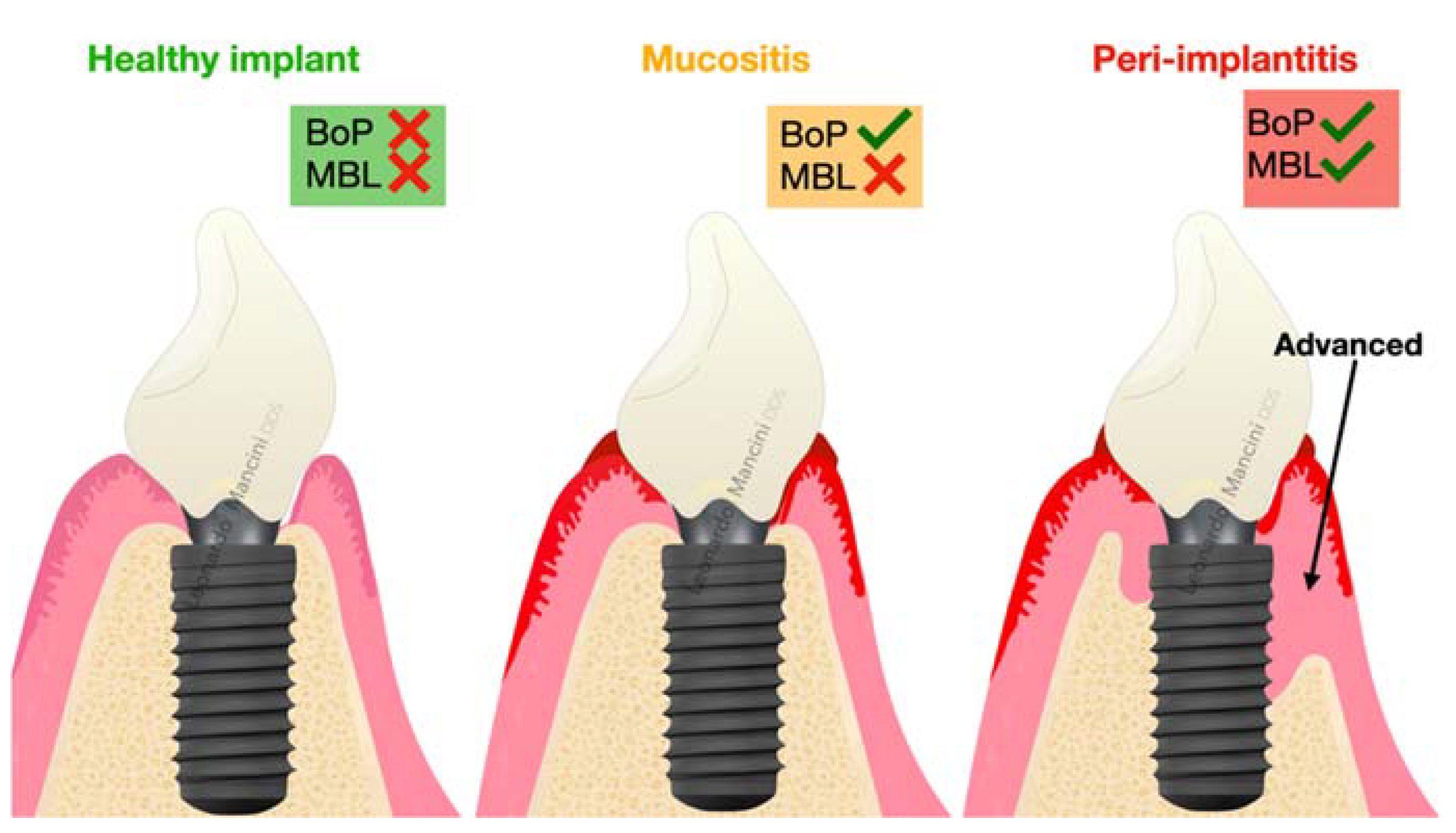
Peri-Implant Health and Diagnostic Considerations
Funding
Conflicts of Interest
References
- Roccuzzo, A.; Imber, J.C.; Marruganti, C.; Salvi, G.E.; Ramieri, G.; Roccuzzo, M. Clinical outcomes of dental implants in patients with and without history of periodontitis: A 20-year prospective study. J. Clin. Periodontol. 2022; Online ahead of print. [Google Scholar] [CrossRef]
- French, D.; Grandin, H.M.; Ofec, R. Retrospective cohort study of 4591 dental implants: Analysis of risk indicators for bone loss and prevalence of peri-implant mucositis and peri-implantitis. J. Periodontol. 2019, 90, 691–700. [Google Scholar] [CrossRef]
- Karoussis, I.K.; Brägger, U.; Salvi, G.E.; Bürgin, W.; Lang, N.P. Effect of implant design on survival and success rates of titanium oral implants: A 10-year prospective cohort study of the ITI Dental Implant System. Clin. Oral. Implant. Res. 2004, 15, 8–17. [Google Scholar] [CrossRef]
- Salvi, G.E.; Brägger, U. Mechanical and technical risks in implant therapy. Int. J. Oral Maxillofac. Implant. 2009, 24, 69–85. [Google Scholar]
- Romanos, G.E.; Delgado-Ruiz, R.; Sculean, A. Concepts for prevention of complications in implant therapy. Periodontol. 2000 2019, 81, 7–17. [Google Scholar] [CrossRef]
- Berglundh, T.; Armitage, G.; Araujo, M.G.; Avila-Ortiz, G.; Blanco, J.; Camargo, P.M.; Chen, S.; Cochran, D.; Derks, J.; Figuero, E.; et al. Peri-implant diseases and conditions: Consensus report of workgroup 4 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J. Clin. Periodontol. 2018, 45 (Suppl. S20), S286–S291. [Google Scholar] [CrossRef] [PubMed]
- Romandini, M.; Berglundh, J.; Derks, J.; Sanz, M.; Berglundh, T. Diagnosis of peri-implantitis in the absence of baseline data: A diagnostic accuracy study. Clin. Oral. Implant. Res. 2021, 32, 297–313. [Google Scholar] [CrossRef] [PubMed]
