Local and Remote Ischemic Preconditioning Improves Sprint Interval Exercise Performance in Team Sport Athletes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
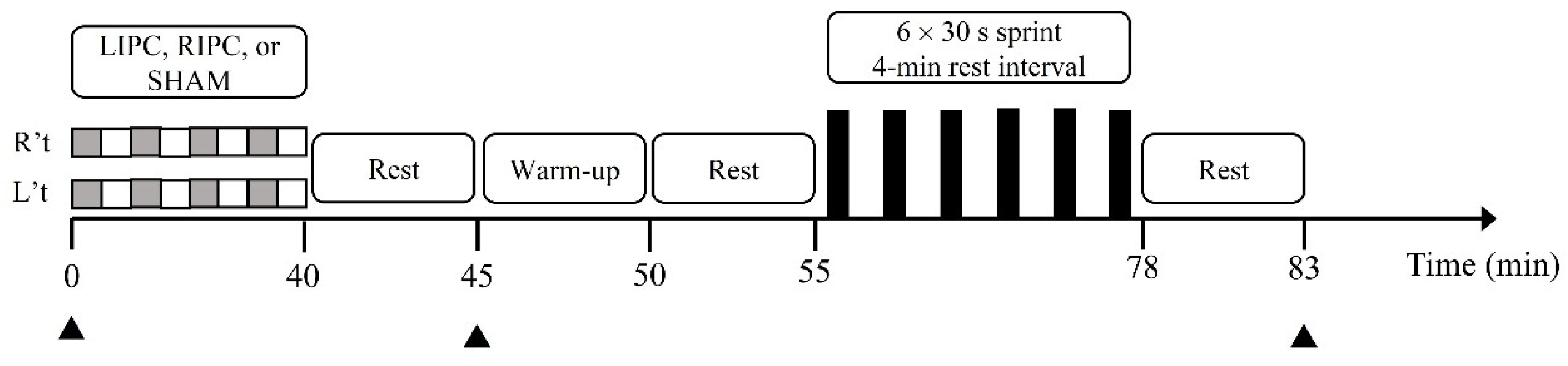
2.2. Experimental Design and Protocols
2.3. Incremental Cycling Test
2.4. IPC Protocols
2.5. Wingate-Based 6 × 30-s Sprint Interval Exercise
2.6. Blood Sampling and Analysis
2.7. Statistical Analysis
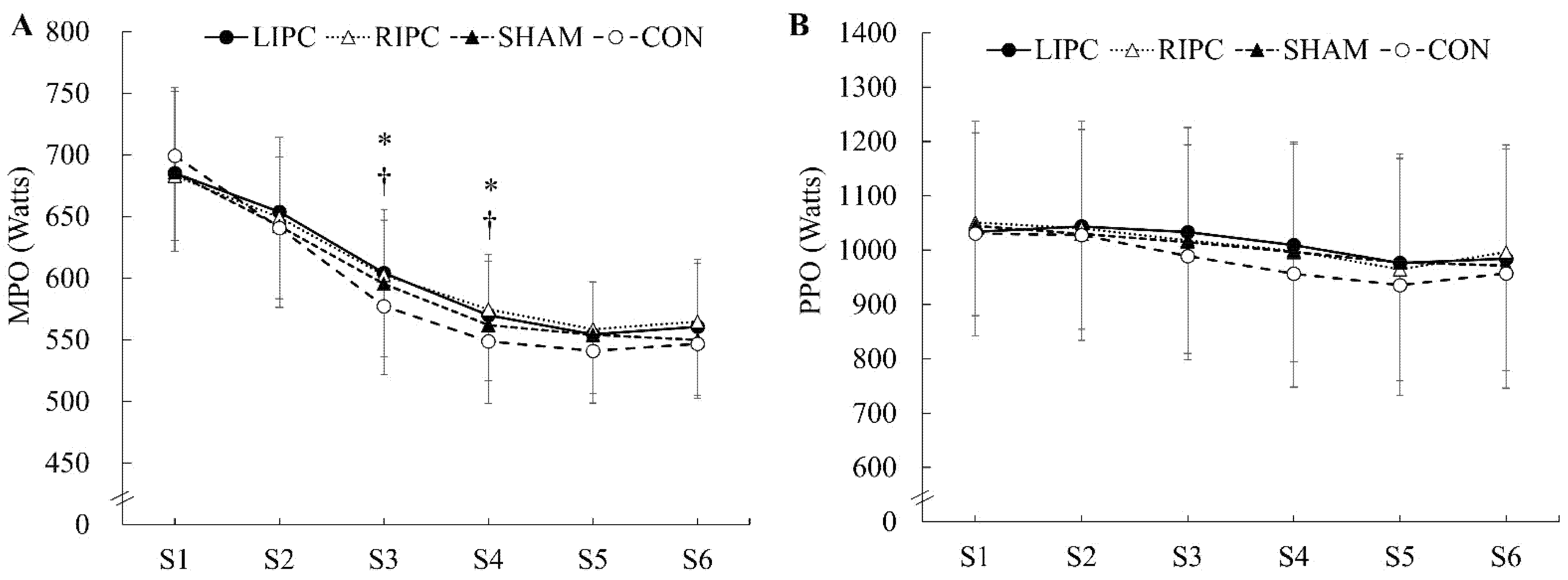
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- De Groot, P.C.; Thijssen, D.H.; Sanchez, M.; Ellenkamp, R.; Hopman, M.T. Ischemic preconditioning improves maximal performance in humans. Eur. J. Appl. Physiol. 2010, 108, 141–146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Incognito, A.V.; Burr, J.F.; Millar, P.J. The effects of ischemic preconditioning on human exercise performance. Sports Med. 2016, 46, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Salvador, A.F.; De Aguiar, R.A.; Lisboa, F.D.; Pereira, K.L.; Cruz, R.S.; Caputo, F. Ischemic preconditioning and exercise performance: A systematic review and meta-analysis. Int. J. Sports Physiol. Perform. 2016, 11, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Bishop, D.; Spencer, M. Determinants of repeated-sprint ability in well-trained team-sport athletes and endurance-trained athletes. J. Sports Med. Phys. Fitness 2004, 44, 1–7. [Google Scholar] [PubMed]
- Patterson, S.D.; Bezodis, N.E.; Glaister, M.; Pattison, J.R. The effect of ischemic preconditioning on repeated sprint cycling performance. Med. Sci. Sports Exerc. 2015, 47, 1652–1658. [Google Scholar] [CrossRef] [PubMed]
- Griffin, P.J.; Hughes, L.; Gissane, C.; Patterson, S.D. Effects of local versus remote ischemic preconditioning on repeated sprint running performance. J. Sports Med. Phys. Fitness 2019, 59, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, T.N.; Sabino-Carvalho, J.L.; Lopes, T.R.; Ribeiro, I.C.; Succi, J.E.; AC, D.A.S.; Silva, B.M. Ischemic preconditioning and repeated sprint swimming: A placebo and nocebo study. Med. Sci. Sports Exerc. 2016, 48, 1967–1975. [Google Scholar] [CrossRef] [PubMed]
- Gibson, N.; Mahony, B.; Tracey, C.; Fawkner, S.; Murray, A. Effect of ischemic preconditioning on repeated sprint ability in team sport athletes. J. Sports Sci. 2015, 33, 1182–1188. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cocking, S.; Ihsan, M.; Jones, H.; Hansen, C.; Timothy Cable, N.; Thijssen, D.; Wilson, M.G. Repeated sprint cycling performance is not enhanced by ischaemic preconditioning or muscle heating strategies. Eur. J. Sport Sci. 2021, 21, 166–175. [Google Scholar] [CrossRef] [Green Version]
