The Effect of Nut Consumption on Diet Quality, Cardiometabolic and Gastrointestinal Health in Children: A Systematic Review of Randomized Controlled Trials
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol and Registration
2.1.1. Eligibility Criteria
2.1.2. Types of Studies
2.1.3. Types of Participants
2.1.4. Types of Intervention
2.1.5. Exclusion Criteria
2.1.6. Types of Outcome Measures
2.1.7. Limitations
2.2. Databases and Search Strategy
2.3. Study Selection
2.4. Data Extraction
2.5. Quality Assessment
2.6. Data Analysis
3. Results
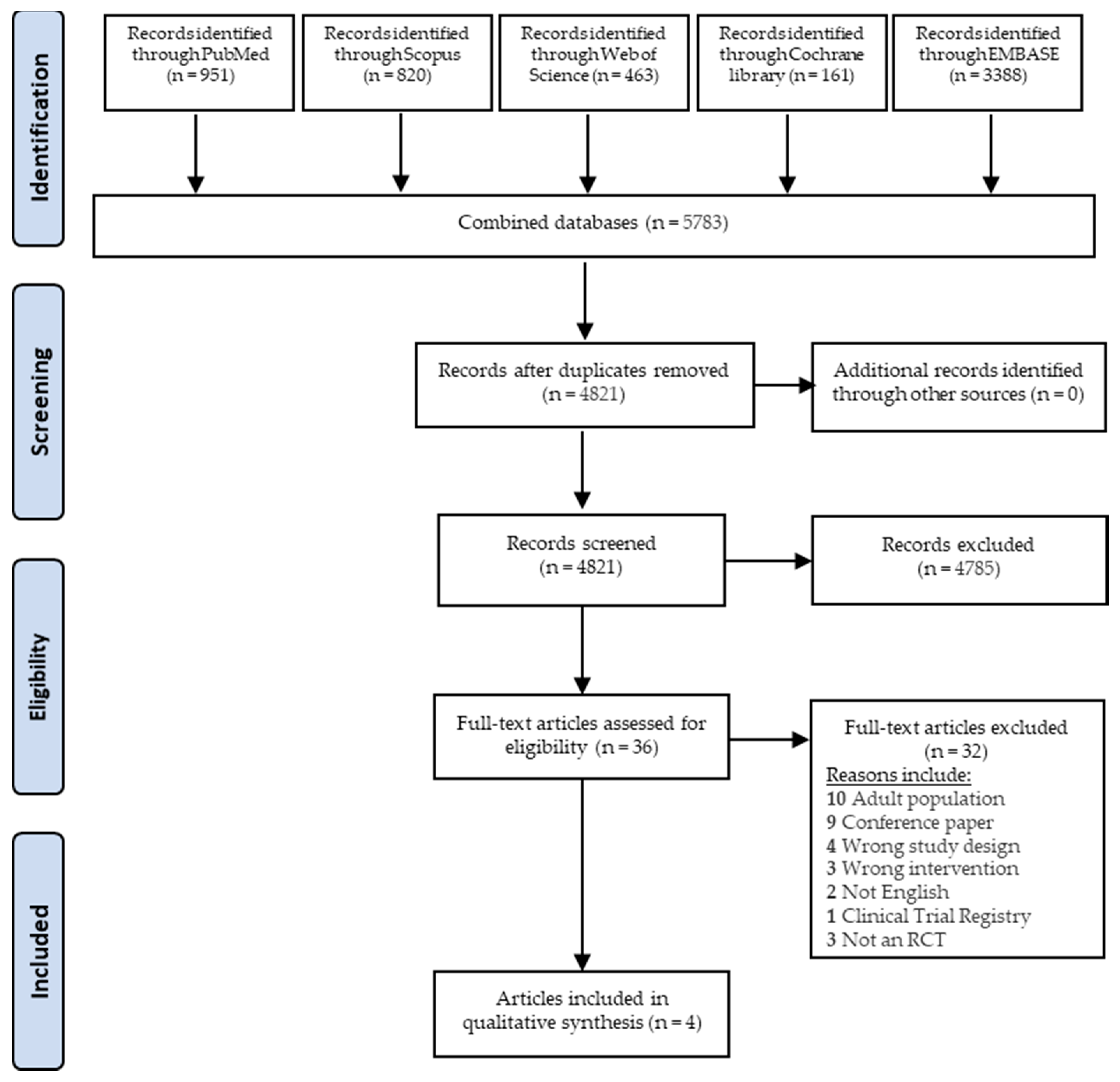
3.1. Study Selection
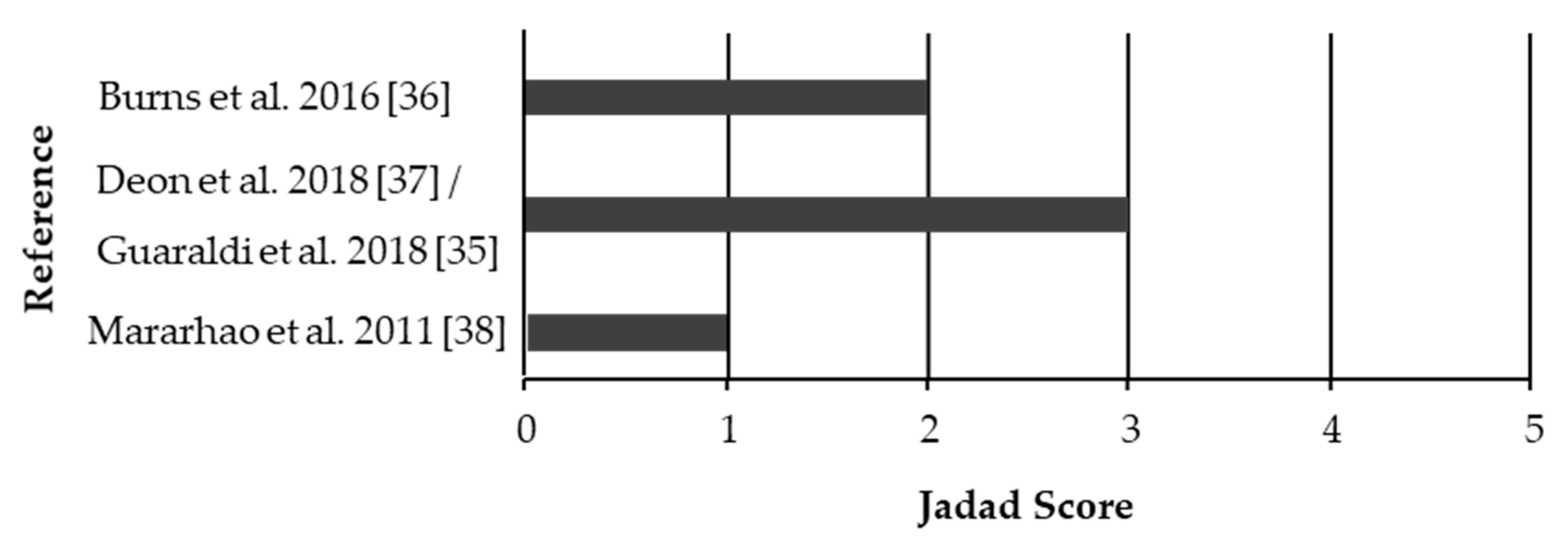
3.2. Quality Assesment
3.3. Study Characteristics
3.4. Dietary Profiles
3.5. Cardiometabolic Health
3.6. Gastrointestinal Health
4. Discussion
4.1. Diet Quality
4.2. Cardiometabolic Health
4.3. Gastrointestinal Health
4.4. Limitations and Future Studies
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Barbour, J.A.; Howe, P.R.; Buckley, J.D.; Wright, G.C.; Bryan, J.; Coates, A.M. Lower energy intake following consumption of Hi-oleic and regular peanuts compared with iso-energetic consumption of potato crisps. Appetite 2014, 82, 124–130. [Google Scholar] [CrossRef]
- Dikariyanto, V.; Berry, S.E.; Pot, G.K.; Francis, L.; Smith, L.; Hall, W.L. Tree nut snack consumption is associated with better diet quality and CVD risk in the UK adult population: National Diet and Nutrition Survey (NDNS) 2008–2014. Public Health Nutr. 2020, 23, 3160–3169. [Google Scholar] [CrossRef] [PubMed]
- Rehm, C.D.; Drewnowski, A. Replacing American snacks with tree nuts increases consumption of key nutrients among US children and adults: Results of an NHANES modeling study. Nutr. J. 2017, 16, 17. [Google Scholar] [CrossRef] [PubMed]
- Bolling, B.W.; Chen, C.-Y.O.; McKay, D.L.; Blumberg, J.B. Tree nut phytochemicals: Composition, antioxidant capacity, bioactivity, impact factors. A systematic review of almonds, Brazils, cashews, hazelnuts, macadamias, pecans, pine nuts, pistachios and walnuts. Nutr. Res. Rev. 2011, 24, 244–275. [Google Scholar] [CrossRef] [PubMed]
- Coates, A.M.; Howe, P.R. Edible nuts and metabolic health. Curr. Opin. Lipidol. 2007, 18, 25–30. [Google Scholar] [CrossRef]
- Coates, A.M.; Hill, A.M.; Tan, S.Y. Nuts and Cardiovascular Disease Prevention. Curr. Atheroscler. Rep. 2018, 20, 48. [Google Scholar] [CrossRef] [PubMed]
- De Souza, R.G.M.; Schincaglia, R.M.; Pimentel, G.D.; Mota, J.F. Nuts and Human Health Outcomes: A Systematic Review. Nutrients 2017, 9, 1311. [Google Scholar] [CrossRef]
