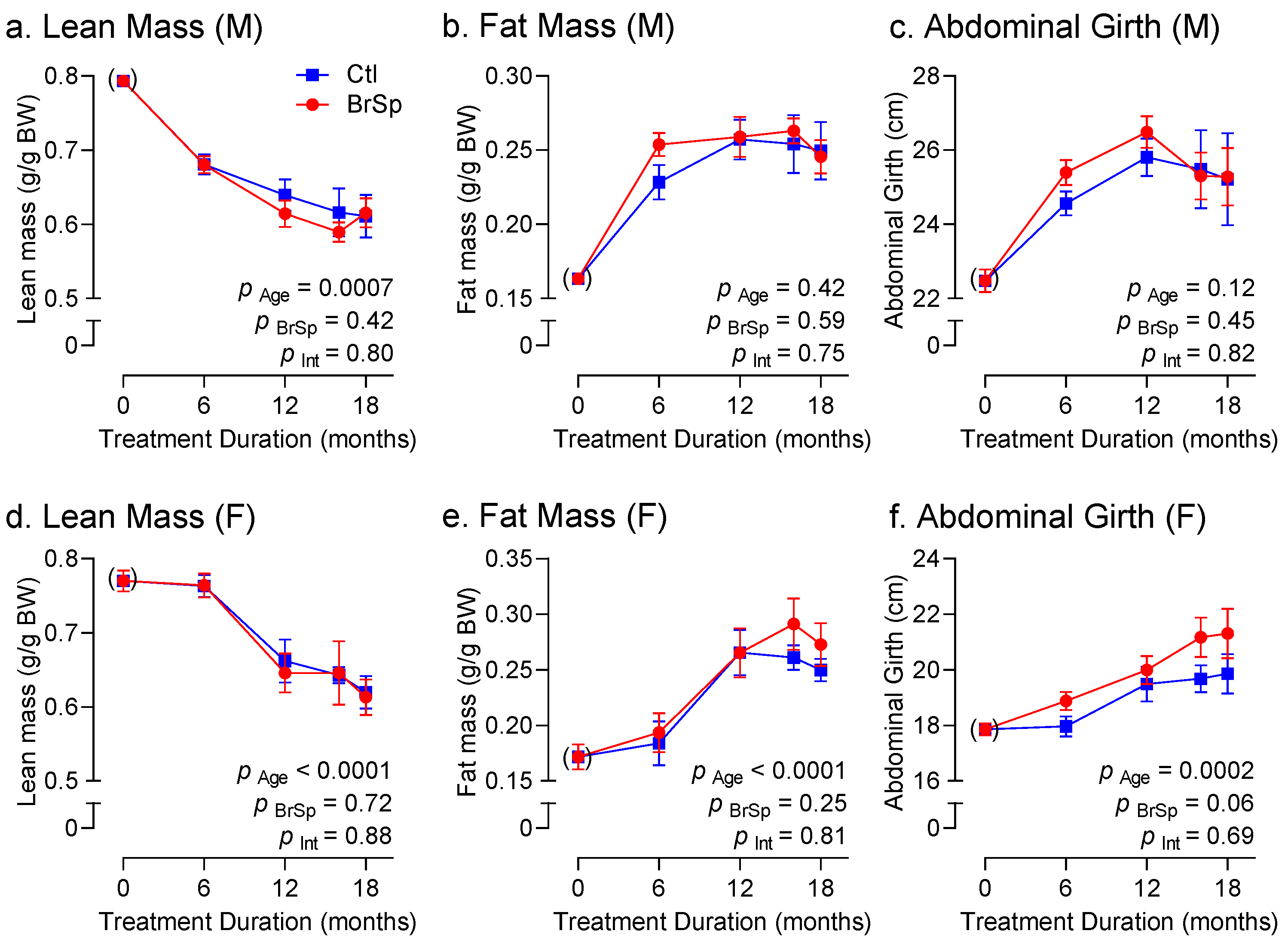
3.3. Body Weight and Composition
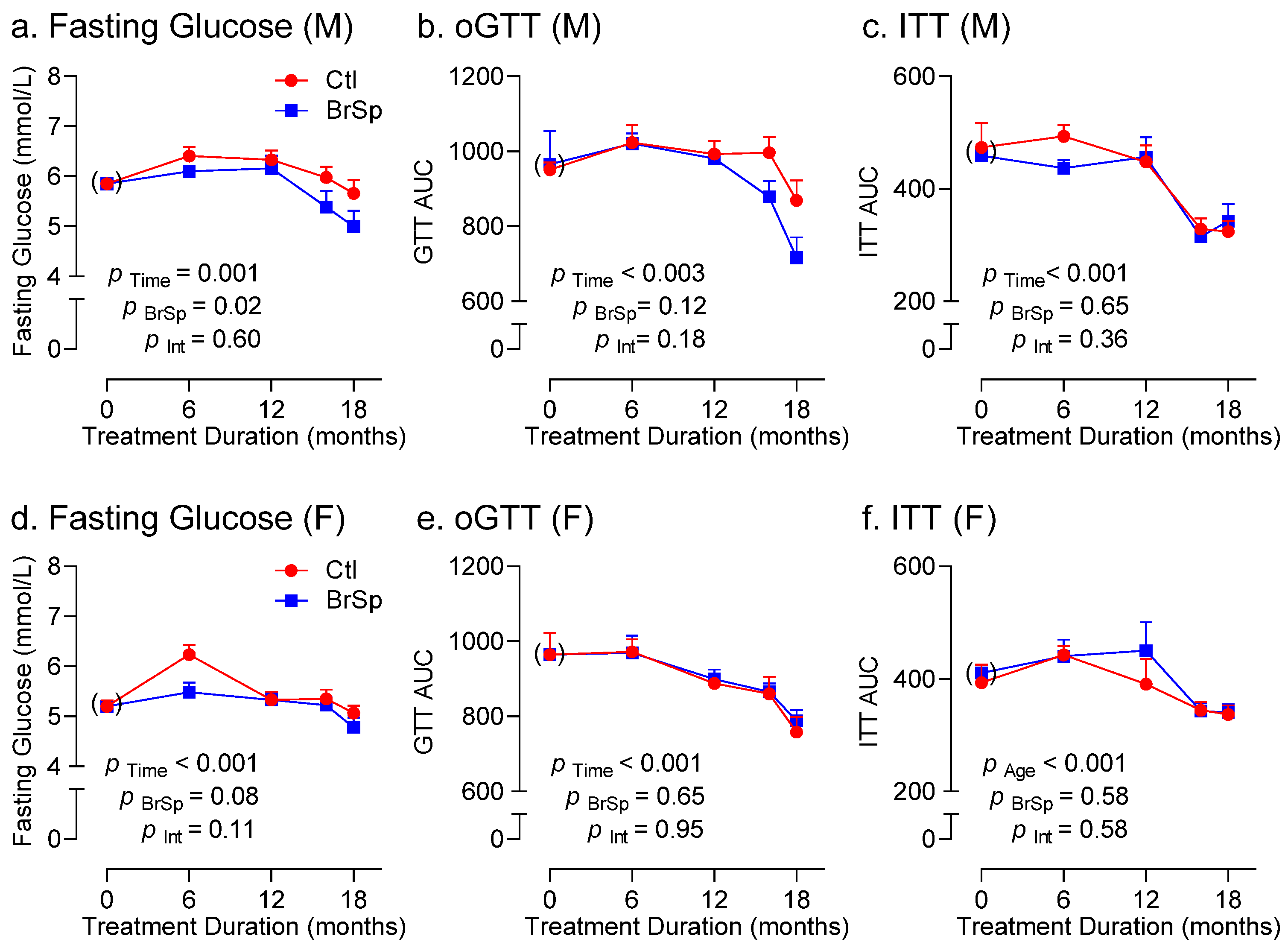
3.4. Glucose Homeostasis
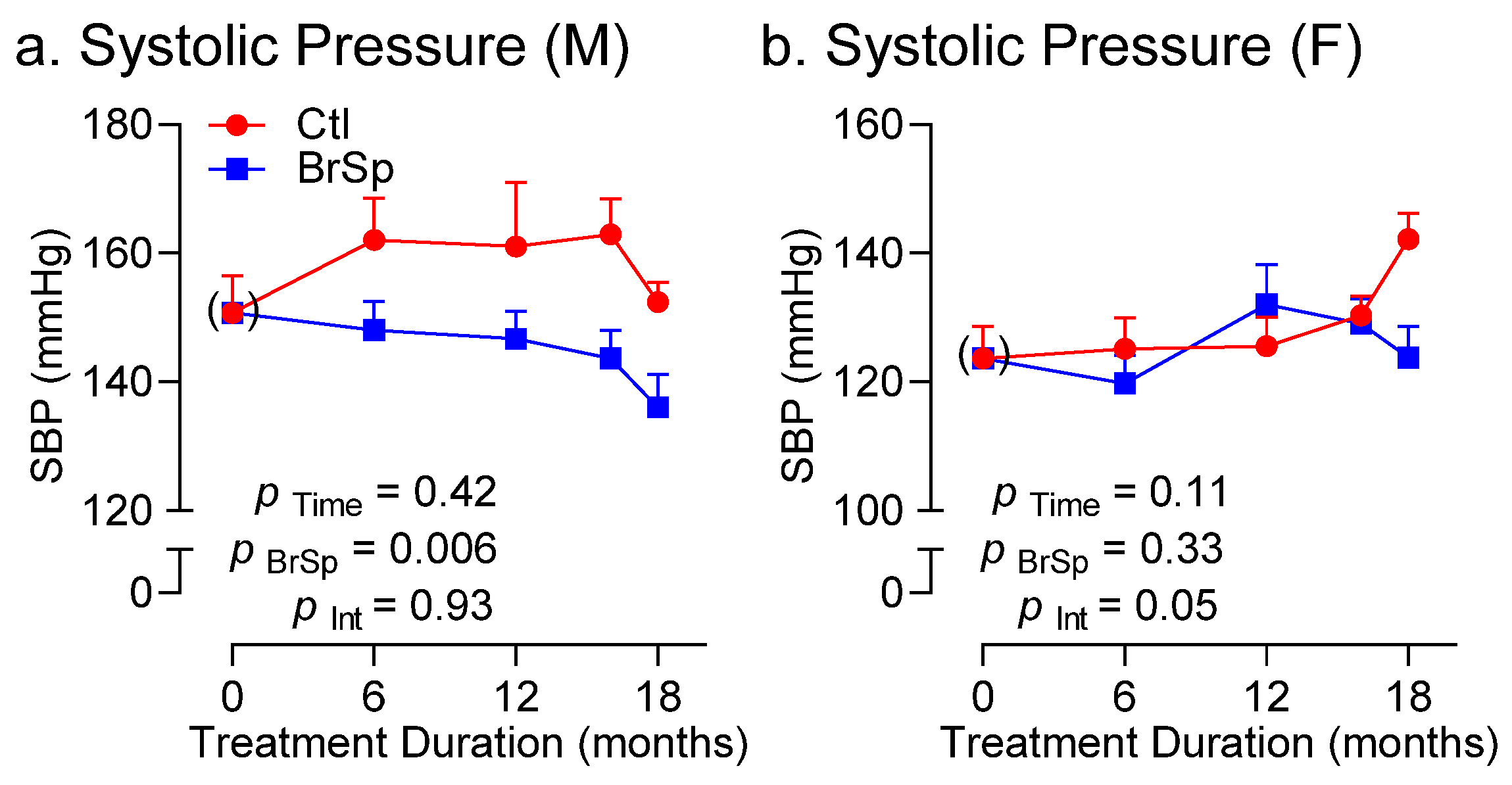
3.5. Blood Pressure and Cardiac Function
3.6. Behavioral Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bose, C.; Alves, I.; Singh, P.; Palade, P.T.; Carvalho, E.; Borsheim, E.; Jun, S.R.; Cheema, A.; Boerma, M.; Awasthi, S.; et al. Sulforaphane prevents age-associated cardiac and muscular dysfunction through Nrf2 signaling. Aging Cell 2020, 19, e13261. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, M.; Miquel, J. An update of the oxidation-inflammation theory of aging: The involvement of the immune system in oxi-inflamm-aging. Curr. Pharm. Des. 2009, 15, 3003–3026. [Google Scholar] [CrossRef] [PubMed]
- Cervantes Gracia, K.; Llanas-Cornejo, D.; Husi, H. CVD and Oxidative Stress. J. Clin. Med. 2017, 6, 22. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Liu, I.Y.; Bi, X.; Thompson, R.F.; Doctrow, S.R.; Malfroy, B.; Baudry, M. Reversal of age-related learning deficits and brain oxidative stress in mice with superoxide dismutase/catalase mimetics. Proc. Natl. Acad. Sci. USA 2003, 100, 8526–8531. [Google Scholar] [CrossRef]
- Masood, A.; Nadeem, A.; Mustafa, S.J.; O’Donnell, J.M. Reversal of oxidative stress-induced anxiety by inhibition of phosphodiesterase-2 in mice. J. Pharmacol. Exp. Ther. 2008, 326, 369–379. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Anil Kumar, N.V.; Zucca, P.; Varoni, E.M.; Dini, L.; Panzarini, E.; Rajkovic, J.; Tsouh Fokou, P.V.; Azzini, E.; Peluso, I.; et al. Lifestyle, Oxidative Stress, and Antioxidants: Back and Forth in the Pathophysiology of Chronic Diseases. Front. Physiol. 2020, 11, 694. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Jahandideh, F.; Wu, J. Food-derived bioactive peptides on inflammation and oxidative stress. Biomed. Res. Int. 2014, 2014, 608979. [Google Scholar] [CrossRef]
- Bai, Y.; Cui, W.; Xin, Y.; Miao, X.; Barati, M.T.; Zhang, C.; Chen, Q.; Tan, Y.; Cui, T.; Zheng, Y.; et al. Prevention by sulforaphane of diabetic cardiomyopathy is associated with up-regulation of Nrf2 expression and transcription activation. J. Mol. Cell Cardiol. 2013, 57, 82–95. [Google Scholar] [CrossRef]
