Indoor Mobility, Frailty, and Disability in Community-Dwelling Older Adults: A Mediation Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Measures
2.3. Statistical Analysis
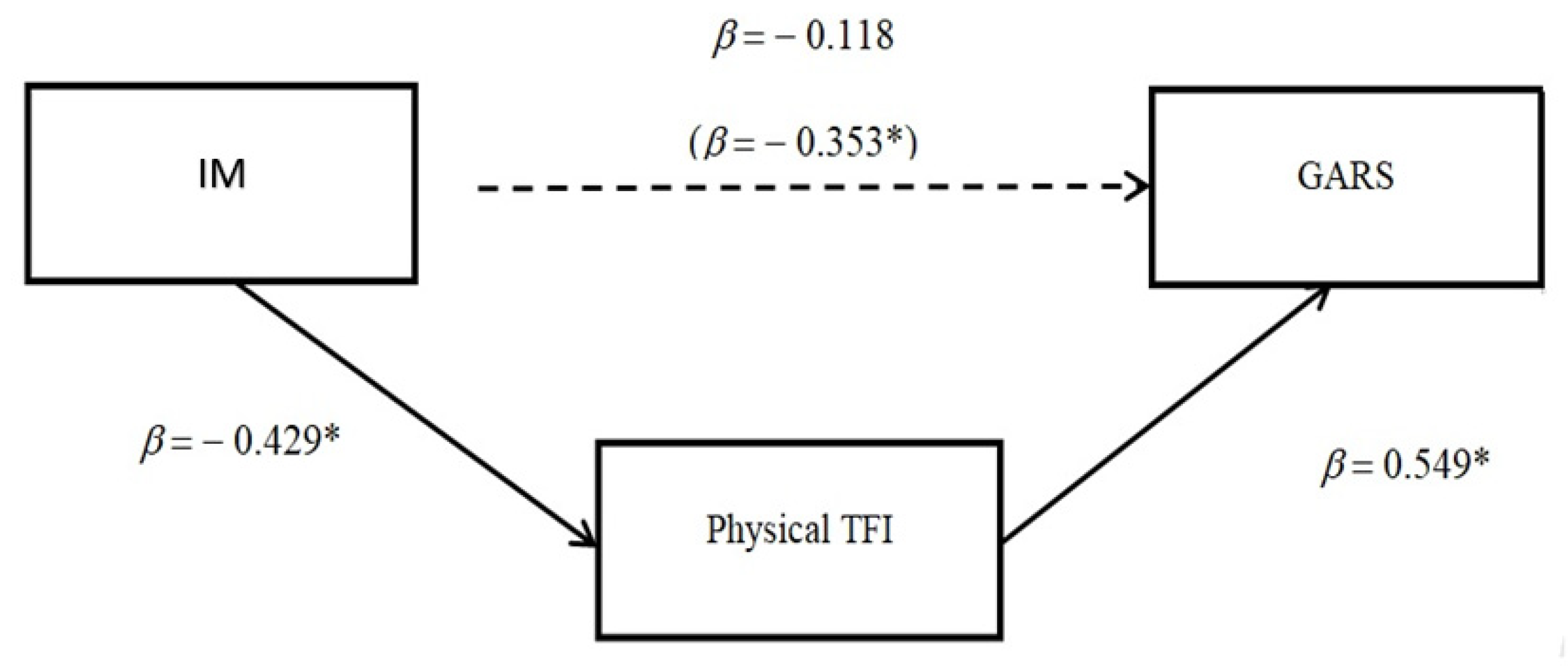
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lutz, W.; Sanderson, W.; Scherbov, S. The coming acceleration of global population ageing. Nature 2008, 451, 716–719. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Prince, M.; Thiyagarajan, J.A.; De Carvalho, I.A.; Bernabei, R.; Chan, P.; Gutierrez-Robledo, L.M.; Michel, J.P.; Morley, J.E.; Ong, P.; et al. Frailty: An Emerging Public Health Priority. J. Am. Med. Dir. Assoc. 2016, 17, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Clegg, A.; Young, J.; Iliffe, S.; Rikkert, M.O.; Rockwood, K. Frailty in elderly people. Lancet 2013, 381, 752–762. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef] [PubMed]
- Canevelli, M.; Cesari, M.; van Kan, G.A. Frailty and cognitive decline: How do they relate? Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 43–50. [Google Scholar] [CrossRef] [PubMed]
- Robertson, D.A.; Savva, G.M.; Kenny, R.A. Frailty and cognitive impairment—A review of the evidence and causal mechanisms. Ageing Res. Rev. 2013, 12, 840–851. [Google Scholar] [CrossRef] [PubMed]
- Langhammer, B.; Bergland, A.; Rydwik, E. The Importance of Physical Activity Exercise among Older People. BioMed Res. Int. 2018, 2018, 7856823. [Google Scholar] [CrossRef] [PubMed]
- Gregg, E.W.; Cauley, J.A.; Stone, K.; Thompson, T.J.; Bauer, D.C.; Cummings, S.R.; Ensrud, K.E.; Study of Osteoporotic Fractures Research Group. Relationship of changes in physical activity and mortality among older women. JAMA 2003, 289, 2379–2386. [Google Scholar] [CrossRef]