- Ros, E. Health Benefits of Nut Consumption. Nutrients 2010, 2, 652–682. [Google Scholar] [CrossRef]
- O’Neil, C.E.; Keast, D.R.; Nicklas, T.A.; Fulgoni, V.L. Out-of-hand nut consumption is associated with improved nutrient intake and health risk markers in US children and adults: National Health and Nutrition Examination Survey 1999–2004. Nutr. Res. 2012, 32, 185–194. [Google Scholar] [CrossRef]
- Lee, Y.; Lee, S.; Lee, K.; Lee, K.-H.; Baik, I. Eating patterns of children’s favorite foods and its related factors among elementary, middle, and high school students in Korea. Nutr. Res. Pr. 2017, 11, 517–524. [Google Scholar] [CrossRef]
- Temme, E.H.M.; Bakker, H.M.; Seves, S.M.; Verkaik-Kloosterman, J.; Dekkers, A.L.; Van Raaij, J.M.; Ocké, M.C. How may a shift towards a more sustainable food consumption pattern affect nutrient intakes of Dutch children? Public Health Nutr. 2015, 18, 2468–2478. [Google Scholar] [CrossRef] [PubMed]
- Moreno, J.P.; Johnston, C.A.; El-Mubasher, A.A.; Papaioannou, M.A.; Tyler, C.; Gee, M.; Foreyt, J.P. Peanut consumption in adolescents is associated with improved weight status. Nutr. Res. 2013, 33, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.C.; Tey, S.L.; Gray, A.; Chisholm, A.; Smith, C.; Fleming, E.; Parnell, W. Nut consumption is associated with better nutrient intakes: Results from the 2008/09 New Zealand Adult Nutrition Survey. Br. J. Nutr. 2015, 115, 105–112. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, C.E.; Keast, D.R.; Fulgoni, V.L.; Nicklas, T.A. Tree nut consumption improves nutrient intake and diet quality in US adults: An analysis of National Health and Nutrition Examination Survey (NHANES) 1999–2004. Asia Pac. J. Clin. Nutr. 2010, 19, 142–150. [Google Scholar] [PubMed]
- Steffen, L.M.; Van Horn, L.; Daviglus, M.L.; Zhou, X.; Reis, J.P.; Loria, C.M.; Jacobs, D.R.; Duffey, K.J. A modified Mediterranean diet score is associated with a lower risk of incident metabolic syndrome over 25 years among young adults: The CARDIA (Coronary Artery Risk Development in Young Adults) study. Br. J. Nutr. 2014, 112, 1654–1661. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Nut consumption and risk of cardiovascular disease, total cancer, all-cause and cause-specific mortality: A systematic review and dose-response meta-analysis of prospective studies. BMC Med. 2016, 14, 207. [Google Scholar] [CrossRef]