- Egner, P.A.; Chen, J.G.; Wang, J.B.; Wu, Y.; Sun, Y.; Lu, J.H.; Zhu, J.; Zhang, Y.H.; Chen, Y.S.; Friesen, M.D.; et al. Bioavailability of Sulforaphane from two broccoli sprout beverages: Results of a short-term, cross-over clinical trial in Qidong, China. Cancer Prev. Res. 2011, 4, 384–395. [Google Scholar] [CrossRef]
- Black, A.M.; Armstrong, E.A.; Scott, O.; Juurlink, B.J.H.; Yager, J.Y. Broccoli sprout supplementation during pregnancy prevents brain injury in the newborn rat following placental insufficiency. Behav. Brain Res. 2015, 291, 289–298. [Google Scholar] [CrossRef]
- Nguyen, A.T.; Bahry, A.M.; Shen, K.Q.; Armstrong, E.A.; Yager, J.Y. Consumption of broccoli sprouts during late gestation and lactation confers protection against developmental delay induced by maternal inflammation. Behav. Brain Res. 2016, 307, 239–249. [Google Scholar] [CrossRef]
- Rubattu, S.; Di Castro, S.; Cotugno, M.; Bianchi, F.; Mattioli, R.; Baima, S.; Stanzione, R.; Madonna, M.; Bozzao, C.; Marchitti, S.; et al. Protective effects of Brassica oleracea sprouts extract toward renal damage in high-salt-fed SHRSP: Role of AMPK/PPARalpha/UCP2 axis. J. Hypertens 2015, 33, 1465–1479. [Google Scholar] [CrossRef]
- Xu, Z.; Wang, S.; Ji, H.; Zhang, Z.; Chen, J.; Tan, Y.; Wintergerst, K.; Zheng, Y.; Sun, J.; Cai, L. Broccoli sprout extract prevents diabetic cardiomyopathy via Nrf2 activation in db/db T2DM mice. Sci. Rep. 2016, 6, 30252. [Google Scholar] [CrossRef]
- Puspitasari, A.; Handayani, N. Broccoli sprouts juice prevents lens protein aggregation in streptozotocin-induced diabetic rat. Int. J. Ophthalmol. 2019, 12, 1380–1385. [Google Scholar] [CrossRef]
- Hagg, S.; Jylhava, J. Sex differences in biological aging with a focus on human studies. eLife 2021, 10, e63425. [Google Scholar] [CrossRef]
- Yagishita, Y.; Fahey, J.W.; Dinkova-Kostova, A.T.; Kensler, T.W. Broccoli or Sulforaphane: Is It the Source or Dose That Matters? Molecules 2019, 24, 3593. [Google Scholar] [CrossRef]
- Carter, R.J.; Morton, J.; Dunnett, S.B. Motor coordination and balance in rodents. Curr. Protoc. Neurosci. 2001, 15, 8.12.1–8.12.14. [Google Scholar] [CrossRef]
- Ardesch, D.J.; Balbi, M.; Murphy, T.H. Automated touch sensing in the mouse tapered beam test using Raspberry Pi. J. Neurosci. Methods 2017, 291, 221–226. [Google Scholar] [CrossRef]