- Lee, I.M.; Shiroma, E.J.; Lobelo, F.; Puska, P.; Blair, S.N.; Katzmarzyk, P.T.; Lancet Physical Activity Series Working Group. Effect of physical inactivity on major non-communicable diseases worldwide: An analysis of burden of disease and life expectancy. Lancet 2012, 380, 219–229. [Google Scholar] [CrossRef]
- Rejeski, W.J.; Mihalko, S.L. Physical activity and quality of life in older adults. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2001, 56 (Suppl. 2), 23–35. [Google Scholar] [CrossRef] [Green Version]
- Bull, F.C.; Al-Ansari, S.S.; Biddle, S.; Borodulin, K.; Buman, M.P.; Cardon, G.; Carty, C.; Chaput, J.-P.; Chastin, S.; Chou, R.; et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. Br. J. Sports Med. 2020, 54, 1451–1462. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.E.; Rejeski, W.J.; Blair, S.N.; Duncan, P.W.; Judge, J.O.; King, A.C.; Macera, C.A.; Castaneda-Sceppa, C. Physical activity and public health in older adults: Recommendation from the American College of Sports Medicine and the American Heart Association. Circulation 2007, 116, 1094–1105. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Mutrie, N.; Bauman, A.; Pratt, M.; Hallal, P.R.C.; Powell, K.E. Physical activity guidelines 2020: Comprehensive and inclusive recommendations to activate populations. Lancet 2020, 396, 1780–1782. [Google Scholar] [CrossRef]
- ISTAT I Tempi Della Quotidianità, Anno 2014. Available online: https://www.istat.it/it/files/2016/11/Report_Tempidivita_2014.pdf (accessed on 1 May 2021).
