Exercise and Reduced Nicotine Content Cigarettes in Adult Female Smokers: A Pilot Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility and Recruitment
2.2. Study Design
2.2.1. Nicotine Protocol
Control Group
RNC with and without Exercise
2.2.2. Metabolic Control Protocol Prior to Assessment
2.2.3. Outcome Variables Assessment
Body Fat, Fitness, and Metabolism
Withdrawal Symptoms
Plasma Cardiometabolic Health Measures
2.3. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Cardiometabolic Health
3.3. Smoking Cessation and Withdrawal Symptoms
4. Discussions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- US Department of Health and Human Services National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health. Let’s Make the next Generation Tobacco-Free: Your Guide to the 50th Anniversary Surgeon General’s Report on Smoking and Health; Government Printing Office: Washington, DC, USA, 2014.
- Bergman, B.C.; Perreault, L.; Hunerdosse, D.; Kerege, A.; Playdon, M.; Samek, A.M.; Eckel, R.H. Novel and Reversible Mechanisms of Smoking-Induced Insulin Resistance in Humans. Diabetes 2012, 61, 3156–3166. [Google Scholar] [CrossRef] [PubMed]
- Facchini, F.S.; Hollenbeck, C.B.; Jeppesen, J.; Chen, Y.D.; Reaven, G.M. Insulin resistance and cigarette smoking. Lancet Lond. Engl. 1992, 339, 1128–1130. [Google Scholar] [CrossRef]
- In The Family Smoking Prevention and Tobacco Control Act. In Proceedings of the 111th Congress, USA, 2 April 2009; p. 123.
- Muscat, J. Reduced Nicotine Content Cigarettes in Smokers of Lower Socioeconomic Status. Published 2015. Available online: https://clinicaltrials.gov/ct2/show/NCT01928719 (accessed on 30 May 2016).
- Donny, E.C.; Denlinger, R.L.; Tidey, J.W. Randomized Trial of Reduced-Nicotine Standards for Cigarettes. N. Engl. J. Med. 2015, 373, 1340–1349. [Google Scholar] [CrossRef]
- Jha, P.; Ramasundarahettige, C.; Landsman, V.; Rostron, B.; Thun, M.; Anderson, R.N.; McAfee, T.; Peto, R. 21st-Century Hazards of Smoking and Benefits of Cessation in the United States. N. Engl. J. Med. 2013, 368, 341–350. [Google Scholar] [CrossRef]
- Lu, Q.; Gottlieb, E.; Rounds, S. Effects of cigarette smoke on pulmonary endothelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 2018, 314, L743–L756. [Google Scholar] [CrossRef]
- McEvoy, J.W.; Blaha, M.J.; DeFilippis, A.P.; Lima, J.A.C.; Bluemke, D.A.; Hundley, W.G.; Min, J.K.; Shaw, L.J.; Lloyd-Jones, D.M.; Barr, R.G.; et al. Cigarette smoking and cardiovascular events: Role of inflammation and subclinical atherosclerosis from the MultiEthnic Study of Atherosclerosis. Arter. Thromb. Vasc. Biol. 2015, 35, 700–709. [Google Scholar] [CrossRef]
- Mahmud, A.; Feely, J. Effect of smoking on arterial stiffness and pulse pressure amplification. Hypertens Dallas Tex 1979 2003, 41, 183–187. [Google Scholar] [CrossRef]
- Williamson, D.F.; Madans, J.; Anda, R.F.; Kleinman, J.C.; Giovino, G.A.; Byers, T. Smoking cessation and severity of weight gain in a national cohort. N. Engl. J. Med. 1991, 324, 739–745. [Google Scholar] [CrossRef]
- Malin, S.K.; Haus, J.M.; Solomon, T.P.J.; Blaszczak, A.; Kashyap, S.R.; Kirwan, J.P. Insulin sensitivity and metabolic flexibility following exercise training among different obese insulin-resistant phenotypes. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E1292–E1298. [Google Scholar] [CrossRef]
- Malin, S.K.; Liu, Z.; Barrett, E.J.; Weltman, A. Exercise resistance across the prediabetes phenotypes: Impact on insulin sensitivity and substrate metabolism. Rev. Endocr. Metab. Disord. 2016, 17, 81–90. [Google Scholar] [CrossRef]