- Guasch-Ferre, M.; Liu, X.; Malik, V.S.; Sun, Q.; Willett, W.C.; Manson, J.E.; Rexrode, K.M.; Li, Y.; Hu, F.B.; Bhupathiraju, S.N. Nut Consumption and Risk of Cardiovascular Disease. J. Am. Coll. Cardiol. 2017, 70, 2519–2532. [Google Scholar] [CrossRef]
- Colpo, E.; Vilanova, C.D.D.A.; Reetz, L.G.B.; Duarte, M.M.M.F.; Farias, I.L.G.; Muller, E.I.; Muller, A.L.H.; Flores Érico, M.M.; Wagner, R.; Rocha, J.B.T. A Single Consumption of High Amounts of the Brazil Nuts Improves Lipid Profile of Healthy Volunteers. J. Nutr. Metab. 2013, 2013, 1–7. [Google Scholar] [CrossRef]
- Del Gobbo, L.C.; Falk, M.C.; Feldman, R.; Lewis, K.; Mozaffarian, D. Effects of tree nuts on blood lipids, apolipoproteins, and blood pressure: Systematic review, meta-analysis, and dose-response of 61 controlled intervention trials. Am. J. Clin. Nutr. 2015, 102, 1347–1356. [Google Scholar] [CrossRef]
- Hu, F.B.; Stampfer, M.J. Nut consumption and risk of coronary heart disease: A review of epidemiologic evidence. Curr. Atheroscler. Rep. 1999, 1, 204–209. [Google Scholar] [CrossRef]
- Fitzgerald, E.; Lambert, K.; Stanford, J.; Neale, E.P. The effect of nut consumption (tree nuts and peanuts) on the gut microbiota of humans: A systematic review. Br. J. Nutr. 2020, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Cândido, F.G.; Valente, F.X.; Grześkowiak, Ł.M.; Moreira, A.P.B.; Rocha, D.M.U.P.; de Cássia Gonçalves Alfenas, R. Impact of dietary fat on gut microbiota and low-grade systemic inflammation: Mechanisms and clinical implications on obesity. Int. J. Food Sci. Nutr. 2018, 69, 125–143. [Google Scholar] [CrossRef] [PubMed]
- Le Chatelier, E.; Nielsen, T.; Qin, J.; Prifti, E.; Hildebrand, F.; Falony, G.; Almeida, M.; Arumugam, M.; Batto, J.-M.; Kennedy, S.; et al. Richness of human gut microbiome correlates with metabolic markers. Nature 2013, 500, 541–546. [Google Scholar] [CrossRef] [PubMed]
- McWilliam, V.; Koplin, J.J.; Lodge, C.; Tang, M.; Dharmage, S.C.; Allen, K.J. The Prevalence of Tree Nut Allergy: A Systematic Review. Curr. Allergy Asthma Rep. 2015, 15, 54. [Google Scholar] [CrossRef] [PubMed]
- Weinberger, T.; Sicherer, S. Current perspectives on tree nut allergy: A review. J. Asthma Allergy 2018, 11, 41–51. [Google Scholar] [CrossRef]
- Simopoulos, A.P. The Mediterranean Diets: What Is So Special about the Diet of Greece? The Scientific Evidence. J. Nutr. 2001, 131, 3065S–3073S. [Google Scholar] [CrossRef] [PubMed]
