Short-Term Associations between Size-Fractioned Particles and Cardiopulmonary Function in COPD Patients: A Panel Study in Shanghai, China, during 2014–2021
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample and Data
2.2. Measures of Variables
2.3. Models and Data Analysis Procedure
3. Results
3.1. Descriptive Statistics
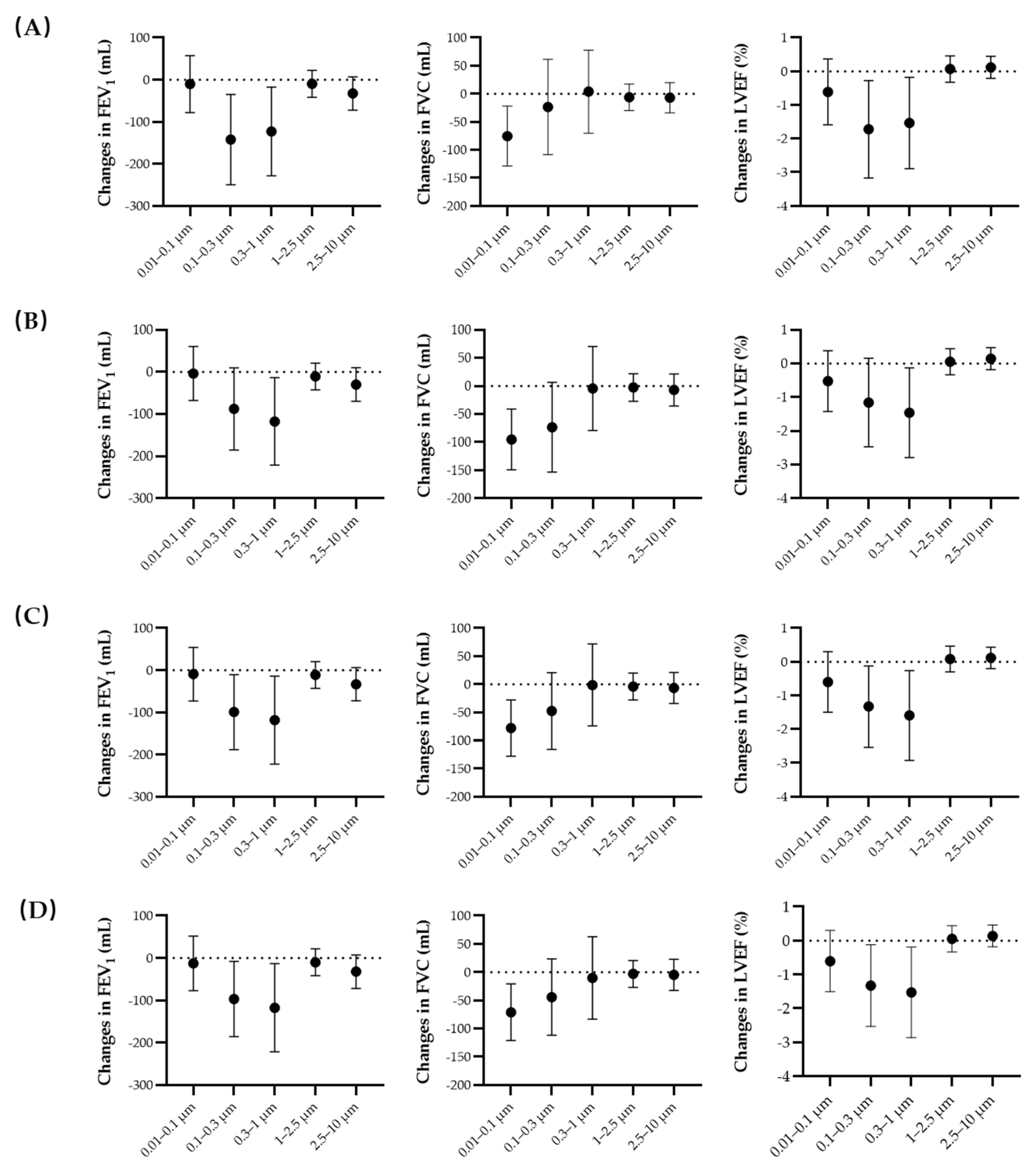
3.2. Regression Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Adeloye, D.; Song, P.; Zhu, Y.; Campbell, H.; Sheikh, A.; Rudan, I.; NIHR RESPIRE Global Respiratory Health Unit. Global, regional, and national prevalence of, and risk factors for, chronic obstructive pulmonary disease (COPD) in 2019: A systematic review and modelling analysis. Lancet Respir. Med. 2022, 10, 447–458. [Google Scholar] [CrossRef]
- GBD 2019 Diseases and Injuries Collaborators. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Zou, J.; Sun, T.; Song, X.; Liu, Y.M.; Lei, F.; Chen, M.M.; Chen, Z.; Zhang, P.; Ji, Y.X.; Zhang, X.J.; et al. Distributions and trends of the global burden of COPD attributable to risk factors by SDI, age, and sex from 1990 to 2019: A systematic analysis of GBD 2019 data. Respir. Res. 2022, 23, 90. [Google Scholar] [CrossRef] [PubMed]
- Cengiz ElçioĞlu, B.; Kamat, S.; Yurdakul, S.; Şahin, Ş.T.; Sarper, A.; Yıldız, P.; Aytekin, S. Assessment of Subclinical Left Ventricular Systolic Dysfunction and Structural Changes in Patients with Chronic Obstructive Pulmonary Disease. Intern. Med. J. 2021. [Google Scholar] [CrossRef]
- Spruit, M.A.; Singh, S.J.; Garvey, C.; ZuWallack, R.; Nici, L.; Rochester, C.; Hill, K.; Holland, A.E.; Lareau, S.C.; Man, W.D.; et al. An official American Thoracic Society/European Respiratory Society statement: Key concepts and advances in pulmonary rehabilitation. Am. J. Respir. Crit. Care Med. 2013, 188, e13–e64. [Google Scholar] [CrossRef]
- Boschetto, P.; Beghé, B.; Fabbri, L.M.; Ceconi, C. Link between chronic obstructive pulmonary disease and coronary artery disease: Implication for clinical practice. Respirology 2012, 17, 422–431. [Google Scholar] [CrossRef]
- Coccia, M. Evolution and structure of research fields driven by crises and environmental threats: The COVID-19 research. Scientometrics 2021, 126, 9405–9429. [Google Scholar] [CrossRef]
- Coccia, M. Factors determining the diffusion of COVID-19 and suggested strategy to prevent future accelerated viral infectivity similar to COVID. Sci. Total Environ. 2020, 729, 138474. [Google Scholar] [CrossRef]
- Kaur, M.; Chandel, J.; Malik, J.; Naura, A.S. Particulate matter in COPD pathogenesis: An overview. Inflamm. Res. 2022, 71, 797–815. [Google Scholar] [CrossRef]