- Turner, K.M.; Burne, T.H. Comprehensive behavioural analysis of Long Evans and Sprague-Dawley rats reveals differential effects of housing conditions on tests relevant to neuropsychiatric disorders. PLoS ONE 2014, 9, e93411. [Google Scholar] [CrossRef]
- Prut, L.; Belzung, C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: A review. Eur. J. Pharmacol. 2003, 463, 3–33. [Google Scholar] [CrossRef]
- Schmitt, U.; Hiemke, C. Combination of open field and elevated plus-maze: A suitable test battery to assess strain as well as treatment differences in rat behavior. Prog. Neuropsychopharmacol. Biol. Psychiatry 1998, 22, 1197–1215. [Google Scholar] [CrossRef]
- Fedarko, N.S. The biology of aging and frailty. Clin. Geriatr. Med. 2011, 27, 27–37. [Google Scholar] [CrossRef]
- Maio, M.T.; Hannan, J.L.; Komolova, M.; Adams, M.A. Caloric restriction prevents visceral adipose tissue accumulation and maintains erectile function in aging rats. J. Sex. Med. 2012, 9, 2273–2283. [Google Scholar] [CrossRef]
- Ghezzi, A.C.; Cambri, L.T.; Botezelli, J.D.; Ribeiro, C.; Dalia, R.A.; de Mello, M.A. Metabolic syndrome markers in wistar rats of different ages. Diabetol. Metab. Syndr. 2012, 4, 16. [Google Scholar] [CrossRef]
- Carrascosa, J.M.; Ruiz, P.; Martinez, C.; Pulido, J.A.; Satrustegui, J.; Andres, A. Insulin receptor kinase activity in rat adipocytes is decreased during aging. Biochem. Biophys. Res. Commun. 1989, 160, 303–309. [Google Scholar] [CrossRef]
- Escriva, F.; Agote, M.; Rubio, E.; Molero, J.C.; Pascual-Leone, A.M.; Andres, A.; Satrustegui, J.; Carrascosa, J.M. In vivo insulin-dependent glucose uptake of specific tissues is decreased during aging of mature Wistar rats. Endocrinology 1997, 138, 49–54. [Google Scholar] [CrossRef][Green Version]
- Bornstein, A.B.; Rao, S.S.; Marwaha, K. Left Ventricular Hypertrophy. [Updated 2021 Dec 19]. In StatPearls [Internet]; StatPearls Publishing LLC: Treasure Island, FL, USA, 2022. [Google Scholar]
- Franco, O.H.; Peeters, A.; Bonneux, L.; de Laet, C. Blood pressure in adulthood and life expectancy with cardiovascular disease in men and women: Life course analysis. Hypertension 2005, 46, 280–286. [Google Scholar] [CrossRef]
- Hardy, S.T.; Loehr, L.R.; Butler, K.R.; Chakladar, S.; Chang, P.P.; Folsom, A.R.; Heiss, G.; MacLehose, R.F.; Matsushita, K.; Avery, C.L. Reducing the Blood Pressure-Related Burden of Cardiovascular Disease: Impact of Achievable Improvements in Blood Pressure Prevention and Control. J. Am. Heart Assoc. 2015, 4, e002276. [Google Scholar] [CrossRef] [PubMed]
