Application of Integrated Fixed-Film Activated Sludge in a Conventional Wastewater Treatment Plant
Abstract
:1. Introduction
2. Material and Methods
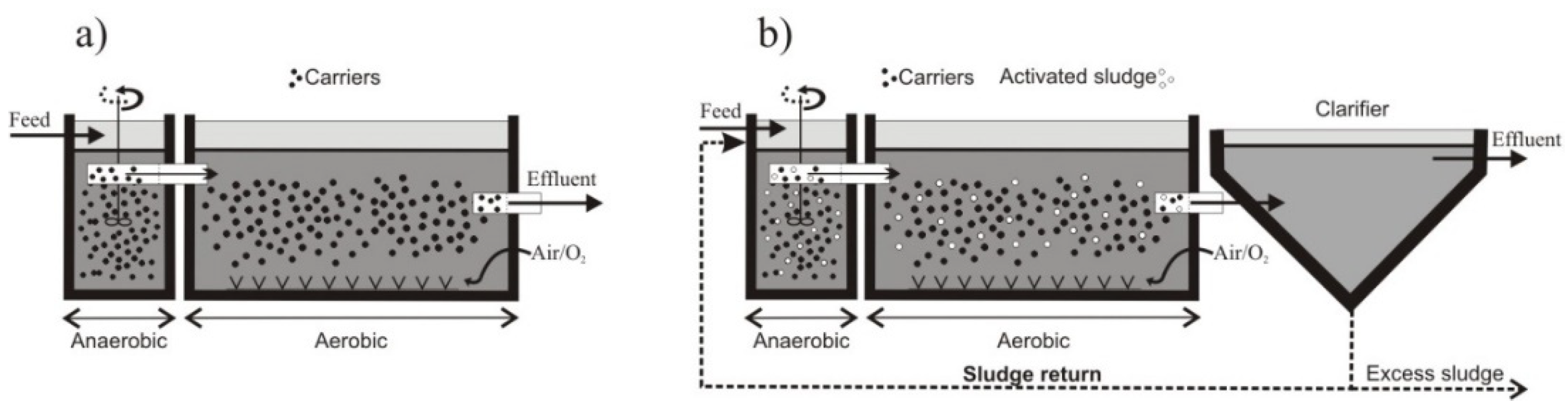
2.1. Activated Sludge System Characteristics
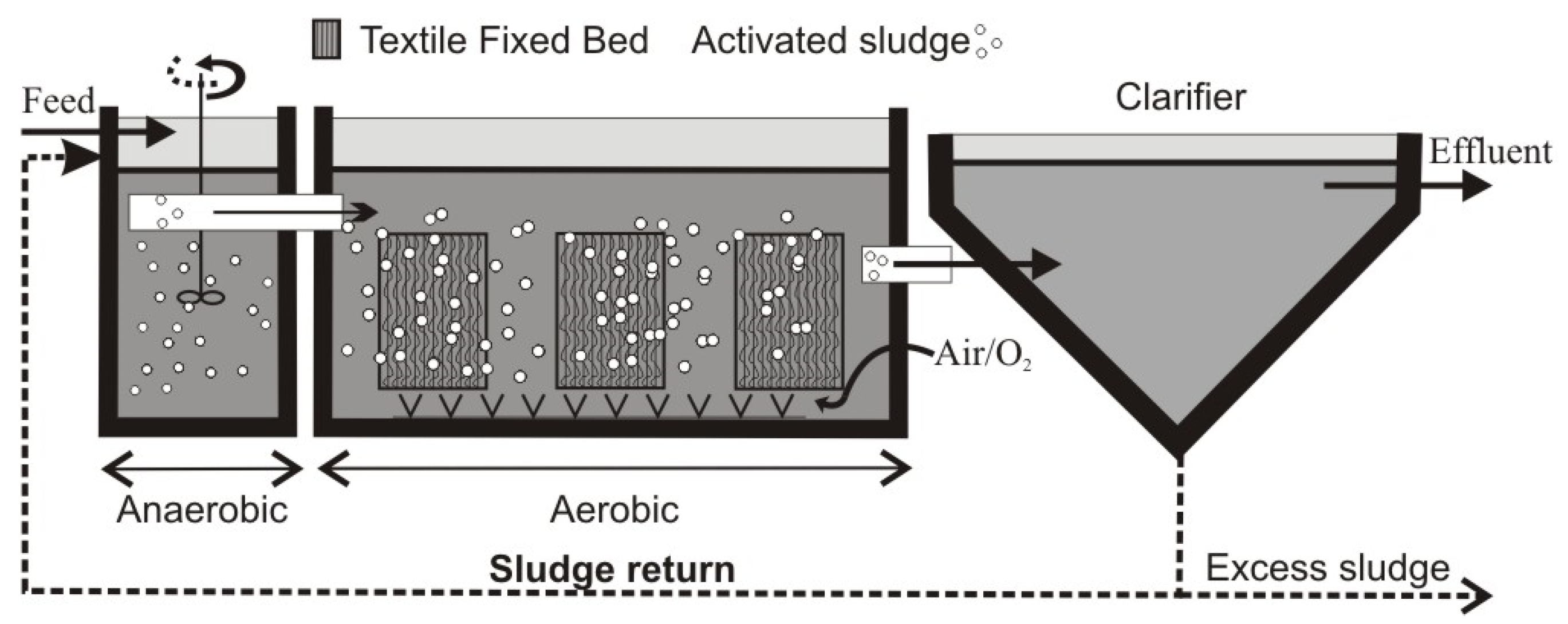
2.2. IFAS System
2.3. Analytical Methods
3. Results and Discussion
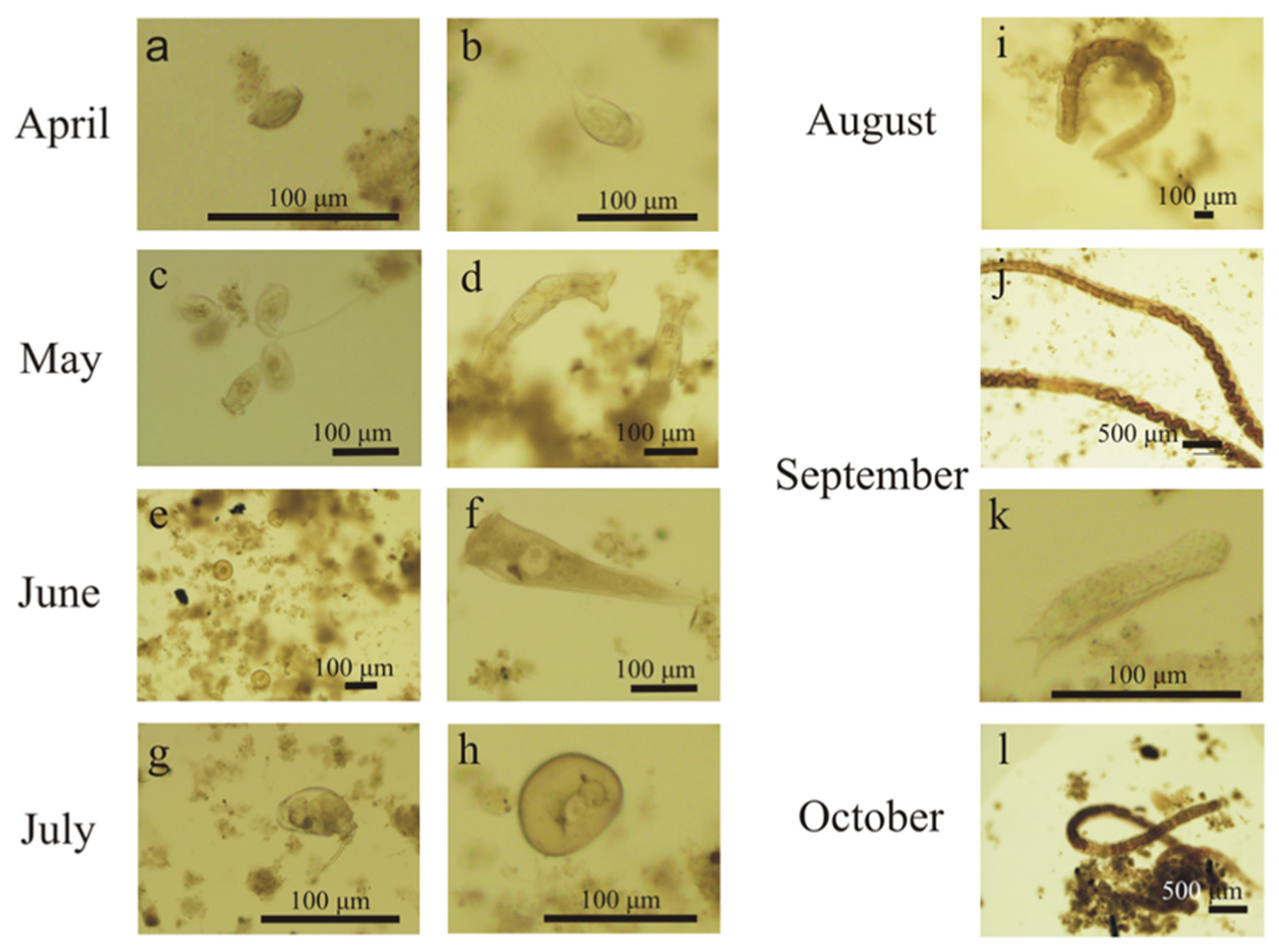
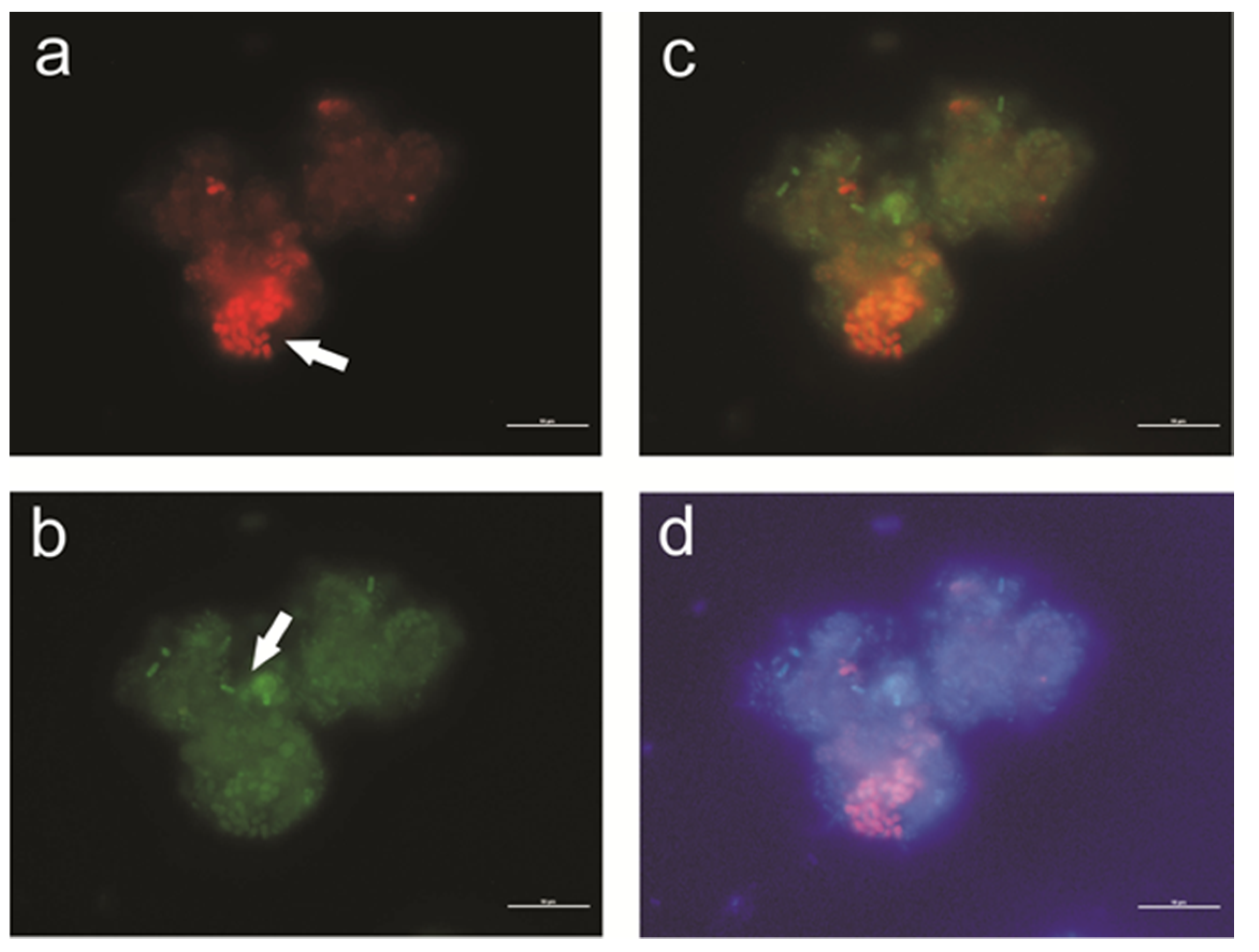
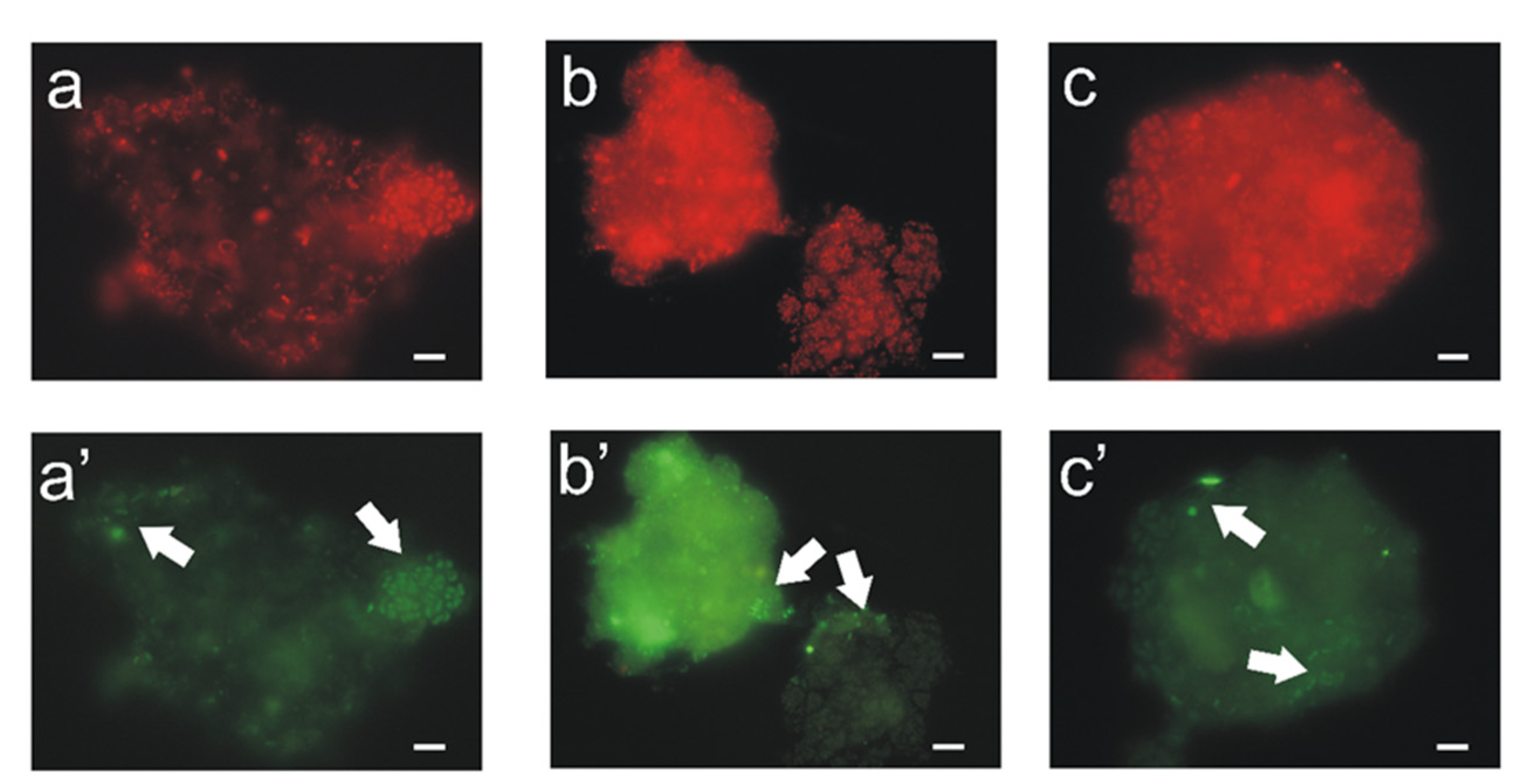
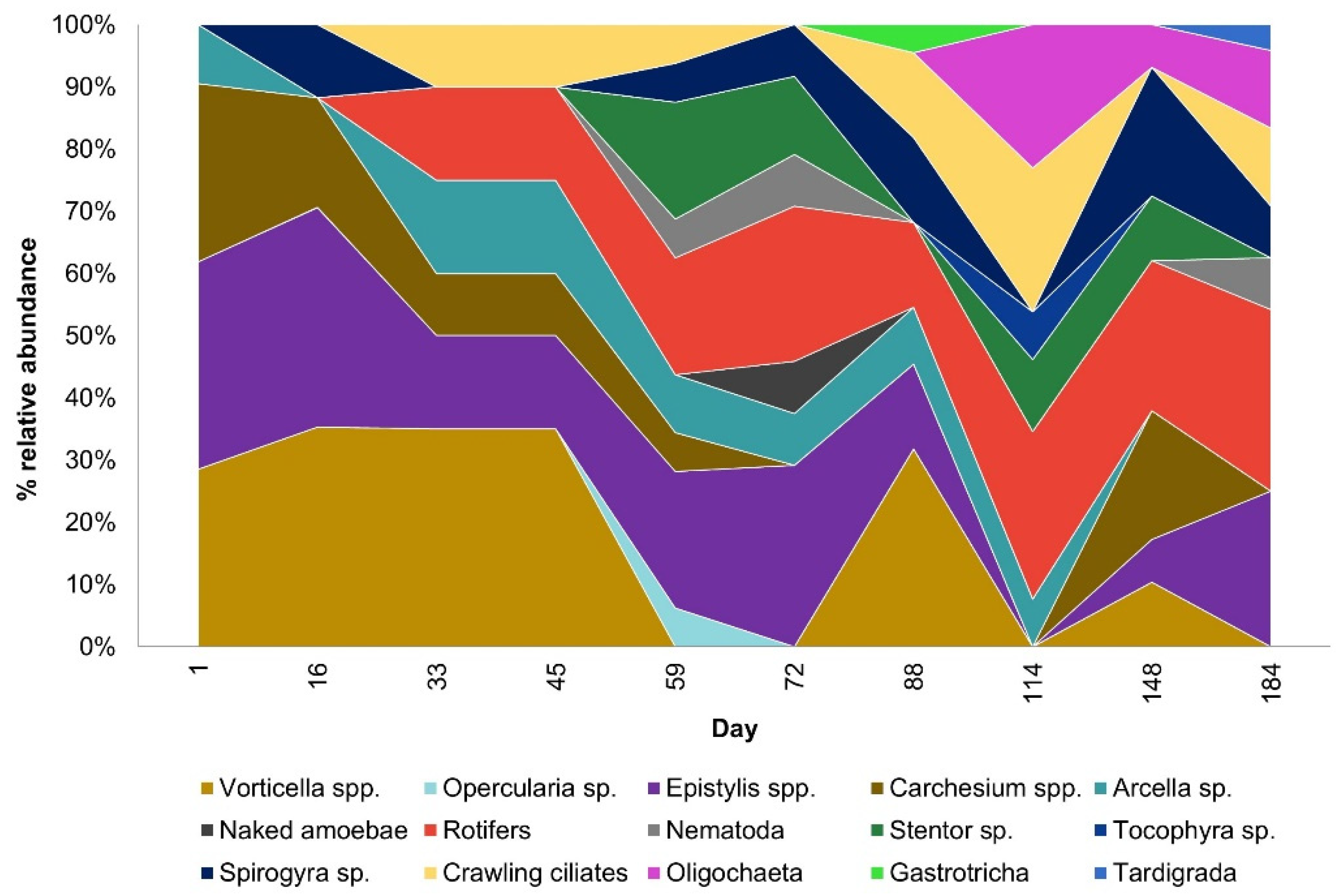
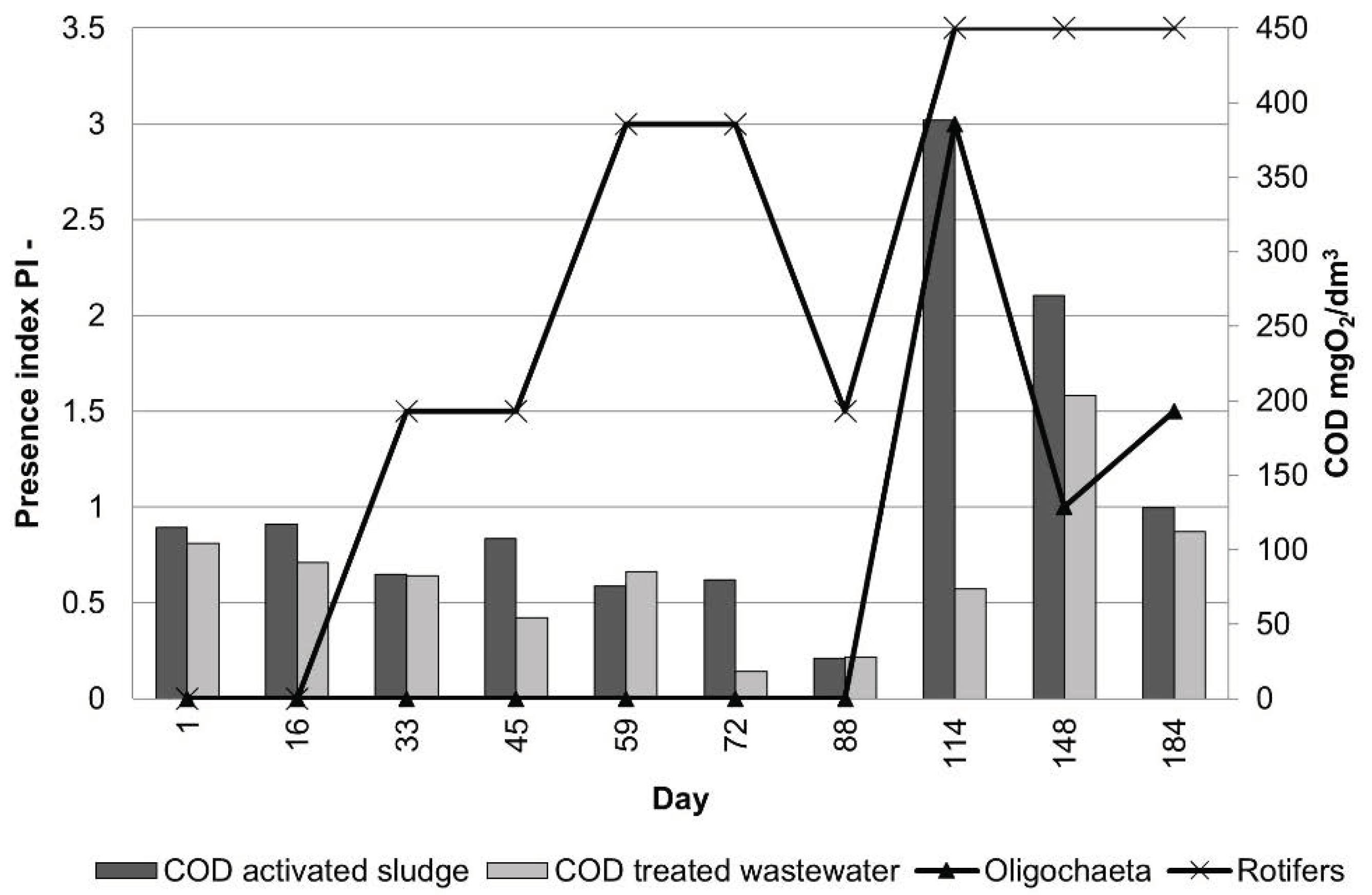
3.1. Activated Sludge Microorganisms Settled on Textile Beds
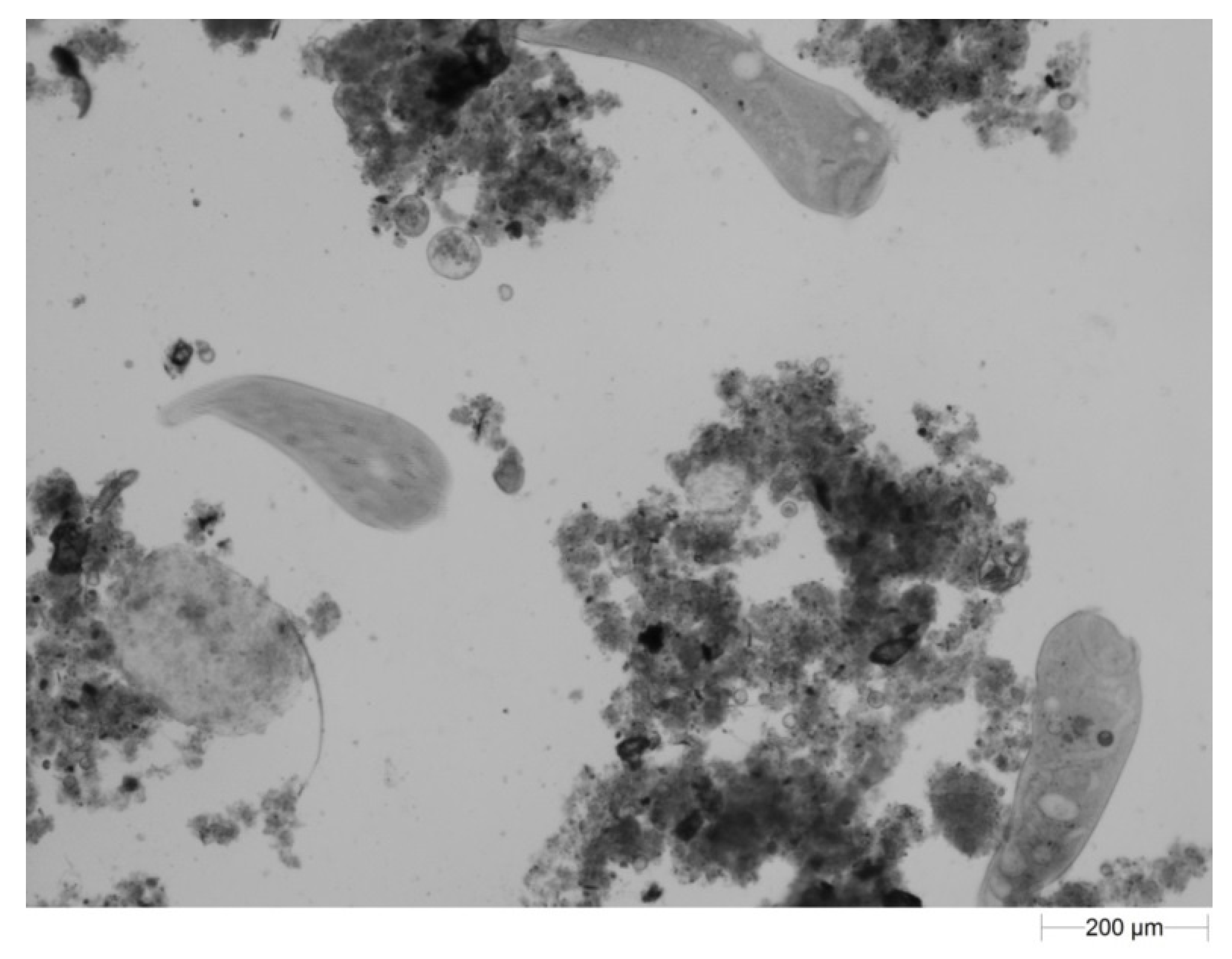
3.2. Activated Suspended Sludge Microorganisms in the Bulk Water
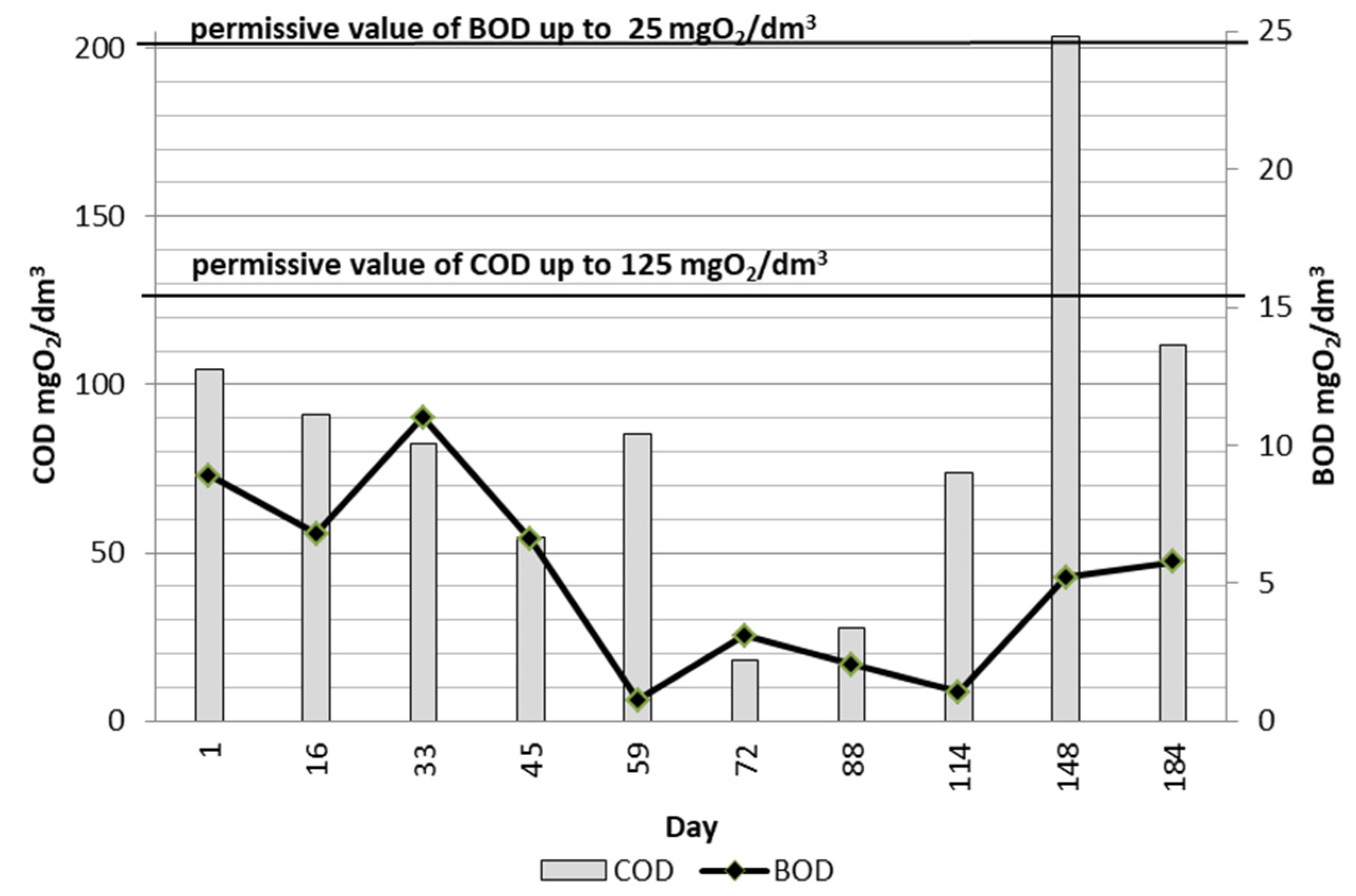
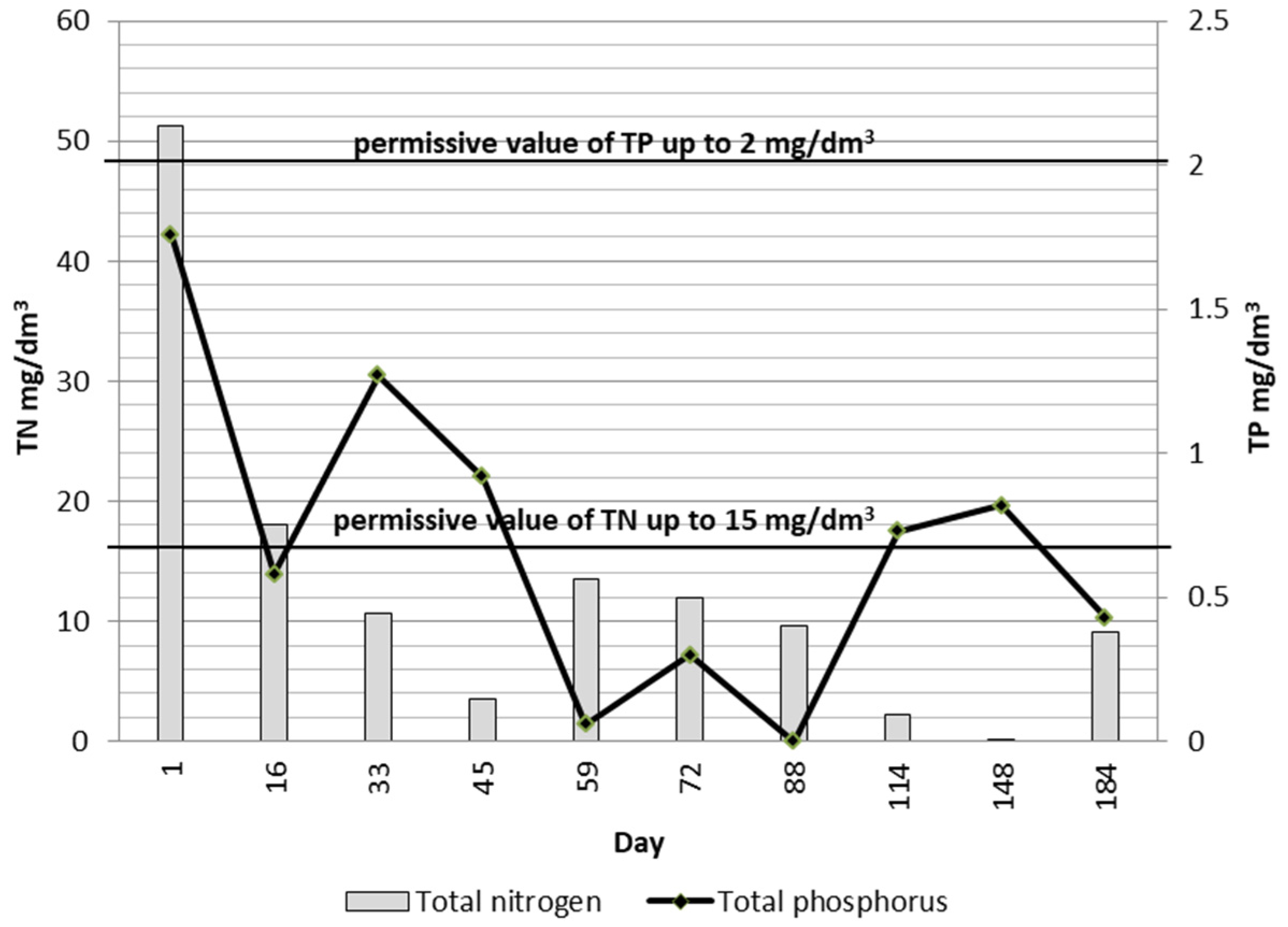
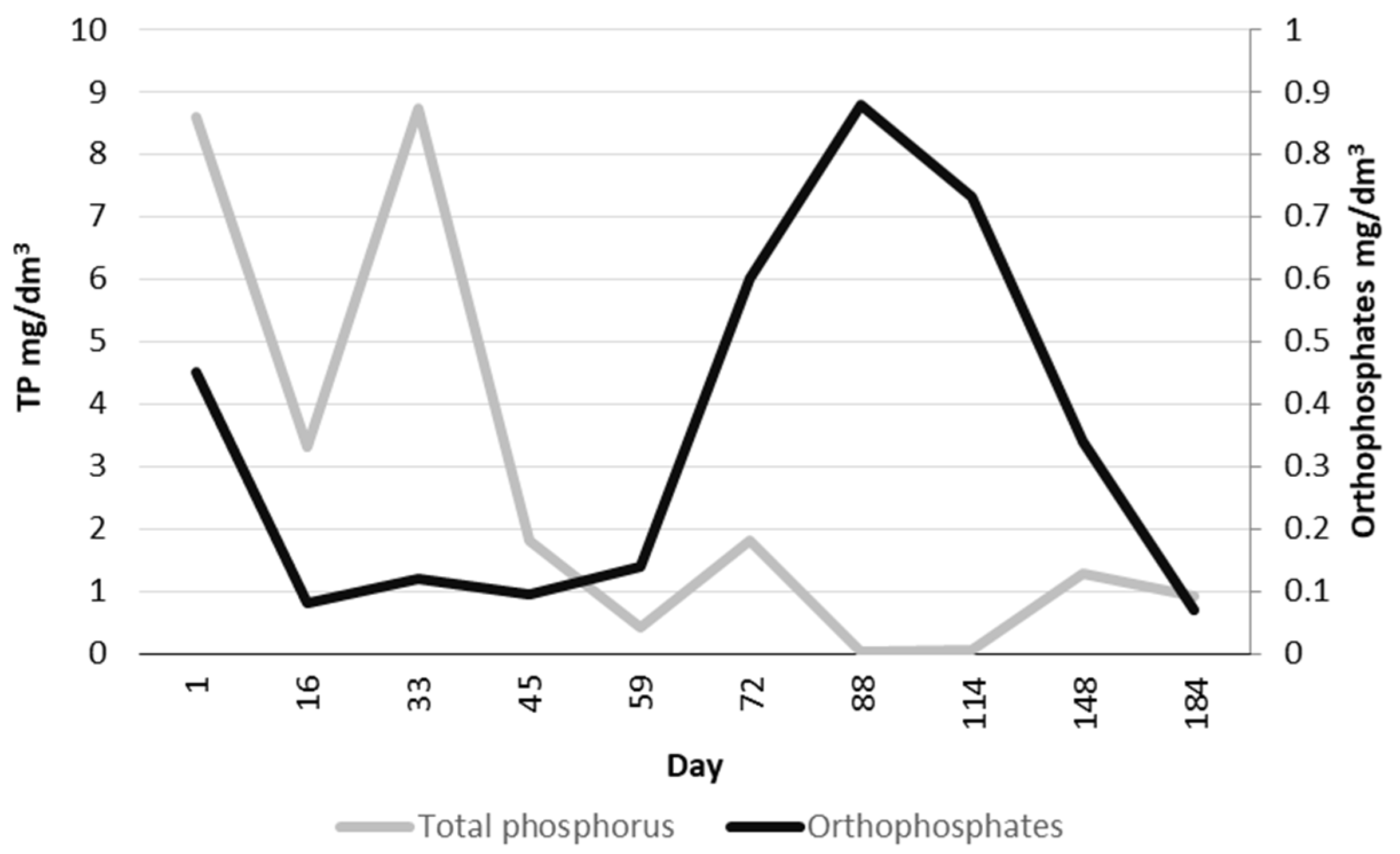
3.3. Treatment Efficiency
3.4. Wastewater Control Information
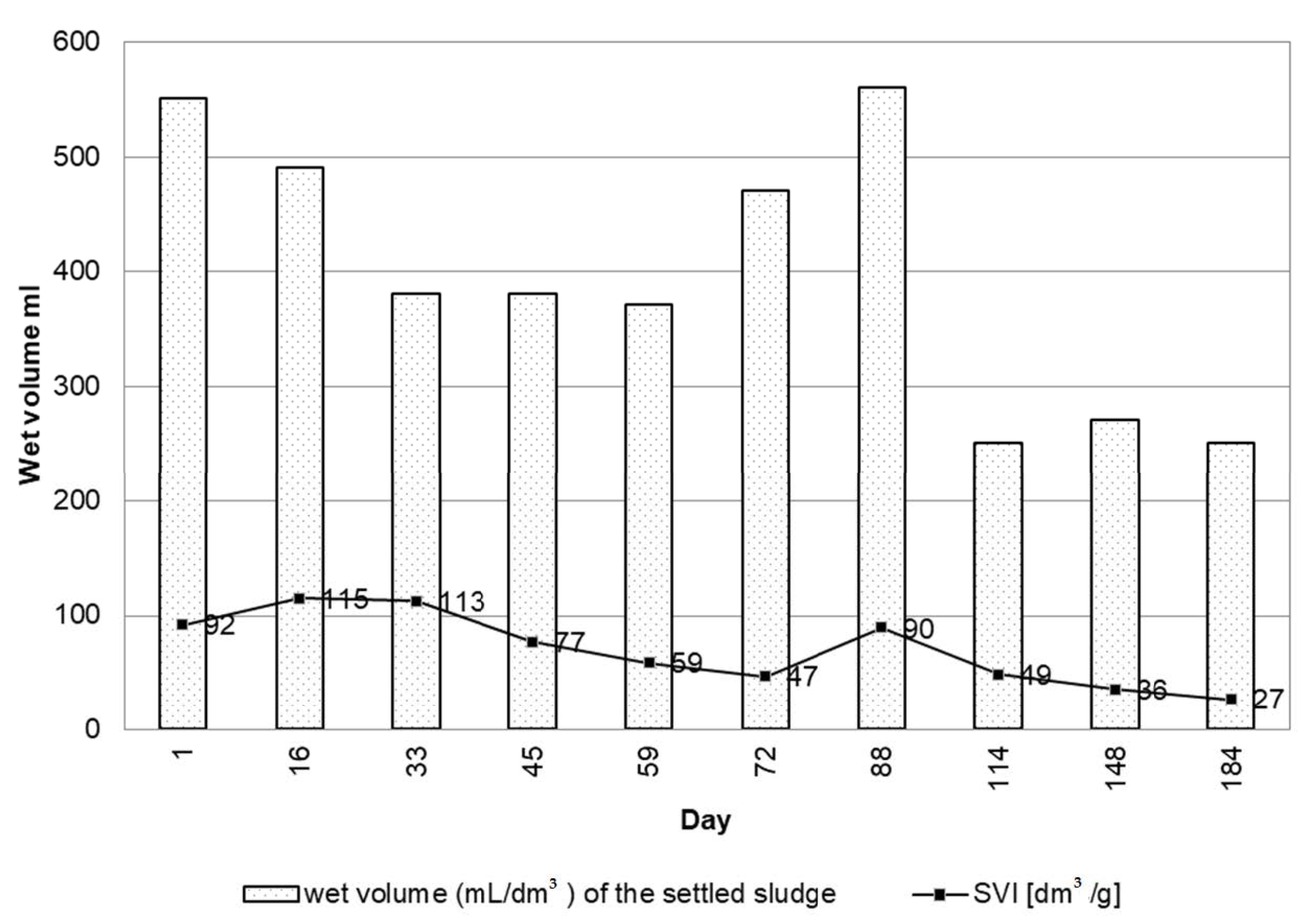
3.5. Activated Sludge Settleability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Iloms, E.; Ololade, O.O.; Ogola, H.J.; Selvarajan, R. Investigating Industrial Effluent Impact on Municipal Wastewater Treatment Plant in Vaal, South Africa. Int. J. Environ. Res. 2020, 17, 1096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pawęska, K.; Bawiec, A. Activated Sludge Technology Combined with Hydroponic Lagoon as a Technology Suitable for Treatment of Wastewater Delivered by Slurry Tanks. J. Ecol. Eng. 2017, 18, 29–37. [Google Scholar] [CrossRef]
- Bugajski, P.M.; Nowobilska-Majewska, E.; Kurek, K. The Variability of Pollution Load of Organic, Biogenic and Chromium Ions in Wastewater Inflow to the Treatment Plant in Nowy Targ. J. Water Land Dev. 2017, 35, 11–17. [Google Scholar] [CrossRef]