- Malin, S.K.; Rynders, C.A.; Weltman, J.Y.; Barrett, E.J.; Weltman, A. Exercise Intensity Modulates Glucose-Stimulated Insulin Secretion when Adjusted for Adipose, Liver and Skeletal Muscle Insulin Resistance. PLoS ONE 2016, 11, e0154063. [Google Scholar] [CrossRef] [PubMed]
- Stamford, B.A.; Matter, S.; Fell, R.D.; Papanek, P. Effects of smoking cessation on weight gain, metabolic rate, caloric consumption, and blood lipids. Am. J. Clin. Nutr. 1986, 43, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Gennuso, K.P.; Thraen-Borowski, K.M.; Schlam, T.R.; LaRowe, T.L.; Fiore, M.C.; Baker, T.B.; Colbert, L.H. Smokers’ physical activity and weight gain one year after a successful versus unsuccessful quit attempt. Prev. Med. 2014, 67, 189–192. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kawachi, I.; Colditz, G.A.; Stampfer, M.J.; Willett, W.C.; Manson, J.E.; Rosner, B.; Hunter, D.J.; Hennekens, C.H.; Speizer, F.E. Smoking cessation in relation to total mortality rates in women. A prospective cohort study. Ann. Intern. Med. 1993, 119, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- French, S.A.; Hennrikus, D.J.; Jeffery, R.W. Smoking status, dietary intake, and physical activity in a sample of working adults. Health Psychol. Off. J. Div. Health Psychol. Am. Psychol. Assoc. 1996, 15, 448–454. [Google Scholar] [CrossRef]
- Roberts, V.; Maddison, R.; Simpson, C.; Bullen, C.; Prapavessis, H. The acute effects of exercise on cigarette cravings, withdrawal symptoms, affect, and smoking behaviour: Systematic review update and meta-analysis. Psychopharmacology 2012, 222, 1–15. [Google Scholar] [CrossRef]
- Navaneethan, S.D.; Fealy, C.E.; Scelsi, A.C.; Arrigain, S.; Malin, S.K.; Kirwan, J.P. A Trial of Lifestyle Modification on Cardiopulmonary, Inflammatory, and Metabolic Effects among Obese with Chronic Kidney Disease. Am. J. Nephrol. 2015, 42, 274–281. [Google Scholar] [CrossRef]
- Malin, S.K.; Rynders, C.A.; Weltman, J.Y.; Jackson Roberts, L.; Barrett, E.J.; Weltman, A. Endothelial function following glucose ingestion in adults with prediabetes: Role of exercise intensity. Obes. Silver Spring Md. 2016, 15, 15–21. [Google Scholar] [CrossRef]
- Welsch, S.K.; Smith, S.S.; Wetter, D.W.; Jorenby, D.E.; Fiore, M.C.; Baker, T.B. Development and validation of the Wisconsin Smoking Withdrawal Scale. Exp. Clin. Psychopharmacol. 1999, 7, 354–361. [Google Scholar] [CrossRef]
- Heatherton, T.F.; Kozlowski, L.T.; Frecker, R.C.; Fagerström, K.O. The Fagerström Test for Nicotine Dependence: A revision of the Fagerström Tolerance Questionnaire. Br. J. Addict. 1991, 86, 1119–1127. [Google Scholar] [CrossRef]
- Jarvis, M.J.; Belcher, M.; Vesey, C.; Hutchison, D.C. Low cost carbon monoxide monitors in smoking assessment. Thorax 1986, 41, 886–887. [Google Scholar] [CrossRef] [PubMed]
- Hatsukami, D.K.; Kotlyar, M.; Hertsgaard, L.A.; Zhang, Y.; Carmella, S.G.; Jensen, J.A.; Allen, S.S.; Shields, P.G.; Murphy, S.E.; Stepanov, I.; et al. Reduced nicotine content cigarettes: Effects on toxicant exposure, dependence and cessation. Addiction 2010, 105, 343–355. [Google Scholar] [CrossRef] [PubMed]
- Department of Health and Human Services. The Health Consequences of Smoking—50 Years of Progress: A Report of the Surgeon General. 2014. Available online: https://www.surgeongeneral.gov/library/reports/50-years-of-progress/index.html (accessed on 12 May 2017).