- Lu, W.; Tian, Q.; Xu, R.; Zhong, C.; Qiu, L.; Zhang, H.; Shi, C.; Liu, Y.; Zhou, Y. Short-term exposure to ambient air pollution and pneumonia hospital admission among patients with COPD: A time-stratified case-crossover study. Respir. Res. 2022, 23, 71. [Google Scholar] [CrossRef]
- Liu, C.; Chen, R.; Sera, F.; Vicedo-Cabrera, A.M.; Guo, Y.; Tong, S.; Coelho, M.; Saldiva, P.H.N.; Lavigne, E.; Matus, P.; et al. Ambient Particulate Air Pollution and Daily Mortality in 652 Cities. N. Engl. J. Med. 2019, 381, 705–715. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, S.; Al-Kindi, S.G.; Brook, R.D. Air Pollution and Cardiovascular Disease: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2018, 72, 2054–2070. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Li, H.; Peng, L.; Wang, Y.; Wang, W. Effects of particulate matter on hospital admissions for respiratory diseases: An ecological study based on 12.5 years of time series data in Shanghai. Environ. Health 2022, 21, 12. [Google Scholar] [CrossRef] [PubMed]
- Paulin, L.; Hansel, N. Particulate air pollution and impaired lung function. F1000Research 2016, 5, 201. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Ma, R.; Wang, Y.; Jiang, N.; Zhang, Y.; Li, T. The relationship between particulate matter and lung function of children: A systematic review and meta-analysis. Environ. Pollut. 2022, 309, 119735. [Google Scholar] [CrossRef] [PubMed]
- Buteau, S.; Goldberg, M.S. A structured review of panel studies used to investigate associations between ambient air pollution and heart rate variability. Environ. Res. 2016, 148, 207–247. [Google Scholar] [CrossRef]
- Huang, C.; Tang, M.; Li, H.; Wen, J.; Wang, C.; Gao, Y.; Hu, J.; Lin, J.; Chen, R. Particulate matter air pollution and reduced heart rate variability: How the associations vary by particle size in Shanghai, China. Ecotox. Environ. Saf. 2021, 208, 111726. [Google Scholar] [CrossRef]
- Lammers, A.; Janssen, N.A.H.; Boere, A.J.F.; Berger, M.; Longo, C.; Vijverberg, S.J.H.; Neerincx, A.H.; Maitland-van der Zee, A.H.; Cassee, F.R. Effects of short-term exposures to ultrafine particles near an airport in healthy subjects. Environ. Int. 2020, 141, 105779. [Google Scholar] [CrossRef]
- Strak, M.; Janssen, N.A.; Godri, K.J.; Gosens, I.; Mudway, I.S.; Cassee, F.R.; Lebret, E.; Kelly, F.J.; Harrison, R.M.; Brunekreef, B.; et al. Respiratory health effects of airborne particulate matter: The role of particle size, composition, and oxidative potential-the RAPTES project. Environ. Health Perspect. 2012, 120, 1183–1189. [Google Scholar] [CrossRef]
- Brook, R.D.; Rajagopalan, S.; Pope, C.A., 3rd; Brook, J.R.; Bhatnagar, A.; Diez-Roux, A.V.; Holguin, F.; Hong, Y.; Luepker, R.V.; Mittleman, M.A.; et al. Particulate matter air pollution and cardiovascular disease: An update to the scientific statement from the American Heart Association. Circulation 2010, 121, 2331–2378. [Google Scholar] [CrossRef]