- Jianlong, W.; Hanchang, S.; Yi, Q. Wastewater Treatment in a Hybrid Biological Reactor (HBR): Effect of Organic Loading Rates. Process Biochem. 2000, 36, 297–303. [Google Scholar] [CrossRef]
- Low, E.W.; Chase, H.A. Reducing Production of Excess Biomass during Wastewater Treatment. Water Res. 1999, 33, 1119–1132. [Google Scholar] [CrossRef]
- Guo, W.-Q.; Yang, S.-S.; Xiang, W.-S.; Wang, X.-J.; Ren, N.-Q. Minimization of Excess Sludge Production by In-Situ Activated Sludge Treatment Processes—A Comprehensive Review. Biotechnol. Adv. 2013, 31, 1386–1396. [Google Scholar] [CrossRef] [PubMed]
- Tandoi, V.; Rossetti, S.; Wanner, J. Activated Sludge Separation Problems: Theory, Control Measures, Practical Experiences; IWA Publishing: London, UK, 2017. [Google Scholar]
- Jin, B.; Wilén, B.-M.; Lant, P. A Comprehensive Insight into Floc Characteristics and Their Impact on Compressibility and Settleability of Activated Sludge. Chem. Eng. J. 2003, 95, 221–234. [Google Scholar] [CrossRef]
- Jenkins, D.; Richard, M.G.; Daigger, G.T. Manual on the Causes and Control of Activated Sludge Bulking, Foaming, and Other Solids Separation Problems; CRC Press: Boca Raton, FL, USA; Taylor & Francis: Abingdon, UK, 2003. [Google Scholar]
- Ødegaard, H. MBBR and IFAS Systems. Advances in Wastewater Treatment; IWA Publishing: London, UK, 2018. [Google Scholar]
- Malovany, A.; Trela, J.; Plaza, E. Mainstream Wastewater Treatment in Integrated Fixed Film Activated Sludge (IFAS) Reactor by Partial Nitritation/Anammox Process. Bioresour. Technol. 2015, 198, 478–487. [Google Scholar] [CrossRef]
- Vergine, P.; Salerno, C.; Berardi, G.; Pollice, A. Sludge Cake and Biofilm Formation as Valuable Tools in Wastewater Treatment by Coupling Integrated Fixed-Film Activated Sludge (IFAS) with Self Forming Dynamic Membrane BioReactors (SFD-MBR). Bioresour. Technol. 2018, 268, 121–127. [Google Scholar] [CrossRef]