- Spiteri, K.; Broom, D.; Bekhet, A.H.; de Caro, J.X.; Laventure, B.; Grafton, K. Barriers and motivators of physical activity participation in middle-aged and older adults—A systematic review. J. Aging Phys. Act. 2019, 27, 929–944. [Google Scholar] [CrossRef]
- Klimek, M.; Peter, R.S.; Denkinger, M.; Dallmeier, D.; Rapp, K.; Rothenbacher, D.; Klenk, J. The relationship of weather with daily physical activity and the time spent out of home in older adults from Germany–the ActiFE study. Eur. Rev. Aging Phys. Act. 2022, 19, 6. [Google Scholar] [CrossRef]
- Hashidate, H.; Shimada, H.; Shiomi, T.; Shibata, M.; Sawada, K.; Sasamoto, N. Measuring indoor life-space mobility at home in older adults with difficulty to perform outdoor activities. J. Geriatr. Phys. Ther. 2013, 36, 109–114. [Google Scholar] [CrossRef]
- Feskanich, D.; Willett, W.; Colditz, G. Walking and leisure-time activity and risk of hip fracture in postmenopausal women. JAMA 2002, 288, 2300–2306. [Google Scholar] [CrossRef]
- Gallucci, A.; Trimarchi, P.D.; Abbate, C.; Tuena, C.; Pedroli, E.; Lattanzio, F.; Stramba-Badiale, M.; Cesari, M.; Giunco, F. ICT technologies as new promising tools for the managing of frailty: A systematic review. Aging Clin. Exp. Res. 2020, 33, 1453–1464. [Google Scholar] [CrossRef]
- Dwyer, T.; Pezic, A.; Sun, C.; Cochrane, J.; Venn, A.; Srikanth, V.; Jones, G.; Shook, R.; Sui, X.; Ortaglia, A. Objectively measured daily steps and subsequent long term all-cause mortality: The tasped prospective cohort study. PLoS ONE 2015, 10, e0141274. [Google Scholar]
- Hansen, B.H.; Dalene, K.E.; Ekelund, U.; Wang Fagerland, M.; Kolle, E.; Steene-Johannessen, J.; Tarp, J.; Alfred Anderssen, S. Step by step: Association of device-measured daily steps with all-cause mortality—A prospective cohort Study. Scand. J. Med. Sci. Sports 2020, 30, 1705–1711. [Google Scholar] [CrossRef]
- Harris, T.; Limb, E.S.; Hosking, F.; Carey, I.; DeWilde, S.; Furness, C.; Wahlich, C.; Ahmad, S.; Kerry, S.; Whincup, P.; et al. Effect of pedometer-based walking interventions on long-term health outcomes: Prospective 4-year follow-up of two randomised controlled trials using routine primary care data. PLoS Med. 2019, 16, e1002836. [Google Scholar] [CrossRef] [PubMed]
- Calamia, M.; De Vito, A.; Bernstein, J.P.; Weitzner, D.S.; Carmichael, O.T.; Keller, J.N. Pedometer-assessed steps per day as a predictor of cognitive performance in older adults. Neuropsychology 2018, 32, 941–949. [Google Scholar] [CrossRef] [PubMed]
- De Silva, N.A.; Gregory, M.A.; Venkateshan, S.S.; Verschoor, C.P.; Kuspinar, A. Examining the Association between Life-Space Mobility and Cognitive Function in Older Adults: A Systematic Review. J. Aging Res. 2019, 2019, 3923574. [Google Scholar] [CrossRef] [PubMed]
- Aranyavalai, T.; Jalayondeja, C.; Jalayondeja, W.; Pichaiyongwongdee, S.; Kaewkungwal, J.; Laskin, J. Association between walking 5000 step/day and fall incidence over six months in urban community-dwelling older people. BMC Geriatr. 2020, 20, 194. [Google Scholar] [CrossRef]
