Nature Exposure and Its Effects on Immune System Functioning: A Systematic Review
Abstract
1. Introduction
1.1. Nature Exposure and Immune System Functioning
1.2. Immunoregulation through Biodiversity
1.3. Immunoregulation through Inhalation of Air-Borne, Volatile Substances
1.4. Health-Promoting Ecosystem Services and Their Effects on the Immune System
2. Methodology
2.1. Search Strategy
2.2. Study Selection
2.3. Quality Assessment
3. Results
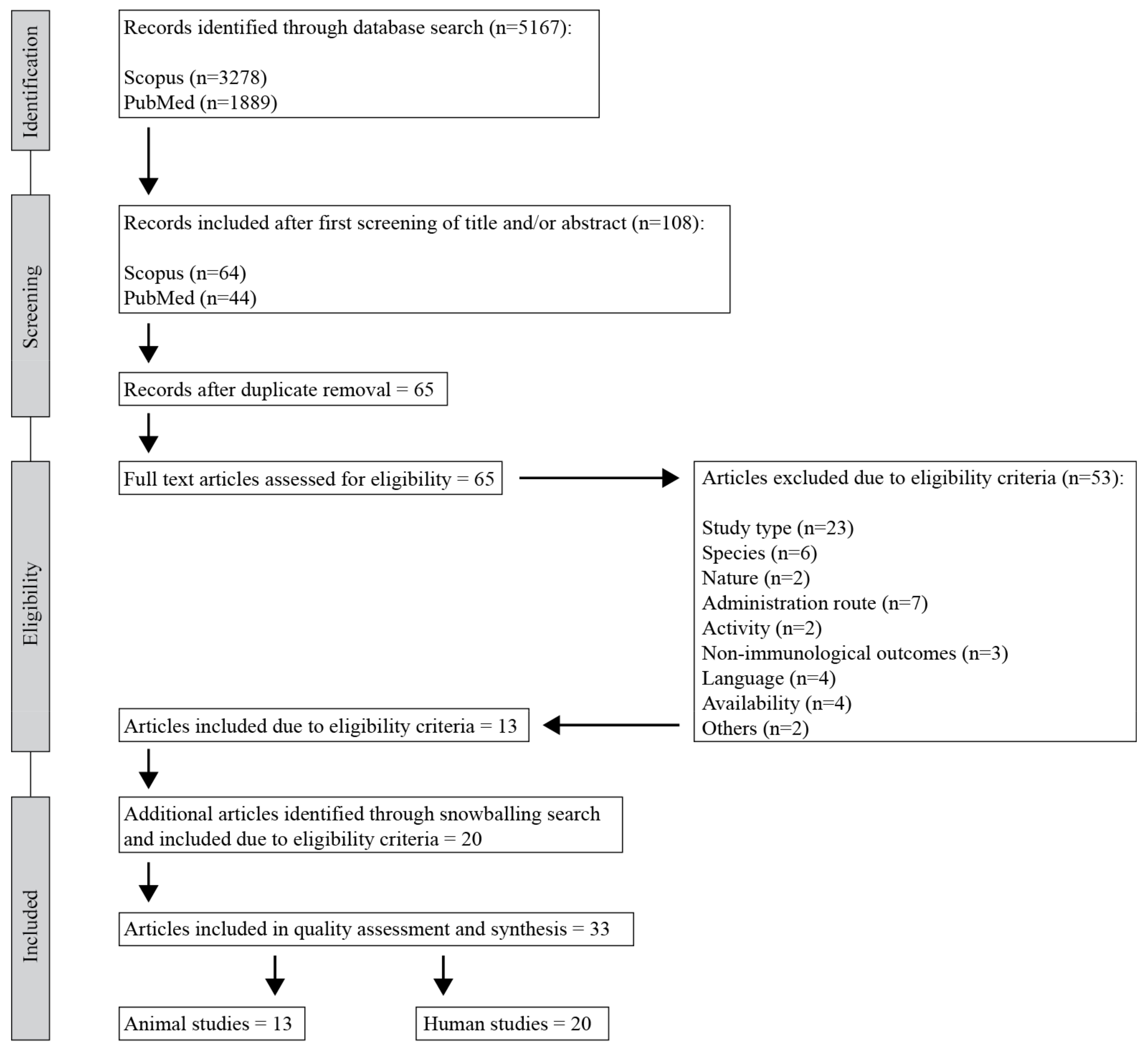
3.1. Article Selection
3.2. Characteristics of Included Studies
3.2.1. Characteristics of Human Studies
| Main Author | Year | Country | Study Design | Sample Size (Intervention/Control) | Sample Characteristics | Sample Age | Sample Sex | Intervention | Control | Duration |
|---|---|---|---|---|---|---|---|---|---|---|
| Forest bathing | ||||||||||
| Han et al. [45] | 2016 | South Korea | Pre-post (2 groups) CCT | 61 (33/28) | Adults with chronic pain | 25–49 | Mixed | Forest bathing (pine, oak maple forest) | Normal daily routine | 2 days |
| Im et al. [44] | 2016 | South Korea | Pre-post (2 groups crossover) RCT | 41 | Healthy students | 18–35 | Mixed | Forest environment (pine tree forest) | Urban environment | 2 h |
| Jia et al. [46] | 2016 | China | Pre-post (2 groups) CCT | 18 (10/8) | COPD patients | 61–79 | Mixed | Forest bathing | Urban stay | 3 days |
| Kim et al. [52] | 2015 | South Korea | Pre-post (1 group) | 11 | Adults with breast cancer | 25–60 | Female | Forest therapy | / | 14 days |
| Li et al. [53] | 2007 | Japan | Pre-post (1 group) | 12 | Healthy adults (office workers) | 37–55 | Male | Forest bathing | / | 3 days (2–4 h/day) |
| Li et al. [55] | 2008a | Japan | Pre-post (1 group) | 13 | Healthy adults (nurses) | 25–43 | Female | Forest bathing | / | 3 days (2–4 h/day) |
| Li et al. [54] | 2008b | Japan | Pre-post (1 group crossover) | 12 | Healthy adults | 35–56 | Male | Forest bathing | Urban stay | 3 days (2–4 h/day) |
| Lyu et al. [47] | 2019 | China | Pre-post (2 groups) CCT | 60 (45/15) | Healthy adults | 19–24 | Male | Forest bathing (bamboo forest) | Urban stay | 3 days |
| Mao et al. [50] | 2012a | China | Pre-post (2 groups) CCT | 20 (10/10) | Healthy students | 20–21 | Male | Forest bathing (broad-leaved forest) | Urban stay | 2 days |
| Mao et al. [51] | 2012b | China | Pre-post (2 groups) CCT | 24 (12/12) | Elderly patients with hypertension | 60–75 | Mixed | Forest bathing (broad-leaved forest) | Urban stay | 7 days |
| Mao et al. [49] | 2017 | China | Pre-post (2 groups) CCT | 33 (23/10) | Elderly patients with chronic heart failure | 66–79 | Mixed | Forest bathing (broad-leaved forest) | Urban stay | 4 days |
| Mao et al. [48] | 2018 | China | Pre-post (2 groups) CCT | 20 (10/10) | Elderly patients with chronic heart failure | 66–79 | Mixed | Second forest bathing trip (broad-leaved forest) after 4 weeks break | Urban stay after previous forest bathing trip 4 weeks ago | 4 days |
| Seo et al. [56] | 2015 | South Korea | Pre-post (1 group) | 21 | Children with asthma | 7–12 | Mixed | Forest bathing (fir tree forest) | / | 4 days |
| Seo et al. [56] | 2015 | South Korea | Pre-post (1 group) | 27 | Children with atopic dermatitis | 7–12 | Mixed | Forest bathing (fir tree forest) | / | 4 days |
| Tsao et al. [57] | 2018 | Taiwan | Retrospective study (pre-post 2 groups) | 200 (90/110) | Healthy adults | 34–56 | Mixed | Forest workers | Urban residents | 1 year |
| Tsao et al. [57] | 2018 | Taiwan | Pre-post (1 group) | 11 | Healthy adults | / | / | Forest bathing (coniferous forest) | / | 5 days |
| BVOC inhalation | ||||||||||
| Li et al. [58] | 2009 | Japan | Pre-post (1 group) | 12 | Healthy adults | 37–60 | Male | Inhalation of phytoncides (vaporized hinoki cypress stem oil) in urban hotel room | / | 3 days |
| Fragrance inhalation | ||||||||||
| Kiecolt-Glaser et al. [59] | 2008 | Ohio, USA | Pre-post (1 group crossover) | 56 | Healthy adults | 18–43 | Mixed | Inhalation of fragrances (lavender, lemon) | Inhalation of water vapour | 1.25 h |
| Komori et al. [60] | 1995 | Japan | Pre-post (2 groups) | 20 (12/8) | Adults with depression | 26–53 | Male | Inhalation of citrus fragrance mix (limonene, citral, other EOs) | only positive control group | 4–11 weeks |
| Trellakis et al. [61] | 2012 | Germany | Pre-post (1 group crossover) | 32 | Healthy adults | 20–45 | Mixed | Inhalation of stimulant fragrances (grapefruit, fennel, pepper) | No fragrance exposure | 3 days (30 min/day) |
| Inhalation of relaxant fragrances (lavender, patchouli, rose) | ||||||||||
| Waterfall exposure | ||||||||||
| Gaisberger et al. [35] | 2012 | Austria | Pre-post (2 groups) CCT | 54 (27/27) | Children with allergic asthma | 8–15 | Mixed | Waterfall exposure (WF+) in national park | No waterfall exposure (WF-) in national park | 3 weeks (1 h/day) |
| Grafetstätter et al. [34] | 2017 | Austria | Pre-post (3 groups) CCT | 91 (33/32/26) | Adults with stress (Pre-treated with oral cholera vaccination) | 19–61 | Mixed | Hiking in national park with waterfall exposure (WF+) | Hiking in national park without waterfall exposure (WF-) | 1 week (1 h/day) |
3.2.2. Characteristics of Animal Studies
| Main Author | Year | Country | Study Design | Sample Size (Number per Group) | Sample Characteristics | Sample Age | Sample Sex | Intervention | Control | Duration |
|---|---|---|---|---|---|---|---|---|---|---|
| BVOC inhalation | ||||||||||
| Ahn et al. [62] | 2018a | South Korea | Animal | 35 (7) | Mice Pre-treated with LPS | 7 weeks | Male | Housing with BVOC wood panels (C. obtusa, P. densiflora) LPS | Housing without wood panels LPS | 4 weeks |
| Ahn et al. [63] | 2018b | South Korea | Animal | 49 (7) | Mice Pre-treated with OVA | 5 weeks | / | Housing with BVOC wood panels (C. obtusa, P. densiflora, P. koraiensis, L.kaempferi) OVA | Housing without wood panels OVA | 27 days |
| Yang et al. [64] | 2015 | South Korea | Animal | / | Mice Dinitrochlorbenzene (DNCB)-induced atopic dermatitis (AD)-like disease model | 7 weeks | / | Exposure to BVOC (C. obtusa) | Exposure to vehicle | 8 weeks |
| Eucalyptol inhalation | ||||||||||
| Bastos et al. [74] | 2011 | Brazil | Animal | ca. 35 (7–10) | Guineau pigs Pre-treated with OVA | / | Male | Eucalyptol (1,8-cineol) inhalation OVA | Saline inhalation OVA | 15 min |
| Kennedy-Feitosa et al. [65] | 2019 | Brazil | Animal | 40 (10) | Mice Pre-exposed to cigarette smoke (CS) | Male | Eucalyptol (1,8-cineol) inhalation CS | Vehicle inhalation CS | 120 days (15 min/day) | |
| Lee et al. [66] | 2016 | South Korea | Animal | / | Mice Pre-sensitised to Der p (house dust mite allergen; HDM) | 6 weeks | Female | Eucalyptol (1,8-cineol) inhalation Der p | Vehicle inhalation Der p | / |
| Limonene inhalation | ||||||||||
| Bibi et al. [67] | 2015 | Israel | Animal | 30 (10) | Mice Pre-treated with OVA | 8 weeks | Female | Housing with Limonene-treated wood bedding OVA | Housing with untreated wood bedding OVA | 30 days |
| Hirota et al. [68] | 2012 | Japan | Animal | 30 (10) | Mice Pre-sensitised to Der f (house dust mite allergen; HDM) | 6 weeks | Male | Limonene inhalation Der f | No inhalation Der f | 31 days |
| Keinan et al. [73] | 2005 | Israel | Animal | 40 (10) | Rats Pre-treated with OVA | 4 weeks | / | Limonene inhalation (ozone scavenger) Eucalyptol inhalation (inert to ozone) OVA | No inhalation OVA | 1 week |
| Limonene/ozone inhalation | ||||||||||
| Hansen et al. [69] | 2013 | Denmark | Animal | ca. 40 (9–10) | Mice Pre-treated with OVA | 5-6 weeks | Female | Limonene inhalation Limonene + ozone inhalation OVA | No inhalation Ozone inhalation OVA | 14 weeks |
| Hansen et al. [70] | 2016 | Denmark | Animal | 40 (10) | Mice Pre-treated with OVA | 6 weeks | Female | Limonene inhalation | Air inhalation | 3 days (60 min/day) |
| Linalool inhalation | ||||||||||
| Naka-mura et al. [72] | 2009 | Japan | Animal | 12 (4) | Rats Stressed by restraining in tube | 7–8 weeks | Male | Linalool inhalation Stress | No inhalation Stress | 2 h |
| Fragrance inhalation | ||||||||||
| Fujiwara et al. [71] | 1998 | Japan | Animal | 84 (12) | Mice Stressed with high pressure | 8–10 weeks | Male | Fragrance exposure (lemon, oak moss, labdanum, tuberose) Stress | No fragrance exposure Stress | 24 h |