- Gibson, N.; White, J.; Neish, M.; Murray, A. Effect of ischemic preconditioning on land-based sprinting in team-sport athletes. Int. J. Sports Physiol. Perform. 2013, 8, 671–676. [Google Scholar] [CrossRef]
- Thompson, K.M.A.; Whinton, A.K.; Ferth, S.; Spriet, L.L.; Burr, J.F. Ischemic preconditioning: No influence on maximal sprint acceleration performance. Int. J. Sports Physiol. Perform. 2018, 13, 986–990. [Google Scholar] [CrossRef]
- Lisbôa, F.D.; Turnes, T.; Cruz, R.S.; Raimundo, J.A.; Pereira, G.S.; Caputo, F. The time dependence of the effect of ischemic preconditioning on successive sprint swimming performance. J. Sci. Med. Sport 2017, 20, 507–511. [Google Scholar] [CrossRef]
- Cruz, R.S.; de Aguiar, R.A.; Turnes, T.; Salvador, A.F.; Caputo, F. Effects of ischemic preconditioning on short-duration cycling performance. Appl. Physiol. Nutr. Metab. 2016, 41, 825–831. [Google Scholar] [CrossRef]
- Paixao, R.C.; da Mota, G.R.; Marocolo, M. Acute effect of ischemic preconditioning is detrimental to anaerobic performance in cyclists. Int. J. Sports Med. 2014, 35, 912–915. [Google Scholar] [CrossRef]
- Bogdanis, G.C.; Nevill, M.E.; Boobis, L.H.; Lakomy, H.K.; Nevill, A.M. Recovery of power output and muscle metabolites following 30 s of maximal sprint cycling in man. J. Physiol. 1995, 482, 467–480. [Google Scholar] [CrossRef]
- Gibala, M.J.; McGee, S.L. Metabolic adaptations to short-term high-intensity interval training: A little pain for a lot of gain? Exerc. Sport Sci. Rev. 2008, 36, 58–63. [Google Scholar] [CrossRef]
- Cocking, S.; Landman, T.; Benson, M.; Lord, R.; Jones, H.; Gaze, D.; Thijssen, D.; George, K. The impact of remote ischemic preconditioning on cardiac biomarker and functional response to endurance exercise. Scand. J. Med. Sci. Sports 2017, 27, 1061–1069. [Google Scholar] [CrossRef] [Green Version]
- Jean-St-Michel, E.; Manlhiot, C.; Li, J.; Tropak, M.; Michelsen, M.M.; Schmidt, M.R.; McCrindle, B.W.; Wells, G.D.; Redington, A.N. Remote preconditioning improves maximal performance in highly trained athletes. Med. Sci. Sports Exerc. 2011, 43, 1280–1286. [Google Scholar] [CrossRef] [Green Version]