- Fryar, C.D.; Ostchega, Y.; Hales, C.M.; Zhang, G.; Kruszon-Moran, D. Hypertension Prevalence and Control Among Adults: United States, 2015–2016. NCHS Data Brief. 2017, 289, 1–8. [Google Scholar]
- Sengupta, P. The Laboratory Rat: Relating Its Age With Human’s. Int. J. Prev. Med. 2013, 4, 624–630. [Google Scholar] [PubMed]
- Lu, K.H.; Hopper, B.R.; Vargo, T.M.; Yen, S.S. Chronological changes in sex steroid, gonadotropin and prolactin secretions in aging female rats displaying different reproductive states. Biol. Reprod. 1979, 21, 193–203. [Google Scholar] [CrossRef]
- Wu, L.; Noyan Ashraf, M.H.; Facci, M.; Wang, R.; Paterson, P.G.; Ferrie, A.; Juurlink, B.H. Dietary approach to attenuate oxidative stress, hypertension, and inflammation in the cardiovascular system. Proc. Natl. Acad. Sci. USA 2004, 101, 7094–7099. [Google Scholar] [CrossRef]
- Piper, M.D.; Skorupa, D.; Partridge, L. Diet, metabolism and lifespan in Drosophila. Exp. Gerontol. 2005, 40, 857–862. [Google Scholar] [CrossRef]
- Golbidi, S.; Daiber, A.; Korac, B.; Li, H.; Essop, M.F.; Laher, I. Health Benefits of Fasting and Caloric Restriction. Curr. Diab. Rep. 2017, 17, 123. [Google Scholar] [CrossRef]
- Watkins, R.E.; Coates, R.; Ferroni, P. Measurement of aging anxiety in an elderly Australian population. Int. J. Aging Hum. Dev. 1998, 46, 319–332. [Google Scholar] [CrossRef]
- Moreno-Torres, A.; Pujol, J.; Soriano-Mas, C.; Deus, J.; Iranzo, A.; Santamaria, J. Age-related metabolic changes in the upper brainstem tegmentum by MR spectroscopy. Neurobiol. Aging 2005, 26, 1051–1059. [Google Scholar] [CrossRef]
- Melancon, M.O.; Lorrain, D.; Dionne, I.J. Exercise and sleep in aging: Emphasis on serotonin. Pathol. Biol. 2014, 62, 276–283. [Google Scholar] [CrossRef]
- Wallace, J.E.; Krauter, E.E.; Campbell, B.A. Motor and reflexive behavior in the aging rat. J. Gerontol. 1980, 35, 364–370. [Google Scholar] [CrossRef]
- Avgustinovich, D.F.; Lipina, T.V.; Bondar, N.P.; Alekseyenko, O.V.; Kudryavtseva, N.N. Features of the genetically defined anxiety in mice. Behav. Genet. 2000, 30, 101–109. [Google Scholar] [CrossRef]
