Vis LED Photo-Fenton Degradation of 124-Trichlorobenzene at a Neutral pH Using Ferrioxalate as Catalyst
Abstract
:1. Introduction
2. Material and Methods
2.1. Chemicals
2.2. Experimental Device and Procedure
2.3. Analytical Methods
2.4. Experimental Design
3. Results and Discussion
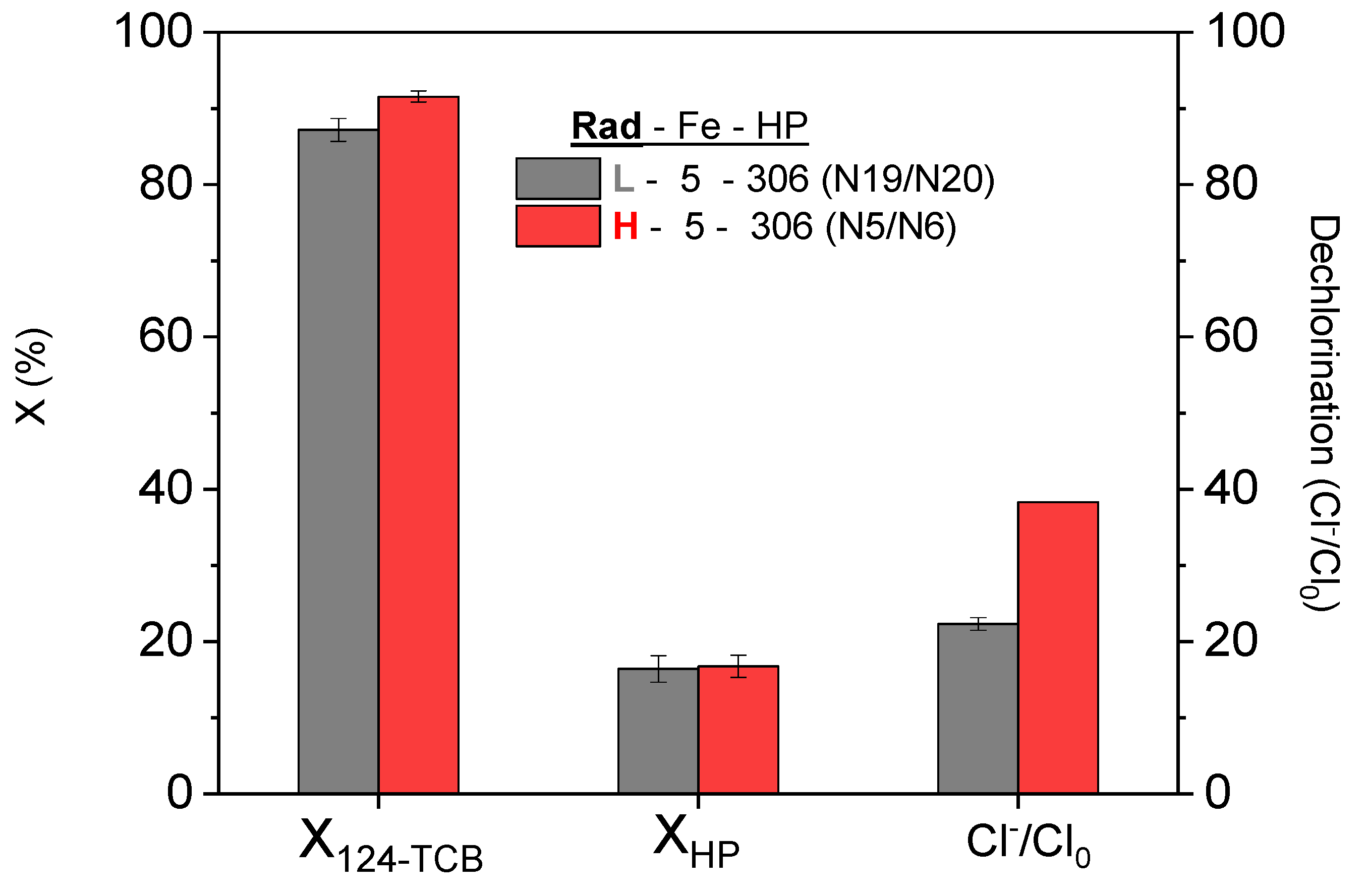
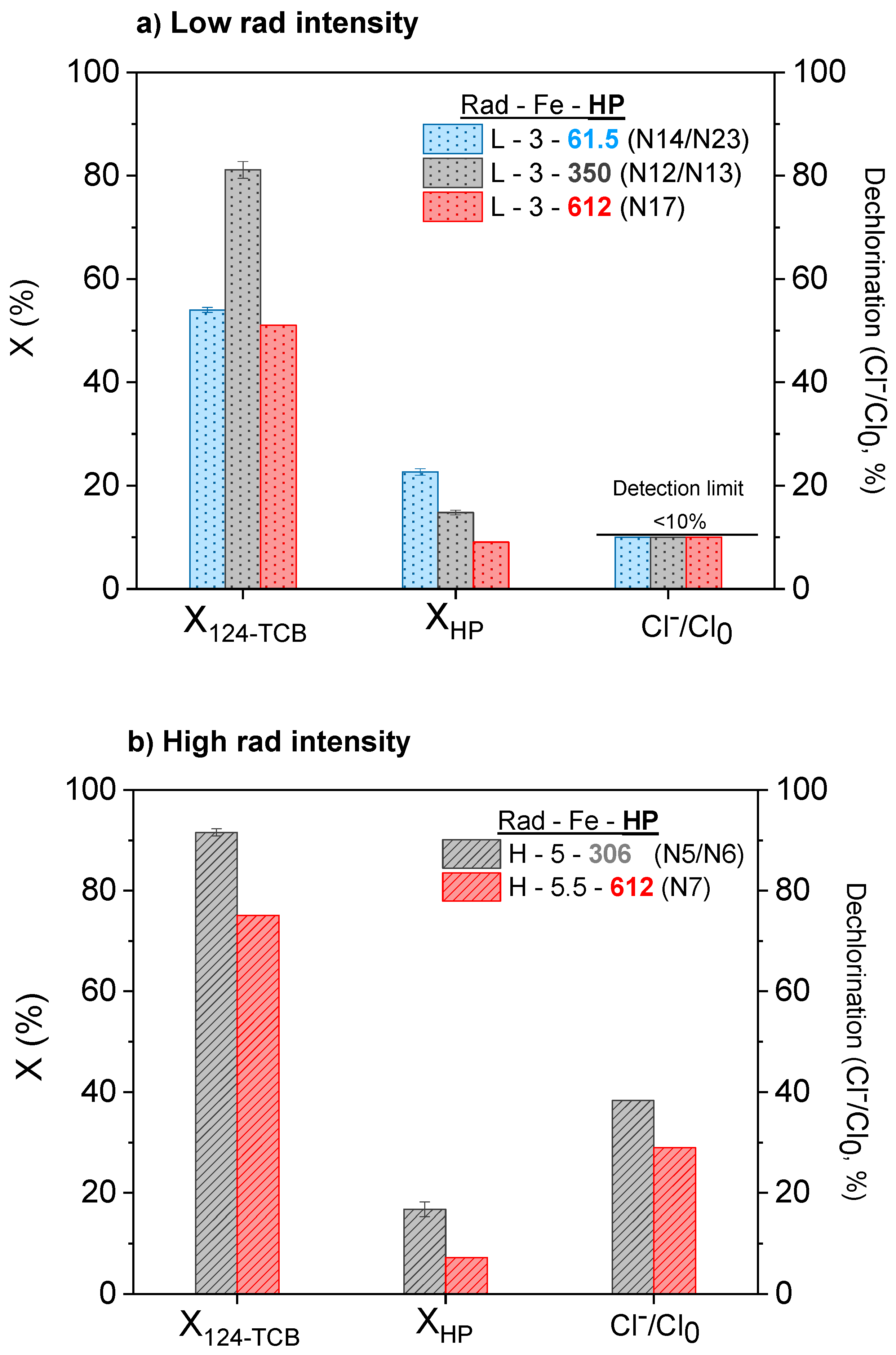
3.1. Influence of the Irradiation Level
3.2. Influence of the Catalyst Concentration
3.3. Influence of the Oxidant Concentration
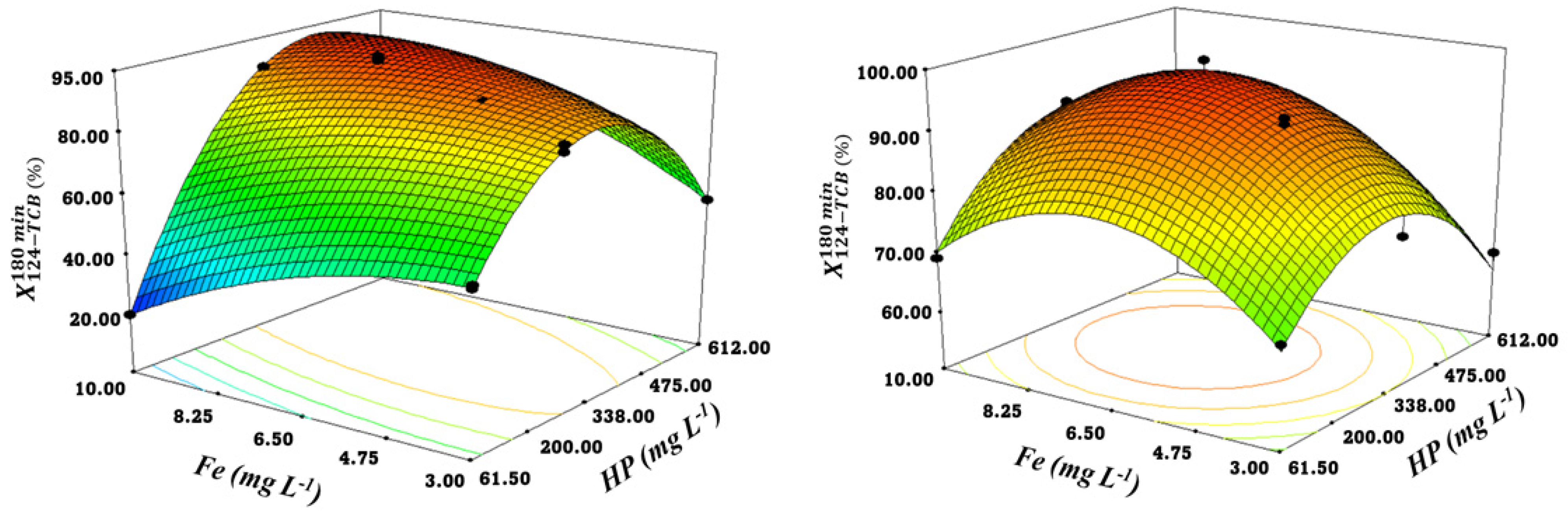
3.4. Response Surface Models Analysis
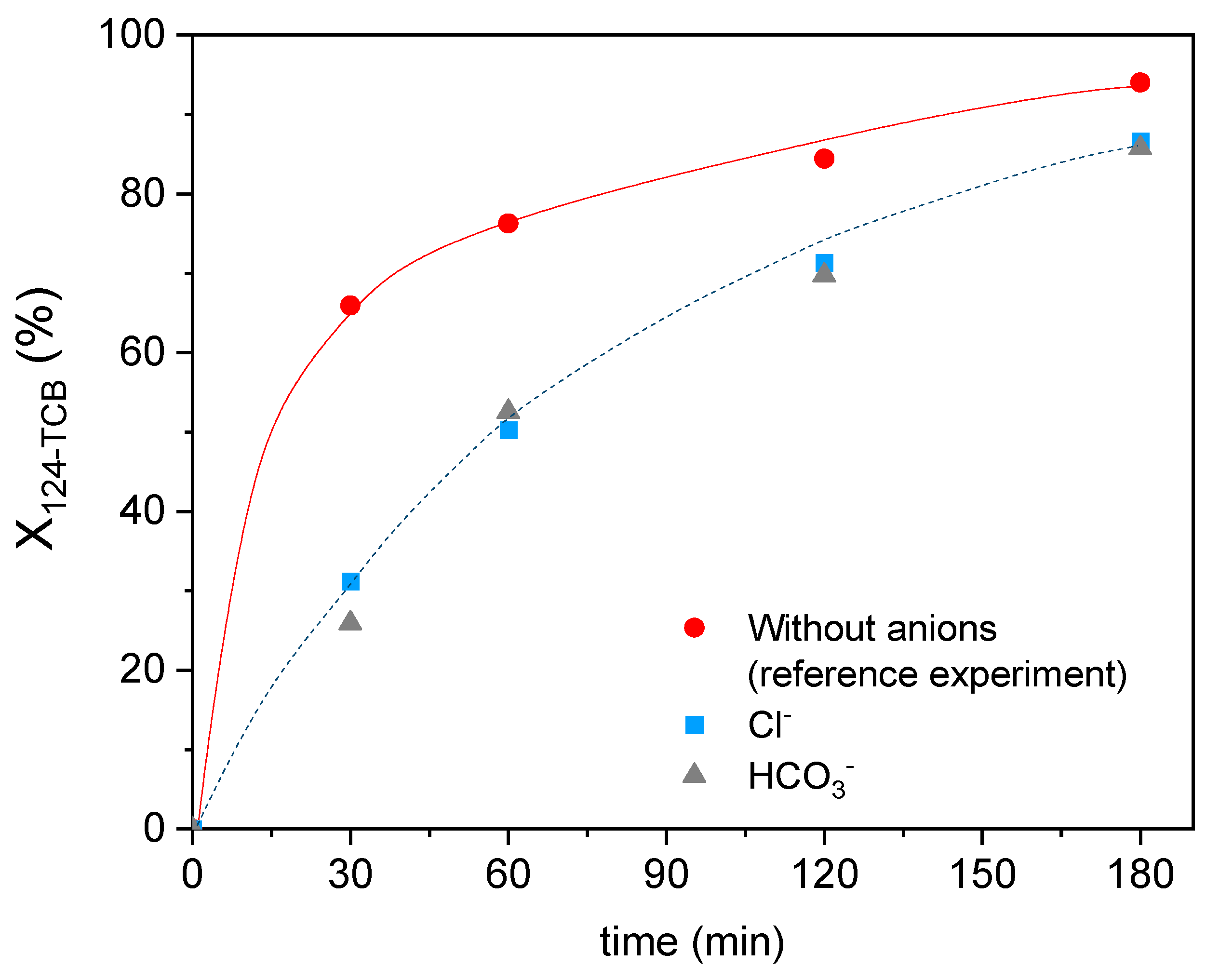
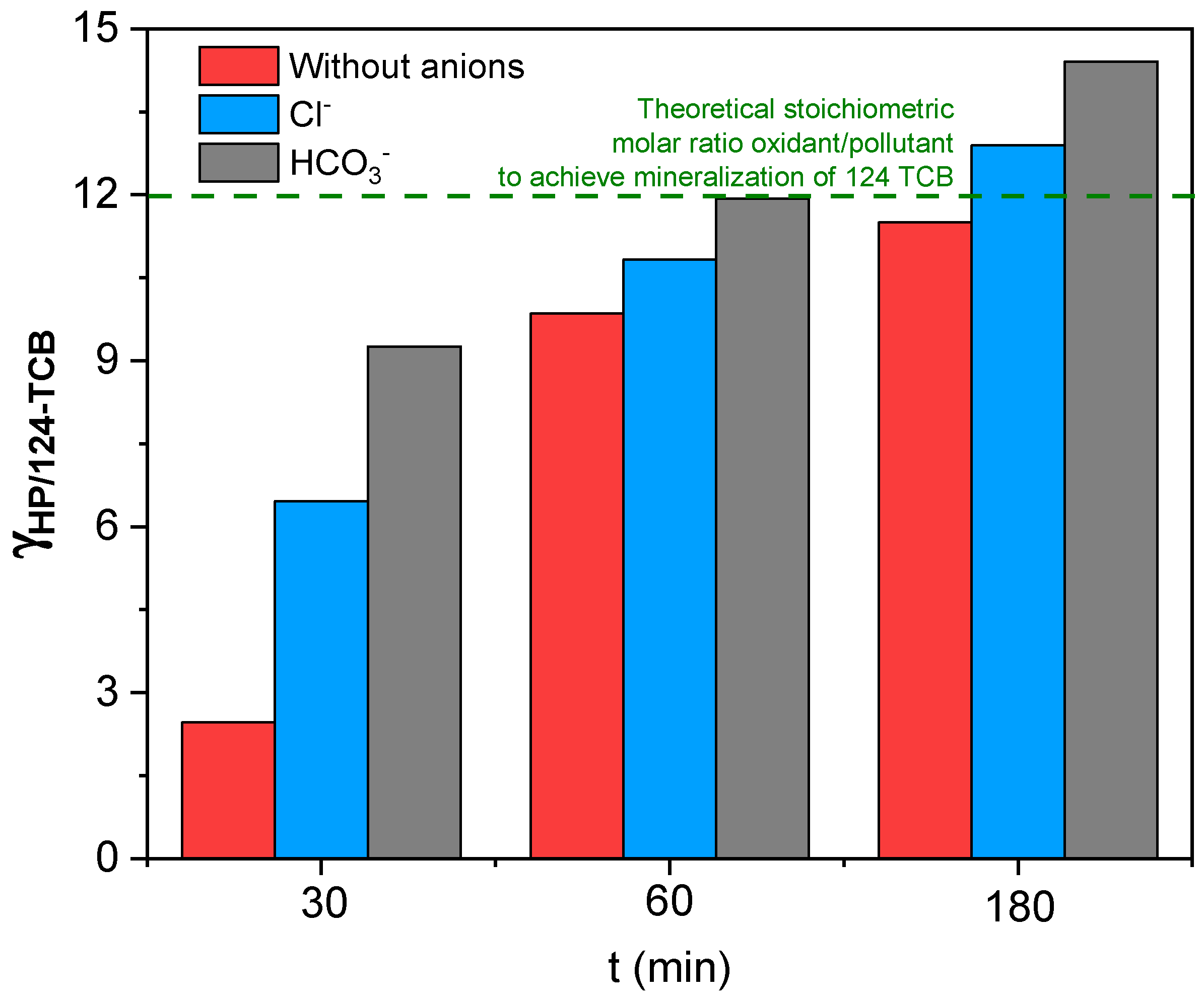
3.5. Effect of Common Anions in Groundwater
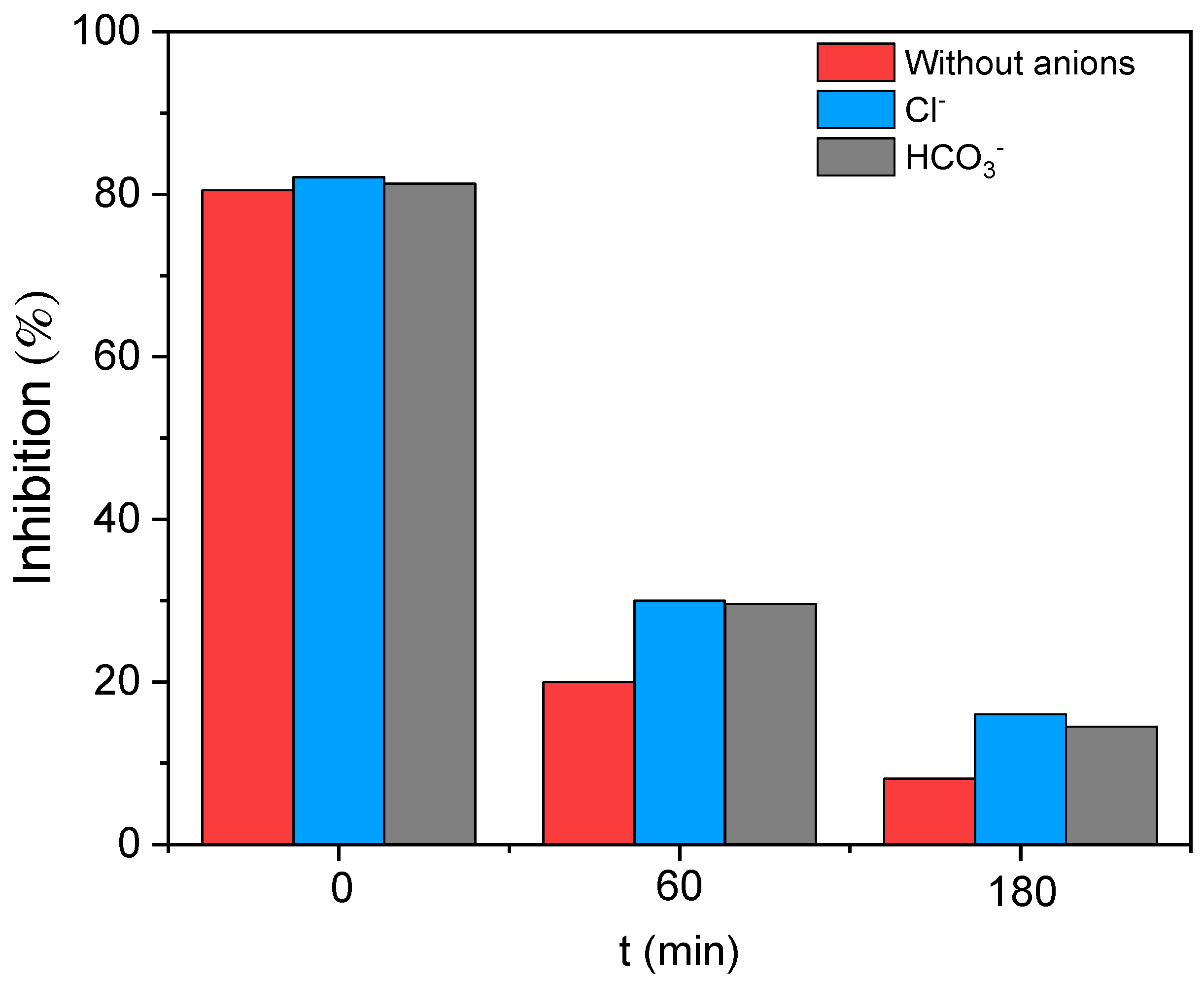
3.6. Toxicity Evaluation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Directive, E. Directive 2013/39/EU of the European Parliament and of the Council of 12 August 2013 amending Directives 2000/60/EC and 2008/105/EC as regards priority substances in the field of water policy. Off. J. Eur. Union L 2013, 226, 1–17. [Google Scholar]
- van Wijk, D.; Cohet, E.; Gard, A.; Caspers, N.; van Ginkel, C.; Thompson, R.; de Rooij, C.; Garny, V.; Lecloux, A. 1,2,4-Trichlorobenzene marine risk assessment with special emphasis on the Osparcom region North Sea. Chemosphere 2006, 62, 1294–1310. [Google Scholar] [CrossRef] [PubMed]
- Djohan, D.; Yu, Q.; Connell, D.W. Partition Isotherms of Chlorobenzenes in a Sediment–Water System. Water Air Soil. Pollut. 2005, 161, 157–173. [Google Scholar] [CrossRef]
- Lecloux, A.J. Scientific activities of Euro Chlor in monitoring and assessing neutrally and man-made organohalogens. Chemosphere 2003, 52, 521–529. [Google Scholar] [CrossRef]
- Dominguez, C.M.; Checa-Fernandez, A.; Romero, A.; Santos, A. Degradation of HCHs by thermally activated persulfate in soil system: Effect of temperature and oxidant concentration. J. Environ. Chem. Eng. 2021, 9, 105668. [Google Scholar] [CrossRef]
- Khan, S.; He, X.; Khan, J.A.; Khan, H.M.; Boccelli, D.L.; Dionysiou, D.D. Kinetics and mechanism of sulfate radical- and hydroxyl radical-induced degradation of highly chlorinated pesticide lindane in UV/peroxymonosulfate system. Chem. Eng. J. 2017, 318, 135–142. [Google Scholar] [CrossRef]