- Vyncke, K.E.; Libuda, L.; De Vriendt, T.; Moreno, L.A.; Van Winckel, M.; Manios, Y.; Gottrand, F.; Molnar, D.; Vanaelst, B.; Sjöström, M.; et al. Dietary fatty acid intake, its food sources and determinants in European adolescents: The HELENA (Healthy Lifestyle in Europe by Nutrition in Adolescence) Study. Br. J. Nutr. 2012, 108, 2261–2273. [Google Scholar] [CrossRef] [PubMed]
- Sjöberg, A.; Hallberg, L.; Höglund, D.; Hulthén, L. Meal pattern, food choice, nutrient intake and lifestyle factors in The Göteborg Adolescence Study. Eur. J. Clin. Nutr. 2003, 57, 1569–1578. [Google Scholar] [CrossRef] [PubMed]
- Emmett, P.M.; Jones, L.R. Diet, growth, and obesity development throughout childhood in the Avon Longitudinal Study of Parents and Children. Nutr. Rev. 2015, 73, 175–206. [Google Scholar] [CrossRef] [PubMed]
- Nowak-Węgrzyn, A.; Conover-Walker, M.K.; Wood, R.A. Food-allergic reactions in schools and preschools. Arch. Pediatr. Adolesc. Med. 2001, 155, 790–795. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Koplin, J.J.; Dharmage, S.C.; Field, M.J.; Sawyer, S.M.; McWilliam, V.; Peters, R.L.; Gurrin, L.C.; Vuillermin, P.J.; Douglass, J.; et al. Prevalence of clinic-defined food allergy in early adolescence: The SchoolNuts study. J. Allergy Clin. Immunol. 2018, 141, 391–398.e4. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; A Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: Explanation and elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef] [PubMed]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control. Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
- Guaraldi, F.; Deon, V.; Del Bo’, C.; Vendrame, S.; Porrini, M.; Riso, P.; Guardamagna, O. Effect of short-term hazelnut consumption on DNA damage and oxidized LDL in children and adolescents with primary hyperlipidemia: A randomized controlled trial. J. Nutr. Biochem. 2018, 57, 206–211. [Google Scholar] [CrossRef]
- Burns, A.M.; Zitt, M.A.; Rowe, C.C.; Langkamp-Henken, B.; Mai, V.; Ukhanova, M.; Christman, M.; Dahl, W.J. Diet quality improves for parents and children when almonds are incorporated into their daily diet: A randomized, crossover study. Nutr. Res. 2016, 36, 80–89. [Google Scholar] [CrossRef]
- Deon, V.; Del Bo’, C.; Guaraldi, F.; Abello, F.; Belviso, S.; Porrini, M.; Riso, P.; Guardamagna, O. Effect of hazelnut on serum lipid profile and fatty acid composition of erythrocyte phospholipids in children and adolescents with primary hyperlipidemia: A randomized controlled trial. Clin. Nutr. 2018, 37, 1193–1201. [Google Scholar] [CrossRef]