3.3. Quality Assessment
3.3.1. Quality Assessment of Human Studies
3.3.2. Quality Assessment of Animal Studies
3.4. Outcomes and Synthesis
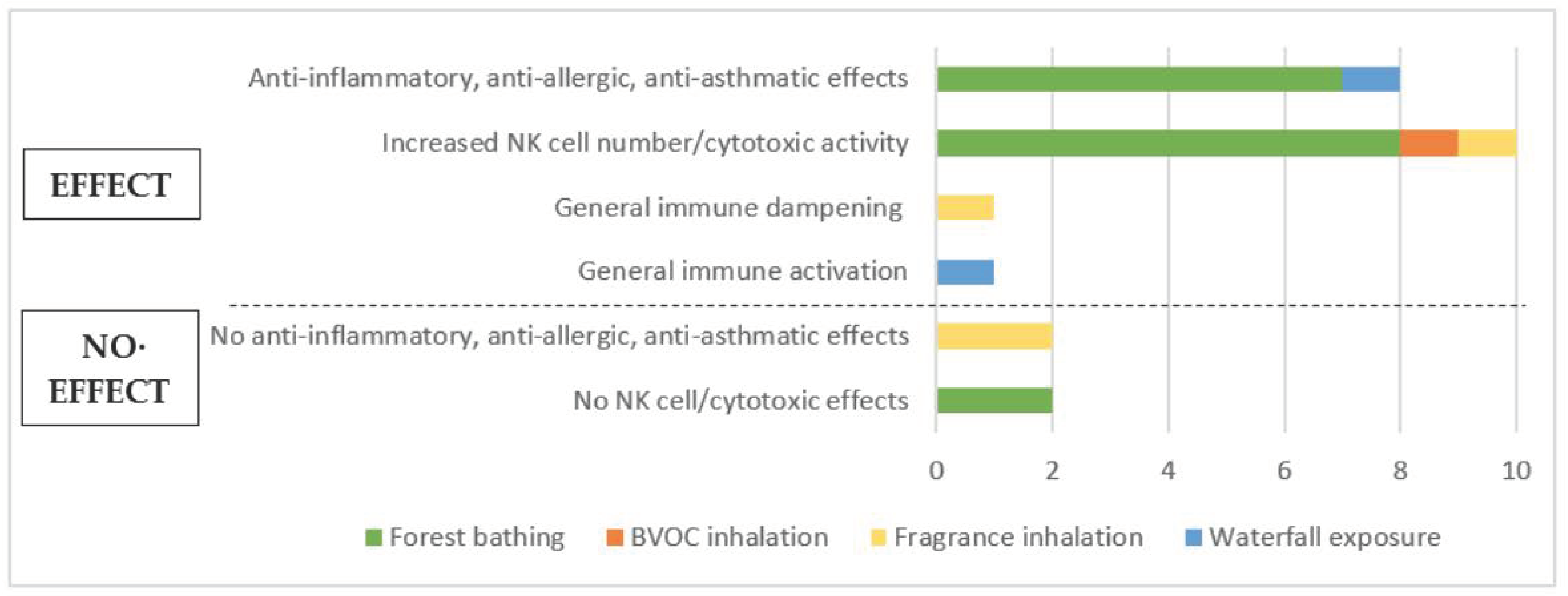
3.4.1. Outcomes of Human Studies
3.4.2. Synthesis of Human Studies
3.5. Outcomes and Synthesis of Animal Studies
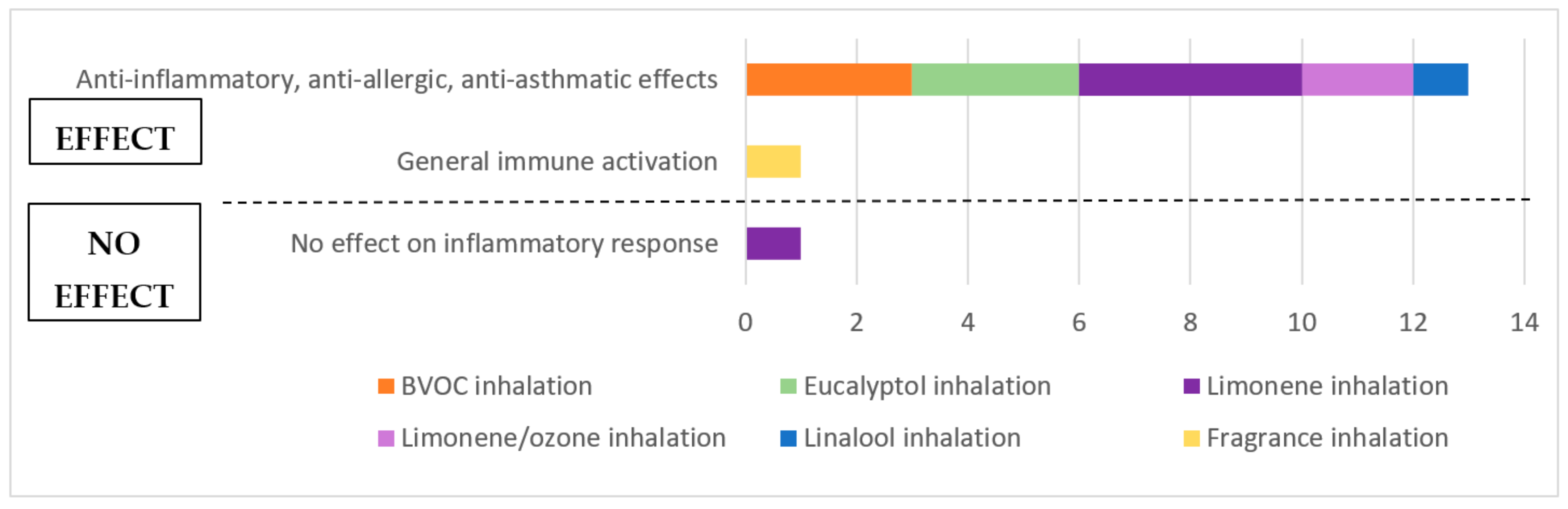
3.5.1. Outcomes of Animal Studies
3.5.2. Synthesis of Animal Studies
4. Limitations of the Review
5. Discussion
5.1. A Baseline for Future Research
5.2. Study Shortcomings and Recommendations for Future Study Designs
5.3. What Is the Optimal Type, Length, Season and Dosage of Nature Exposure?
5.4. Understanding Biomedical Mechanisms
5.5. Nature-Based Clinical Applications
6. Outlook
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Whitmee, S.; Haines, A.; Beyrer, C.; Boltz, F.; Capon, A.G.; De Souza Dias, B.F.D.S.; Ezeh, A.; Frumkin, H.; Gong, P.; Head, P.; et al. Safeguarding human health in the Anthropocene epoch: Report of The Rockefeller Foundation–Lancet Commission on planetary health. Lancet 2015, 386, 1973–2028. [Google Scholar] [CrossRef]
- Myers, S.S. Planetary health: Protecting human health on a rapidly changing planet. Lancet 2017, 390, 2860–2868. [Google Scholar] [CrossRef]
- United Nations Departement of Economic and Social Affairs. Population Division World Urbanization Prospects: The 2018 Revision; UN DESA: New York, NY, USA, 2018. [Google Scholar]
- Frumkin, H.; Bratman, G.N.; Breslow, S.J.; Cochran, B., Jr.; Khan, P.H.; Lawler, J.J.; Levin, P.S.; Tandon, P.S.; Varanasi, U.; Wolf, K.L.; et al. Nature Contact and Human Health: A Research Agenda. Environ. Health Perspect. 2017, 125, 075001. [Google Scholar] [CrossRef] [PubMed]
- Lauesen, L.M. Natural Environment BT. In Encyclopedia of Corporate Social Responsibility; Idowu, S.O., Capaldi, N., Zu, L., Das Gupta, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1734–1742. ISBN 978-3-642-28036-8. [Google Scholar]
- Mitten, D.; Brymer, E. Outdoor and Environmental Education: Nature and Wellbeing. In Encyclopedia of Teacher Education; Springer: Singapore, 2019; pp. 1–7. [Google Scholar] [CrossRef]
- WHO. WHO Guidelines on Physical Activity and Sedentary Behaviour; WHO: Geneva, Switzerland, 2020; ISBN 9789241550536. [Google Scholar]
- European Environment Agency. Healthy Environment, Healthy Lives: How the Environment Influences Health and Well-being in Europe; EEA: Copenhagen, Denmark, 2020; ISBN 9789294802125. [Google Scholar]
- Landrigan, P.J.; Fuller, R.; Acosta, N.J.R.; Adeyi, O.; Arnold, R.; Basu, N.; Baldé, A.B.; Bertollini, R.; Bose-O’Reilly, S.; Boufford, J.I.; et al. The Lancet Commission on pollution and health. Lancet 2018, 391, 462–512. [Google Scholar] [CrossRef]
- Watts, N.; Amann, M.; Ayeb-Karlsson, S.; Belesova, K.; Bouley, T.; Boykoff, M.; Byass, P.; Cai, W.; Campbell-Lendrum, D.; Chambers, J.; et al. The Lancet Countdown on health and climate change: From 25 years of inaction to a global transformation for public health. Lancet 2018, 391, 581–630. [Google Scholar] [CrossRef]
- Frumkin, H.; Haines, A. Global Environmental Change and Noncommunicable Disease Risks. Annu. Rev. Public Health 2019, 40, 261–282. [Google Scholar] [CrossRef]
- World Health Organization. Preventing noncommunicable diseases (NCDs) by reducing environmental risk factors. 2017. [Google Scholar]
- Fong, K.C.; Hart, J.E.; James, P. A Review of Epidemiologic Studies on Greenness and Health: Updated Literature Through 2017. Curr. Environ. Health Rep. 2018, 5, 77–87. [Google Scholar] [CrossRef]
- Corazon, S.S.; Sidenius, U.; Poulsen, D.V.; Gramkow, M.C.; Stigsdotter, U.K. Psycho-Physiological Stress Recovery in Outdoor Nature-Based Interventions: A Systematic Review of the Past Eight Years of Research. Int. J. Environ. Res. Public Health 2019, 16, 1711. [Google Scholar] [CrossRef]
- Hartig, T.; Mitchell, R.; De Vries, S.; Frumkin, H. Nature and Health. Annu. Rev. Public Health 2014, 35, 207–228. [Google Scholar] [CrossRef]
- Kuo, M. How might contact with nature promote human health? Promising mechanisms and a possible central pathway. Front. Psychol. 2015, 6, 1093. [Google Scholar] [CrossRef]
- Haluza, D.; Schönbauer, R.; Cervinka, R. Green Perspectives for Public Health: A Narrative Review on the Physiological Effects of Experiencing Outdoor Nature. Int. J. Environ. Res. Public Health 2014, 11, 5445–5461. [Google Scholar] [CrossRef] [PubMed]
- McMahan, E.A.; Estes, D. The effect of contact with natural environments on positive and negative affect: A meta-analysis. J. Posit. Psychol. 2015, 10, 507–519. [Google Scholar] [CrossRef]
- Twohig-Bennett, C.; Jones, A. The health benefits of the great outdoors: A systematic review and meta-analysis of greenspace exposure and health outcomes. Environ. Res. 2018, 166, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Craig, J.; Logan, A.C.; Prescott, S.L. Natural environments, nature relatedness and the ecological theater: Connecting satellites and sequencing to shinrin-yoku. J. Physiol. Anthr. 2016, 35, 1. [Google Scholar] [CrossRef]
- Kim, T.; Song, B.; Cho, K.S.; Lee, I. Therapeutic Potential of Volatile Terpenes and Terpenoids from Forests for Inflammatory Diseases. Int. J. Mol. Sci. 2020, 21, 2187. [Google Scholar] [CrossRef]
- Peterfalvi, A.; Miko, E.; Nagy, T.; Reger, B.; Simon, D.; Miseta, A.; Czéh, B.; Szereday, L. Much More Than a Pleasant Scent: A Review on Essential Oils Supporting the Immune System. Molecules 2019, 24, 4530. [Google Scholar] [CrossRef]
- Janeway, C.J.; Travers, P.; Walport, M. Immunobiology: The Immune System in Health and Disease, 5th ed.; Garland Science: New York, NY, USA, 2001; ISBN 0-8153-3642-X. [Google Scholar]
- Rook, G. Regulation of the immune system by biodiversity from the natural environment: An ecosystem service essential to health. Proc. Natl. Acad. Sci. USA 2013, 110, 18360–18367. [Google Scholar] [CrossRef]
- Aerts, R.; Honnay, O.; Van Nieuwenhuyse, A. Biodiversity and human health: Mechanisms and evidence of the positive health effects of diversity in nature and green spaces. Br. Med Bull. 2018, 127, 5–22. [Google Scholar] [CrossRef]
- World Health Organisation. Connecting Global Priorities: Biodiversity and Human Health. A State of Knowledge Review; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Hanski, I.; Von Hertzen, L.; Fyhrquist, N.; Koskinen, K.; Torppa, K.; Laatikainen, T.; Karisola, P.; Auvinen, P.; Paulin, L.; Mäkelä, M.J.; et al. Environmental biodiversity, human microbiota, and allergy are interrelated. Proc. Natl. Acad. Sci. USA 2012, 109, 8334–8339. [Google Scholar] [CrossRef]
- Ruokolainen, L.; Von Hertzen, L.C.; Fyhrquist, N.; Laatikainen, T.J.; Lehtomäki, J.; Auvinen, P.; Karvonen, A.M.; Hyvärinen, A.M.; Tillmann, V.; Niemelä, O.; et al. Green areas around homes reduce atopic sensitization in children. Allergy 2015, 70, 195–202. [Google Scholar] [CrossRef]
- Von Hertzen, L.; Hanski, I.; Haahtela, T. Natural immunity. Biodiversity loss and inflammatory diseases are two global megatrends that might be related. EMBO Rep. 2011, 12, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Laothawornkitkul, J.; Taylor, J.E.; Paul, N.D.; Hewitt, N. Biogenic volatile organic compounds in the Earth system: Tansley review. New Phytol. 2009, 183, 27–51. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Tang, J.; Yu, X. Environmental and physiological controls on diurnal and seasonal patterns of biogenic volatile organic compound emissions from five dominant woody species under field conditions. Environ. Pollut. 2020, 259, 113955. [Google Scholar] [CrossRef] [PubMed]
- Quintans, J.S.; Shanmugan, S.; Heimfarth, L.; Araújo, A.A.S.; Almeida, J.R.G.d.S.; Picot, L.; Quintans-Júnior, L.J. Monoterpenes modulating cytokines—A review. Food Chem. Toxicol. 2019, 123, 233–257. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.S.; Lim, Y.-R.; Lee, K.; Lee, J.; Lee, J.H.; Lee, I. Terpenes from Forests and Human Health. Toxicol. Res. 2017, 33, 97–106. [Google Scholar] [CrossRef]
- Grafetstätter, C.; Gaisberger, M.; Freidl, J.; Ritter, M.; Kolarž, P.; Pichler, C.; Thalhamer, J.; Hartl, A. Does waterfall aerosol influence mucosal immunity and chronic stress? A randomized controlled clinical trial. J. Physiol. Anthr. 2017, 36, 10. [Google Scholar] [CrossRef]