- Saltnes, T.; Sørensen, G.; Eikås, S. Biological Nutrient Removal in a Continuous Biofilm Process. Water Pract. Technol. 2017, 12, 797–805. [Google Scholar] [CrossRef]
- Randall, C.W.; Sen, D. Full-Scale Evaluation of an Integrated Fixed-Film Activated Sludge (IFAS) Process for Enhanced Nitrogen Removal. Water Sci. Technol. 1996, 33, 155–162. [Google Scholar] [CrossRef]
- Shao, Y.; Shi, Y.; Mohammed, A.; Liu, Y. Wastewater Ammonia Removal Using an Integrated Fixed-Film Activated Sludge-Sequencing Batch Biofilm Reactor (IFAS-SBR): Comparison of Suspended Flocs and Attached Biofilm. Int. Biodeterior. Biodegrad. 2017, 116, 38–47. [Google Scholar] [CrossRef]
- Piechna, P.; Żubrowska-Sudoł, M. Respirometric Activity of Activated Sludge and Biofilm in Ifas-Mbbr System. J. Ecol. Eng. 2017, 18, 145–151. [Google Scholar] [CrossRef] [Green Version]
- Veuillet, F.; Lacroix, S.; Bausseron, A.; Gonidec, E.; Ochoa, J.; Christensson, M.; Lemaire, R. Integrated Fixed-Film Activated Sludge ANITATM Mox Process—A New Perspective for Advanced Nitrogen Removal. Water Sci. Technol. 2014, 69, 915–922. [Google Scholar] [CrossRef] [PubMed]
- Rusten, B.; Merete Nielsen, I.; Welander, T.; Rasmussen, V. Increasing the Capacity of Activated Sludge Plants by Using AS/MBBRTM Hybrid Process with Kaldnes Biofilm Carriers. In Proceedings of the 5th IWA Conference on Biofilms Systems, Cape Town, South Africa, 14–17 September 2003. [Google Scholar]
- Singh, N.K.; Kazmi, A.A.; Starkl, M. Treatment performance and microbial diversity under dissolved oxygen stress conditions: Insights from a single stage IFAS reactor treating municipal wastewater. J. Taiwan Inst. Chem. Eng. 2016, 65, 197–203. [Google Scholar] [CrossRef]
