Influence of Female Sex Hormones on Ultra-Running Performance and Post-Race Recovery: Role of Testosterone
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Population and Ethical Approval
2.3. Hematological Variables
2.4. Body Mass Index (BMI) and Body Composition Assessment
2.5. Loss of Strength Measurement
2.6. Statistical Methods
3. Results
4. Discussion
5. Limitations
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- The State of Running 2019 | RunRepeat. Available online: https://runrepeat.com/state-of-running (accessed on 4 September 2021).
- Stöhr, A.; Nikolaidis, P.T.; Villiger, E.; Sousa, C.V.; Scheer, V.; Hill, L.; Knechtle, B. An analysis of participation and performance of 2067 100-km ultra-marathons worldwide. Int. J. Environ. Res. Public Health 2021, 18, 362. [Google Scholar] [CrossRef]
- Handelsman, D.J.; Hirschberg, A.L.; Bermon, S. Circulating testosterone as the hormonal basis of sex differences in athletic performance. Endocr. Rev. 2018, 39, 803–829. [Google Scholar] [CrossRef] [Green Version]
- Hirschberg, A.L. Female hyperandrogenism and elite sport. Endocr. Connect. 2020, 9, R81–R92. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Notelovitz, M. Androgen effects on bone and muscle. Fertil. Steril. 2002, 77, 34–41. [Google Scholar] [CrossRef]
- Herbst, K.L.; Bhasin, S. Testosterone action on skeletal muscle. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Hartgens, F.; Kuipers, H. Effects of androgenic-anabolic steroids in athletes. Sports Med. 2004, 34, 513–554. [Google Scholar] [CrossRef]
- Stone, M.H.; Cardinale, M.; Stone, M.H. Is Testosterone Influencing Explosive Performance? Artic. J. Strength Cond. Res. 2006, 20, 103–107. [Google Scholar] [CrossRef]
- Costello, J.T.; Bieuzen, F.; Bleakley, C.M. Where are all the female participants in Sports and Exercise Medicine research? Eur. J. Sport Sci. 2014, 14, 847–851. [Google Scholar] [CrossRef] [Green Version]
- De Jonge, X.A.K.J. Effects of the menstrual cycle on exercise performance. Sports Med. 2003, 33, 833–851. [Google Scholar] [CrossRef]
- Schaumberg, M.A.; Jenkins, D.G.; Janse de Jonge, X.A.K.; Emmerton, L.M.; Skinner, T.L. Three-step method for menstrual and oral contraceptive cycle verification. J. Sci. Med. Sport 2017, 20, 965–969. [Google Scholar] [CrossRef] [Green Version]
- Scheid, J.; De Souza, M.J. Menstrual Irregularities and Energy Deficiency in Physically Active Women: The Role of Ghrelin, PYY and Adipocytokines. Cytokines Growth Mediat. Phys. Act. 2015, 55, 82–102. [Google Scholar] [CrossRef]
- Hagmar, M.; Berglund, B.; Brismar, K.; Hirschberg, A.L. Hyperandrogenism May Explain Reproductive Dysfunction in Olympic Athletes. Med. Sci. Sports Exerc. 2009, 41, 1241–1248. [Google Scholar] [CrossRef]
- Hirschberg, A.L.; Elings Knutsson, J.; Helge, T.; Godhe, M.; Ekblom, M.; Bermon, S.; Ekblom, B. Effects of moderately increased testosterone concentration on physical performance in young women: A double blind, randomised, placebo controlled study. Br. J. Sports Med. 2019. [Google Scholar] [CrossRef]
- Rickenlund, A.; Thorén, M.; Carlström, K.; von Schoultz, B.; Hirschberg, A.L. Diurnal Profiles of Testosterone and Pituitary Hormones Suggest Different Mechanisms for Menstrual Disturbances in Endurance Athletes. J. Clin. Endocrinol. Metab. 2004, 89, 702–707. [Google Scholar] [CrossRef] [Green Version]