- Gaisberger, M.; Šanović, R.; Dobias, H.; Kolarž, P.; Moder, A.; Thalhamer, J.; Selimović, A.; Huttegger, I.; Ritter, M.; Hartl, A. Effects of Ionized Waterfall Aerosol on Pediatric Allergic Asthma. J. Asthma 2012, 49, 830–838. [Google Scholar] [CrossRef]
- Coutts, C.; Hahn, M. Green Infrastructure, Ecosystem Services, and Human Health. Int. J. Environ. Res. Public Health 2015, 12, 9768–9798. [Google Scholar] [CrossRef]
- World Health Organisation. Evolution of WHO Air Quality Guidelines: Past, Present and Future; WHO Regional Office for Europe: Copenhagen, Denmark, 2017. [Google Scholar]
- Wu, K.; Yang, X.; Chen, D.; Gu, S.; Lu, Y.; Jiang, Q.; Wang, K.; Ou, Y.; Qian, Y.; Shao, P.; et al. Estimation of biogenic VOC emissions and their corresponding impact on ozone and secondary organic aerosol formation in China. Atm. Res. 2020, 231, 104656. [Google Scholar] [CrossRef]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; PRISMA-P Group. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef]
- Oh, B.; Lee, K.J.; Zaslawski, C.; Yeung, A.; Rosenthal, D.; Larkey, L.; Back, M. Health and well-being benefits of spending time in forests: Systematic review. Environ. Health Prev. Med. 2017, 22, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Effective Public Health Practice Project. Quality Assessment Tool for Quantitative Studies; Effective Public Health Practice Project: Hamilton, ON, USA, 1998. [Google Scholar]
- Kilkenny, C.; Browne, W.J.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Improving Bioscience Research Reporting: The ARRIVE Guidelines for Reporting Animal Research. PLoS Biol. 2010, 8, e1000412. [Google Scholar] [CrossRef] [PubMed]
- Hooijmans, C.R.; Rovers, M.M.; De Vries, R.B.M.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef]
- Im, S.G.; Choi, H.; Jeon, Y.-H.; Song, M.-K.; Kim, W.; Woo, J.-M. Comparison of Effect of Two-Hour Exposure to Forest and Urban Environments on Cytokine, Anti-Oxidant, and Stress Levels in Young Adults. Int. J. Environ. Res. Public Health 2016, 13, 625. [Google Scholar] [CrossRef]
- Han, J.-W.; Choi, H.; Jeon, Y.-H.; Yoon, C.-H.; Woo, J.-M.; Kim, W. The Effects of Forest Therapy on Coping with Chronic Widespread Pain: Physiological and Psychological Differences between Participants in a Forest Therapy Program and a Control Group. Int. J. Environ. Res. Public Health 2016, 13, 255. [Google Scholar] [CrossRef] [PubMed]
- Jia, B.B.; Yang, Z.X.; Mao, G.X.; Lyu, Y.D.; Wen, X.L.; Xu, W.H.; Lyu, X.L.; Cao, Y.B.; Wang, G.F. Health Effect of Forest Bathing Trip on Elderly Patients with Chronic Obstructive Pulmonary Disease. Biomed. Environ. Sci. 2016, 29, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Lvu, B.; Zeng, C.; Xie, S.; Li, D.; Lin, W.; Li, N.; Jiang, M.; Liu, S.; Chen, Q. Benefits of A Three-Day Bamboo Forest Therapy Session on the Psychophysiology and Immune System Responses of Male College Students. Int. J. Environ. Res. Public Health 2019, 16, 4991. [Google Scholar] [CrossRef]
- Mao, G.X.; Cao, Y.B.; Yang, Y.; Chen, Z.M.; Dong, J.H.; Chen, S.S.; Wu, Q.; Lyu, X.L.; Jia, B.B.; Yan, J.; et al. Additive Benefits of Twice Forest Bathing Trips in Elderly Patients with Chronic Heart Failure. Biomed. Environ. Sci 2018, 31, 159–162. [Google Scholar] [CrossRef]
- Mao, G.; Cao, Y.; Wang, B.; Wang, S.; Chen, Z.; Wang, J.; Xing, W.; Ren, X.; Lv, X.; Dong, J.; et al. The Salutary Influence of Forest Bathing on Elderly Patients with Chronic Heart Failure. Int. J. Environ. Res. Public Health 2017, 14, 368. [Google Scholar] [CrossRef]
- Mao, G.X.; Lan, X.G.; Cao, Y.B.; Chen, Z.M.; He, Z.H.; Lv, Y.D.; Wang, Y.Z.; Hu, X.L.; Wang, G.F.; Yan, J. Effects of short-term forest bathing on human health in a broad-leaved evergreen forest in Zhejiang Province, China. Biomed. Environ. Sci. 2012, 25, 317–324. [Google Scholar] [CrossRef]
- Mao, G.-X.; Cao, Y.-B.; Lan, X.-G.; He, Z.-H.; Chen, Z.-M.; Wang, Y.-Z.; Hu, X.-L.; Lv, Y.-D.; Wang, G.-F.; Yan, J. Therapeutic effect of forest bathing on human hypertension in the elderly. J. Cardiol. 2012, 60, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Jeong, H.; Park, S.; Lee, S. Forest adjuvant anti-cancer therapy to enhance natural cytotoxicity in urban women with breast cancer: A preliminary prospective interventional study. Eur. J. Integr. Med. 2015, 7, 474–478. [Google Scholar] [CrossRef]
- Li, Q.; Morimoto, K.; Nakadai, A.; Inagaki, H.; Katsumata, M.; Shimizu, T.; Hirata, Y.; Hirata, K.; Suzuki, H.; Miyazaki, Y.; et al. Forest Bathing Enhances Human Natural Killer Activity and Expression of Anti-Cancer Proteins. Int. J. Immunopathol. Pharmacol. 2007, 20, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Morimoto, K.; Kobayashi, M.; Inagaki, H.; Katsumata, M.; Hirata, Y.; Hirata, K.; Suzuki, H.; Li, Y.; Wakayama, Y.; et al. Visiting a Forest, but Not a City, Increases Human Natural Killer Activity and Expression of Anti-Cancer Proteins. Int. J. Immunopathol. Pharmacol. 2008, 21, 117–127. [Google Scholar] [CrossRef]
- Li, Q.; Morimoto, K.; Kobayashi, M.; Inagaki, H.; Katsumata, M.; Hirata, Y.; Hirata, K.; Shimizu, T.; Li, Y.; Wakayama, Y.; et al. A forest bathing trip increases human natural killer activity and expression of anti-cancer proteins in female subjects. J. Biol. Regul. Homeost. Agents 2008, 22, 45–55. [Google Scholar] [PubMed]
- Seo, S.C.; Park, S.J.; Park, C.-W.; Yoon, W.S.; Choung, J.T.; Yoo, Y. Clinical and immunological effects of a forest trip in children with asthma and atopic dermatitis. Iran. J. Allergy Asthma Immunol. 2015, 14, 28–36. [Google Scholar] [PubMed]
- Tsao, T.-M.; Tsai, M.-J.; Hwang, J.-S.; Cheng, W.-F.; Wu, C.-F.; Chou, C.-C.; Su, T.-C. Health effects of a forest environment on natural killer cells in humans: An observational pilot study. Oncotarget 2018, 9, 16501–16511. [Google Scholar] [CrossRef]
- Li, Q.; Kobayashi, M.; Wakayama, Y.; Inagaki, H.; Katsumata, M.; Hirata, Y.; Hirata, K.; Shimizu, T.; Kawada, T.; Park, B.; et al. Effect of Phytoncide from Trees on Human Natural Killer Cell Function. Int. J. Immunopathol. Pharmacol. 2009, 22, 951–959. [Google Scholar] [CrossRef]
- Kiecolt-Glaser, J.K.; Graham, J.E.; Malarkey, W.B.; Porter, K.; Lemeshow, S.; Glaser, R. Olfactory influences on mood and autonomic, endocrine, and immune function. Psychoneuroendocrinology 2008, 33, 328–339. [Google Scholar] [CrossRef]
- Komori, T.; Fujiwara, R.; Tanida, M.; Nomura, J.; Yokoyama, M.M. Effects of Citrus Fragrance on Immune Function and Depressive States. Neuroimmunomodulation 1995, 2, 174–180. [Google Scholar] [CrossRef]
- Trellakis, S.; Fischer, C.; Rydleuskaya, A.; Tagay, S.; Bruderek, K.; Greve, J.; Lang, S.; Brandau, S. Subconscious olfactory influences of stimulant and relaxant odors on immune function. Eur. Arch. Oto-Rhino-Laryngol. 2011, 269, 1909–1916. [Google Scholar] [CrossRef] [PubMed]
- Ahn, C.; Lee, J.-H.; Kim, J.-W.; Park, M.-J.; Lee, S.-S.; Jeung, E.-B. Alleviation effects of natural volatile organic compounds from Pinus densiflora and Chamaecyparis obtusa on systemic and pulmonary inflammation. Biomed. Rep. 2018, 9, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Ahn, C.; Jang, Y.-J.; Kim, J.-W.; Park, M.-J.; Yoo, Y.-M.; Jeung, E.-B. Anti-asthmatic effects of volatile organic compounds from Chamaecyparis obtusa, Pinus densiflora, Pinus koraiensis, or Larix kaempferi wood panels. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2019, 69, 933–941. [Google Scholar] [CrossRef]
- Yang, H.; Ahn, C.; Choi, I.-G.; Choi, W.-S.; Park, M.-J.; Lee, S.-S.; Choi, D.; Jeung, E.-B. Estimation of the environmental effect of natural volatile organic compounds from Chamaecyparis obtusa and their effect on atopic dermatitis-like skin lesions in mice. Mol. Med. Rep. 2012, 12, 345–350. [Google Scholar] [CrossRef] [PubMed]
- Kennedy-Feitosa, E.; Cattani-Cavalieri, I.; Barroso, M.V.; Romana-Souza, B.; Brito-Gitirana, L.; Valença, S.S. Eucalyptol promotes lung repair in mice following cigarette smoke-induced emphysema. Phytomedicine 2019, 55, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.-S.; Park, D.-E.; Song, W.-J.; Park, H.; Kang, H.; Cho, S.-H.; Sohn, S.-W. Effect of 1.8-Cineole in Dermatophagoides pteronyssinus-Stimulated Bronchial Epithelial Cells and Mouse Model of Asthma. Biol. Pharm. Bull. 2016, 39, 946–952. [Google Scholar] [CrossRef] [PubMed]