- Renvert, S.; Persson, G.R.; Pirih, F.Q.; Camargo, P.M. Peri-implant health, peri-implant mucositis, and peri-implantitis: Case definitions and diagnostic considerations. J. Periodontol. 2018, 89 (Suppl. S1), S304–S312. [Google Scholar] [CrossRef] [PubMed]
- Derks, J.; Ichioka, Y.; Dionigi, C.; Trullenque-Eriksson, A.; Berglundh, J.; Tomasi, C.; Graziani, F. 2Prevention and management of peri-implant mucositis and peri-implantitis: A systematic review of outcome measures used in clinical studies in the last 10 years. J. Clin. Periodontol. 2022; Online ahead of print. [Google Scholar] [CrossRef]
- Graziani, F.; Figuero, E.; Herrera, D. Systematic review of quality of reporting, outcome measurements and methods to study efficacy of preventive and therapeutic approaches to peri-implant diseases. J. Clin. Periodontol. 2012, 39 (Suppl. S12), 224–244. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mancini, L. Peri-Implant Health and Diagnostic Considerations. Int. J. Environ. Res. Public Health 2022, 19, 12008. https://doi.org/10.3390/ijerph191912008
Mancini L. Peri-Implant Health and Diagnostic Considerations. International Journal of Environmental Research and Public Health. 2022; 19(19):12008. https://doi.org/10.3390/ijerph191912008
Chicago/Turabian StyleMancini, Leonardo. 2022. "Peri-Implant Health and Diagnostic Considerations" International Journal of Environmental Research and Public Health 19, no. 19: 12008. https://doi.org/10.3390/ijerph191912008
APA StyleMancini, L. (2022). Peri-Implant Health and Diagnostic Considerations. International Journal of Environmental Research and Public Health, 19(19), 12008. https://doi.org/10.3390/ijerph191912008