- Shiels, M.S.; Katki, H.A.; Freedman, N.D.; Purdue, M.P.; Wentzensen, N.; Trabert, B.; Kitahara, C.M.; Furr, M.; Li, Y.; Kemp, T.J.; et al. Cigarette Smoking and Variations in Systemic Immune and Inflammation Markers. JNCI J. Natl. Cancer Inst. 2014, 106, dju294. [Google Scholar] [CrossRef] [PubMed]
- Gilbertson, N.M.; Eichner, N.Z.M.; Francois, M.; Gaitán, J.M.; Heiston, E.M.; Weltman, A.; Malin, S.K. Glucose Tolerance is Linked to Postprandial Fuel Use Independent of Exercise Dose. Med. Sci. Sports Exerc. 2018, 50, 2058–2066. [Google Scholar] [CrossRef]
- Keshel, T.E.; Coker, R.H. Exercise Training and Insulin Resistance: A Current Review. J. Obes. Weight Loss Ther. 2015, 5, S5-003. [Google Scholar] [CrossRef]
- Slentz, C.A.; Tanner, C.J.; Bateman, L.A.; Durheim, M.T.; Huffman, K.M.; Houmard, J.A.; Kraus, W.E. Effects of exercise training intensity on pancreatic beta-cell function. Diabetes Care 2009, 32, 1807–1811. [Google Scholar] [CrossRef]
- Tobacco Control Research Branch of the National Cancer Institute. Do I Have Nicotine Withdrawal? Smokefree.gov. 2015. Available online: http://smokefree.gov/withdrawal (accessed on 26 May 2016).
- Lee, E.C.; Whitehead, A.L.; Jacques, R.M.; Julious, S.A. The statistical interpretation of pilot trials: Should significance thresholds be reconsidered? BMC Med. Res. Methodol. 2014, 14, 41. [Google Scholar] [CrossRef]
- Garcia, F.S.; Derefinko, K.J.; Bursac, Z.; Klesges, R.C.; Ebbert, J.O.; Womack, C.R.; Krukowski, R.A. Fit & quit: An efficacy trial of two behaviorial post-cessation weight gain interventions. Contemp. Clin. Trials 2019, 76, 31–40. [Google Scholar] [CrossRef]