- Santos, A.; Fernández, J.; Guadaño, J.; Lorenzo, D.; Romero, A. Chlorinated organic compounds in liquid wastes (DNAPL) from lindane production dumped in landfills in Sabiñanigo (Spain). Environ. Pollut. 2018, 242, 1616–1624. [Google Scholar] [CrossRef]
- Li, J.-H.; Sun, X.-F.; Yao, Z.-T.; Zhao, X.Y. Remediation of 1,2,3-trichlorobenzene contaminated soil using a combined thermal desorption–molten salt oxidation reactor system. Chemosphere 2014, 97, 125–129. [Google Scholar] [CrossRef]
- Wang, M.-J.; Jones, K.C. Behaviour and fate of chlorobenzenes (CBs) introduced into soil-plant systems by sewage sludge application: A review. Chemosphere 1994, 28, 1325–1360. [Google Scholar] [CrossRef]
- Li, H.; Wang, Y.; Liu, F.; Tong, L.; Li, K.; Yang, H.; Zhang, L. Volatile organic compounds in stormwater from a community of Beijing, China. Environ. Pollut. 2018, 239, 554–561. [Google Scholar] [CrossRef]
- Bullot, L.; Vieira-Sellaï, L.; Chaplais, G.; Simon-Masseron, A.; Daou, T.J.; Patarin, J.; Fiani, E. Adsorption of 1,2-dichlorobenzene and 1,2,4-trichlorobenzene in nano- and microsized crystals of MIL-101(Cr): Static and dynamic gravimetric studies. Environ. Sci. Pollut. Res. 2017, 24, 26562–26573. [Google Scholar] [CrossRef] [PubMed]
- Pei, Z.; Li, L.; Sun, L.; Zhang, S.; Shan, X.-Q.; Yang, S.; Wen, B. Adsorption characteristics of 1,2,4-trichlorobenzene, 2,4,6-trichlorophenol, 2-naphthol and naphthalene on graphene and graphene oxide. Carbon 2013, 51, 156–163. [Google Scholar] [CrossRef]
- Zhao, Y.; He, P.; Zhang, Y.-H.; Ma, S. Removal of trichlorobenzene using ‘oxygen-enriched’ highly active absorbent. Environ. Technol. 2011, 32, 27–35. [Google Scholar] [CrossRef]
- Andreozzi, R.; Caprio, V.; Insola, A.; Marotta, R. Advanced oxidation processes (AOP) for water purification and recovery. Catal. Today 1999, 53, 51–59. [Google Scholar] [CrossRef]
- Đurkić, T.; Jazić, J.M.; Isakovski, M.K.; Maletić, S.; Tubić, A.; Dalmacija, B.; Agbaba, J. Ultraviolet/Hydrogen Peroxide Oxidative Degradation of 1,2,3-Trichlorobenzene: Influence of Water Matrix and Toxicity Assessment. Environ. Eng. Sci. 2019, 36, 947–957. [Google Scholar] [CrossRef]
- Vagi, M.C.; Petsas, A.S. Recent advances on the removal of priority organochlorine and organophosphorus biorecalcitrant pesticides defined by Directive 2013/39/EU from environmental matrices by using advanced oxidation processes: An overview (2007–2018). J. Environ. Chem. Eng. 2020, 8, 102940. [Google Scholar] [CrossRef]