- Ohlwein, S.; Kappeler, R.; Kutlar Joss, M.; Künzli, N.; Hoffmann, B. Health effects of ultrafine particles: A systematic literature review update of epidemiological evidence. Int. J. Public Health 2019, 64, 547–559. [Google Scholar] [CrossRef] [PubMed]
- Strak, M.; Boogaard, H.; Meliefste, K.; Oldenwening, M.; Zuurbier, M.; Brunekreef, B.; Hoek, G. Respiratory health effects of ultrafine and fine particle exposure in cyclists. Occup. Environ. Med. 2010, 67, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, G.; Marks, G.B.; Morawska, L. Health effects of daily airborne particle dose in children: Direct association between personal dose and respiratory health effects. Environ. Pollut. 2013, 180, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Koppelman, G.H.; Hoek, G.; Kerckhoffs, J.; Vonk, J.M.; Vermeulen, R.; Gehring, U. Ultrafine particles, particle components and lung function at age 16 years: The PIAMA birth cohort study. Environ. Int. 2021, 157, 106792. [Google Scholar] [CrossRef]
- van Nunen, E.; Hoek, G.; Tsai, M.Y.; Probst-Hensch, N.; Imboden, M.; Jeong, A.; Naccarati, A.; Tarallo, S.; Raffaele, D.; Nieuwenhuijsen, M.; et al. Short-term personal and outdoor exposure to ultrafine and fine particulate air pollution in association with blood pressure and lung function in healthy adults. Environ. Res. 2021, 194, 110579. [Google Scholar] [CrossRef]
- Sun, Y.; Song, X.; Han, Y.; Ji, Y.; Gao, S.; Shang, Y.; Lu, S.E.; Zhu, T.; Huang, W. Size-fractioned ultrafine particles and black carbon associated with autonomic dysfunction in subjects with diabetes or impaired glucose tolerance in Shanghai, China. Part. Fibre. Toxicol. 2015, 12, 8. [Google Scholar] [CrossRef]
- Pan, L.; Wu, S.; Li, H.; Xu, J.; Dong, W.; Shan, J.; Yang, X.; Chen, Y.; Shima, M.; Deng, F.; et al. The short-term effects of indoor size-fractioned particulate matter and black carbon on cardiac autonomic function in COPD patients. Environ. Int. 2018, 112, 261–268. [Google Scholar] [CrossRef]
- Fang, L.; Gao, P.; Bao, H.; Tang, X.; Wang, B.; Feng, Y.; Cong, S.; Juan, J.; Fan, J.; Lu, K.; et al. Chronic obstructive pulmonary disease in China: A nationwide prevalence study. Lancet Respir. Med. 2018, 6, 421–430. [Google Scholar] [CrossRef]
- Vogelmeier, C.F.; Criner, G.J.; Martinez, F.J.; Anzueto, A.; Barnes, P.J.; Bourbeau, J.; Celli, B.R.; Chen, R.; Decramer, M.; Fabbri, L.M.; et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease 2017 Report. GOLD Executive Summary. Am. J. Respir. Crit. Care Med. 2017, 195, 557–582. [Google Scholar] [CrossRef]
- Doiron, D.; de Hoogh, K.; Probst-Hensch, N.; Fortier, I.; Cai, Y.; De Matteis, S.; Hansell, A.L. Air pollution, lung function and COPD: Results from the population-based UK Biobank study. Eur. Respir. J. 2019, 54, 1802140. [Google Scholar] [CrossRef]