- Maranhão, P.A.; Kraemer-Aguiar, L.G.; De Oliveira, C.L.; Kuschnir, M.C.; Vieira, Y.R.; Souza, M.D.G.C.D.; Koury, J.C.; Bouskela, E. Brazil nuts intake improves lipid profile, oxidative stress and microvascular function in obese adolescents: A randomized controlled trial. Nutr. Metab. 2011, 8, 32. [Google Scholar] [CrossRef]
- Andersen, R.; Biltoft-Jensen, A.; Andersen, E.W.; Ege, M.; Christensen, T.; Ygil, K.H.; Thorsen, A.V.; Damsgaard, C.T.; Astrup, A.; Michaelsen, K.F.; et al. Effects of school meals based on the New Nordic Diet on intake of signature foods: A randomised controlled trial. The OPUS School Meal Study. Br. J. Nutr. 2015, 114, 772–779. [Google Scholar] [CrossRef]
- Tey, S.L.; Brown, R.C.; Gray, A.; Chisholm, A.; Delahunty, C. Nuts Improve Diet Quality Compared to Other Energy-Dense Snacks While Maintaining Body Weight. J. Nutr. Metab. 2011, 2011, 1–11. [Google Scholar] [CrossRef]
- Murakami, K. Associations between nutritional quality of meals and snacks assessed by the Food Standards Agency nutrient profiling system and overall diet quality and adiposity measures in British children and adolescents. Nutrients 2018, 49, 57–65. [Google Scholar] [CrossRef]
- Forjaz, C.L.D.M.; Bel-Serrat, S.; Manios, Y.; Molnar, D.; Kafatos, A.; Cuenca-García, M.; Huybrechts, I.; Sette, S.; Widhalm, K.; Stehle, P.; et al. Dietary protein and amino acids intake and its relationship with blood pressure in adolescents: The HELENA STUDY. Eur. J. Public Health 2015, 25, 450–456. [Google Scholar] [CrossRef]
- Arouca, A.; Michels, N.; Moreno, L.A.; González-Gil, E.M.; Marcos, A.; Gómez, S.; Díaz, L.E.; Widhalm, K.; Molnár, D.; Manios, Y.; et al. Associations between a Mediterranean diet pattern and inflammatory biomarkers in European adolescents. Eur. J. Nutr. 2017, 57, 1747–1760. [Google Scholar] [CrossRef] [PubMed]
- Bel-Serrat, S.; Mouratidou, T.; Börnhorst, C.; Peplies, J.; De Henauw, S.; Molnár, D.; Siani, A.; Tornaritis, M.; Veidebaum, T. Food consumption and cardiovascular risk factors in European children: The IDEFICS study. Pediatr. Obes. 2012, 8, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Berding, K.; Holscher, H.D.; Arthur, A.E.; Donovan, S.M. Fecal microbiome composition and stability in 4- to 8-year old children is associated with dietary patterns and nutrient intake. J. Nutr. Biochem. 2018, 56, 165–174. [Google Scholar] [CrossRef] [PubMed]
- Lamuela-Raventós, R.; Onge, M.-P.S. Prebiotic nut compounds and human microbiota. Crit. Rev. Food Sci. Nutr. 2017, 57, 3154–3163. [Google Scholar] [CrossRef]