- Barbash, A.M.; Hoag, G.E.; Nadim, F. Oxidation and Removal of 1,2,4-Trichlorobenzene using Sodium Persulfate in a Sorption-Desorption Experiment. Water Air Soil Pollut. 2006, 172, 67–80. [Google Scholar] [CrossRef]
- Masten, S.J.; Galbraith, M.J.; Davies, S.H.R. Oxidation of 1,3,5-trichlorobenzene using advanced oxidation processes. Ozone-Sci. Eng. 1997, 18, 535–547. [Google Scholar] [CrossRef]
- Ormad, P.; Puig, A.; Sarasa, J.; Roche, P.; Martin, A.; Ovelleiro, L. Ozonation of waste-water resulting from the production of organochlorine plaguicides derived from DDT and Trichlorobenzene. Ozone-Sci. Eng. 1994, 16, 487–503. [Google Scholar] [CrossRef]
- Uchida, H.; Katoh, S.; Watanabe, M. Photocatalytic Decomposition of Trichlorobenzene Using TiO2Supported on Nickel-Poly(tetrafluoroethylene) Composite Plate. Chem. Lett. 1995, 24, 261–262. [Google Scholar] [CrossRef]
- Uchida, H.; Katoh, S.; Watanabe, M. Photocatalytic degradation of trichlorobenzene using immobilized TiO2 films containing poly(tetrafluoroethylene) and platinum metal catalyst. Electrochim. Acta 1998, 43, 2111–2116. [Google Scholar] [CrossRef]
- Nakamura, A.; Hirano, K.; Iji, M. Decomposition of Trichlorobenzene with Different Radicals Generated by Alternating Current Electrolysis in Aqueous Solution. Chem. Lett. 2005, 34, 802–803. [Google Scholar] [CrossRef]
- Lin, S.; Su, G.; Zheng, M.; Ji, D.; Jia, M.; Liu, Y. Synthesis of flower-like Co3O4-CeO2 composite oxide and its application to catalytic degradation of 1,2,4-trichlorobenzene. Appl. Catal. B Environ. 2012, 123, 440–447. [Google Scholar] [CrossRef]
- Lin, S.; Su, G.; Zheng, M.; Jia, M.; Qi, C.; Li, W. The degradation of 1,2,4-trichlorobenzene using synthesized Co3O4 and the hypothesized mechanism. J. Hazard. Mater. 2011, 192, 1697–1704. [Google Scholar] [CrossRef] [PubMed]
- Đurkić, T.; Jazić, J.M.; Watson, M.; Bašić, B.; Prica, M.; Tubić, A.; Maletić, S.; Agbaba, J. Application of UV-activated persulfate and peroxymonosulfate processes for the degradation of 1,2,3-trichlorobenzene in different water matrices. Environ. Sci. Pollut. Res. 2020, 28, 59165–59179. [Google Scholar] [CrossRef]
- Oller, I.; Malato, S. Photo-Fenton applied to the removal of pharmaceutical and other pollutants of emerging concern. Curr. Opin. Green Sustain. Chem. 2021, 29, 100458. [Google Scholar] [CrossRef]
