Biocompatibility and Effectiveness of a Novel, Organic Olive Oil-Based Denture Adhesive: A Multicenter Randomized and Placebo-Controlled Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
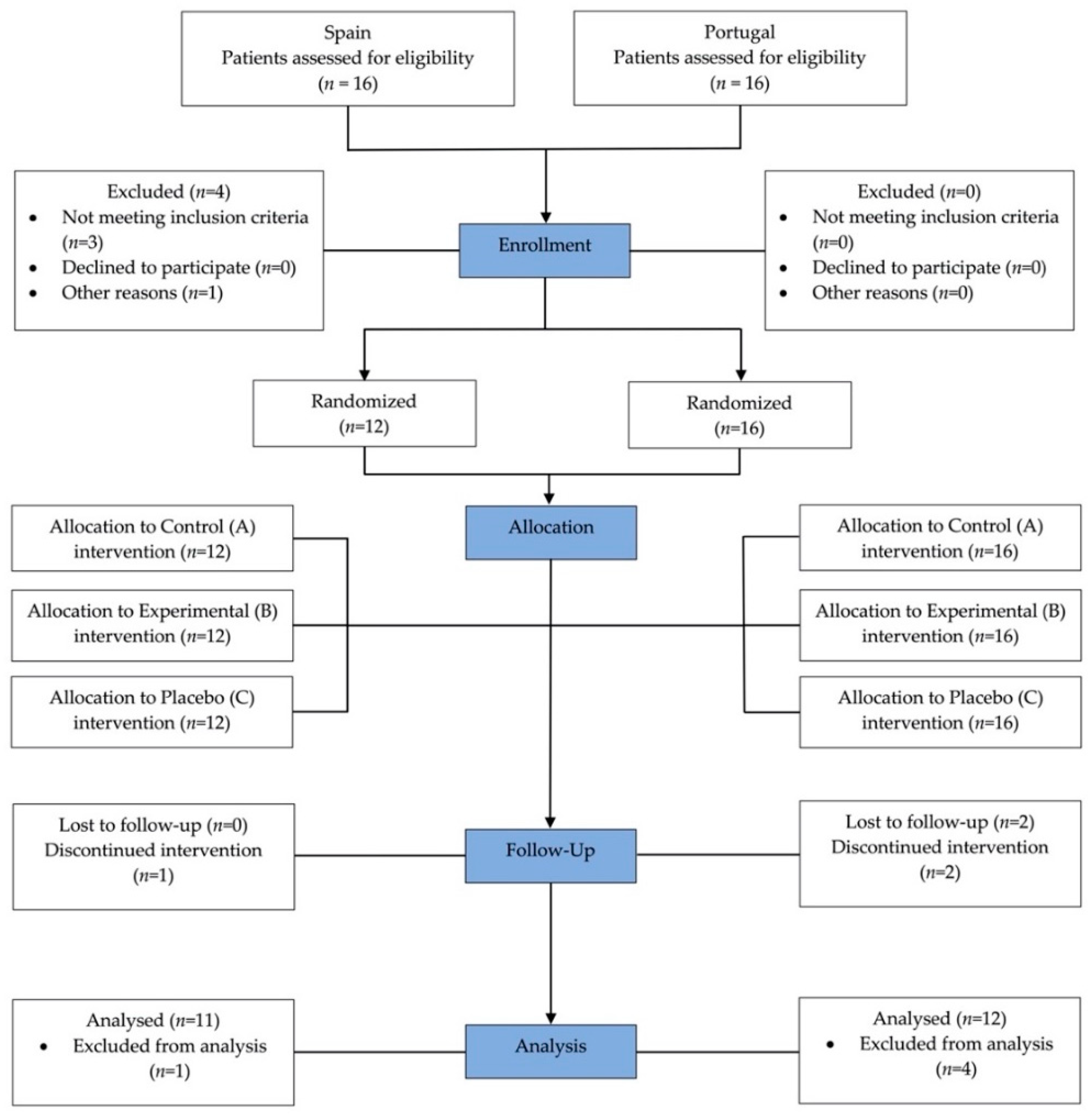
2.1. Study Design
2.2. Study Participants
2.3. Study Intervention
2.4. Experimental Design
2.5. Study Materials
- Control adhesive (A): conventional denture adhesive, (Kukident Pro®, Procter and Gamble, Cincinnati, OH, USA) cream form, composed of Polyvinyl Alcohol-Methyl acrylicate (PVM/MA) copolymers, liquid paraffin, sodium celluloses, petrol, colorings, preservatives, and aromatic particles.