- Bibi, H.; Reany, O.; Waisman, D.; Keinan, E. Prophylactic treatment of asthma by an ozone scavenger in a mouse model. Bioorganic Med. Chem. Lett. 2015, 25, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Hirota, R.; Nakamura, H.; Bhatti, S.A.; Ngatu, N.R.; Muzembo, B.A.; Dumavibhat, N.; Eitoku, M.; Sawamura, M.; Suganuma, N. Limonene inhalation reduces allergic airway inflammation inDermatophagoides farinae-treated mice. Inhal. Toxicol. 2012, 24, 373–381. [Google Scholar] [CrossRef]
- Hansen, J.S.; Nielsen, G.D.; Sørli, J.B.; Clausen, P.A.; Wolkoff, P.; Larsen, S.T. Adjuvant and Inflammatory Effects in Mice After Subchronic Inhalation of Allergen and Ozone-Initiated Limonene Reaction Products. J. Toxicol. Environ. Health Part A 2013, 76, 1085–1095. [Google Scholar] [CrossRef]
- Hansen, J.S.; Nørgaard, A.W.; Koponen, I.K.; Sørli, J.B.; Paidi, M.D.; Hansen, S.W.K.; Clausen, P.A.; Nielsen, G.D.; Wolkoff, P.; Larsen, S.T. Limonene and its ozone-initiated reaction products attenuate allergic lung inflammation in mice. J. Immunotoxicol. 2016, 13, 793–803. [Google Scholar] [CrossRef]
- Fujiwara, R.; Komori, T.; Noda, Y.; Kuraoka, T.; Shibata, H.; Shizuya, K.; Miyahara, S.; Ohmori, M.; Nomura, J.; Yokoyama, M.M. Effects of a long-term inhalation of fragrances on the stress-induced immunosuppression in mice. Neuroimmunomodulation 1998, 5, 318–322. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, A.; Fujiwara, S.; Matsumoto, I.; Abe, K. Stress Repression in Restrained Rats by (R)-(−)-Linalool Inhalation and Gene Expression Profiling of Their Whole Blood Cells. J. Agric. Food Chem. 2009, 57, 5480–5485. [Google Scholar] [CrossRef] [PubMed]
- Keinan, E.; Alt, A.; Amir, G.; Bar-Yoseph, R.; Bibi, H.; Shoseyov, D. Natural ozone scavenger prevents asthma in sensitized rats. Bioorg. Med. Chem. 2005, 13, 557–562. [Google Scholar] [CrossRef] [PubMed]
- Bastos, V.P.; Gomes, A.S.; Lima, F.J.; Brito, T.S.; Soares, P.M.; Pinho, J.P.; Silva, C.S.; Santos, A.A.; Souza, M.H.; Magalhães, P.J. Inhaled 1,8-Cineole Reduces Inflammatory Parameters in Airways of Ovalbumin-Challenged Guinea Pigs. Basic Clin. Pharmacol. Toxicol. 2010, 108, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-E.; Lee, E.-H.; Park, H.-J.; Kim, Y.-J.; Jung, H.-Y.; Ahn, D.-H.; Cho, Y.-J. Inhibition of inflammatory responses in lipopolysaccharide-induced RAW 264.7 cells byPinus densifloraroot extract. J. Appl. Biol. Chem. 2018, 61, 275–281. [Google Scholar] [CrossRef]
- Wheeler, B.; Lovell, R.; Higgins, S.L.; White, M.P.; Alcock, I.; Osborne, N.J.; Husk, K.; Sabel, C.; Depledge, M. Beyond greenspace: An ecological study of population general health and indicators of natural environment type and quality. Int. J. Health Geogr. 2015, 14, 1–17. [Google Scholar] [CrossRef]
- White, M.P.; Elliott, L.R.; Gascon, M.; Roberts, B.; Fleming, L.E. Blue space, health and well-being: A narrative overview and synthesis of potential benefits. Environ. Res. 2020, 191, 110169. [Google Scholar] [CrossRef]
- Gascon, M.; Zijlema, W.; Vert, C.; White, M.P.; Nieuwenhuijsen, M.J. Outdoor blue spaces, human health and well-being: A systematic review of quantitative studies. Int. J. Hyg. Environ. Health 2017, 220, 1207–1221. [Google Scholar] [CrossRef]
- Song, C.; Ikei, H.; Miyazaki, Y. Elucidation of a Physiological Adjustment Effect in a Forest Environment: A Pilot Study. Int. J. Environ. Res. Public Health 2015, 12, 4247–4255. [Google Scholar] [CrossRef]
- Acosta Navarro, J.C.; Smolander, S.; Struthers, H.; Zorita, E.; Ekman, A.M.L.; Kaplan, J.O.; Guenther, A.; Arneth, A.; Riipinen, I. Global emissions of terpenoid VOCs from terrestrial vegetation in the last millennium. J. Geophys. Res. Atmos. 2014, 119, 6867–6885. [Google Scholar] [CrossRef]
- Guenther, A.B.; Jiang, X.; Heald, C.L.; Sakulyanontvittaya, T.; Duhl, T.; Emmons, L.K.; Wang, X. The Model of Emissions of Gases and Aerosols from Nature version 2.1 (MEGAN2.1): An extended and updated framework for modeling biogenic emissions. Geosci. Model Dev. 2012, 5, 1471–1492. [Google Scholar] [CrossRef]
- Meneguzzo, F.; Albanese, L.; Bartolini, G.; Zabini, F. Temporal and Spatial Variability of Volatile Organic Compounds in the Forest Atmosphere. Int. J. Environ. Res. Public Health 2019, 16, 4915. [Google Scholar] [CrossRef] [PubMed]
- White, M.P.; Alcock, I.; Grellier, J.; Wheeler, B.W.; Hartig, T.; Warber, S.L.; Bone, A.; Depledge, M.H.; Fleming, L.E. Spending at least 120 minutes a week in nature is associated with good health and wellbeing. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Shanahan, D.F.; Bush, R.; Gaston, K.J.; Lin, B.B.; Dean, J.; Barber, E.; Fuller, R.A. Health Benefits from Nature Experiences Depend on Dose. Sci. Rep. 2016, 6, 28551. [Google Scholar] [CrossRef]
- Riedler, J.; Braun-Fahrländer, C.; Eder, W.; Schreuer, M.; Waser, M.; Maisch, S.; Carr, D.; Schierl, R.; Nowak, D.; Von Mutius, E. Exposure to farming in early life and development of asthma and allergy: A cross-sectional survey. Lancet 2001, 358, 1129–1133. [Google Scholar] [CrossRef]
- Ege, M.J.; Herzum, I.; Büchele, G.; Krauss-Etschmann, S.; Lauener, R.; Roponen, M.; Hyvärinen, A.; Vuitton, D.A.; Riedler, J.; Brunekreef, B.; et al. Prenatal exposure to a farm environment modifies atopic sensitization at birth. J. Allergy Clin. Immunol. 2008, 122, 407–412. [Google Scholar] [CrossRef]
- Gale, C.R.; Martyn, C.N. Migrant studies in multiple sclerosis. Prog. Neurobiol. 1995, 47, 425–448. [Google Scholar] [CrossRef]
- Ahlgren, C.; Odén, A.; Lycke, J. A nationwide survey of the prevalence of multiple sclerosis in immigrant populations of Sweden. Mult. Scler. J. 2012, 18, 1099–1107. [Google Scholar] [CrossRef]
- Correale, J.; Farez, M.F. The impact of parasite infections on the course of multiple sclerosis. J. Neuroimmunol. 2011, 233, 6–11. [Google Scholar] [CrossRef]
- Oray, M.; Abu Samra, K.; Ebrahimiadib, N.; Meese, H.; Foster, C.S. Long-term side effects of glucocorticoids. Expert Opin. Drug Saf. 2016, 15, 457–465. [Google Scholar] [CrossRef]
- Michot, J.; Bigenwald, C.; Champiat, S.; Collins, M.; Carbonnel, F.; Postel-Vinay, S.; Berdelou, A.; Varga, A.; Bahleda, R.; Hollebecque, A.; et al. Immune-related adverse events with immune checkpoint blockade: A comprehensive review. Eur. J. Cancer 2016, 54, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Rohr, A.C.; Wilkins, C.K.; Clausen, P.A.; Hammer, M.; Nielsen, G.D.; Wolkoff, P.; Spengler, J.D. Upper airway and pulmonary effects of oxidation products of (+)- α -pinene, d -limonene and isoprene in balb/c mice. Inhal. Toxicol. 2002, 14, 663–684. [Google Scholar] [CrossRef] [PubMed]
- Cakmak, S.; Dales, R.E.; Liu, L.; Kauri, L.M.; Lemieux, C.L.; Hebbern, C.; Zhu, J. Residential exposure to volatile organic compounds and lung function: Results from a population-based cross-sectional survey. Environ. Pollut. 2014, 194, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; Wei, C.-N.; Harada, K.; Minamoto, K.; Okamoto, Y.; Otsuka, M.; Ueda, A. Evaluation of Allergenicity of Constituents of Myoga Using the Murine Local Lymph Node Assay. Int. J. Immunopathol. Pharmacol. 2010, 23, 463–470. [Google Scholar] [CrossRef]
- Pesonen, M.; Suomela, S.; Kuuliala, O.; Henriks-Eckerman, M.-L.; Aalto-Korte, K. Occupational contact dermatitis caused byD-limonene. Contact Dermat. 2014, 71, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Dharmagunawardena, B.; Takwale, A.; Sanders, K.J.; Cannan, S.; Rodger, A.; Ilchyshyn, A. Gas chromatography: An investigative tool in multiple allergies to essential oils. Contact Dermat. 2002, 47, 288–292. [Google Scholar] [CrossRef]
- Kumar, P.; Druckman, A.; Gallagher, J.; Gatersleben, B.; Allison, S.; Eisenman, T.S.; Hoang, U.; Hama, S.; Tiwari, A.; Sharma, A.; et al. The nexus between air pollution, green infrastructure and human health. Environ. Int. 2019, 133, 105181. [Google Scholar] [CrossRef]
- Shanahan, D.F.; Lin, B.B.; Bush, R.; Gaston, K.J.; Dean, J.H.; Barber, E.; Fuller, R.A. Toward Improved Public Health Outcomes from Urban Nature. Am. J. Public Heal. 2015, 105, 470–477. [Google Scholar] [CrossRef]