- Johnson, T.L.; McQuarrie, J.P.; Shaw, A.R. Integrated Fixed-film Activated Sludge (IFAS): The new choice for nitrogen removal upgrades in the United States. In Proceedings of the Water Environment Federation Annual Conference (WEFTEC), New Orleans, LA, USA, 2–6 October 2004; Volume 16, pp. 296–318. [Google Scholar]
- Amann, R.I.; Ludwig, W.; Schleifer, K.-H. Phylogenetic Identification and in Situ Detection of Individual Microbial Cells without Cultivation. Microbiol. Rev. 1995, 59, 143–169. [Google Scholar] [CrossRef] [PubMed]
- Kocwa-Haluch, R.; Woźniakiewicz, T. Microscopic analysis of activated sludge and its role in the control of the technological process of wastewater treatment. Czasopismo Techniczne. Środowisko 2011, 108, 141–162. [Google Scholar]
- Drzewicki, A.; Fyda, J. Bioindicative Aspects of Activated Sludge in Wastewater Treatment; Wydawnictwo Seidel-Przywecki: Warsaw, Poland, 2020. [Google Scholar]
- Kwon, S.D.; Kim, T.S.; Yu, G.H.; Jung, J.H.; Park, H.D. Bacterial community composition and diversity of a full-scale integrated fixed-film activated sludge system as investigated by pyrosequencing. J. Microbiol. Biotechnol. 2010, 20, 1717–1723. [Google Scholar] [CrossRef]
- Suarez, C.; Persson, F.; Hermansson, M. Predation of nitritation–anammox biofilms used for nitrogen removal from wastewater. FEMS Microbiol. Ecol. 2015, 91, fiv124. [Google Scholar] [CrossRef] [Green Version]
- Dos Santos, L.A.; Ferreira, V.; Pereira, M.O.; Nicolau, A. Relationship between protozoan and metazoan communities and operation and performance parameters in a textile sewage activated sludge system. Eur. J. Protistol. 2014, 50, 319–328. [Google Scholar] [CrossRef] [Green Version]