- Chen, S.; Chen, T.; Kishimoto, H.; Yatsugi, H.; Kumagai, S. Associations of objectively measured patterns of sedentary behavior and physical activity with frailty status screened by the frail scale in Japanese community-dwelling older adults. J. Sci. Med. Sport 2020, 19, 166–174. [Google Scholar]
- Mulasso, A.; Brustio, P.R.; Rainoldi, A.; Zia, G.; Feletti, L.; N’dja, A.; Del Signore, S.; Poggiogalle, E.; Luisi, F.; Donini, L.M. A comparison between an ICT tool and a traditional physical measure for frailty evaluation in older adults. BMC Geriatr. 2019, 19, 88. [Google Scholar] [CrossRef]
- Watanabe, D.; Yoshida, T.; Watanabe, Y.; Yamada, Y.; Kimura, M.; Kyoto-Kameoka Study Group. Objectively Measured Daily Step Counts and Prevalence of Frailty in 3,616 Older Adults. J. Am. Geriatr. Soc. 2020, 68, 2310–2318. [Google Scholar] [CrossRef]
- Yuki, A.; Otsuka, R.; Tange, C.; Nishita, Y.; Tomida, M.; Ando, F.; Shimokata, H.; Arai, H. Daily physical activity predicts frailty development among community-dwelling older Japanese adults. J. Am. Med. Dir. Assoc. 2019, 20, 1032–1036. [Google Scholar] [CrossRef]
- Mankowski, R.T.; Anton, S.D.; Axtell, R.; Chen, S.H.; Fielding, R.A.; Glynn, N.W.; Hsu, F.C.; King, A.C.; Layne, A.S.; Leeuwenburgh, C.; et al. Device-measured physical activity as a predictor of disability in mobility-limited older adults. J. Am. Geriatr. Soc. 2017, 65, 2251–2256. [Google Scholar] [CrossRef] [PubMed]
- Gobbens, R.J.; van Assen, M.A.L.M.; Luijkx, K.G.; Schols, J.M.G.A. The Predictive Validity of the Tilburg Frailty Indicator: Disability, Health Care Utilization, and Quality of Life in a Population at Risk. Gerontologist 2012, 52, 619–631. [Google Scholar] [CrossRef] [PubMed]
- Mulasso, A.; Roppolo, M.; Gobbens, R.J.; Rabaglietti, E. The Italian Version of the Tilburg Frailty Indicator: Analysis of Psychometric Properties. Res. Aging 2016, 38, 842–863. [Google Scholar] [CrossRef] [PubMed]
- Kempen, G.I.; Miedema, I.; Ormel, J.; Molenaar, W. The assessment of disability with the Groningen Activity Restriction Scale. Conceptual framework and psychometric properties. Soc. Sci. Med. 1996, 43, 1601–1610. [Google Scholar] [CrossRef]
- Gobbens, R.J.; van Assen, M.A.L.M.; Luijkx, K.G.; Wijnen-Sponselee, M.T.; Schols, J.M.G.A. The Tilburg Frailty Indicator: Psychometric properties. J. Am. Med. Dir. Assoc. 2010, 11, 344–355. [Google Scholar] [CrossRef]
- Magistro, D.; Brustio, P.R.; Ivaldi, M.; Esliger, D.W.; Zecca, M.; Rainoldi, A.; Boccia, G. Validation of the ADAMO Care Watch for step counting in older adults. PLoS ONE 2018, 13, e0190753. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Routledge: London, UK, 2013. [Google Scholar]
- Baron, R.M.; Kenny, D.A. The moderator-mediator variable distinction in social psychological research: Conceptual, strategic, and statistical considerations. J. Pers. Soc. Psychol. 1986, 51, 1173–1182. [Google Scholar] [CrossRef]
- Brustio, P.R.; Magistro, D.; Zecca, M.; Liubicich, M.E.; Rabaglietti, E. Fear of falling and activities of daily living function: Mediation effect of dual-task ability. Aging Ment. Health 2018, 22, 856–861. [Google Scholar] [CrossRef]
- Huisingh-Scheetz, M.; Wroblewski, K.; Kocherginsky, M.; Huang, E.; Dale, W.; Waite, L.; Schumm, L.P. The relationship between physical activity and frailty among US older adults based on hourly accelerometry data. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2018, 73, 622–629. [Google Scholar] [CrossRef]