- Steffen, W.; Broadgate, W.; Deutsch, L.; Gaffney, O.; Ludwig, C. The trajectory of the Anthropocene: The Great Acceleration. Anthr. Rev. 2014, 2, 81–98. [Google Scholar] [CrossRef]
- Maes, J.; Teller, A.; Nessi, S.; Bulgheroni, C.; Konti, A.; Sinkko, T.; Tonini, D.; Pant, R. Mapping and Assessment of Ecosystems and Their Services: An EU Ecosystem Assessment; European Comission: Maastricht, The Netherlands, 2020; ISBN 9789276178330. [Google Scholar]
- Altizer, S.; Ostfeld, R.S.; Johnson, P.T.J.; Kutz, S.; Harvell, C.D. Climate Change and Infectious Diseases: From Evidence to a Predictive Framework. Science 2013, 341, 514–519. [Google Scholar] [CrossRef]
- Rohr, J.R.; Dobson, A.P.; Johnson, P.T.; Kilpatrick, A.M.; Paull, S.H.; Raffel, T.R.; Ruiz-Moreno, D.; Thomas, M.B. Frontiers in climate change–disease research. Trends Ecol. Evol. 2011, 26, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Houwenhuyse, S.; Macke, E.; Reyserhove, L.; Bulteel, L.; Decaestecker, E. Back to the future in a petri dish: Origin and impact of resurrected microbes in natural populations. Evol. Appl. 2018, 11, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Legendre, M.; Bartoli, J.; Shmakova, L.; Jeudy, S.; Labadie, K.; Adrait, A.; Lescot, M.; Poirot, O.; Bertaux, L.; Bruley, C.; et al. Thirty-thousand-year-old distant relative of giant icosahedral DNA viruses with a pandoravirus morphology. Proc. Natl. Acad. Sci. USA 2014, 111, 4274–4279. [Google Scholar] [CrossRef] [PubMed]
- Ziska, L.H.; Makra, L.; Harry, S.K.; Bruffaerts, N.; Hendrickx, M.; Coates, F.; Saarto, A.; Thibaudon, M.; Oliver, G.; Damialis, A.; et al. Temperature-related changes in airborne allergenic pollen abundance and seasonality across the northern hemisphere: A retrospective data analysis. Lancet Planet. Health 2019, 3, e124–e131. [Google Scholar] [CrossRef]
| Overall Score | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Selection Bias | Study Design | Con-founders | Blinding | Data Collection | Dropouts | Without Blinding * | |||
| Gaisberger et al. | 2012 | 2 | 1 | 1 | 3 | 1 | 3 | 3 | 2 |
| Grafetstätter et al. | 2017 | 3 | 1 | 2 | 3 | 1 | 3 | 3 | 3 |
| Han et al. | 2016 | 2 | 1 | 1 | 3 | 1 | 3 | 3 | 2 |
| Im et al. | 2016 | 2 | 1 | 1 | 3 | 1 | 3 | 3 | 2 |
| Jia et al. | 2016 | 3 | 1 | 1 | 3 | 1 | 3 | 3 | 3 |
| Kiecolt-Glaser et al. | 2008 | 2 | 2 | 3 | 1 | 3 | 1 | 3 | 3 |
| Kim et al. | 2015 | 2 | 2 | NA | 3 | 1 | 1 | 2 | 1 |
| Komori et al. | 1995 | 3 | 2 | 2 | 3 | 1 | 3 | 3 | 3 |
| Li et al. | 2007 | 3 | 2 | NA | 3 | 1 | 2 | 3 | 2 |
| Li et al. | 2008a | 3 | 2 | NA | 3 | 1 | 3 | 3 | 3 |
| Li et al. | 2008b | 3 | 2 | NA | 3 | 1 | 2 | 3 | 2 |
| Li et al. | 2009 | 3 | 2 | NA | 3 | 1 | 2 | 3 | 2 |
| Lyu et al. | 2019 | 3 | 1 | 2 | 3 | 1 | 2 | 3 | 2 |
| Mao et al. | 2012a | 3 | 1 | 1 | 3 | 1 | 2 | 3 | 2 |
| Mao et al. | 2012b | 3 | 1 | 1 | 3 | 1 | 2 | 3 | 2 |
| Mao et al. | 2017 | 2 | 1 | 1 | 3 | 1 | 1 | 2 | 1 |
| Mao et al. | 2018 | 2 | 1 | 1 | 3 | 1 | 1 | 2 | 1 |
| Seo et al. | 2015 | 2 | 2 | NA | 3 | 1 | 2 | 2 | 1 |
| Trellakis et al. | 2012 | 2 | 2 | NA | 2 | 1 | 1 | 1 | 1 |
| Tsao et al. | 2018 | 3 | 2 | 1 | 3 | 1 | 3 | 3 | 3 |
| (a) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | ||
| Title | Abstract | Introduction | Methods | ||||||||
| Background | Objectives and Hypotheses | Ethical Statement | Study Design (Number of Experimental Groups, Blinding, Experimental Unit) | Experimental Procedure | Animal Details (Species, Sex, Age, Source, Weight) | Housing and Husbandry Conditions | Sample size Calculation (Number per Group, Number of Independent Replicates) | ||||
| Ahn et al. | 2018a | 2 | 1 | 1 | 1 | 1 | 3 | 1 | 1 | 1 | 2 |
| Ahn et al. | 2018b | 2 | 1 | 1 | 1 | 2 | 2 | 1 | 2 | 1 | 2 |
| Bastos et al. | 2011 | 1 | 1 | 1 | 1 | 1 | 2 | 1 | 2 | 3 | 3 |
| Bibi et al. | 2015 | 1 | 2 | 1 | 1 | 2 | 2 | 1 | 2 | 1 | 2 |
| Fujiwara et al. | 1998 | 1 | 1 | 2 | 2 | 3 | 2 | 2 | 2 | 2 | 1 |
| Hansen et al. | 2013 | 2 | 1 | 1 | 2 | 1 | 2 | 1 | 1 | 1 | 2 |
| Hansen et al. | 2016 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 2 |
| Hirota et al. | 2012 | 1 | 2 | 2 | 3 | 2 | 1 | 1 | 1 | 1 | 2 |
| Keinan et al. | 2005 | 1 | 1 | 1 | 1 | 2 | 3 | 1 | 3 | 3 | 3 |
| Kennedy-Feitosa et al. | 2019 | 1 | 1 | 2 | 2 | 2 | 2 | 1 | 1 | 1 | 2 |
| Lee et al. | 2016 | 2 | 1 | 1 | 1 | 2 | 3 | 1 | 1 | 3 | 3 |
| Nakamura et al. | 2009 | 2 | 2 | 2 | 3 | 3 | 2 | 1 | 2 | 1 | 2 |
| Yang et al. | 2015 | 1 | 1 | 1 | 1 | 1 | 3 | 3 | 2 | 1 | 3 |
| (b) | |||||||||||
| 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 19 | 20 | ||
| Methods | Results | Discussion | |||||||||
| Allocation to Experimen-tal Groups (Randomi-sation, Matching) | Experi-mental Outcomes | Statistical Analysis | Baseline Data Monitoring | Number of Animals Analysed | Outcome Report and Estimation (Individual Values, Standard Deviation) | Adverse Events | Interpreta-tion and Limitations | Generalis-ability and Translational Relevance | Funding | ||
| Ahn et al. | 2018a | 3 | 1 | 1 | 3 | 3 | 2 | 3 | 2 | 2 | 1 |
| Ahn et al. | 2018b | 3 | 1 | 1 | 3 | 3 | 2 | 3 | 2 | 2 | 3 |
| Bastos et al. | 2011 | 3 | 1 | 1 | 3 | 3 | 2 | 3 | 2 | 2 | 2 |
| Bibi et al. | 2015 | 3 | 1 | 1 | 3 | 1 | 2 | 3 | 2 | 1 | 1 |
| Fujiwara et al. | 1998 | 3 | 3 | 2 | 3 | 3 | 2 | 3 | 2 | 2 | 3 |
| Hansen et al. | 2013 | 3 | 1 | 1 | 1 | 2 | 1 | 3 | 1 | 1 | 1 |
| Hansen et al. | 2016 | 2 | 1 | 1 | 1 | 1 | 2 | 3 | 1 | 1 | 1 |
| Hirota et al. | 2012 | 2 | 1 | 1 | 3 | 1 | 2 | 3 | 2 | 2 | 2 |
| Keinan et al. | 2005 | 3 | 2 | 2 | 3 | 3 | 2 | 3 | 3 | 2 | 3 |
| Kennedy-Feitosa et al. | 2019 | 3 | 1 | 1 | 3 | 1 | 2 | 3 | 1 | 1 | 1 |
| Lee et al. | 2016 | 3 | 1 | 1 | 3 | 3 | 2 | 3 | 2 | 2 | 1 |
| Nakamura et al. | 2009 | 3 | 1 | 1 | 3 | 1 | 2 | 3 | 2 | 2 | 3 |
| Yang et al. | 2015 | 3 | 1 | 1 | 3 | 3 | 2 | 3 | 2 | 2 | 1 |
| Outcome | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Main Author | Year | Title | Measure | Intervention (Compared to Baseline) | p-Value | Control (Compared to Baseline) | p-Value | Comparison (Intervention Compared to Control) | p-Value | Conclusion | Comment | |
| Forest bathing | ||||||||||||
| Han et al. [45] | 2016 | The effects of forest therapy on coping with chronic widespread pain | NK cell activity | Increase | p < 0.001 | NS | Increase | NA | NK cell activity increases after a forest bathing trip in adults with chronic pain. | Significant baseline differences between control and intervention group in NK cell activity. | ||
| Im et al. [44] | 2016 | Comparison of effect of two-hour exposure to forest and urban environments on cytokine, anti-oxidant and stress levels in young adults | IL-6 level in serum | NS | Pro-inflammatory cytokine level (IL-8 and TNFα, but not IL-6) is reduced in healthy students after a forest bathing trip. | No pre-intervention data shown. | ||||||
| IL-8 level in serum | Decrease | p < 0.001 | ||||||||||
| TNFα level in serum | Decrease | p < 0.001 | ||||||||||
| Glutathione peroxidase (GPx) level in serum | Increase | p < 0.05 | ||||||||||
| Jia et al. [46] | 2016 | Health effects of forest bathing trip on elderly patients with chronic obstructive pulmonary disease | CD8+ T cell number and % | NS | NS | NS | Forest bathing reduces pro-inflammatory cytokine levels, but not proportions of CD8+ T, NK or NKT cells or GrB expression in COPD patients. | Result on perforin expression is questionable, since there is also an effect in control group. | ||||
| NK cell number and % (CD3-CD56+) | NS | NS | NS | |||||||||
| NKT cell number and % (CD3+CD56+) | NS | NS | NS | |||||||||
| Perforin expression in CD8+ T cells (flow cytometry) | Decrease | p < 0.001 | Decrease | NA | Decrease | p < 0.01 | ||||||
| Perforin expression in NK cells (flow cytometry) | Decrease | p < 0.001 | Decrease | NA | NS | |||||||
| Perforin expression in NKT cells (flow cytometry) | Decrease | p < 0.001 | Decrease | NA | Decrease | p < 0.05 | ||||||
| Granzyme B expression in CD8+ T cells (flow cytometry) | NS | NS | NS | |||||||||
| Granzyme B expression in NK cells (flow cytometry) | NS | NS | NS | |||||||||
| Granzyme B expression in NKT cells (flow cytometry) | NS | NS | NS | |||||||||
| IL-6 level in serum | Decrease | p < 0.01 | NS | Decrease | p < 0.05 | |||||||