- Zhou, L.; Tao, Y.; Li, H.; Niu, Y.; Li, L.; Kan, H.; Xie, J.; Chen, R. Acute effects of fine particulate matter constituents on cardiopulmonary function in a panel of COPD patients. Sci. Total Environ. 2021, 770, 144753. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.; Gustafsson, P.; et al. Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Gottdiener, J.S.; Bednarz, J.; Devereux, R.; Gardin, J.; Klein, A.; Manning, W.J.; Morehead, A.; Kitzman, D.; Oh, J.; Quinones, M.; et al. American Society of Echocardiography recommendations for use of echocardiography in clinical trials. J. Am. Coll. Cardiol. 2004, 17, 1086–1119. [Google Scholar] [CrossRef] [PubMed]
- Ponikowski, P.; Voors, A.A.; Anker, S.D.; Bueno, H.; Cleland, J.G.; Coats, A.J.; Falk, V.; González-Juanatey, J.R.; Harjola, V.P.; Jankowska, E.A.; et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 2016, 18, 891–975. [Google Scholar]
- Khlystov, A.; Stanier, C.; Pandis, S.N. An Algorithm for Combining Electrical Mobility and Aerodynamic Size Distributions Data when Measuring Ambient Aerosol Special Issue of Aerosol Science and Technology on Findings from the Fine Particulate Matter Supersites Program. Aerosol Sci. Technol. 2004, 38, 229–238. [Google Scholar] [CrossRef]
- Meng, X.; Ma, Y.; Chen, R.; Zhou, Z.; Chen, B.; Kan, H. Size-fractionated particle number concentrations and daily mortality in a Chinese city. Environ. Health Perspect. 2013, 121, 1174–1178. [Google Scholar] [CrossRef]
- Zhao, A.; Chen, R.; Wang, C.; Zhao, Z.; Yang, C.; Lu, J.; Chen, X.; Kan, H. Associations between size-fractionated particulate air pollution and blood pressure in a panel of type II diabetes mellitus patients. Environ. Int. 2015, 80, 19–25. [Google Scholar] [CrossRef]
- Chen, R.; Zhao, Z.; Sun, Q.; Lin, Z.; Zhao, A.; Wang, C.; Xia, Y.; Xu, X.; Kan, H. Size-fractionated particulate air pollution and circulating biomarkers of inflammation, coagulation, and vasoconstriction in a panel of young adults. Epidemiology 2015, 26, 328–336. [Google Scholar] [CrossRef]
- Duan, R.; Niu, H.; Yu, T.; Huang, K.; Cui, H.; Chen, C.; Yang, T.; Wang, C. Adverse effects of short-term personal exposure to fine particulate matter on the lung function of patients with chronic obstructive pulmonary disease and asthma: A longitudinal panel study in Beijing, China. Environ. Sci. Pollut. R. 2021, 28, 47463–47473. [Google Scholar] [CrossRef]
- Chen, T.; Chen, F.; Wang, K.; Ma, X.; Wei, X.; Wang, W.; Huang, P.; Yang, D.; Xia, Z.; Zhao, Z. Acute respiratory response to individual particle exposure (PM1.0, PM2.5 and PM10) in the elderly with and without chronic respiratory diseases. Environ. Pollut. 2021, 271, 116329. [Google Scholar] [CrossRef]