- Gokdemir, O.; Cetinkaya, C.; Gumus, H.; Aksu, I.; Kiray, M.; Ates, M.; Kiray, A.; Baykara, B.; Baykara, B.; Sisman, A.R.; et al. The effect of exercise on anxiety- and depression-like behavior of aged rats. Biotech. Histochem. 2020, 95, 8–17. [Google Scholar] [CrossRef]
| Groups | N | Total | % |
|---|---|---|---|
| Male-Ctl | 11 | 13 | 84.6% |
| Male-BrSp | 8 | 12 | 66.7% |
| Female-Ctl | 8 | 11 | 72.7% |
| Female-BrSp | 7 | 12 | 58.3% |
| Ctl | BrSp | p-Value | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 Months | 6 Months | 12 Months | 16 Months | 18 Months | 0 Months | 6 Months | 12 Months | 16 Months | 18 Months | Time | BrSp | Int | ||
| Male Offspring | IVSd, mm | 1.7 ± 0.0 | 2.2 ± 0.1 | 2.4 ± 0.1 | 2.2 ± 0.1 | 2.4 ± 0.1 | 1.7 ± 0.0 | 2.2 ± 0.1 | 2.4 ± 0.1 | 2.1 ± 0.0 | 2.2 ± 0.1 | 0.008 | 0.37 | 0.51 |
| IVSs, mm | 2.3 ± 0.3 | 3.6 ± 0.2 | 3.8 ± 0.2 | 3.6 ± 0.1 | 4.0 ± 0.2 | 2.3 ± 0.3 | 3.6 ± 0.1 | 4.0 ± 0.1 | 3.5 ± 0.1 | 3.8 ± 0.2 | 0.02 | 0.85 | 0.40 | |
| LVIDd, mm | 7.9 ± 0.6 | 8.4 ± 0.2 | 8.8 ± 0.2 | 9.2 ± 0.3 | 8.8 ± 0.2 | 8.0 ± 0.3 | 8.3 ± 0.2 | 8.1 ± 0.2 | 8.2 ± 0.5 | 8.4 ± 0.4 | 0.61 | 0.04 | 0.31 | |
| LVIDs, mm | 3.7 ± 0.6 | 4.2 ± 0.3 | 4.4 ± 0.3 | 4.5 ± 0.3 | 4.0 ± 0.4 | 3.7 ± 0.6 | 3.8 ± 0.3 | 3.5 ± 0.1 | 3.9 ± 0.4 | 3.7 ± 0.3 | 0.75 | 0.18 | 0.60 | |
| LVPWd, mm | 2.2 ± 0.2 | 2.2 ± 0.1 | 2.3 ± 0.1 | 2.0 ± 0.2 | 2.3 ± 0.2 | 2.2 ± 0.2 | 2.3 ± 0.1 | 2.1 ± 0.1 | 2.1 ± 0.2 | 2.2 ± 0.1 | 0.72 | 0.88 | 0.78 | |
| LVPWs, mm | 3.4 ± 0.1 | 3.6 ± 0.2 | 3.7 ± 0.1 | 3.7 ± 0.2 | 3.8 ± 0.2 | 3.5 ± 0.0 | 3.8 ± 0.1 | 3.6 ± 0.1 | 3.6 ± 0.1 | 3.8 ± 0.1 | 0.81 | 0.88 | 0.79 | |
| Corrected LV mass, g | 1.0 ± 0.1 | 1.3 ± 0.05 | 1.5 ± 0.06 | 1.4 ± 0.09 | 1.4 ± 0.07 | 1.0 ± 0.1 | 1.2 ± 0.04 | 1.3 ± 0.06 | 1.2 ± 0.06 | 1.3 ± 0.07 | 0.03 | 0.04 | 0.17 | |
| Female Offspring | IVSd, mm | 1.9 ± 0.1 | 1.9 ± 0.1 | 1.8 ± 0.0 | 1.9 ± 0.1 | 1.9 ± 0.1 | 1.9 ± 0.1 | 1.9 ± 0.1 | 1.8 ± 0.0 | 1.9 ± 0.1 | 1.9 ± 0.0 | 0.32 | 0.60 | 0.95 |
| IVSs, mm | 3.2 ± 0.2 | 3.2 ± 0.1 | 3.4 ± 0.1 | 3.4 ± 0.1 | 3.3 ± 0.1 | 3.2 ± 0.2 | 3.2 ± 0.1 | 3.3 ± 0.1 | 3.2 ± 0.1 | 3.3 ± 0.1 | 0.69 | 0.49 | 0.99 | |
| LVIDd, mm | 6.1 ± 0.2 | 6.7 ± 0.2 | 6.8 ± 0.2 | 7.3 ± 0.2 | 7.4 ± 0.2 | 6.1 ± 0.2 | 6.2 ± 0.3 | 7.1 ± 0.1 | 7.1 ± 0.3 | 7.0 ± 0.2 | 0.003 | 0.46 | 0.25 | |
| LVIDs, mm | 2.8 ± 0.1 | 3.0 ± 0.2 | 2.8 ± 0.2 | 3.4 ± 0.2 | 3.3 ± 0.3 | 2.8 ± 0.1 | 2.8 ± 0.3 | 3.1 ± 0.1 | 3.6 ± 0.3 | 3.3 ± 0.3 | 0.04 | 0.76 | 0.47 | |