- Lalonde, F.; Curnier, D.Y. Can anaerobic performance be improved by remote ischemic preconditioning? J. Strength Cond. Res. 2015, 29, 80–85. [Google Scholar] [CrossRef]
- Barbosa, T.C.; Machado, A.C.; Braz, I.D.; Fernandes, I.A.; Vianna, L.C.; Nobrega, A.C.; Silva, B.M. Remote ischemic preconditioning delays fatigue development during handgrip exercise. Scand. J. Med. Sci. Sports 2015, 25, 356–364. [Google Scholar] [CrossRef]
- Da Silva Novaes, J.; da Silva Telles, L.G.; Monteiro, E.R.; da Silva Araujo, G.; Vingren, J.L.; Silva Panza, P.; Reis, V.M.; Laterza, M.C.; Vianna, J.M. Ischemic preconditioning improves resistance training session performance. J. Strength Cond. Res. 2020. online ahead of print. [Google Scholar] [CrossRef]
- Da Silva Telles, L.G.; Carelli, L.C.; Bráz, I.D.; Junqueira, C.; Monteiro, E.R.; Reis, V.M.; Vianna, J.M.; da Silva Novaes, J. Effects of ischemic preconditioning as a warm-up on leg press and bench press performance. J. Hum. Kinet. 2020, 75, 267–277. [Google Scholar] [CrossRef]
- Kraus, A.S.; Pasha, E.P.; Machin, D.R.; Alkatan, M.; Kloner, R.A.; Tanaka, H. Bilateral upper limb remote ischemic preconditioning improves anaerobic power. Open Sports Med. J. 2015, 9, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Buchheit, M.; Laursen, P.B. High-intensity interval training, solutions to the programming puzzle: Part I: Cardiopulmonary emphasis. Sports Med. 2013, 43, 313–338. [Google Scholar] [CrossRef]
- Cheng, C.F.; Hsu, W.C.; Kuo, Y.H.; Chen, T.W.; Kuo, Y.C. Acute effect of inspiratory resistive loading on sprint interval exercise performance in team-sport athletes. Respir. Physiol. Neurobiol. 2020, 282, 103531. [Google Scholar] [CrossRef]
- Costa, F.; Christensen, N.J.; Farley, G.; Biaggioni, I. NO modulates norepinephrine release in human skeletal muscle: Implications for neural preconditioning. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 280, R1494–R1498. [Google Scholar] [CrossRef]
- Rassaf, T.; Totzeck, M.; Hendgen-Cotta, U.B.; Shiva, S.; Heusch, G.; Kelm, M. Circulating nitrite contributes to cardioprotection by remote ischemic preconditioning. Circ. Res. 2014, 114, 1601–1610. [Google Scholar] [CrossRef]