- Ziembowicz, S.; Kida, M. Limitations and future directions of application of the Fenton-like process in micropollutants degradation in water and wastewater treatment: A critical review. Chemosphere 2022, 296, 134041. [Google Scholar] [CrossRef]
- Xu, M.; Wu, C.; Zhou, Y. Advancements in the Fenton Process for Wastewater Treatment. In Advanced Oxidation Processes—Applications, Trends, and Prospects; IntechOpen: London, UK, 2020; p. 61. [Google Scholar] [CrossRef]
- Parsons, S. Advanced Oxidation Processes for Water and Wastewater Treatment; IWA Publishing: London, UK, 2005. [Google Scholar] [CrossRef]
- Ahile, U.J.; Wuana, R.A.; Itodo, A.U.; Sha’Ato, R.; Dantas, R.F. A review on the use of chelating agents as an alternative to promote photo-Fenton at neutral pH: Current trends, knowledge gap and future studies. Sci. Total Environ. 2019, 710, 134872. [Google Scholar] [CrossRef]
- Polo-López, M.I.; Pérez, J.A.S. Perspectives of the solar photo-Fenton process against the spreading of pathogens, antibiotic-resistant bacteria and genes in the environment. Curr. Opin. Green Sustain. Chem. 2020, 27, 100416. [Google Scholar] [CrossRef]
- Abdelhaleem, A.; Chu, W. Prediction of carbofuran degradation based on the hydroxyl radical’s generation using the FeIII impregnated N doped-TiO2/H2O2/visible LED photo-Fenton-like process. Chem. Eng. J. 2020, 382, 122930. [Google Scholar] [CrossRef]
- Carra, I.; Pérez, J.A.S.; Malato, S.; Autin, O.; Jefferson, B.; Jarvis, P. Application of high intensity UVC-LED for the removal of acetamiprid with the photo-Fenton process. Chem. Eng. J. 2014, 264, 690–696. [Google Scholar] [CrossRef]
- de Souza, Z.S.B.; Silva, M.P.; Fraga, T.J.; Motta Sobrinho, M.A. A comparative study of photo-Fenton process assisted by neutral sunlight, UV-A, or visible LED light irradiation for degradation of real textile wastewater: Factorial designs, kinetics, cost assessment, and phytotoxicity studies. Environ. Sci. Pollut. Res. 2021, 28, 23912–23928. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, Y.; Lu, J.; Yuan, Z.; Bond, P.L.; Guo, J. Efficient inactivation of antibiotic resistant bacteria and antibiotic resistance genes by photo-Fenton process under visible LED light and neutral pH. Water Res. 2020, 179, 115878. [Google Scholar] [CrossRef] [PubMed]