- Wu, S.; Ni, Y.; Li, H.; Pan, L.; Yang, D.; Baccarelli, A.A.; Deng, F.; Chen, Y.; Shima, M.; Guo, X. Short-term exposure to high ambient air pollution increases airway inflammation and respiratory symptoms in chronic obstructive pulmonary disease patients in Beijing, China. Environ. Int. 2016, 94, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, H.; Pan, L.; Xu, J.; Yang, X.; Dong, W.; Shan, J.; Wu, S.; Deng, F.; Chen, Y.; et al. Chemical constituents and sources of indoor PM2.5 and cardiopulmonary function in patients with chronic obstructive pulmonary disease: Estimation of individual and joint effects. Environ. Res. 2021, 197, 111191. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Zhou, Y.; Liu, S.; Chen, X.; Zou, W.; Zhao, D.; Li, X.; Pu, J.; Huang, L.; Chen, J.; et al. Association between exposure to ambient particulate matter and chronic obstructive pulmonary disease: Results from a cross-sectional study in China. Thorax 2017, 72, 788–795. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Xiao, S.; Gao, W.; Zhou, Y.; Zhou, J.; Yang, D.; Ye, X. Short-term associations between size-fractionated particulate air pollution and COPD mortality in Shanghai, China. Environ. Pollut. 2020, 257, 113483. [Google Scholar] [CrossRef]
- Chuang, K.J.; Chan, C.C.; Chen, N.T.; Su, T.C.; Lin, L.Y. Effects of particle size fractions on reducing heart rate variability in cardiac and hypertensive patients. Environ. Health Perspect. 2005, 113, 1693–1697. [Google Scholar] [CrossRef]
- Rivas, I.; Vicens, L.; Basagaña, X.; Tobías, A.; Katsouyanni, K.; Walton, H.; Hüglin, C.; Alastuey, A.; Kulmala, M.; Harrison, R.M.; et al. Associations between sources of particle number and mortality in four European cities. Environ. Int. 2021, 155, 106662. [Google Scholar] [CrossRef]
- Coccia, M. How do low wind speeds and high levels of air pollution support the spread of COVID-19? Atmos. Pollut. Res. 2021, 12, 437–445. [Google Scholar] [CrossRef]
- Morakinyo, O.M.; Mokgobu, M.I.; Mukhola, M.S.; Hunter, R.P. Health Outcomes of Exposure to Biological and Chemical Components of Inhalable and Respirable Particulate Matter. Int. J. Environ. Res. Public Health 2016, 13, 592. [Google Scholar] [CrossRef]

| Item | Measurement |
|---|---|
| Age, years, mean ± SD | 73.6 ± 9.5 |
| Gender, N (%) | |
| Male | 171 (81.0%) |
| Female | 40 (19.0%) |
| BMI, Kg/m2, mean ± SD | 22.9 ± 3.6 |
| Smoking status, N (%) | |
| Never | 111 (52.6%) |
| Former | 48 (22.8%) |
| Current | 52 (24.6%) |
| Medication use, N (%) | 171 (81.0%) |
| Cardiopulmonary function measures, mean ± SD | |
| FEV1, mL | 1094.3 ± 556.5 |
| FVC, mL | 722.4 ± 689.5 |
| LVEF, % | 64.5 ± 5.4 |
| Exposure | Mean | SD | P25 | P50 | P75 | IQR |
|---|---|---|---|---|---|---|