- Kida, M.; Fujiwara, H.; Ishida, M.; Kawai, C.; Ohura, M.; Miura, I.; Yabuuchi, Y. Ischemic preconditioning preserves creatine phosphate and intracellular pH. Circulation 1991, 84, 2495–2503. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andreas, M.; Schmid, A.I.; Keilani, M.; Doberer, D.; Bartko, J.; Crevenna, R.; Moser, E.; Wolzt, M. Effect of ischemic preconditioning in skeletal muscle measured by functional magnetic resonance imaging and spectroscopy: A randomized crossover trial. J. Cardiovasc. Magn. Reson. 2011, 13, 32. [Google Scholar] [CrossRef] [Green Version]
- Bogdanis, G.C.; Nevill, M.E.; Boobis, L.H.; Lakomy, H.K. Contribution of phosphocreatine and aerobic metabolism to energy supply during repeated sprint exercise. J. Appl. Physiol. 1996, 80, 876–884. [Google Scholar] [CrossRef] [PubMed]
- Griffin, P.J.; Ferguson, R.A.; Gissane, C.; Bailey, S.J.; Patterson, S.D. Ischemic preconditioning enhances critical power during a 3 min all-out cycling test. J. Sports Sci. 2018, 36, 1038–1043. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Midgley, A.W.; McNaughton, L.R.; Wilkinson, M. Is there an optimal training intensity for enhancing the maximal oxygen uptake of distance runners?: Empirical research findings, current opinions, physiological rationale and practical recommendations. Sports Med. 2006, 36, 117–132. [Google Scholar] [CrossRef] [PubMed]
- Paradis-Deschênes, P.; Joanisse, D.R.; Mauriège, P.; Billaut, F. Ischemic preconditioning enhances aerobic adaptations to sprint-interval training in athletes without altering systemic hypoxic signaling and immune function. Front. Sports Act. Living 2020, 2, 41. [Google Scholar] [CrossRef] [PubMed]
- Marocolo, M.; da Mota, G.R.; Pelegrini, V.; Appell Coriolano, H.J. Are the beneficial effects of ischemic preconditioning on performance partly a placebo effect? Int. J. Sports Med. 2015, 36, 822–825. [Google Scholar] [CrossRef]
- Marocolo, M.; Willardson, J.M.; Marocolo, I.C.; da Mota, G.R.; Simão, R.; Maior, A.S. Ischemic preconditioning and placebo intervention improves resistance exercise performance. J. Strength Cond. Res. 2016, 30, 1462–1469. [Google Scholar] [CrossRef]
- Cheung, C.P.; Slysz, J.T.; Burr, J.F. Ischemic preconditioning: Improved cycling performance despite nocebo expectation. Int. J. Sports Physiol. Perform. 2019, 15, 354–360. [Google Scholar] [CrossRef]
- Jensen, K.B.; Kaptchuk, T.J.; Kirsch, I.; Raicek, J.; Lindstrom, K.M.; Berna, C.; Gollub, R.L.; Ingvar, M.; Kong, J. Nonconscious activation of placebo and nocebo pain responses. Proc. Natl. Acad. Sci. USA 2012, 109, 15959–15964. [Google Scholar] [CrossRef] [Green Version]