- Experimental denture adhesive (B): novel adhesive, (OlivaFix® Gold, Bonyf AG, Lichtenstein) cream formulated with 30% extra virgin organic olive oil, with no zinc, mineral oil, or Vaseline.
- Placebo adhesive (C): Vaseline (Vaseline, Senti2®, Madrid, Spain).
2.6. Outcome Variables
2.7. Pilot Study–Inter- and Intra-Observer Reliability
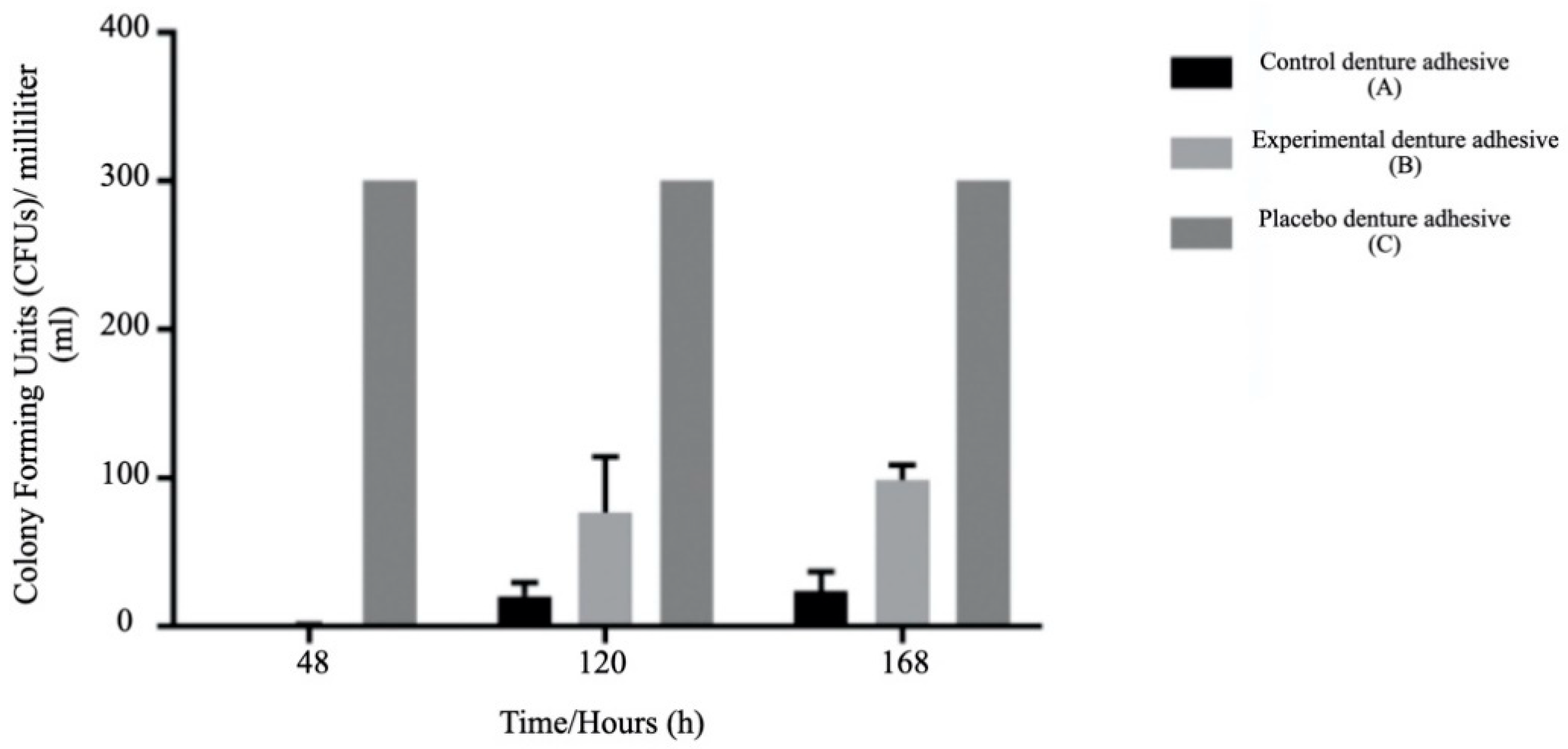
2.8. Effect of Denture Adhesives on Candida Albicans Growth
2.9. Statistically Data Analysis
3. Results
3.1. In Vivo Assessment
3.2. In Vitro Assessment
4. Discussion
5. Conclusions
- There were no differences in the force required to dislodge the denture under traction between the experimental and the control denture adhesives.