- Amanatidou, E.; Samiotis, G.; Trikoilidou, E.; Tzelios, D.; Michailidis, A. Influence of Wastewater Treatment Plants’ Operational Conditions on Activated Sludge Microbiological and Morphological Characteristics. Environ. Technol. 2016, 37, 265–278. [Google Scholar] [CrossRef] [PubMed]
- Fiałkowska, E.; Fyda, J.; Pajdak-Stós, A.; Wiąckowski, K. Activated Sludge: Biology and Microscopic Analysis; Wydawnictwo Seidel-Przywecki: Warsaw, Poland, 2010; Volume 108, pp. 141–162. [Google Scholar]
- Liang, P.; Huang, X.; Qian, Y. Excess Sludge Reduction in Activated Sludge Process through Predation of Aeolosoma Hemprichi. Biochem. Eng. J. 2006, 28, 117–122. [Google Scholar] [CrossRef]
- Höfle, G.; Reinecke, S.; Laude, U.; Spitzner, D. Amethystin, the Coloring Principle of Stentor Amethystinus. J. Nat. Prod. 2014, 77, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Foissner, W.; Wölfl, S. Revision of the Genus Stentor Oken (Protozoa, Ciliophora) and Description of S. araucanus Nov. Spec, from South American Lakes. J. Plankton Res. 1994, 16, 255–289. [Google Scholar] [CrossRef] [Green Version]
- Martín-Cereceda, M.; Serrano, S.; Guinea, A. A comparative study of ciliated protozoa communities in activated-sludge plants. FEMS Microbiol. Ecol. 1996, 21, 267–276. [Google Scholar] [CrossRef]
- Lanzoni, O.; Plotnikov, A.; Khlopko, Y.; Munz, G.; Petroni, G.; Potekhin, A. The Core Microbiome of Sessile Ciliate Stentor Coeruleus Is Not Shaped by the Environment. Sci. Rep. 2019, 9, 11452. [Google Scholar] [CrossRef]
- Pucciarelli, S.; Buonanno, F.; Pellegrini, G.; Pozzi, S.; Ballarini, P.; Miceli, C. Biomonitoring of Lake Garda: Identification of Ciliate Species and Symbiotic Algae Responsible for the “Black-Spot” Bloom during the Summer of 2004. Environ. Res. 2008, 107, 194–200. [Google Scholar] [CrossRef]