- Holscher, H.D.; Guetterman, H.M.; Swanson, K.S.; An, R.; Matthan, N.R.; Lichtenstein, A.H.; Novotny, J.A.; Baer, D.J. Walnut Consumption Alters the Gastrointestinal Microbiota, Microbially Derived Secondary Bile Acids, and Health Markers in Healthy Adults: A Randomized Controlled Trial. J. Nutr. 2018, 148, 861–867. [Google Scholar] [CrossRef]
- Bamberger, C.; Rossmeier, A.; Lechner, K.; Wu, L.; Waldmann, E.; Fischer, S.; Stark, R.G.; Altenhofer, J.; Henze, K.; Parhofer, K.G. A Walnut-Enriched Diet Affects Gut Microbiome in Healthy Caucasian Subjects: A Randomized, Controlled Trial. Nutrients 2018, 10, 244. [Google Scholar] [CrossRef]
- Holscher, H.D.; Taylor, A.M.; Swanson, K.S.; A Novotny, J.; Baer, D.J. Almond Consumption and Processing Affects the Composition of the Gastrointestinal Microbiota of Healthy Adult Men and Women: A Randomized Controlled Trial. Nutrients 2018, 10, 126. [Google Scholar] [CrossRef]
- Hernández-Alonso, P.; Cañueto, D.; Giardina, S.; Salas-Salvadó, J.; Cañellas, N.; Correig, X.; Bulló, M. Effect of pistachio consumption on the modulation of urinary gut microbiota-related metabolites in prediabetic subjects. J. Nutr. Biochem. 2017, 45, 48–53. [Google Scholar] [CrossRef]
- Schulz, K.F.; The CONSORT Group; Altman, D.G.; Moher, D. CONSORT 2010 Statement: Updated guidelines for reporting parallel group randomised trials. BMC Med. 2010, 8, 18. [Google Scholar] [CrossRef]
| Parameter | Description |
|---|---|
| P-Population | Humans, children ≤ 18 years old |
| I-Intervention/variable of interest | Nut intake (whole, chopped, pastes) |
| C-Comparator | No nut intake, Received dietary advice, Placebo |
| O-Outcome | Cardiometabolic Health, Diet Quality, Adiposity and Gut Health |
| S-Study design | Randomized controlled trials |
| First Author (Year) Country | Number (N) and Characteristics of Participants | Study Design, (Length of Intervention) | Intervention Group(s) | Control Group | Health Outcomes from Nut Consumption |
|---|---|---|---|---|---|
| Burns et al. (2016) [36] USA | N = 29, Healthy males and females (15/14) 4 ± 0.2 years | Randomized, 2-phase crossover study (Each phase was 3 weeks with a 6-week wash-out) | 15 g of almonds (whole, skin on) or the equivalent amount in almond butter per day | No almonds or products containing almonds | Almond group compared to control group after intervention (3 weeks): ↓ GSRS: constipation score, (d = −1.11) Almond group at final week compared to baseline: Significant changes in the prevalence of various bacterial signatures at the genus and species level, * Almond group compared to control