- Differences in individuals’ evaluation of the dentures’ retention and stability were not statistically significant among the three products.
- The experimental adhesive showed a better effectiveness time than the control and placebo.
- The control denture adhesive improved the ability to speak and chew, taste and odor, and ease of removal with significant differences.
- The experimental denture adhesive showed the best antimycotic effect against the growth of Candida albicans compared to the control and placebo.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Thalji, G.; McGraw, K.; Cooper, L.F. Maxillary Complete Denture Outcomes: A Systematic Review of Patient-Based Outcomes. Int. J. Oral Maxillofac. Implants 2016, 31, 169–181. [Google Scholar] [CrossRef] [PubMed]
- De Baat, C.; Van’t Hof, M.; Van Zeghbroeck, L.; Özcan, M.; Kalk, W. An international multicenter study on the effectiveness of a denture adhesive in maxillary dentures using disposable gnathometers. Clin. Oral Investig. 2007, 11, 237–243. [Google Scholar] [CrossRef][Green Version]
- De Oliveira Junior, N.M.; Rodriguez, L.S.; Marin, D.O.M.; Paleari, A.G.; Pero, A.C.; Compagnoni, M.A. Masticatory performance of complete denture wearers after using two adhesives: A crossover randomized clinical trial. J. Prosthet. Dent. 2014, 112, 1182–1187. [Google Scholar] [CrossRef]
- Coates, A.J. Usage of denture adhesives. J. Dent. 2000, 28, 137–140. [Google Scholar] [CrossRef]
- Uysal, H.; Altay, O.T.; Alparslan, N.; Bilge, A. Comparison of four different denture cushion adhesives-a subjective study. J. Oral Rehabil. 1998, 25, 209–213. [Google Scholar] [CrossRef] [PubMed]
- Shay, K. Denture hygiene: A review and update. J. Contemp. Dent Pract. 2000, 1, 1–8. [Google Scholar] [CrossRef]
- Tarbet, W.J.; Grossman, E. Observations of denture-supporting tissue during six months of denture adhesive wearing. J. Am. Dent Assoc. 1980, 101, 789–791. [Google Scholar] [CrossRef] [PubMed]
- Berg, E. A clinical comparison of four denture adhesives. Int. J. Prosthodont. 1991, 4, 449–456. [Google Scholar]
- Grasso, J.; Gay, T.; Rendell, J.; Baker, R.; Knippernberg, S.; Finkeldey, J.; Zou, X.; Winston, J.L. Effect of denture adhesive on retention of the mandibular and maxillary dentures during function. J. Clin. Dent. 2000, 11, 98–103. [Google Scholar]
- Pradies, G.; Sanz, I.; Evans, O.; Martinez, F.; Sanz, M. Clinical study comparing the efficacy of two denture adhesives in complete denture patients. Int. J. Prosthodont. 2009, 22, 361–367. [Google Scholar]
- Polyzois, G.; Lagouvardos, P.; Frangou, M.; Stefaniotis, T. Efficacy of denture adhesives in maxillary dentures using gnathodynamometry: A comparative study. Odontology 2011, 99, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Polyzois, G.L.; De Baat, C. Attitudes and usage of denture adhesives by complete denture wearers: A survey in Greece and the Netherlands. Gerodontology 2012, 29, e807–e814. [Google Scholar] [CrossRef]
- Koronis, S.; Pizatos, E.; Polyzois, G.; Lagouvardos, P. Clinical evaluation of three denture cushion adhesives by complete denture wearers. Gerodontology 2012, 29, e161–e169. [Google Scholar] [CrossRef]