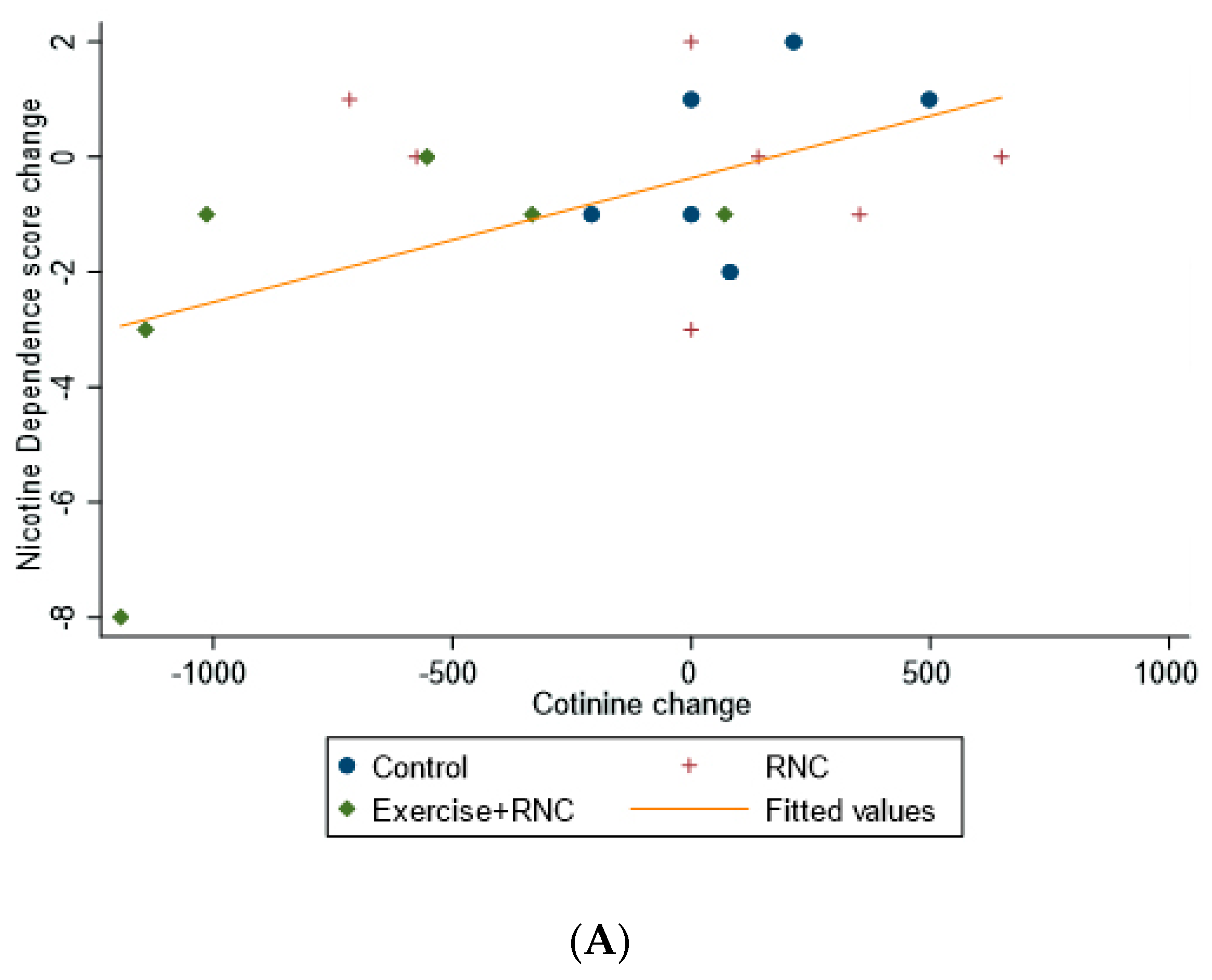
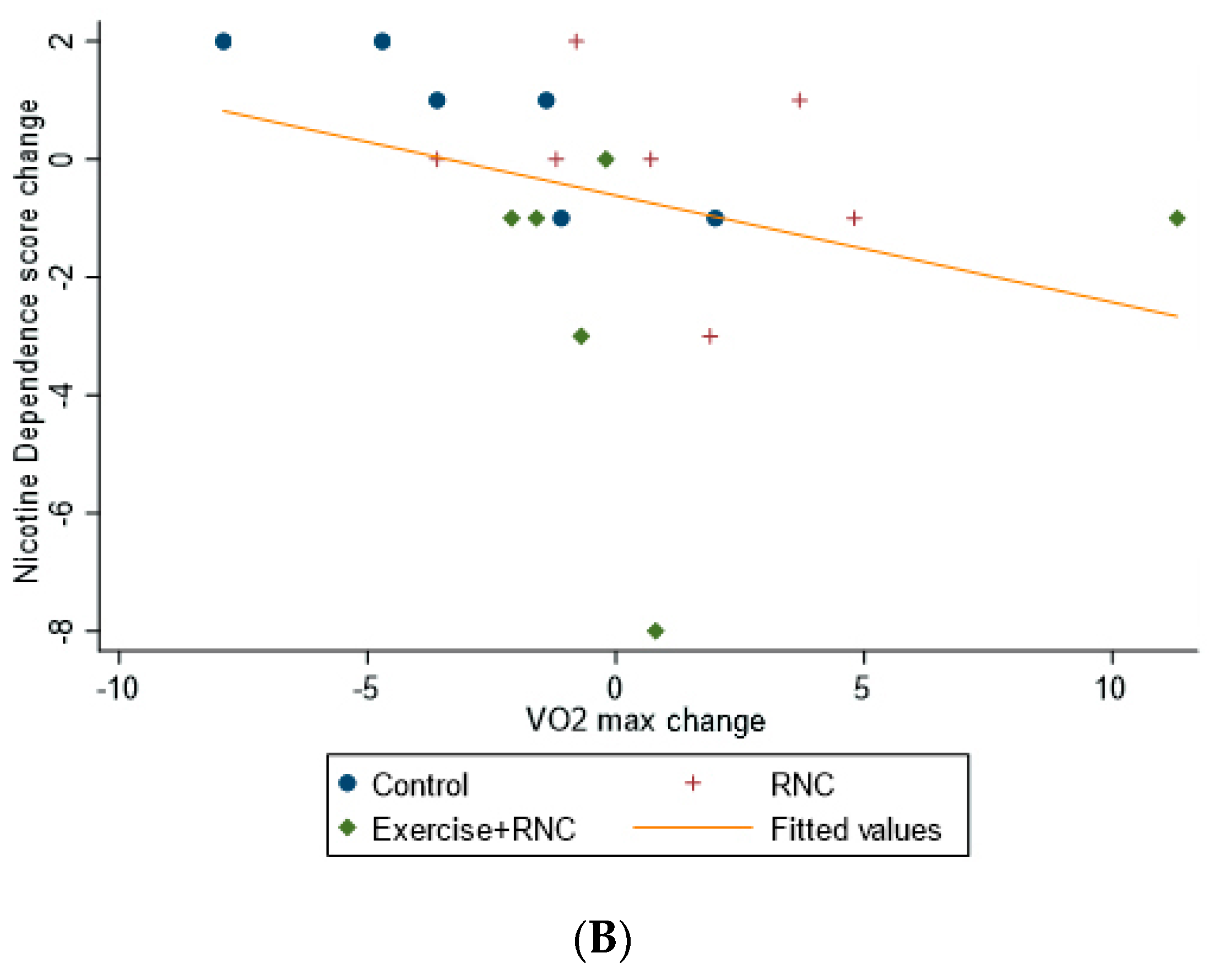
| Outcome Variables | Control (N = 7) | RNC (N = 7) | RNC + Exercise (N = 6) | Time X Treatment ANOVA (p-Value) | Time X Treatment Effect Size (Partial η2) |
|---|---|---|---|---|---|
| Demographics | |||||
| Age | 45.0 ± 12.0 | 38.6 ± 13.0 | 45.7 ± 7.9 | 0.80 | 0.03 |
| Body Fat, Fitness, and Metabolism | |||||
| Weight (kg) | 0.07 | 0.29 | |||
| Baseline | 74.7 ± 17.3 | 88.3 ± 12.8 | 81.2 ± 14.8 | ||
| 12 Weeks | 79.0 ± 16.0 | 88.5 ± 12.5 | 80.8 ± 15.4 | ||
| BMI (kg/m2) | 0.51 | 0.09 | |||
| Baseline | 29.2 ± 6.4 | 32.6 ± 4.3 | 31.4 ± 6.9 | ||
| 12 Weeks | 29.7 ± 6.7 | 32.4 ± 4.1 | 31.2 ± 7.0 | ||
| Fat% | 0.90 | 0.01 | |||
| Baseline | 38.1 ± 9.6 | 40.0 ± 5.2 | 43.7 ± 5.0 | ||
| 12 Weeks | 38.2 ± 10.6 | 39.5 ± 6.2 | 43.4 ± 4.3 | ||
| VO2max (mL/kg/min) | 0.20 | 0.18 | |||
| Baseline | 26.8 ± 8.9 | 20.4 ± 4.7 | 22.5 ± 2.8 | ||
| 12 Weeks | 24.0 ± 8.4 | 21.2 ± 4.4 | 23.8 ± 4.7 | ||
| RMR (kcal/day) | 0.54 | 0.08 | |||
| Baseline | 1344.8 ± 202.0 | 1483.3 ± 184.5 | 1302.0 ± 163.4 | ||
| 12 Weeks | 1362.3 ± 219.7 | 1479.7 ± 164.3 | 1301.2 ± 171.0 | ||
| RER | 0.44 | 0.10 | |||
| Baseline | 0.83 ± 0.04 | 0.84 ± 0.05 | 0.82 ± 0.03 | ||
| 12 Weeks | 0.84 ± 0.05 | 0.89 ± 0.08 | 0.82 ± 0.05 | ||
| Metabolic Health and Hormones: | |||||
| Insulin (uIU/mL) ** | 0.13 | 0.25 | |||
| Baseline | 14.6 ± 8.0 | 9.8 ± 7.6 | 6.8 ± 2.2 | ||
| 12 Weeks | 19.2 ± 11.7 a | 15.6 ± 7.9 a | 6.3 ± 2.6 | ||
| Glucose (mg/dL) | 0.50 | 0.10 | |||
| Baseline | 94.5 ± 11.7 | 93.7 ± 7.2 | 95.1 ± 9.3 | ||
| 12 Weeks | 102.8 ± 15.3 | 102.3 ± 17.4 | 94.3 ± 5.5 | ||
| HbA1C (%) | 0.63 | 0.06 | |||