- Lagori, G.; Fornaini, C.; Rocca, J.P.; Merigo, E. Use of photo-Fenton’s reaction by 400-nm LED light for endodontic disinfection: A preliminary in vitro study on Enterococcus faecalis. J. Photochem. Photobiol. B Biol. 2017, 171, 85–89. [Google Scholar] [CrossRef]
- Martínez-Pachón, D.; Espinosa-Barrera, P.; Rincón-Ortíz, J.; Moncayo-Lasso, A. Advanced oxidation of antihypertensives losartan and valsartan by photo-electro-Fenton at near-neutral pH using natural organic acids and a dimensional stable anode-gas diffusion electrode (DSA-GDE) system under light emission diode (LED) lighting. Environ. Sci. Pollut. Res. 2018, 26, 4426–4437. [Google Scholar] [CrossRef]
- Pliego, G.; García-Muñoz, P.; Zazo, J.A.; Casas, J.A.; Rodriguez, J. Improving the Fenton process by visible LED irradiation. Environ. Sci. Pollut. Res. 2016, 23, 23449–23455. [Google Scholar] [CrossRef]
- Checa-Fernandez, A.; Santos, A.; Romero, A.; Dominguez, C. Application of Chelating Agents to Enhance Fenton Process in Soil Remediation: A Review. Catalysts 2021, 11, 722. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, M. A critical review of the application of chelating agents to enable Fenton and Fenton-like reactions at high pH values. J. Hazard. Mater. 2018, 362, 436–450. [Google Scholar] [CrossRef]
- Conte, L.O.; Schenone, A.V.; Alfano, O.M. Photo-Fenton degradation of the herbicide 2,4-D in aqueous medium at pH conditions close to neutrality. J. Environ. Manag. 2016, 170, 60–69. [Google Scholar] [CrossRef]
- Giménez, B.N.; Conte, L.O.; Alfano, O.M.; Schenone, A.V. Paracetamol removal by photo-Fenton processes at near-neutral pH using a solar simulator: Optimization by D-optimal experimental design and toxicity evaluation. J. Photochem. Photobiol. A Chem. 2020, 397, 112584. [Google Scholar] [CrossRef]
- Malato, S.; Fernandez-Ibañez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- Montgomery, D.C. Design and Analysis of Experiments; John Wiley & Sons: Hoboken, NJ, USA, 2017. [Google Scholar]
- Boutra, B.; Sebti, A.; Trari, M. Response surface methodology and artificial neural network for optimization and modeling the photodegradation of organic pollutants in water. Int. J. Environ. Sci. Technol. 2022. [Google Scholar] [CrossRef]