| LVPWd, mm | 2.3 ± 0.2 | 1.7 ± 0.1 | 2.1 ± 0.1 | 2.1 ± 0.2 | 1.9 ± 0.1 | 2.3 ± 0.2 | 2.0 ± 0.1 | 2.0 ± 0.1 | 1.9 ± 0.1 | 2.1 ± 0.1 | 0.37 | 0.57 | 0.11 | |
| LVPWs, mm | 3.8 ± 0.1 | 3.1 ± 0.1 | 3.5 ± 0.1 | 3.6 ± 0.2 | 3.6 ± 0.2 | 3.8 ± 0.1 | 3.2 ± 0.1 | 3.5 ± 0.1 | 3.2 ± 0.1 | 3.5 ± 0.2 | 0.08 | 0.50 | 0.33 | |
| Corrected LV mass, g | 0.72 ± 0.03 | 0.69 ± 0.03 | 0.77 ± 0.04 | 0.92 ± 0.06 | 0.82 ± 0.04 | 0.72 ± 0.03 | 0.72 ± 0.04 | 0.79 ± 0.04 | 0.77 ± 0.03 | 0.82 ± 0.05 | 0.006 | 0.74 | 0.05 | |
| Ctl | BrSp | p-Value | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 Months | 6 Months | 12 Months | 16 Months | 18 Months | 0 Months | 6 Months | 12 Months | 16 Months | 18 Months | Time | BrSp | Int | ||
| Male Offspring | LV end-diastolic volume, µL | 340.6 ± 25.9 | 389.6 ± 24.5 | 427.5 ± 19.4 | 471.8 ± 29.5 | 404.1 ± 32.1 | 340.6 ± 25.9 | 380.1 ± 20.2 | 355.5 ± 14.6 | 369.9 ± 44.5 | 412.2 ± 41.8 | 0.51 | 0.14 | 0.11 |
| LV end-systolic volume, µL | 63.2 ± 19.0 | 84.4 ± 15.1 | 96.6 ± 15.7 | 96.6 ± 13.6 | 76.4 ± 13.2 | 63.2 ± 19.0 | 66.2 ± 10.9 | 54.3 ± 4.9 | 69.8 ± 14.8 | 59.9 ± 10.6 | 0.79 | 0.12 | 0.50 | |
| Hear rate (bpm) | 394.3 ± 7.8 | 332.1 ± 14.6 | 333.6 ± 12.6 | 328.9 ± 5.4 | 325.8 ± 17.7 | 426.2 ± 0.7 | 351.1 ± 9.8 | 315.8 ± 12.1 | 288.2 ± 15.8 | 309.5 ± 17.7 | 0.06 | 0.30 | 0.14 | |
| Stroke volume, mL | 277.4 ± 10.2 | 305.2 ± 13.1 | 330.9 ± 14.8 | 375.2 ± 28.0 | 327.6 ± 21.6 | 277.4 ± 10.2 | 313.9 ± 13.0 | 301.2 ± 10.8 | 300.1 ± 33.7 | 328.4 ± 24.3 | 0.55 | 0.19 | 0.13 | |
| Cardiac output, mL/min | 113.6 ± 2.9 | 100.5 ± 4.5 | 110.4 ± 6.7 | 123.0 ± 8.5 | 106.0 ± 8.4 | 113.6 ± 2.9 | 109.7 ± 4.6 | 94.5 ± 3.9 | 84.1 ± 6.9 ** | 102.9 ± 8.4 | 0.92 | 0.08 | 0.003 | |
| Cardiac output, mL/min.g | 0.18 ± 0.00 | 0.13 ± 0.01 | 0.14 ± 0.01 | 0.15 ± 0.01 | 0.14 ± 0.01 | 0.19 ± 0.00 | 0.14 ± 0.01 | 0.12 ± 0.01 | 0.10 ± 0.00 | 0.15 ± 0.02 | 0.17 | 0.34 | 0.004 | |
| Mitral Flow Doppler | ||||||||||||||
| E, mm/s | 918.4 ± 57.1 | 936.4 ± 49.7 | 886.2 ± 28.4 | 892.4 ± 76.4 | 917.1 ± 79.4 | 918.4 ± 57.1 | 838.1 ± 34.7 | 861.5 ± 40.5 | 807.1 ± 67.9 | 912.1 ± 90.2 | 0.72 | 0.24 | 0.77 | |
| A, mm/s | 998.1 ± 50.1 | 747.4 ± 72.2 | 754.4 ± 69.7 | 758.2 ± 86.6 | 827.0 ± 81.7 | 964.4 ± 0.0 | 718.9 ± 39.6 | 665.3 ± 51.7 | 485.4 ± 34.5 | 714.5 ± 72.8 | 0.18 | 0.10 | 0.48 | |