| Baseline | 5.4 ± 0.5 | 5.2 ± 0.3 | 5.5 ± 0.3 | ||
| 12 Weeks | 5.5 ± 0.6 | 5.2 ± 0.4 | 5.5 ± 0.3 | ||
| HOMA-IR * | 0.32 | 0.19 | |||
| Baseline | 2.8 ± 1.2 | 2.2 ± 1.6 | 1.6 ± 0.7 | ||
| 12 Weeks | 4.3 ± 3.2 | 4.1 ± 2.7 | 1.6 ± 0.7 | ||
| Cortisol (μg/dL) | 0.06 | 0.30 | |||
| Baseline | 10.7 ± 3.5 | 8.7 ± 3.2 | 11.3 ± 5.2 | ||
| 12 Weeks | 16.5 ± 3.7 b | 11.1 ± 2.9 | 11.0 ± 3.2 | ||
| hs-CRP (mg/L) | 0.35 | 0.13 | |||
| Baseline | 3.0 ± 2.6 | 2.0 ± 1.7 | 6.3 ± 7.9 | ||
| 12 Weeks | 7.4 ± 9.2 | 1.6 ± 1.2 | 8.3 ± 11.2 | ||
| Leptin (ng/mL) | 0.94 | 0.01 | |||
| Baseline | 18.5 ± 12.9 | 16.2 ± 9.6 | 16.5 ± 10.3 | ||
| 12 Weeks | 20.4 ± 13.9 | 18.0 ± 8.7 | 19.5 ± 9.5 | ||
| TC (mg/dL) ** | 0.12 | 0.23 | |||
| Baseline | 170.7 ± 24.2 | 176.4 ± 35.6 | 206.7 ± 30.7 | ||
| 12 Weeks | 175.3 ± 29.1 | 158.1 ± 27.4 | 208.7 ± 31.1 | ||
| TG (mg/dL) | 0.09 | 0.26 | |||
| Baseline | 102.2 ± 80.0 | 123.4 ± 61.1 | 106.8 ± 79.1 | ||
| 12 Weeks | 134.8 ± 90.8 | 114.4 ± 51.0 | 110.3 ± 67.6 | ||
| HDL (mg/dL) ** | 0.44 | 0.10 | |||
| Baseline | 53.3 ± 7.9 | 43.1 ± 7.5 | 54.2 ± 13.7 | ||
| 12 Weeks | 55.3 ± 10.9 | 41.6 ± 6.3 | 56.8 ± 11.6 | ||
| LDL (mg/dL) * | 0.35 | 0.12 | |||
| Baseline | 110.2 ± 14.0 | 112.9 ± 35.5 | 134.7 ± 23.7 | ||
| 12 Weeks | 97.5 ± 21.9 | 97.6 ± 28.2 | 133.7 ± 26.4 | ||
| TC/HDL | 0.59 | 0.06 | |||
| Baseline | 3.3 ± 0.7 | 4.3 ± 1.4 | 4.0 ± 1.1 | ||
| 12 Weeks | 3.3 ± 0.9 | 3.9 ± 1.0 | 3.8 ± 0.9 | ||
| Outcome Variables | Control | RNC | RNC + Exercise | Time X Treatment ANOVA (p-Value) | Time X Treatment Effect Size (Partial η2) |
|---|---|---|---|---|---|
| Withdrawal symptoms: | |||||
| Wisconsin smoking withdrawal score | 0.82 | 0.02 | |||