- Lojo-López, M.; Andrades, J.; Egea-Corbacho, A.; Coello, M.; Quiroga, J. Degradation of simazine by photolysis of hydrogen peroxide Fenton and photo-Fenton under darkness, sunlight and UV light. J. Water Process Eng. 2021, 42, 102115. [Google Scholar] [CrossRef]
- Schenone, A.; Conte, L.; Botta, M.A.; Alfano, O.M. Modeling and optimization of photo-Fenton degradation of 2,4-D using ferrioxalate complex and response surface methodology (RSM). J. Environ. Manag. 2015, 155, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Lide, D.R. CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Jewett, J.W.; Serway, R. Physics for scientists and engineers with modern physics. Vectors 2008, 1. [Google Scholar]
- Lorenzo, D.; Santos, A.; Sánchez-Yepes, A.; Conte, L.; Domínguez, C.M. Abatement of 1,2,4-Trichlorobencene by Wet Peroxide Oxidation Catalysed by Goethite and Enhanced by Visible LED Light at Neutral pH. Catalysts 2021, 11, 139. [Google Scholar] [CrossRef]
- Murov, S.L.; Carmichael, I.; Hug, G.L. Handbook of Photochemistry; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- Santos, A.; Fernandez, J.; Rodriguez, S.; Dominguez, C.; Lominchar, M.; Lorenzo, D.; Romero, A. Abatement of chlorinated compounds in groundwater contaminated by HCH wastes using ISCO with alkali activated persulfate. Sci. Total Environ. 2017, 615, 1070–1077. [Google Scholar] [CrossRef]
- ISO, D. 11348-3; Water Quality—Determination of the Inhibitory Effect of Water Samples on the Light Emission of Vibrio Fischeri (Luminescent Bacteria Test)—part 3: Method using Freeze-Dried Bacteria. International Organization for Standardization: Geneve, Switzerland, 2007.
- Conte, L.O.; Schenone, A.V.; Alfano, O.M. Ferrioxalate-assisted solar photo-Fenton degradation of a herbicide at pH conditions close to neutrality. Environ. Sci. Pollut. Res. 2017, 24, 6205–6212. [Google Scholar] [CrossRef]
- Grčić, I.; Vujevic, D.; Koprivanac, N. The use of D-optimal design to model the effects of process parameters on mineralization and discoloration kinetics of Fenton-type oxidation. Chem. Eng. J. 2010, 157, 408–419. [Google Scholar] [CrossRef]
- Dean, A.; Voss, D.; Draguljić, D. Response Surface Methodology. In Design and Analysis of Experiments; Dean, A., Voss, D., Draguljić, D., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 565–614. [Google Scholar]