| IL-8 level in serum | Decrease | p < 0.05 | NS | Decrease | p < 0.01 | |||||||
| IFN-y level in serum | Decrease | p < 0.01 | NS | Decrease | p < 0.05 | |||||||
| IL-1b level in serum | NS | NS | Decrease | p < 0.05 | ||||||||
| CRP level in serum | NS | NS | Decrease | p < 0.05 | ||||||||
| TNFα level in serum | NS | NS | NS | |||||||||
| Kim et al. [52] | 2015 | Forest adjuvant anti-cancer therapy to enhance natural cytotoxicity in urban women with breast cancer: A preliminary prospective interventional study | NK cell number (CD3-CD56+) | Increase | p < 0.01 | Forest therapy enhances natural cytotoxicity in breast cancer patients by increasing NK cells and cytotoxic mediators. | ||||||
| Perforin level in serum (ELISA) | Increase | p < 0.02 | ||||||||||
| Granzyme B level in serum (ELISA) | Increase | p < 0.02 | ||||||||||
| Li et al. [53] | 2007 | Forest bathing enhances human natural killer activity and expression of anti-cancer proteins | NK cell number and % (CD16+) | Increase | p < 0.01 | Forest bathing enhances NK cell activity and numbers in healthy male adults. | ||||||
| Cytolytic NK cell activity (Cr-release assay) | Increase | p < 0.01 | ||||||||||
| % of T cells (CD3+) | Decrease | NA | ||||||||||
| % of perforin- expressing cells | Increase | p < 0.01 | ||||||||||
| % of granzyme A/B- expressing cells | Increase | p < 0.01 | ||||||||||
| % of granulysin-expressing cells | Increase | p < 0.01 | ||||||||||
| Li et al. [55] | 2008a | A forest bathing trip increases human natural killer activity and expression of anti-cancer proteins in female subjects | NK cell number and % (CD16+) | Increase | p < 0.01 | Forest bathing enhances NK cell activity and numbers in healthy female adults. | ||||||
| Cytolytic NK cell activity (Cr-release assay) | Increase | p < 0.01 | ||||||||||
| % of T cells (CD3+) | Decrease | p < 0.05 | ||||||||||
| % of perforin- expressing cells | Increase | p < 0.01 | ||||||||||
| % of granzyme A/B- expressing cells | Increase | p < 0.01 | ||||||||||
| % of granulysin- expressing cells | Increase | p < 0.01 | ||||||||||
| Li et al. [54] | 2008b | Visiting a forest, but not a city, increases human natural killer activity and expression of anti-cancer proteins | NK cell number and % (CD16+) | Increase | p < 0.01 | NS | Increase | p < 0.05 | Forest bathing enhances NK cell activity and numbers in healthy adults. | |||
| Cytolytic NK cell activity (Cr-release assay) | Increase | p < 0.01 | NS | Increase | p < 0.05 | |||||||
| % of T cells (CD3+) | NS | NS | ||||||||||
| % of perforin- expressing cells | Increase | p < 0.01 | NS | |||||||||
| % of granzyme A/B- expressing cells | Increase | p < 0.01 | NS | |||||||||
| % of granulysin- expressing cells | Increase | p < 0.01 | NS | |||||||||
| Lyu et al. [47] | 2019 | Benefits of a three-day bamboo forest therapy session on the psychophysiology and immune system responses of male college students | Cytolytic NK cell activity | Increase | p < 0.05 | NS | NA | Forest bathing in a bamboo forest enhances NK cell activity and percentages in healthy adults. | ||||
| % of NK cells (CD16+CD56+) | Increase | p < 0.05 | NS | NA | ||||||||
| Perforin level (ELISA) | Increase | p < 0.05 | NS | NA | ||||||||
| Granulysin level (ELISA) | NS | NS | NA | |||||||||
| Granzyme A/B level (ELISA) | Increase | p < 0.05 | NS | NA | ||||||||
| Mao et al. [50] | 2012a | Effects of short-term forest bathing on human health in a broad-leaved evergreen forest in Zhejiang Province, China | IL-6 level in serum (radioimmunoassay) | NS | Increase | NA | Decrease | p < 0.05 | Forest bathing decreases pro-inflammatory cytokine levels (IL-6 and TNFα) in healthy young adults but has no effect on immune cell distribution. | Questionable effect (increase) in control group compared to baseline for IL-6 and TNFα levels. | ||
| TNFα level in serum (radioimmunoassay) | Decrease | NA | Increase | NA | Decrease | p < 0.05 | ||||||
| HCRP level in serum | NS | |||||||||||
| % of B cells (CD5+CD19+) | Increase | p < 0.05 | No pre-intervention data shown for leukocyte distributions. | |||||||||
| % of T cells (CD3+) | NS | |||||||||||
| % of Th cells (CD3+CD4+) | NS | |||||||||||
| % of cytotoxic T cells (CD3+CD8+) | NS | |||||||||||
| % of NK cells (CD3-CD16+CD56+) | NS | |||||||||||
| Mao et al. [51] | 2012b | Therapeutic effect of forest bathing on human hypertension in the elderly | IL-6 level in serum (radioimmunoassay) | Decrease | p < 0.05 | NS | NS | Forest bathing decreases pro-inflammatory cytokine level (IL-6, but not TNFα) in elderly patients with hypertension. | ||||
| TNFα level in serum (radioimmunoassay) | NS | NS | NS | |||||||||
| Mao et al. [49] | 2017 | The salutary influence of forest bathing on elderly patients with chronic heart failure | IL-6 level in serum (ELISA) | NS | NS | Decrease | p < 0.05 | Forest bathing decreases pro-inflammatory cytokine level (IL-6, but not TNFα) in elderly patients with chronic heart failure. | No effect in intervention group compared to baseline. | |||
| TNFα level in serum (ELISA) | NS | NS | NS | |||||||||
| HCRP level in serum | NS | NS | NS | |||||||||
| Mao et al. [48] | 2018 | Additive benefits of twice forest bathing trips in elderly patients with chronic heart failure | IL-6 level in serum (ELISA) | NS | NS | NS | A second forest bathing trip further decreases pro-inflammatory cytokine level (TNFα, but not IL-6) in elderly patients with chronic heart failure. | |||||
| TNFα level in serum (ELISA) | Decrease | p < 0.05 | NS | Decrease | p < 0.05 | |||||||
| Seo et al. [56] | 2015 | Clinical and immunological effects of a forest trip in children with asthma and atopic dermatitis | Asthma: | Forest environment improves clinical symptoms in asthmatic children. | ||||||||
| Forced vital capacity (spirometry, FCV) | Increase | p < 0.05 | ||||||||||
| Fractional exhaled nitric oxide (FeNO) | Decrease | NA | ||||||||||
| Seo et al. [56] | 2015 | Clinical and immunological effects of a forest trip in children with asthma and atopic dermatitis | Atopic dermatitis: | Forest environment improves clinical symptoms and has immunological effects in chronic allergic skin disease. | ||||||||
| Atopic dermatitis index (SCORAD) | Decrease | NA | ||||||||||
| Thymus and activation-regulated chemokine/CCL17 level | NS | |||||||||||
| Macrophage-derived chemokine/CCL22 level | Decrease | p < 0.01 | ||||||||||
| Tsao et al. (forest workers) [57] | 2018 | Health effects of a forest environment on natural killer cells in humans: an observational pilot study | % of NK cells in blood (CD3-CD56+) | Increase | p < 0.05 | Living in a forest environment increases NK cell percentage, but not the amount of activated NK cells. | ||||||
| % of activated NK cells in blood (CD3-CD56+CD69+) | NS | |||||||||||
| Tsao et al. (forest bathing) [57] | 2018 | Health effects of a forest environment on natural killer cells in humans: an observational pilot study | % of NK cells in blood on d5 | NS | Short-term forest trip enhances fraction of activated NK cells in healthy adults, and effect lasts for at least 4 days. | |||||||
| % of activated NK cells in blood on d5 | Increase | p < 0.01 | ||||||||||
| % of NK cells in blood on d9 (4 days after intervention) | NS | |||||||||||
| % of activated NK cells in blood on d9 (4 days after intervention) | Increase | p < 0.01 | ||||||||||
| BVOC inhalation | ||||||||||||
| Li et al. [58] | 2009 | Effect of phytoncide from trees on human natural killer cell function | Cytolytic NK cell activity (Cr-release assay) | Increase | p < 0.05 | Phytoncide exposure enhances NK cell activity and % in healthy adults. | ||||||
| % of NK cells (CD16+) | Increase | p < 0.01 | ||||||||||
| % of T cells (CD3+) | Decrease | p < 0.01 | ||||||||||
| % of perforin- expressing cells | Increase | p < 0.05 | ||||||||||
| % of granzyme A/B- expressing cells | Increase | p < 0.01 p < 0.05 | ||||||||||
| % of granulysin- expressing cells | Increase | p < 0.05 | ||||||||||
| Fragrance inhalation | ||||||||||||