| PNC, particles/cm3 | ||||||
| 0.01–0.1 μm | 4229.8 | 2310.6 | 2647.3 | 3769.1 | 5212.3 | 2565.0 |
| 0.1–0.3 μm | 2424.2 | 1279.5 | 1476.0 | 2182.3 | 3144.9 | 1668.9 |
| 0.3–1 μm | 275.6 | 206.1 | 127.9 | 227.9 | 362.8 | 234.9 |
| 1–2.5 μm | 4.4 | 12.2 | 0.0 | 1.9 | 3.9 | 3.9 |
| 2.5–10 μm | 0.2 | 0.4 | 0.0 | 0.0 | 0.2 | 0.2 |
| Air pollutants, μg/m3 | ||||||
| PM2.5 | 37.7 | 27.0 | 20.8 | 32.5 | 50.5 | 29.7 |
| NO2 | 44.0 | 19.7 | 30.0 | 40.9 | 54.1 | 24.1 |
| SO2 | 10.4 | 7.7 | 5.7 | 8.5 | 12.9 | 7.2 |
| O3 (8 h mean) | 96.1 | 45.9 | 63.5 | 88.5 | 121.6 | 58.1 |
| CO, mg/m3 | 0.78 | 0.29 | 0.59 | 0.74 | 0.92 | 0.33 |
| Weather conditions | ||||||
| Temperature, °C | 17.6 | 8.5 | 10.2 | 18.6 | 24.5 | 14.3 |
| Relative humidity, % | 73.9 | 12.7 | 65.0 | 75.0 | 83.0 | 18.0 |
| Wind Speed, m/s | 2.5 | 0.9 | 1.9 | 2.4 | 3.0 | 1.1 |
| FEV1 (mL) | FVC (mL) | LVEF (%) | |
|---|---|---|---|
| PNC0.01–0.1 | −9.9 (−73.5, 53.7) | −78.4 (−129.1, −27.7) * | −0.6 (−1.5, 0.3) |
| PNC0.1–0.3 | −97.5 (−186.1, −8.9) * | −41.6 (−110.8, 27.7) | −1.3 (−2.5, −0.1) * |
| PNC0.3–1 | −119.5 (−223.2, −15.7) * | −4.1 (−77.8, 69.5) | −1.5 (−2.9, −0.2) * |
| PNC1–2.5 | −10.0 (−41.6, 21.5) | −2.7 (−26.6, 21.2) | 0.1 (−0.3, 0.4) |
| PNC2.5–10 | −32.4 (−71.8, 7.0) | −6.4 (−33.9, 21.2) | 0.1 (−0.2, 0.5) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, L.; Tao, Y.; Su, X.; Chen, X.; Li, L.; Fu, Q.; Xie, J.; Chen, R. Short-Term Associations between Size-Fractioned Particles and Cardiopulmonary Function in COPD Patients: A Panel Study in Shanghai, China, during 2014–2021. Int. J. Environ. Res. Public Health 2022, 19, 12473. https://doi.org/10.3390/ijerph191912473
Zhou L, Tao Y, Su X, Chen X, Li L, Fu Q, Xie J, Chen R. Short-Term Associations between Size-Fractioned Particles and Cardiopulmonary Function in COPD Patients: A Panel Study in Shanghai, China, during 2014–2021. International Journal of Environmental Research and Public Health. 2022; 19(19):12473. https://doi.org/10.3390/ijerph191912473
Chicago/Turabian StyleZhou, Lu, Yingmin Tao, Xiaozhen Su, Xiyin Chen, Liang Li, Qingyan Fu, Juan Xie, and Renjie Chen. 2022. "Short-Term Associations between Size-Fractioned Particles and Cardiopulmonary Function in COPD Patients: A Panel Study in Shanghai, China, during 2014–2021" International Journal of Environmental Research and Public Health 19, no. 19: 12473. https://doi.org/10.3390/ijerph191912473
APA StyleZhou, L., Tao, Y., Su, X., Chen, X., Li, L., Fu, Q., Xie, J., & Chen, R. (2022). Short-Term Associations between Size-Fractioned Particles and Cardiopulmonary Function in COPD Patients: A Panel Study in Shanghai, China, during 2014–2021. International Journal of Environmental Research and Public Health, 19(19), 12473. https://doi.org/10.3390/ijerph191912473