- Goncalves, T.M.S.V.; Viu, F.C.; Goncalves, L.M.; Garcia, R.C.M.R. Denture adhesives improve mastication in denture wearers. Int. J. Prosthodont. 2014, 27, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Pachore, N.J.; Patel, J.R.; Sethuraman, R.; Naveen, Y.G. A comparative analysis of the effect of three types of denture adhesives on the retention of maxillary denture bases: An in vivo study. J. Indian Prosthodont. Soc. 2014, 14, 369–375. [Google Scholar] [CrossRef]
- Oliveira, M.C.; Oliveira, V.M.; Vieira, A.C.; Rambob, I. In vivo assessment of the effect of an adhesive for complete dentures on colonisation of Candida species. Gerodontology 2010, 27, 303–307. [Google Scholar] [CrossRef]
- Sampaio-Maia, B.; Figueiral, M.H.; Sousa-Rodrigues, P.; Fernandes, M.H.; Scully, C. The effect of denture adhesives on Candida albicans growth in vitro. Gerodontology 2012, 29, e348–e356. [Google Scholar] [CrossRef] [PubMed]
- Budtz-Jorgensen, E.; Stenderup, A.; Grabowski, M. An epidemiologic study of yeasts in elderly denture wearers. Commun. Dent. Oral Epidemiol. 1975, 3, 115–119. [Google Scholar] [CrossRef] [PubMed]
- Stafford, G.D.; Russell, C. Efficiency of denture adhesives and their possible influence on oral microorganisms. J. Dent. Res. 1971, 50, 832–836. [Google Scholar] [CrossRef]
- Makihira, S.; Nikawa, H.; Satonobu, S.V.; Jin, C.; Hamada, T. Growth of Candida species on commercial denture adhesives in vitro. Int. J. Prosthodont. 2001, 14, 48–52. [Google Scholar]
- Jimenez-Lopez, C.; Carpena, M.; Lourenço-Lopes, C.; Gallardo-Gomez, M.; Lorenzo, J.M.; Barba, F.J.; Prieto, M.A.; Simal-Gandara, J. Bioactive Compounds and Quality of Extra Virgin Olive Oil. Foods 2020, 9, 1014. [Google Scholar]
- Mota, A.H.; Silva, C.O.; Nicolai, M.; Baby, A.; Palma, L.; Rijo, P.; Ascensão, L.; Reis, C.P. Design and evaluation of novel topical formulation with olive oil as natural functional active. Pharm. Dev. Technol. 2018, 23, 794–805. [Google Scholar]
- Nardi, G.M.; Fais, S.; Casu, C.; Mazur, M.; Di Giorgio, R.; Grassi, R.; Grassi, F.R.; Orrù, G. Mouthwash Based on Ozonated Olive Oil in Caries Prevention: A Preliminary In-Vitro Study. Int. J. Environ. Res. Public Health 2020, 17, 9106. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, V.; Hasanoglu Erbasar, G.N.; Cigerim, L.; Altay Turgut, H.; Cerit, A. Effect of St. John’s wort oil and olive oil on the postoperative complications after third molar surgery: Randomized, double-blind clinical trial. Clin. Oral Investig. 2021, 25, 2429–2438. [Google Scholar] [CrossRef] [PubMed]
- Schulz, K.F.; Altman, D.G.; Moher, D.; Group, C. CONSORT 2010 statement: Updated guidelines for reporting parallel group randomized trials. BMC Med. 2010, 8, 18. [Google Scholar] [CrossRef] [PubMed]
- Maeda, Y.; Yang, T.-C.; Ikebe, K.; Andoh, T. Frequency of relining procedures during the maintenance period of removable prostheses: An experiential report. Int. J. Prosthodont. 2014, 27, 151–152. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ozcan, M.; Kulak, Y.; de Baat, C.; Arikan, A.; Ucankale, M. The effect of a new denture adhesive on bite force until denture dislodgement. J Prosthodont. 2005, 14, 122–126. [Google Scholar] [CrossRef] [PubMed]