| Kiecolt-Glaser et al. [59] | 2008 | Olfactory influences on mood and autonomic, endocrine and immune function | Delayed hypersensitivity to candida (DTH) | Increase | NA | Increase | NA | Decrease | p = 0.02 (lavender) p = 0.06 (lemon) | Greater DTH response after water inhalation indicates better immune response than in fragrance groups, but no difference in cytokine levels detectable. | * Differing effects in blinded and informed groups for blastogenesis responses. | |
| PBL proliferation (blastogenesis) | NA* | |||||||||||
| IL-6 level in PBLs (ELISA) | NS | |||||||||||
| IL-10 level in PBLs (ELISA) | NS | |||||||||||
| Komori et al. [60] | 1995 | Effects of citrus fragrance on immune function and depressive states | Deviation from normal CD4/CD8 ratio | Decrease | NA | Citrus fragrance has a beneficial effect on immune cell distribution in depressive patients and can reduce the dose antidepressants needed. | ||||||
| Deviation from normal NK cell activity (Cr-release assay) | Decrease | NA | ||||||||||
| Trellakis et al. [61] | 2012 | Subconscious olfactory influences of stimulant and relaxant odors on immune function | IL-8 level in serum (ELISA) | NS | No significant effect of any stimulatory or relaxing fragrance exposure on immune parameters in healthy adults. | |||||||
| IL-6 level in serum (ELISA) | NS | |||||||||||
| TNFα level in serum (ELISA) | NS | |||||||||||
| CCL3 (MIP-1a) level in serum (ELISA) | NS | |||||||||||
| CCL4 (MIP-1b) level in serum (ELISA) | NS | |||||||||||
| CCL5 (RANTES) level in serum (ELISA) | NS | |||||||||||
| CXCL8 (IL-8) release by neutrophils | NS | |||||||||||
| Waterfall exposure | ||||||||||||
| Gaisberger et al. [35] | 2012 | Effects of ionized waterfall aerosol on pediatric allergic asthma | IL-5 level in serum (ELISpot) | Decrease | p < 0.05 | NS | Decrease | NS | Exposure to waterfalls reduces pro-inflammatory cytokines and allergic asthma symptoms, enhances lung function and induces Treg cells. | |||
| IL-10 level in serum (ELISpot) | Increase | p < 0.05 | NS | Increase | NS | |||||||
| IL-13 level in serum (ELISpot) | Decrease | p < 0.01 | Decrease | p < 0.01 | NS | |||||||
| IL-10 expression (PCR) | Increase | NA | Increase | NA | NS | |||||||
| IL-13 expression (PCR) | Decrease | NA | NS | NA | Decrease | p < 0.05 | ||||||
| IFNg expression (PCR) | Increase | NA | Increase | NA | NS | |||||||
| Treg cells (%) | Increase | p < 0.01 | Increase | p < 0.05 | NS | |||||||
| Eosinophilic cationic protein (ECP) levels in serum | Decrease | p < 0.05 | NS | NS | ||||||||
| Fractional exhaled nitric oxide (FeNO) at d20 | Decrease | p < 0.001 | Decrease | p < 0.001 | NA | |||||||
| Fractional exhaled nitric oxide (FeNO) at d80 | Decrease | p < 0.01 | NS | NA | ||||||||
| Peak expiratory flow rate (PEF) | Increase | p < 0.01 | Increase | p < 0.01 | NA | |||||||
| Other spirometric parameters (FEV, FEV%FVC, FEF25, FEF50, MMEF2575) | Increase | p < 0.05 p < 0.01 | NS | NA | ||||||||
| Grafetstätter et al. [34] | 2017 | Does waterfall aerosol influence mucosal immunity and chronic stress? A randomized controlled clinical trial | IgA level in saliva (d6) | Increase | NA | Increase | NA | Increase | p = 0.001 | Exposure to waterfalls activates the immune system and improves lung function. | ||
| IgA level in saliva (d66) | Increase | NA | NS | Increase | p < 0.05 | |||||||
| Peak expiratory flow rate (PEF) (d6) | Increase | p = 0.023 | Increase | NA | NS | |||||||
| Outcome | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Main Author | Year | Title | Measure | Pre-treatment | p-Value | Intervention (Compared to Pre-treatment) | p-Value | Conclusion | Comment |
| BVOC inhalation | |||||||||
| Ahn et al. [62] | 2018a | Alleviation effects of natural volatile organic compounds from Pinus densiflora and Chamaecyparis obtuda on systemic and pulmonary inflammation | IgE level in serum (ELISA) | Increase | p < 0.05 | Decrease | p < 0.05 | BVOCs (VOCCo, VOCPd) excert anti-inflammatory effects in mice. | |
| Prostaglandin E2 (PGE2) level in serum (ELISA) | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| COX-2 mRNA expression in PBMCs | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| TNFα mRNA expression in PBMCs | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| IL-1b mRNA expression in PBMCs | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| IL-13 mRNA expression in PBMCs | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| COX-2 mRNA expression in lung tissue | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| NF-kB mRNA expression in lung tissue | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| TNFα mRNA expression in lung tissue | Increase | p < 0.05 | NS | ||||||
| COX-2, NF-kB, IL-1b, TNFα, IL-13 mRNA in BALF cells | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| Ahn et al. [63] | 2018b | Anti-asthmatic effects of volatile organic compounds from Chamaecyparis obtusa, Pinus densiflora, Pinus koraiensis, and Larix kaempferi wood panels | Thickening of bronchiolar wall (hypertrophy) | Increase | NA | Decrease | NA | BVOCs (VOCCo, VOCPd, VOCPk, VOCLk) excert anti-allergic effects in asthmatic mice. | |
| IL-4 level in serum (ELISA) | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| TNFα level in serum (ELISA) | Increase | p < 0.05 | Decrease (C. obtusa) | p < 0.05 | |||||
| IL-4 mRNA expression in bronchioles | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| IL-5 mRNA expression in bronchioles | Increase | p < 0.05 | NS | ||||||
| IL-9 mRNA expression in bronchioles | Increase | p < 0.05 | Decrease (C. obtusa) | p < 0.05 | |||||
| IL-13 mRNA expression in bronchioles | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| Yang et al. [64] | 2015 | Estimation of the environmental effect of natural volatile organic compounds from Chamaecyparis obtusa and their effect on atopic dermatitis-like skin lesions in mice | IgE level in serum | Increase | p < 0.05 | Decrease | p < 0.05 | Exposure to BVOCs (C. obtusa) ameliorates inflammatory skin reactions in mice with atopic dermatitis. | |
| Mast cell infiltration into skin lesions | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| IL-1b mRNA expression in skin lesions | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| IL-6 mRNA expression in skin lesions | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| Eucalyptol inhalation | |||||||||
| Bastos et al. [74] | 2011 | Inhaled 1,8-Cineole reduces inflammatory parameters in airways of ovalbumin-challenged guinea pigs | TNFα level in BALF (ELISA) | Increase | p < 0.05 | Decrease | NS | Eucalyptol (1,8-cineol) inhibits antigen-induced airway inflammation in guinea pigs. | |
| IL-1b level in BALF (ELISA) | Increase | p < 0.05 | Decrease | NS | |||||
| IL-10 level in BALF (ELISA) | Decrease | p < 0.05 | Increase | NS | |||||
| Leukocyte number in BALF (eosinophils and neutrophils) | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| MPO activity | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| Kennedy-Feitosa et al. [65] | 2019 | Eucalyptol promotes lung repair in mice following cigarette smoke-induced emphysema | TNFα level in lung tissue | Increase | p < 0.01 | Decrease | p < 0.01 | Eucalyptol reduces pro-inflammatory cytokines and neutrophil activation marker (MPO) after lung damage by cigarette smoke. | |
| IL-1b level in lung tissue | Increase | p < 0.01 | Decrease | p < 0.05 | |||||
| IL-6 level in lung tissue | Increase | p < 0.01 | Decrease | p < 0.01 | |||||
| TGFß-1 level in lung tissue | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| MPO activity in lung tissue | Increase | p < 0.01 | Decrease | p < 0.05 | |||||
| Lee et al. [66] | 2016 | Effect of 1,8-cineol in Dermatophagiodes pteronyssinus-stimulated bronchial epithelial cells and mouse model of asthma | IL-4 level in BALF (ELISA) | Increase | p < 0.01 | Decrease | p < 0.05 | Eucalyptol reduces pro-inflammatory cytokine expression (IL-4, IL-13, IL-17A) in house dust mite-allergic/asthmatic mice. | |
| IL-13 level in BALF (ELISA) | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| IL-17A level in BALF (ELISA) | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| Neutrophil number in BALF | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| Eosinophil number in BALF | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| Lymphocyte number in BALF | Increase | p < 0.05 | Decrease | p < 0.05 | |||||