- Myers, R.H.; Montgomery, D.C.; Vining, G.G.; Borror, C.M.; Kowalski, S.M. Response Surface Methodology: A Retrospective and Literature Survey. J. Qual. Technol. 2004, 36, 53–77. [Google Scholar] [CrossRef]
- Kusic, H.; Peternel, I.; Ukic, S.; Koprivanac, N.; Bolanca, T.; Papic, S.; Bozic, A.L. Modeling of iron activated persulfate oxidation treating reactive azo dye in water matrix. Chem. Eng. J. 2011, 172, 109–121. [Google Scholar] [CrossRef]
- De Laat, J.; Truong Le, G.; Legube, B. A comparative study of the effects of chloride, sulfate and nitrate ions on the rates of decomposition of H2O2 and organic compounds by Fe(II)/H2O2 and Fe(III)/H2O2. Chemosphere 2004, 55, 715–723. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Effect of inorganic anions on the performance of advanced oxidation processes for degradation of organic contaminants. Chem. Eng. J. 2021, 411, 128392. [Google Scholar] [CrossRef]
- Conte, L.O.; Schenone, A.V.; Giménez, B.N.; Alfano, O.M. Photo-Fenton degradation of a herbicide (2,4-D) in groundwater for conditions of neutral pH and presence of inorganic anions. J. Hazard. Mater. 2019, 372, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, K.L.; Palabrica, V.S. Photobacterium phosphoreum Toxicity Data Index. Water Qual. Res. J. 1991, 26, 361–431. [Google Scholar] [CrossRef]
- Blaschke, U.; Paschke, A.; Rensch, I.; Schüürmann, G. Acute and Chronic Toxicity toward the Bacteria Vibrio fischeri of Organic Narcotics and Epoxides: Structural Alerts for Epoxide Excess Toxicity. Chem. Res. Toxicol. 2010, 23, 1936–1946. [Google Scholar] [CrossRef]

| Run/Level | Experimental Conditions | Results at 180 min | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Run | Level | HP mg L−1 | Fe3+ mg L−1 | Rad (I) W cm−2 | qw × 107 Ecm−2 s−1 | (%) | (%) | (%) | mol HP mol 124-TCB−1 |
| R1 | N9 | 61.5 | 3 | 0.18 | 7.89 | 67.62 | 25 | 15 | 7.41 |
| R2 | N11 | 61.5 | 10 | 0.18 | 7.89 | 69.63 | 29.63 | 28 | 8.84 |
| R3 | N5 | 306 | 5 | 0.18 | 7.89 | 91.1 | 17.79 | 36.5 | 15.57 |
| R4 | N6 b | 306 | 5 | 0.18 | 7.89 | 92.05 | 15.72 | 38.32 | 10.39 |
| R5 | N10 | 350 | 3 | 0.18 | 7.89 | 75.45 | 18.11 | 31 | 11.04 |
| R6 | N3 | 350 | 7 | 0.18 | 7.89 | 98.5 | 21.08 | 39.54 | 12.58 |
| R7 | N1 | 350 | 10 | 0.18 | 7.89 | 87 | 13 | 35.57 | 10.27 |
| R8 | N2 a | 350 | 10 | 0.18 | 7.89 | 88.02 | 12.6 | 36.37 | 11.8 |
| R9 | N8 | 612 | 3 | 0.18 | 7.89 | 64.5 | 9 | 21.9 | 17.39 |