| E/A | 0.93 ± 0.08 | 1.31 ± 0.08 | 1.25 ± 0.11 | 1.08 ± 0.06 | 1.06 ± 0.06 | 0.93 ± 0.08 | 1.18 ± 0.05 | 1.32 ± 0.08 | 1.57 ± 0.13 | 1.28 ± 0.11 | 0.46 | 0.12 | 0.03 | |
| TEI index | 0.52 ± 0.05 | 0.44 ± 0.03 | 0.57 ± 0.04 | 0.46 ± 0.02 | 0.52 ± 0.04 | 0.52 ± 0.05 | 0.46 ± 0.04 | 0.55 ± 0.02 | 0.51 ± 0.06 | 0.57 ± 0.06 | 0.02 | 0.41 | 0.83 | |
| Female Offspring | LV end-diastolic volume, µL | 190.4 ± 9.2 | 240.9 ± 16.6 | 250.4 ± 12.8 | 289.8 ± 21.4 | 294.8 ± 14.7 | 194.0 ± 29.9 | 220.0 ± 17.5 | 258.8 ± 10.8 | 279.2 ± 20.8 | 274.4 ± 14.6 | <0.001 | 0.61 | 0.55 |
| LV end-systolic volume, µL | 20.0 ± 4.0 | 33.9 ± 4.9 | 29.2 ± 5.6 | 47.1 ± 6.6 | 40.2 ± 9.4 | 17.3 ± 4.8 | 28.4 ± 5.4 | 32.9 ± 3.8 | 55.8 ± 12.8 | 44.9 ± 9.3 | 0.007 | 0.62 | 0.62 | |
| Hear rate (bpm) | 505.7 ± 11.6 | 398.5 ± 12.9 | 403.3 ± 15.0 | 370.4 ± 21.1 | 370.3 ± 11.5 | 484.3 ± 10.8 | 383.6 ± 19.8 | 403.3 ± 15.9 | 357.2 ± 16.3 | 332.5 ± 26.7 | 0.004 | 0.29 | 0.76 | |
| Stroke volume, mL | 170.4 ± 5.2 | 207.0 ± 12.7 | 221.2 ± 8.7 | 242.6 ± 17.4 | 258.2 ± 13.8 | 176.7 ± 25.0 | 191.6 ± 15.1 | 225.9 ± 8.6 | 213.3 ± 12.3 | 229.5 ± 9.3 | 0.002 | 0.17 | 0.42 | |
| Cardiac output, mL/min | 86.2 ± 4.6 | 82.7 ± 6.0 | 88.5 ± 3.4 | 89.3 ± 7.0 | 95.9 ± 6.4 | 85.3 ± 10.2 | 72.4 ± 5.9 | 90.6 ± 4.4 | 76.2 ± 5.4 | 76.8 ± 7.2 | 0.09 | 0.04 | 0.24 | |
| Cardiac output, mL/min.g | 0.20 ± 0.02 | 0.17 ± 0.1 | 0.16 ± 0.01 | 0.16 ± 0.01 | 0.18 ± 0.02 | 0.21 ± 0.03 | 0.15 ± 0.01 | 0.17 ± 0.01 | 0.14 ± 0.01 | 0.14 ± 0.2 | 0.48 | 0.06 | 0.22 | |
| Mitral Flow Doppler | ||||||||||||||
| E, mm/s | 824.2 ± 0.0 | 837.1 ± 47.2 | 885.5 ± 56.3 | 827.1 ± 72.4 | 786.4 ± 43.9 | 832.6 ± 0.0 | 780.1 ± 44.6 | 889.0 ± 42.9 | 902.5 ± 53.7 | 797.9 ± 61.1 | 0.13 | 0.99 | 0.77 | |
| A, mm/s | 906.5 ± 0.0 | 854.4 ± 46.5 | 760.4 ± 60.2 | 661.4 ± 54.2 | 789.3 ± 41.2 | 926.7 ± 0.0 | 778.5 ± 51.9 | 764.0 ± 60.7 | 660.0 ± 42.8 | 766.9 ± 83.4 | 0.12 | 0.58 | 0.91 | |
| E/A | 0.91 ± 0.00 | 0.96 ± 0.03 | 1.24 ± 0.20 | 1.27 ± 0.13 | 1.05 ± 0.08 | 0.90 ± 0.00 | 1.10 ± 0.06 | 1.20 ± 0.10 | 1.46 ± 0.11 | 1.30 ± 0.20 | 0.10 | 0.28 | 0.75 | |
| TEI index | 0.54 ± 0.05 | 0.48 ± 0.05 | 0.58 ± 0.04 | 0.55 ± 0.07 | 0.54 ± 0.05 | 0.54 ± 0.05 | 0.44 ± 0.03 | 0.50 ± 0.03 | 0.50 ± 0.07 | 0.50 ± 0.03 | 0.11 | 0.22 | 0.96 | |
| Behavioral Test | Parameters | Male | Female | ||||
|---|---|---|---|---|---|---|---|