- Blodgett, J.; Theou, O.; Kirkland, S.; Andreou, P.; Rockwood, K. The association between sedentary behaviour, moderate–vigorous physical activity and frailty in NHANES cohorts. Maturitas 2015, 80, 187–191. [Google Scholar] [CrossRef]
- Del Pozo-Cruz, B.; Mañas, A.; Martín-García, M.; Marín-Puyalto, J.; García-García, F.J.; Rodriguez-Mañas, L.; Guadalupe-Grau, A.; Ara, I. Frailty is associated with objectively assessed sedentary behaviour patterns in older adults: Evidence from the Toledo Study for Healthy Aging (TSHA). PLoS ONE 2017, 12, e0183911. [Google Scholar]
- Kehler, D.S.; Hay, J.L.; Stammers, A.N.; Hamm, N.C.; Kimber, D.E.; Schultz, A.S.H.; Szwajcer, A.; Arora, R.C.; Tangri, N.; Duhamel, T.A. A systematic review of the association between sedentary behaviors with frailty. Exp. Gerontol. 2018, 114, 1–12. [Google Scholar] [CrossRef]
- Kehler, D.S.; Theou, O. The impact of physical activity and sedentary behaviors on frailty levels. Mech. Ageing Dev. 2019, 180, 29–41. [Google Scholar] [CrossRef]
- Ammar, A.; Brach, M.; Trabelsi, K.; Chtourou, H.; Boukhris, O.; Masmoudi, L.; Bouaziz, B.; Bentlage, E.; How, D.; Ahmed, M.; et al. Effects of COVID-19 Home Confinement on Eating Behaviour and Physical Activity: Results of the ECLB-COVID19 International Online Survey. Nutrients 2020, 12, 1583. [Google Scholar] [CrossRef] [PubMed]
- Paterson, D.H.; Warburton, D.E. Physical activity and functional limitations in older adults: A systematic review related to Canada’s Physical Activity Guidelines. Int. J. Behav. Nutr. Phys. Act. 2010, 7, 38. [Google Scholar] [CrossRef] [Green Version]
- Steen, G.; Sonn, U.; Börjesson Hanson, A.; Steen, B. Cognitive function and functional ability. A cross-sectional and longitudinal study at ages 85 and 95 in a non-demented population. Aging Clin. Exp. Res. 2001, 13, 68–77. [Google Scholar] [CrossRef]
- Baltes, M.M.; Carstensen, L.L. The process of successful ageing. Ageing Soc. 1996, 16, 397–422. [Google Scholar] [CrossRef]
- Miller, M.E.; Rejeski, W.J.; Reboussin, B.A.; Ten Have, T.R.; Ettinger, W.H. Physical activity, functional limitations, and disability in older adults. J. Am. Geriatr. Soc. 2000, 48, 1264–1272. [Google Scholar] [CrossRef]
- Soysal, P.; Stubbs, B.; Lucato, P.; Luchini, C.; Solmi, M.; Peluso, R.; Sergi, G.; Isik, A.T.; Manzato, E.; Maggi, S.; et al. Inflammation and frailty in the elderly: A systematic review and meta-analysis. Ageing Res. Rev. 2016, 31, 1–8. [Google Scholar] [CrossRef]
- Adriaensen, W.; Matheï, C.; van Pottelbergh, G.; Vaes, B.; Legrand, D.; Wallemacq, P.; Degryse, J.M. Significance of serum immune markers in identification of global functional impairment in the oldest old: Cross-sectional results from the BELFRAIL study. Age 2014, 36, 457–467. [Google Scholar] [CrossRef]
- Verghese, J.; Holtzer, R.; Oh-Park, M.; Derby, C.A.; Lipton, R.B.; Wang, C. Inflammatory markers and gait speed decline in older adults. J. Gerontol. Ser. A Biol. Sci. Med. 2011, 66, 1083–1089. [Google Scholar] [CrossRef]