- Klimek, B.; Fyda, J.; Pajdak-Stós, A.; Kocerba, W.; Fiałkowska, E.; Sobczyk, M. Toxicity of Ammonia Nitrogen to Ciliated Protozoa Stentor Coeruleus and Coleps Hirtus Isolated from Activated Sludge of Wastewater Treatment Plants. Bull. Environ. Contam. Toxicol. 2012, 89, 975–977. [Google Scholar] [CrossRef] [Green Version]
- Tartar, V. The Biology of Stentor: International Series of Monographs on Pure and Applied Biology: Zoology; Pergamon Press LTD: Oxford, UK; London, UK; New York, NY, USA; Paris, France, 1961. [Google Scholar]
- González-Pleiter, M.; Velázquez, D.; Casero, M.C.; Tytgat, B.; Verleyen, E.; Leganés, F.; Rosal, R.; Quesada, A.; Fernández-Piñas, F. Microbial Colonizers of Microplastics in an Arctic Freshwater Lake. Sci. Total Environ. 2021, 795, 148640. [Google Scholar] [CrossRef]
- Dubber, D.; Gray, N.F. The influence of fundamental design parameters on ciliates community structure in Irish activated sludge systems. Eur. J. Protistol. 2011, 47, 274–286. [Google Scholar] [CrossRef]
- Fazelipour, M.; Takdastan, A.; Borghei, S.M. Biological removal of nutrients (N & P) from urban wastewater with a modified integrated fixed-film activated sludge-oxic settling anoxic system using an anoxic sludge holding tank. Water. Environ. J. 2021, 35, 830–846. [Google Scholar] [CrossRef]
- .Hamaidi-Chergui, F.; Zoubiri, A.F.; Debib, A.; Hamaidi, M.S.; Kais, H. Occurrence and removal of pathogens and a preliminary study of microfauna in the Medea sewage treatment plant. LARHYSS J. 2016, 26, 183–208. [Google Scholar]
- Klimiuk, E.; Łebkowska, M. Biotechnology in Environmental Protection; Wydawnictwo Naukowe PWN: Warsaw, Poland, 2003. [Google Scholar]