| Baseline | 1.9 ± 0.4 | 1.9 ± 0.6 | 1.9 ± 0.2 | ||
| 12 Weeks | 1.9 ± 0.5 | 2.1 ± 0.4 | 1.8 ± 0.5 | ||
| Anger | 0.43 | 0.10 | |||
| Baseline | 2.5 ± 0.6 | 1.9 ± 0.9 | 1.7 ± 1.0 | ||
| 12 Weeks | 1.9 ± 0.8 | 2.2 ± 1.1 | 1.7 ± 0.8 | ||
| Anxiety | 0.39 | 0.11 | |||
| Baseline | 2.4 ± 0.8 | 2.3 ± 1.0 | 2.5 ± 0.7 | ||
| 12 Weeks | 2.1 ± 0.8 | 2.5 ± 0.5 | 2.0 ± 0.8 | ||
| Concentration | 0.79 | 0.03 | |||
| Baseline | 1.6 ± 1.3 | 2.0 ± 0.8 | 1.6 ± 0.4 | ||
| 12 Weeks | 1.5 ± 0.9 | 1.9 ± 0.6 | 1.3 ± 0.5 | ||
| Craving | 0.07 | 0.27 | |||
| Baseline | 2.1 ± 0.7 | 1.9 ± 0.6 | 2.5 ± 0.6 | ||
| 12 Weeks | 2.0 ± 0.9 | 2.4 ± 0.4 | 1.8 ± 0.9 | ||
| Hunger | 0.67 | 0.05 | |||
| Baseline | 2.2 ± 0.8 | 2.3 ± 1.0 | 1.6 ± 0.5 | ||
| 12 Weeks | 2.2 ± 1.0 | 2.6 ± 0.6 | 2.0 ± 0.9 | ||
| Sadness | 0.98 | 0.003 | |||
| Baseline | 1.2 ± 0.4 | 1.5 ± 0.7 | 1.6 ± 0.6 | ||
| 12 Weeks | 1.4 ± 0.6 | 1.6 ± 0.9 | 1.9 ± 0.5 | ||
| Sleep | 0.71 | 0.04 | |||
| Baseline | 1.9 ± 0.7 | 1.7 ± 0.9 | 1.7 ± 0.8 | ||
| 12 Weeks | 1.8 ± 0.9 | 1.5 ± 0.4 | 1.9 ± 1.0 | ||
| Smoking Cessation Outcomes: | |||||
| Nicotine Dependence score | 0.08 | 0.25 | |||
| Baseline | 4.9 ± 1.9 | 5.4 ± 2.1 | 5.7 ± 2.5 | ||
| 12 Weeks | 5.1 ± 2.5 | 5.3 ± 1.7 | 3.3 ± 2.7 | ||
| Nicotine (ng/mL) | 0.68 | 0.06 | |||
| Baseline | 636.2 ± 491.0 | 200.3 ± 179.8 | 462.6 ± 440.8 | ||
| 12 Weeks | 582.0 ± 500.5 | 305.8 ± 454.2 | 265.4 ± 523.2 | ||
| Nornicotine (ng/mL) | 0.38 | 0.13 | |||
| Baseline | 62.9 ± 43.9 | 33.4 ± 16.5 | 51.4 ± 30.6 | ||
| 12 Weeks | 68.3 ± 48.3 | 42.7 ± 27.8 | 35.8 ± 47.9 | ||
| Cotinine (ng/mL) | 0.01 | 0.43 | |||
| Baseline | 824.5 ± 379.2 | 899.1 ± 333.6 | 1058.2 ± 314.4 | ||