| R10 | N7 | 612 | 5.5 | 0.18 | 7.89 | 75.08 | 7.2 | 28.95 | 11.46 |
| R11 | N4 | 612 | 10 | 0.18 | 7.89 | 75.23 | 12 | 38.39 | 26.68 |
| R12 | N14 | 61.5 | 3 | 0.12 | 4.4 | 52.39 | 22.22 | <10.00 * | 6.69 |
| R13 | N23 | 61.5 | 3 | 0.12 | 4.4 | 53.45 | 23.09 | <10.00 * | 6.34 |
| R14 | N16 | 61.5 | 10 | 0.12 | 4.4 | 19.53 | 28 | <10.00 * | 18.97 |
| R15 | N12 | 250 | 3 | 0.12 | 4.4 | 82.25 | 14.5 | <10.00 * | 9.26 |
| R16 | N13 c | 250 | 3 | 0.12 | 4.4 | 80.63 | 15.11 | <10.00 * | 8.97 |
| R17 | N19 | 306 | 5 | 0.12 | 4.4 | 86.32 | 17.65 | 21.72 | 15.88 |
| R18 | N20 d | 306 | 5 | 0.12 | 4.4 | 87.54 | 15.19 | 22.9 | 12.69 |
| R19 | N18 | 350 | 7.5 | 0.12 | 4.4 | 92.12 | 21 | 26.4 | 37.02 |
| R20 | N21 e | 350 | 7.5 | 0.12 | 4.4 | 91.56 | 19.67 | 23.3 | 34.86 |
| R21 | N22 | 350 | 10 | 0.12 | 4.4 | 85.48 | 17.59 | 20 | 18.08 |
| R22 | N17 | 612 | 3 | 0.12 | 4.4 | 50.24 | 9.08 | <10.00 * | 22.61 |
| R23 | N15 | 612 | 10 | 0.12 | 4.4 | 82.56 | 11.22 | 16 | 22.38 |
| Model | F-Value | p-Value | |||||||
|---|---|---|---|---|---|---|---|---|---|
| <0.0001 | |||||||||
| <0.0005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Conte, L.O.; Dominguez, C.M.; Checa-Fernandez, A.; Santos, A. Vis LED Photo-Fenton Degradation of 124-Trichlorobenzene at a Neutral pH Using Ferrioxalate as Catalyst. Int. J. Environ. Res. Public Health 2022, 19, 9733. https://doi.org/10.3390/ijerph19159733
Conte LO, Dominguez CM, Checa-Fernandez A, Santos A. Vis LED Photo-Fenton Degradation of 124-Trichlorobenzene at a Neutral pH Using Ferrioxalate as Catalyst. International Journal of Environmental Research and Public Health. 2022; 19(15):9733. https://doi.org/10.3390/ijerph19159733
Chicago/Turabian StyleConte, Leandro O., Carmen M. Dominguez, Alicia Checa-Fernandez, and Aurora Santos. 2022. "Vis LED Photo-Fenton Degradation of 124-Trichlorobenzene at a Neutral pH Using Ferrioxalate as Catalyst" International Journal of Environmental Research and Public Health 19, no. 15: 9733. https://doi.org/10.3390/ijerph19159733
APA StyleConte, L. O., Dominguez, C. M., Checa-Fernandez, A., & Santos, A. (2022). Vis LED Photo-Fenton Degradation of 124-Trichlorobenzene at a Neutral pH Using Ferrioxalate as Catalyst. International Journal of Environmental Research and Public Health, 19(15), 9733. https://doi.org/10.3390/ijerph19159733