- Hu, X.; Sobotka, D.; Czerwionka, K.; Zhou, Q.; Xie, L.; Makinia, J. Effects of different external carbon sources and electron acceptors on interactions between denitrification and phosphorus removal in biological nutrient removal processes. J. Zhejiang Univ. Sci. B 2018, 19, 305–316. [Google Scholar] [CrossRef]
- Pastorelli, G.; Canziani, R.; Pedrazzi, L.; Rozzi, A. Phosphorus and nitrogen removal in moving-bed sequencing batch biofilm reactors. Water Sci. Technol. 1999, 40, 169–176. [Google Scholar] [CrossRef]
- Amaral, A.L.; Leal, C.S.; Vaz, A.I.; Vieira, J.C.; Quinteiro, A.C.; Costa, M.L.; Castro, L.M. Use of chemometric analyses to assess biological wastewater treatment plants by protozoa and metazoa monitoring. Environ. Monit. Assess. 2018, 190, 497. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Yang, F.; Zhou, F.; Xue, Y. Control of COD/N Ratio for Nutrient Removal in a Modified Membrane Bioreactor (MBR) Treating High Strength Wastewater. Bioresour. Technol. 2009, 100, 136–141. [Google Scholar] [CrossRef] [PubMed]
- Sobczyk, M.; Pajdak-Stós, A.; Fiałkowska, E.; Sobczyk, Ł.; Fyda, J. Multivariate analysis of activated sludge community in full-scale wastewater treatment plants. Environ. Sci. Pollut. Res. 2021, 28, 3579–3589. [Google Scholar] [CrossRef]



| Pollution Indicator | Research Methodology | Norm |
|---|---|---|
| BOD | Specific method | PN-EN 1899-1:2002 |
| COD | Specific method | PN-ISO 6060:2006 |
| TSS, MLTSS, MLVSS | General method | PN-EN 872:2002 |
| TN | Calculation method | - |
| TP and orthophosphates | Spectrophotometric method with ammonium molybdate | PN-EN ISO 6878:2006 |
| Day of Operation | MLVSS mg/dm3 | F/M mg BOD/mg MLVSS | F/M mg COD/mg MLVSS | COD/BOD mg BOD/mg COD | COD/N mg BOD/mg TN |