- Rendell, J.K.; Gay, T.; Grasso, J.E.; Baker, R.A.; Winston, J.L. The effect of denture adhesive on mandibular movement during chewing. J. Am. Dent. Assoc. 2000, 131, 981–986. [Google Scholar] [CrossRef]
- Fujimori, T.; Hirano, S.; Hayakawa, I. Effects of a denture adhesive on masticatory functions for complete denture wearers—Consideration for the condition of denture-bearing tissues. J. Med. Dent. Sci. 2002, 49, 151–156. [Google Scholar]
- Fløystrand, F.; Koppang, R.; Williams, V.D.; Ørstavik, J. A method for testing denture adhesives. J. Prosthet. Dent. 1991, 66, 501–504. [Google Scholar] [CrossRef]
- Mañes, J.F.; Selva, E.J.; De-Barutell, A.; Bouazza, K. Comparison of the retention strengths of three complete denture adhesives: An in vivo study. Med. Oral Patol. Oral Cir. Bucal. 2011, 16, e132–e136. [Google Scholar] [CrossRef] [PubMed]
- Chung, K.-H.; Chung, C.-Y.; Cagna, D.R.; Cronin, R.J.J. Retention characteristics of attachment systems for implant overdentures. J. Prosthodont. 2004, 13, 221–226. [Google Scholar] [CrossRef]
- Bayer, S.; Steinheuser, D.; Gruner, M.; Keilig, L.; Enkling, N.; Stark, H.; Mues, S. Comparative study of four retentive anchor systems for implant supported overdentures—Retention force changes. Gerodontology 2009, 26, 268–272. [Google Scholar] [CrossRef] [PubMed]
- Mastrolorenzo, A.; Scozzafava, A.; Supuran, C.T. Antifungal activity of silver and zinc complexes of sulfadrug derivatives incorporating arylsulfonylureido moieties. Eur. J. Pharm. Sci. 2000, 11, 99–107. [Google Scholar] [CrossRef]
- Ververis, K.; Karagiannis, T.C. Mechanisms of action of phenolic compounds in olive. J. Diet Suppl. 2012, 9, 96–109. [Google Scholar]
- Dacrory, S.; Abou-Yousef, H.; Abouzeid, R.E.; Kamel, S.; Abdel-Aziz, M.S.; El-Badry, M. Antimicrobial cellulosic hydrogel from olive oil industrial residue. Int. J. Biol. Macromol. 2018, 117, 179–188. [Google Scholar] [CrossRef]
- Souza, J.G.S.; Bertolini, M.; Thompson, A.; Barão, V.A.R.; Dongari-Bagtzoglou, A. Biofilm Interactions of Candida albicans and Mitis Group Streptococci in a Titanium-Mucosal Interface Model. Appl. Environ. Microbiol. 2020, 86, e02950-19. [Google Scholar] [CrossRef] [PubMed]
| Participant-Centered Outcomes Concerning Denture Adhesives | Speaking Improvement | Chewing Ability | Satisfaction Degree | Taste | Effectiveness Time | Opinion on Utility | Cleaning/Removal | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Yes | No | Yes | No | No | Little | Sufficient | Very | Yes | No | Less than 4 h | 4–8 h | 8–12 h | More than 12 h | Zero | Little | Effective | Easy | Difficult | |
| Control denture adhesive (A) | 78.26% (18) | 21.74% (5) | 69.57% (16) | 30.43% (7) | 13.04% (3) | 21.74% (5) | 56.52% (13) | 13.04% (3) | 34.78% (8) | 39.13% (9) | 60.87% (14) | 8.70% (2) | 47.83% (11) | 34.78% (8) | 8.70% (2) | 13.04% (3) | 21.74% (5) | 73.91% (17) | 26.09% (6) |