| LIPC (n = 15) | RIPC (n = 15) | SHAM (n = 15) | CON (n = 15) | |
|---|---|---|---|---|
| Total work (kJ) | 108.3 ± 8.9 * | 108.4 ± 6.9 * | 107.1 ± 8.6 | 106.0 ± 8.6 |
| Percentage decrement score (%) | ||||
| PPO | 5.4 ± 2.3 * | 6.2 ± 3.1 | 6.5 ± 2.9 | 7.6 ± 3.8 |
| MPO | 11.9 ± 4.7 * | 11.9 ± 4.6 * | 13.1 ± 4.1 | 15.2 ± 5.3 |
| Accumulated exercise time (s) | ||||
| O2max | 95.5 ± 52.7 | 99.9 ± 53.9 | 81.2 ± 62.0 | 95.2 ± 59.5 |
| O2max | 34.5 ± 28.0 | 36.7 ± 33.9 | 28.9 ± 32.9 | 37.5 ± 40.1 |
| O2max | 9.1 ± 11.7 | 10.5 ± 13.1 | 7.5 ± 11.5 | 11.6 ± 17.3 |
| Lactate (mmol/L) | ||||
| Baseline | 0.96 ± 0.21 | 1.01 ± 0.23 | 0.95 ± 0.27 | 0.95 ± 0.24 |
| 5 min after treatment | 1.02 ± 0.24 | 1.04 ± 0.17 | 0.95 ± 0.15 | - |
| 5 min after SIE | 11.09 ± 1.73 | 10.83 ± 2.02 | 10.62 ± 2.89 | 11.59 ± 2.21 |
| pH | ||||
| Baseline | 7.37 ± 0.02 | 7.39 ± 0.03 | 7.37 ± 0.02 | 7.38 ± 0.02 |
| 5 min after treatment | 7.39 ± 0.02 | 7.40 ± 0.02 | 7.39 ± 0.02 | - |
| 5 min after SIE | 7.20 ± 0.05 | 7.20 ± 0.05 | 7.20 ± 0.05 | 7.19 ± 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, C.-F.; Kuo, Y.-H.; Hsu, W.-C.; Chen, C.; Pan, C.-H. Local and Remote Ischemic Preconditioning Improves Sprint Interval Exercise Performance in Team Sport Athletes. Int. J. Environ. Res. Public Health 2021, 18, 10653. https://doi.org/10.3390/ijerph182010653
Cheng C-F, Kuo Y-H, Hsu W-C, Chen C, Pan C-H. Local and Remote Ischemic Preconditioning Improves Sprint Interval Exercise Performance in Team Sport Athletes. International Journal of Environmental Research and Public Health. 2021; 18(20):10653. https://doi.org/10.3390/ijerph182010653
Chicago/Turabian StyleCheng, Ching-Feng, Yu-Hsuan Kuo, Wei-Chieh Hsu, Chu Chen, and Chi-Hsueh Pan. 2021. "Local and Remote Ischemic Preconditioning Improves Sprint Interval Exercise Performance in Team Sport Athletes" International Journal of Environmental Research and Public Health 18, no. 20: 10653. https://doi.org/10.3390/ijerph182010653
APA StyleCheng, C.-F., Kuo, Y.-H., Hsu, W.-C., Chen, C., & Pan, C.-H. (2021). Local and Remote Ischemic Preconditioning Improves Sprint Interval Exercise Performance in Team Sport Athletes. International Journal of Environmental Research and Public Health, 18(20), 10653. https://doi.org/10.3390/ijerph182010653