|---|---|---|---|---|---|
| 1 | 4293.36 | 0.257 | 0.796 | 3.093 | 11.809 |
| 16 | 3064.32 | 0.427 | No data | No data | No data |
| 33 | 2426.40 | 0.581 | 1.307 | 2.250 | 10.500 |
| 45 | 3543.84 | 0.490 | 1.023 | 2.087 | 13.091 |
| 59 | 4502.16 | 0.257 | 0.450 | 1.750 | 8.050 |
| 72 | 7185.60 | 0.116 | 0.332 | 2.876 | 8.627 |
| 88 | 4502.16 | 0.274 | 0.503 | 1.835 | 10.702 |
| 114 | 3682.80 | 0.287 | 0.580 | 2.019 | 8.742 |
| 148 | 5381.28 | 0.262 | 0.510 | 1.946 | 7.786 |
| 184 | 6677.28 | 0.121 | 0.287 | 2.375 | 8.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuśnierz, M.; Domańska, M.; Hamal, K.; Pera, A. Application of Integrated Fixed-Film Activated Sludge in a Conventional Wastewater Treatment Plant. Int. J. Environ. Res. Public Health 2022, 19, 5985. https://doi.org/10.3390/ijerph19105985
Kuśnierz M, Domańska M, Hamal K, Pera A. Application of Integrated Fixed-Film Activated Sludge in a Conventional Wastewater Treatment Plant. International Journal of Environmental Research and Public Health. 2022; 19(10):5985. https://doi.org/10.3390/ijerph19105985
Chicago/Turabian StyleKuśnierz, Magdalena, Magdalena Domańska, Kamila Hamal, and Agnieszka Pera. 2022. "Application of Integrated Fixed-Film Activated Sludge in a Conventional Wastewater Treatment Plant" International Journal of Environmental Research and Public Health 19, no. 10: 5985. https://doi.org/10.3390/ijerph19105985
APA StyleKuśnierz, M., Domańska, M., Hamal, K., & Pera, A. (2022). Application of Integrated Fixed-Film Activated Sludge in a Conventional Wastewater Treatment Plant. International Journal of Environmental Research and Public Health, 19(10), 5985. https://doi.org/10.3390/ijerph19105985