| Experimental denture adhesive (B) | 65.22% (15) | 34.78% (8) | 56.52% (13) | 43.48% (10) | 8.70% (2) | 47.83% (11) | 39.13% (9) | 8.70% (2) | 91.30% (21) | 56.52% (13) | 43.48% (10) | 8.70% (2) | 17.39% (4) | 73.91% (17) | 0.00% (0) | 8.70% (2) | 47.83% (11) | 39.13% (9) | 65.22% (15) |
| Placebo denture adhesive (C) | 26.98% (6) | 73.91% (17) | 21.74% (5) | 78.26% (18) | 21.74% (5) | 52.17% (12) | 26.09% (6) | 21.74% (5) | 60.87% (14) | 39.13% (9) | 39.13% (9) | 78.26% (18) | 13.04% (3) | 8.70% (2) | 0.00% (0) | 21.74% (5) | 52.17% (12) | 26.09% (6) | 8.70% (2) |
| Participant-Centered Outcomes Concerning Denture Adhesives | Speaking Improvement | Chewing Ability | Satisfaction Degree | Taste | Effectiveness Time | Cleaning/Removal |
|---|---|---|---|---|---|---|
| p | p | p | p | p | p | |
| Control denture adhesive (A) vs placebo denture adhesive (C) | 0.004 | 0.001 | 0.024 | 0.009 | 0.001 | 0.01 |
| Control denture adhesive (A) vs experimental denture adhesive (B) | 0.003 | 0.001 | 0.073 | 0.027 | 0.001 | 0.026 |
| Experimental denture adhesive (B) vs placebo denture adhesive (C) | 0.004 | 0.001 | 0.09 | 0.365 | 0.001 | 0.037 |
| Groups | Dynamometer | Gnathometer | ||
|---|---|---|---|---|
| Mean ± SD (g) | p-Value | Mean ± SD | p-Value | |
| No denture adhesive | 123.8 ± 38.3 | vs (B)—0.034 vs (A)—0.041 vs (C)—1.000 | 0.8 ± 0.6 | 0.055 |
| Control denture adhesive (A) | 152.7 ± 52.6 | vs initial—0.041 vs (B)—0.995 vs (C)—0.048 | 1.1 ± 0.6 | |
| Experimental denture adhesive (B) | 155.8 ± 51.5 | vs initial—0.034 vs (A)—0.995 vs (C)—0.047 | 1 ± 0.6 | |
| Placebo denture adhesive (C) | 122.7 ± 36.0 | vs initial—1.000 vs (B)—0.047 vs (A)—0.048 | 0.8 ± 0.5 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azevedo, L.; Correia, A.; Almeida, C.F.; Molinero-Mourelle, P.; Correia, M.; Del Río Highsmith, J. Biocompatibility and Effectiveness of a Novel, Organic Olive Oil-Based Denture Adhesive: A Multicenter Randomized and Placebo-Controlled Clinical Trial. Int. J. Environ. Res. Public Health 2021, 18, 3398. https://doi.org/10.3390/ijerph18073398
Azevedo L, Correia A, Almeida CF, Molinero-Mourelle P, Correia M, Del Río Highsmith J. Biocompatibility and Effectiveness of a Novel, Organic Olive Oil-Based Denture Adhesive: A Multicenter Randomized and Placebo-Controlled Clinical Trial. International Journal of Environmental Research and Public Health. 2021; 18(7):3398. https://doi.org/10.3390/ijerph18073398
Chicago/Turabian StyleAzevedo, Luís, André Correia, Carlos F. Almeida, Pedro Molinero-Mourelle, Maria Correia, and Jaime Del Río Highsmith. 2021. "Biocompatibility and Effectiveness of a Novel, Organic Olive Oil-Based Denture Adhesive: A Multicenter Randomized and Placebo-Controlled Clinical Trial" International Journal of Environmental Research and Public Health 18, no. 7: 3398. https://doi.org/10.3390/ijerph18073398
APA StyleAzevedo, L., Correia, A., Almeida, C. F., Molinero-Mourelle, P., Correia, M., & Del Río Highsmith, J. (2021). Biocompatibility and Effectiveness of a Novel, Organic Olive Oil-Based Denture Adhesive: A Multicenter Randomized and Placebo-Controlled Clinical Trial. International Journal of Environmental Research and Public Health, 18(7), 3398. https://doi.org/10.3390/ijerph18073398