- Perrig-Chiello, P.; Perrig, W.J.; Uebelbacher, A.; Stähelin, H.B. Impact of physical and psychological resources on functional autonomy in old age. Psychol. Health Med. 2006, 11, 470–482. [Google Scholar] [CrossRef]
- Gill, T.M.; Han, L.; Gahbauer, E.A.; Leo-Summers, L.; Murphy, T.E. Risk Factors and Precipitants of Severe Disability Among Community-Living Older Persons. JAMA 2020, 3, e206021. [Google Scholar] [CrossRef]
| Variables | n (%) | Mean (SD) |
|---|---|---|
| Age, years | - | 70 (6) |
| Gender, n (%) of female | 18 (56.2) | - |
| BMI (kg/m2) | - | 30 (7) |
| Marital status | - | |
| Married | 19 (59.4) | |
| Unmarried | 3 (9.4) | |
| Widowed | 10 (31.2) | |
| Level of education | - | |
| Primary school, 5 years | 5 (15.6) | |
| Secondary school, 8 years | 9 (28.1) | |
| High school diploma, 13 years | 14 (43.8) | |
| University degree, 18 years | 4 (12.5) | |
| Chronic disease, n (%) of Yes | 19 (59.4) | - |
| Pharmacotherapy, n (%) of Yes | 22 (68.8) | - |
| TFI, points | - | 4 (2) |
| Physical TFI, points | - | 2 (2) |
| Psychosocial TFI, points | - | 2 (1) |
| GARS, points | - | 22 (5) |
| IM, steps per week | - | 26,735 (12,752) |
| Robust People Mean (SD) | Frail People Mean (SD) | ANCOVA | |
|---|---|---|---|
| IM (steps) | 31,466 (10,426) | 21,374 (13,337) | F = 4.639, p = 0.040 partial η2 = 0.142 |
| GARS (points) | 21 (3) | 25 (7) | F = 4.342, p = 0.046 partial η2 = 0.134 |
| IM | GARS | TFI | Physical TFI | Psychosocial TFI | |
|---|---|---|---|---|---|
| IM | 1 | - | - | - | - |
| GARS | −0.353 * (−0.625,−0.005) | 1 | - | - | - |
| TFI | −0.358 * (−0.628,−0.011) | 0.557 ** (0.259, 0.758) | 1 | - | - |
| Physical TFI | −0.429 * (−0.467,−0.218) | 0.599 ** (0.317, 0.758) | 0.832 ** (−0.681, 0.995) | 1 | - |
| Psychosocial TFI | −0.151 (−0.475, 0.209) | 0.307 (−0.046, 0.593) | 0.810 ** (−0.642, 0.903) | 0.349 (−0.000, 0.622) | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brustio, P.R.; Mulasso, A.; D’Emanuele, S.; Zia, G.; Feletti, L.; Del Signore, S.; Rainoldi, A. Indoor Mobility, Frailty, and Disability in Community-Dwelling Older Adults: A Mediation Model. Int. J. Environ. Res. Public Health 2022, 19, 11386. https://doi.org/10.3390/ijerph191811386
Brustio PR, Mulasso A, D’Emanuele S, Zia G, Feletti L, Del Signore S, Rainoldi A. Indoor Mobility, Frailty, and Disability in Community-Dwelling Older Adults: A Mediation Model. International Journal of Environmental Research and Public Health. 2022; 19(18):11386. https://doi.org/10.3390/ijerph191811386
Chicago/Turabian StyleBrustio, Paolo Riccardo, Anna Mulasso, Samuel D’Emanuele, Gianluca Zia, Luca Feletti, Susanna Del Signore, and Alberto Rainoldi. 2022. "Indoor Mobility, Frailty, and Disability in Community-Dwelling Older Adults: A Mediation Model" International Journal of Environmental Research and Public Health 19, no. 18: 11386. https://doi.org/10.3390/ijerph191811386
APA StyleBrustio, P. R., Mulasso, A., D’Emanuele, S., Zia, G., Feletti, L., Del Signore, S., & Rainoldi, A. (2022). Indoor Mobility, Frailty, and Disability in Community-Dwelling Older Adults: A Mediation Model. International Journal of Environmental Research and Public Health, 19(18), 11386. https://doi.org/10.3390/ijerph191811386