| 12 Weeks | 921.8 ± 484.3 | 877.9 ± 456.1 | 363.9 ± 471.1 a | ||
| Anabasine (ng/mL) | 0.37 | 0.17 | |||
| Baseline | 11.8 ± 9.9 | 3.5 ± 1.5 | 8.4 ± 2.3 | ||
| 12 Weeks | 11.5 ± 6.9 | 6.4 ± 4.8 | 5.2 ± 7.7 | ||
| CO (ppm) | 0.95 | 0.01 | |||
| Baseline | 1.8 ± 1.1 | 2.1 ± 1.5 | 2.0 ± 2.0 | ||
| 12 Weeks | 1.3 ± 1.7 | 1.3 ± 1.8 | 1.3 ± 0.7 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, C.; Yao, N.; Miller, S.L.; Macpherson, C.; Hassinger, T.; Love, K.; Malin, S.K. Exercise and Reduced Nicotine Content Cigarettes in Adult Female Smokers: A Pilot Trial. Int. J. Environ. Res. Public Health 2022, 19, 6647. https://doi.org/10.3390/ijerph19116647
Li C, Yao N, Miller SL, Macpherson C, Hassinger T, Love K, Malin SK. Exercise and Reduced Nicotine Content Cigarettes in Adult Female Smokers: A Pilot Trial. International Journal of Environmental Research and Public Health. 2022; 19(11):6647. https://doi.org/10.3390/ijerph19116647
Chicago/Turabian StyleLi, Chaofan, Nengliang Yao, Stephanie L. Miller, Christopher Macpherson, Taryn Hassinger, Kaitlin Love, and Steven K. Malin. 2022. "Exercise and Reduced Nicotine Content Cigarettes in Adult Female Smokers: A Pilot Trial" International Journal of Environmental Research and Public Health 19, no. 11: 6647. https://doi.org/10.3390/ijerph19116647
APA StyleLi, C., Yao, N., Miller, S. L., Macpherson, C., Hassinger, T., Love, K., & Malin, S. K. (2022). Exercise and Reduced Nicotine Content Cigarettes in Adult Female Smokers: A Pilot Trial. International Journal of Environmental Research and Public Health, 19(11), 6647. https://doi.org/10.3390/ijerph19116647