group during intervention (throughout 3 weeks): ↑ HEI component score for total protein foods, (d = 1.01) ↑ HEI component score for fatty acids, (d = 1.0) ↑ HEI component score for seafood and plant proteins, (d = 2.47) ↑ HEI component score for empty calories (high energy/low nutrients), (d = 0.62) ↑ Total HEI Score, (d = 0.84) No change in HEI component score for total fruit, total vegetables, greens and beans, dairy, refined grain, sodium, whole fruit, whole grain. No change in mean energy intake or nutrient intake for protein, fat, monounsaturated fat, total fiber, potassium, energy, carbohydrate, sodium, vitamin C No changes in the microbiota profile (quantiles of Bifidobacteria spp and lactic acid bacteria, microbiota diversity measures (Shannon diversity index, inverse Simpson diversity index) and GSRS (Diarrhea, Abdominal pain, Indigestion, Reflux) |
| Deon et al. (2018) [37] Italy | N = 60, Hyperlipemic males and females (34/26) 11.6 ± 2.6 years | Randomized, single blind, three-arm, parallel- study (8 weeks) | Group 1: hazelnuts with skin (HZN + S), roasted 15–30 g/day, based on body weight Group 2-hazelnuts without skin (HZN-S), roasted 15–30 g/day, based on body weight | Group 3-dietary advice for hyperlipidemia (do not include nuts in diet) | Intake of hazelnuts with and without skin at week 8 compared to baseline: ↑ total Fat% of energy, (d = HZN + S: 1.13, HZN-S: 0.64) ↑ MUFA% of energy, (d = HZN + S: 0.45, HZN-S: 1.35) ↑ PUFA% of energy, (d = HZN + S: 1.82, HZN-S: 1.4) ↑ Omega 3% of energy, (d = HZN + S: 1.63, HZN-S: 0.8) ↑ Omega 6% of energy, (d = HZN + S: 1.95, HZN-S: 1.59) ↓ Carbohydrate intake% of energy, (d = HZN + S: −0.91, HZN-S: −0.42) No change in blood pressure, BMI, weight, daily energy and nutrient intakes (protein, SFA, fiber and cholesterol) No change in serum lipid profile and fatty acid composition of erythrocyte membranes except for: ↑ total MUFA composition of erythrocytes%, (d = HZN + S: 0.48, HZN-S: 0.57) ↑ the ratio of MUFA/PUFA composition of erythrocytes%, (d = HZN + S: 0.57, HZN-S: 0.67) |
| Guaraldi et al. (2018) [35] Italy | N = 60, Hyperlipidemic males and females, (34/26) 11.6 ± 2.6 years | Randomized, single blind, three-arm, parallel study (8 weeks) | Group 1- raw hazelnuts with skin (HZN + S) 15–30 g/day Group 2- hazelnuts without skin (HZN-S) 15–30 g/day | Group 3- dietary advice for hyperlipidemia (do not include nuts in diet) | Intake of hazelnuts with and without skin: ↓ levels of endogenous DNA damage (FGP-sensitive sites (% DNA in tail), (d = HZN + S: −1.13, HZN-S: −1.15) No change in level of DNA strand breaks and oxidatively induced DNA damage No change in levels of ox-LDL, ox-LDL/LDL and ox-LDL/HDL ratio |