| Der p-specific IgG1 in serum (ELISA) | Increase | p < 0.01 | Decrease | p < 0.05 | |||||
| Airway restriction (Penh) | Increase | p < 0.01 | Decrease | p < 0.05 | |||||
| Limonene inhalation | |||||||||
| Bibi et al. [67] | 2015 | Treatment of asthma by an ozone scavenger in a mouse model | Aldehyde (ozone oxydation product) levels in BALF | Increase | NA | Decrease | NA | Prophylactic limonene inhalation protects against allergic asthma in mice. | |
| Aldehyde (ozone oxydation product) levels in lung tissue | Increase | NA | Decrease | NA | |||||
| Aldehyde (ozone oxydation product) levels in spleen | Increase | NA | Decrease | NA | |||||
| Neutrophil number in BALF | Increase | NA | Decrease | p < 0.05 | |||||
| Eosinophil number in BALF | Increase | NA | NS | ||||||
| Infiltration of inflammatory cells into lung tissue | Increase | NA | Decrease | NA | |||||
| Hirota et al. [68] | 2012 | Limonene inhalation reduces allergic airway inflammation in Dermatophagoides farinae-treated mice | Der f-specific IgG in serum (ELISA) | Increase | p < 0.001 | Decrease | p < 0.01 | Limonene reduces pro-inflammatory cytokines and cell numbers in mice pre-sensitized to house dust mite allergen. | |
| Total IgE in serum (ELISA) | Increase | NS | Decrease | NS | |||||
| Eosinophil number in BALF | Increase | p < 0.001 | Decrease | p < 0.001 | |||||
| Lymphocyte number in BALF | Increase | p < 0.001 | Decrease | p < 0.05 | |||||
| Neutrophil number in BALF | Increase | p < 0.001 | Decrease | p < 0.05 | |||||
| Macrophage number in BALF | Increase | p < 0.001 | Decrease | p < 0.05 | |||||
| IL-5 level in BALF | Increase | p < 0.001 | Decrease | p < 0.001 | |||||
| IL-13 level in BALF | Increase | p < 0.001 | Decrease | p < 0.001 | |||||
| Eotaxin level in BALF | Increase | p < 0.001 | Decrease | p < 0.001 | |||||
| MCP-1 level in BALF | Increase | p < 0.001 | Decrease | p < 0.001 | |||||
| TGF-b level in BALF | Increase | p < 0.001 | Decrease | p < 0.05 | |||||
| IFNy level in BALF | Decrease | p < 0.01 | Increase | p < 0.05 | |||||
| Bronchorestriction with Ach | Increase | p < 0.01 | Decrease | p < 0.01 | |||||
| Keinan et al. [73] | 2005 | Natural ozone scavenger prevents asthma in sensitized rats | Limonene inhalation: | Limonene reduces inflammatory cell infiltrates into lung tissue and improves airway restriction in lungs of rats with allergic asthma. | No significances provided in graphical outcome report | ||||
| Inflammatory cell infiltrates | Increase | NA | Decrease | NA | |||||
| Airway restriction (Penh) | Increase | NA | Decrease | NA | |||||
| Eucalyptol inhalation: | Eucalyptol reduces inflammatory cell infiltrates into lung tissue, but to a lesser extent than limonene, but does not improve airway restriction. | ||||||||
| Inflammatory cell infiltrates | Increase | NA | Decrease | NA | |||||
| Airway restriction (Penh) | Increase | NA | NS | ||||||
| Limonene/ozone inhalation | |||||||||
| Hansen et al. [69] | 2013 | Adjuvant and inflammatory effects in mice after subchronic inhalation of allergen and ozone-initiated limonene reaction products | Limonene inhalation: | Limonene inhalation has no significant effect on inflammatory response in pre-sensitized mice. | No naive (baseline before OVA-sensitisation) data shown. | ||||
| OVA-specific IgE in serum (ELISA) | Increase | NA | NS | ||||||
| Eosinophil number in BALF | NS | NS | |||||||
| Lymphocyte number in BALF | Increase | NA | NS | ||||||
| Neutrophil number in BALF | Increase | NA | NS | ||||||
| Macrophage number in BALF | Increase | NA | NS | ||||||
| Ozone inhalation: | Limonene + ozone inhalation: | Limonene/ozone mixture reduces allergen-specific reactions in pre-sensitized mice. | No naive (baseline before OVA-sensitisation) data shown. | ||||||
| OVA-specific IgE in serum (ELISA) | Increase | NA | Increase | p < 0.05 | |||||
| Eosinophil number in BALF | Increase | NA | Decrease | p < 0.05 | |||||
| Lymphocyte number in BALF | Increase | NA | Decrease | NS | |||||
| Neutrophil number in BALF | Increase | NA | Decrease | p < 0.05 | |||||
| Macrophage number in BALF | Increase | NA | NS | ||||||
| Hansen et al. [70] | 2016 | Limonene and its ozone-initiated reaction products attenuate allergic lung inflammation in mice | Air inhalation: | Limonene inhalation: | Limonene potentially reduces airway inflammation in allergic mice, however no significances given. | Unclear graphical and verbal description of outcomes and signifi-cances. No OVA-only control. No comparisons between relevant groups. | |||
| OVA-specific IgE in serum (ELISA) | Increase | NA | Decrease | NA | |||||
| OVA-specific IgG1 in serum (ELISA) | Increase | NA | NS | ||||||
| Eosinophil number in BALF | Increase | NA | NS | ||||||
| Lymphocyte number in BALF | Increase | NA | Increase | NA | |||||
| Neutrophil number in BALF | Increase | NA | Decrease | NA | |||||
| Macrophage number in BALF | Increase | NA | Decrease | NA | |||||
| IL-5 expression in BALF | Increase | NA | Decrease | NA | |||||
| Ozone inhalation: | Limonene + ozone inhalation: | Limonene/ozone mixture potentially attenuates allergic inflammation and ozone-induced pulmonary irritation in allergic mice. | Unclear graphical and verbal description of outcomes and signifi-cances. No OVA-only control. No comparisons between relevant groups. | ||||||
| OVA-specific IgE in serum (ELISA) | Increase | NA | Decrease | NA | |||||
| OVA-specific IgG1 in serum (ELISA) | Increase | NA | Increase | NA | |||||
| Eosinophil number in BALF | Increase | NA | Decrease | p < 0.05 | |||||
| Lymphocyte number in BALF | Increase | NA | Decrease | NA | |||||
| Neutrophil number in BALF | NS | NA | |||||||
| Macrophage number in BALF | Increase | NA | Decrease | NA | |||||
| IL-5 expression in BALF | Increase | NA | Decrease | NA | |||||
| Linalool inhalation | |||||||||
| Nakamura et al. [72] | 2009 | Stress repression in restrained rats by R-(-)-linalool inhalation and gene expression profiling of their whole blood cells | % of neutrophils | Increase | p < 0.05 | Decrease | NA | Linalool inhalation reverts stress-induced changes in neutrophil and lymphocyte fractions. | |
| % of lymphocytes | Decrease | p < 0.05 | Increase | NA | |||||
| Fragrance inhalation | |||||||||
| Fujiwara et al. [71] | 1998 | Effects of a long-term inhalation of fragrances on the stress-induced immunosuppression in mice | Number of plaque forming cells (PFC) in spleen: | Exposure to natural fragrances reverses stress-induced thymic involution and activates the immune system. | |||||
| Lemon inhalation | Decrease | p < 0.05 | Increase | p < 0.05 | |||||
| Oakmoss inhalation | Decrease | p < 0.05 | Increase | p < 0.05 | |||||
| Labdanum inhalation | Decrease | p < 0.05 | Increase | p < 0.05 | |||||
| Tuberose inhalation | Decrease | p < 0.05 | Increase | p < 0.05 | |||||
| Thymic weight: | |||||||||
| Lemon inhalation | Decrease | p < 0.05 | Increase | p < 0.05 | |||||
| Oakmoss inhalation | Decrease | p < 0.05 | Increase | p < 0.05 | |||||
| Labdanum inhalation | Decrease | p < 0.05 | Increase | p < 0.05 | |||||
| Tuberose inhalation | Decrease | p < 0.05 | Increase | p < 0.05 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andersen, L.; Corazon, S.S.; Stigsdotter, U.K. Nature Exposure and Its Effects on Immune System Functioning: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 1416. https://doi.org/10.3390/ijerph18041416
Andersen L, Corazon SS, Stigsdotter UK. Nature Exposure and Its Effects on Immune System Functioning: A Systematic Review. International Journal of Environmental Research and Public Health. 2021; 18(4):1416. https://doi.org/10.3390/ijerph18041416
Chicago/Turabian StyleAndersen, Liisa, Sus Sola Corazon, and Ulrika Karlsson Stigsdotter. 2021. "Nature Exposure and Its Effects on Immune System Functioning: A Systematic Review" International Journal of Environmental Research and Public Health 18, no. 4: 1416. https://doi.org/10.3390/ijerph18041416
APA StyleAndersen, L., Corazon, S. S., & Stigsdotter, U. K. (2021). Nature Exposure and Its Effects on Immune System Functioning: A Systematic Review. International Journal of Environmental Research and Public Health, 18(4), 1416. https://doi.org/10.3390/ijerph18041416