| Ctl | BrSp | p-Value | Ctl | BrSp | p-Value | ||
| Open Field Test | Total distance traveled (m) | 13.6 ± 1.9 | 16.2 ± 2.9 | 0.44 | 20.1 ± 1.2 | 21.8 ± 1.4 | 0.35 |
| Time mobile (s) | 53 ± 5 | 77 ± 10 | 0.03 | 79.11 ± 4.46 | 88 ± 4 | 0.13 | |
| Time in inner zone (s) | 37 ± 10 | 48 ± 12 | 0.49 | 27.69 ± 3.16 | 32 ± 5 | 0.48 | |
| Elevated Plus Maze | Time in open arm (s) | 118 ± 29 | 92 ± 26 | 0.41 | 125 ± 20 | 150 ± 21 | 0.41 |
| Time in closed arm (s) | 125 ± 20 | 150 ± 21 | 0.53 | 99 ± 19 | 100 ± 19 | 0.98 | |
| Entries into open arm | 6.4 ± 0.8 | 7.0 ± 1.1 | 0.66 | 9.4 ± 0.8 | 11.3 ± 1.6 | 0.35 | |
| Entries into closed arm | 5.8 ± 0.7 | 5.9 ± 1.5 | 0.97 | 8.4 ± 1.1 | 7.0 ± 0.8 | 0.30 | |
| Open arm avoidance Index | 53.9 ± 5.8 | 43.8 ± 8.2 | 0.31 | 52.1 ± 5.4 | 43.6 ± 5.7 | 0.30 | |
| Tapered beam | Total foot faults | 3.4 ± 0.6 | 3.5 ± 0.5 | 0.86 | 1.3 ± 0.4 | 0. 9 ± 0.2 | 0.43 |
| Right foot faults | 1.9 ± 0.4 | 2.1 ± 0.6 | 0.80 | 0.5 ± 0.2 | 0.3 ± 0.1 | 0.39 | |
| Left foot faults | 1. 5 ± 0.3 | 1.4 ± 0.4 | 0.97 | 1.0 ± 0.3 | 0.4 ± 0.1 | 0.06 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noble, R.M.N.; Jahandideh, F.; Armstrong, E.A.; Bourque, S.L.; Yager, J.Y. Broccoli Sprouts Promote Sex-Dependent Cardiometabolic Health and Longevity in Long-Evans Rats. Int. J. Environ. Res. Public Health 2022, 19, 13468. https://doi.org/10.3390/ijerph192013468
Noble RMN, Jahandideh F, Armstrong EA, Bourque SL, Yager JY. Broccoli Sprouts Promote Sex-Dependent Cardiometabolic Health and Longevity in Long-Evans Rats. International Journal of Environmental Research and Public Health. 2022; 19(20):13468. https://doi.org/10.3390/ijerph192013468
Chicago/Turabian StyleNoble, Ronan M. N., Forough Jahandideh, Edward A. Armstrong, Stephane L. Bourque, and Jerome Y. Yager. 2022. "Broccoli Sprouts Promote Sex-Dependent Cardiometabolic Health and Longevity in Long-Evans Rats" International Journal of Environmental Research and Public Health 19, no. 20: 13468. https://doi.org/10.3390/ijerph192013468
APA StyleNoble, R. M. N., Jahandideh, F., Armstrong, E. A., Bourque, S. L., & Yager, J. Y. (2022). Broccoli Sprouts Promote Sex-Dependent Cardiometabolic Health and Longevity in Long-Evans Rats. International Journal of Environmental Research and Public Health, 19(20), 13468. https://doi.org/10.3390/ijerph192013468