| Maranhao et al. (2011) [38] Brazil | N = 17 Obese females 15.4 ± 2.0 years | Randomized, non-blinded, two-arm parallel study (16 weeks) | Brazil nuts, 15–25 g/day | Do not eat nuts and consume one capsule (placebo tablet) per day containing lactose | Intake of Brazil nuts after 16 weeks compared to control: ↓ triglycerides (mg/dL), * ↓ total cholesterol (mg/dL), * ↓ concentration of LDL (mg/dL), * No change in biomarkers with antioxidant capacity (GPX-3 and 8-epi-PGF2a) except for: Intake of Brazil nuts after 16 weeks compared to baseline: ↑ selenium (μg/L) (anti-inflammatory nutrient), * ↓ oxidized LDL (ng/mL), * ↑ RBCV max (mm/s) (Improved microvascular reactivity (compared to baseline and compared to control), * No changes in Body mass, BMI, WC, fasting insulin, glucose, HOMA, CRP, HDL-c No change in microcirculatory parameters (functional capillary density, TRBCV max, Afferent diameter, Apical diameter, Efferent diameter) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mead, L.C.; Hill, A.M.; Carter, S.; Coates, A.M. The Effect of Nut Consumption on Diet Quality, Cardiometabolic and Gastrointestinal Health in Children: A Systematic Review of Randomized Controlled Trials. Int. J. Environ. Res. Public Health 2021, 18, 454. https://doi.org/10.3390/ijerph18020454
Mead LC, Hill AM, Carter S, Coates AM. The Effect of Nut Consumption on Diet Quality, Cardiometabolic and Gastrointestinal Health in Children: A Systematic Review of Randomized Controlled Trials. International Journal of Environmental Research and Public Health. 2021; 18(2):454. https://doi.org/10.3390/ijerph18020454
Chicago/Turabian StyleMead, Lauren C, Alison M Hill, Sharayah Carter, and Alison M Coates. 2021. "The Effect of Nut Consumption on Diet Quality, Cardiometabolic and Gastrointestinal Health in Children: A Systematic Review of Randomized Controlled Trials" International Journal of Environmental Research and Public Health 18, no. 2: 454. https://doi.org/10.3390/ijerph18020454
APA StyleMead, L. C., Hill, A. M., Carter, S., & Coates, A. M. (2021). The Effect of Nut Consumption on Diet Quality, Cardiometabolic and Gastrointestinal Health in Children: A Systematic Review of Randomized Controlled Trials. International Journal of Environmental Research and Public Health, 18(2), 454. https://doi.org/10.3390/ijerph18020454