- Bell, D.R.; Blackburn, J.T.; Norcorss, M.F.; Ondrak, K.S.; Hudson, J.D.; Hackney, A.C.; Padua, D.A. Estrogen and muscle stiffness have a negative relationship in females. Knee Surg. Sports Traumatol. Arthrosc. 2012, 20, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Cadegiani, F.A.; Kater, C.E. Basal hormones and biochemical markers as predictors of overtraining syndrome in Male athletes: The EROS-Basal study. J. Athl. Train. 2019, 54, 906–914. [Google Scholar] [CrossRef] [Green Version]
- Martin, D.; Sale, C.; Cooper, S.B.; Elliott-Sale, K.J. Period prevalence and perceived side effects of hormonal contraceptive use and the menstrual cycle in elite athletes. Int. J. Sports Physiol. Perform. 2018, 13, 926–932. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Larsen, B.; Morris, K.; Quinn, K.; Osborne, M.; Minahan, C. Practice does not make perfect: A brief view of athletes’ knowledge on the menstrual cycle and oral contraceptives. J. Sci. Med. Sport 2020, 23, 690–694. [Google Scholar] [CrossRef]
- Cheng, J.; Santiago, K.A.; Abutalib, Z.; Temme, K.E.; Hulme, A.; Goolsby, M.A.; Esopenko, C.L.; Casey, E.K. Menstrual Irregularity, Hormonal Contraceptive Use, and Bone Stress Injuries in Collegiate Female Athletes in the United States. PM&R 2020. [Google Scholar] [CrossRef]
- Oxfeldt, M.; Dalgaard, L.B.; Jørgensen, A.A.; Hansen, M. Hormonal contraceptive use, menstrual dysfunctions, and self-reported side effects in elite athletes in Denmark. Int. J. Sports Physiol. Perform. 2020, 15, 1377–1384. [Google Scholar] [CrossRef]
- Dill, D.B.; Costill, D.L. Calculation of percentage changes in volumes of blood, plasma, and red cells in dehydration. J. Appl. Physiol. 1974, 37, 247–248. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Navarro, I.; Sánchez-Gómez, J.M.; Collado-Boira, E.J.; Hernando, B.; Panizo, N.; Hernando, C. Cardiac Damage Biomarkers and Heart Rate Variability Following a 118-Km Mountain Race: Relationship with Performance and Recovery. J. Sports Sci. Med. 2019, 18, 615. [Google Scholar]
- Dourado, V.Z.; de Oliveira Antunes, L.C.; Tanni, S.E.; de Paiva, S.A.R.; Padovani, C.R.; Godoy, I. Relationship of Upper-Limb and Thoracic Muscle Strength to 6-min Walk Distance in COPD Patients. Chest 2006, 129, 551–557. [Google Scholar] [CrossRef] [Green Version]
- Reuter, S.E.; Massy-Westropp, N.; Evans, A.M. Reliability and validity of indices of hand-grip strength and endurance. Aust. Occup. Ther. J. 2011, 58, 82–87. [Google Scholar] [CrossRef]
- Martínez-Navarro, I.; Sanchez-Gómez, J.M.; Aparicio, I.; Priego-Quesada, J.I.; Pérez-Soriano, P.; Collado, E.; Hernando, B.; Hernando, C. Effect of mountain ultramarathon distance competition on biochemical variables, respiratory and lower-limb fatigue. PLoS ONE 2020, 15, e0238846. [Google Scholar] [CrossRef]
- Luckey, D.W. Nonparametric Statistics for Health Care Research: Statistics for Small Samples and Unusual Distributions. J. Am. Stat. Assoc. 1998, 93, 409–411. [Google Scholar] [CrossRef]
- Guerrero, C.; Collado-Boira, E.; Martinez-Navarro, I.; Hernando, B.; Hernando, C.; Balino, P.; Muriach, M. Impact of Plasma Oxidative Stress Markers on Post-race Recovery in Ultramarathon Runners: A Sex and Age Perspective Overview. Antioxidants 2021, 10, 355. [Google Scholar] [CrossRef]
- Stoica, P.; Söderström, T. On the parsimony principle. Int. J. Control 1982, 36, 409–418. [Google Scholar] [CrossRef]
- Williams, M.N.; Grajales, C.A.G.; Kurkiewicz, D. Assumptions of multiple regression: Correcting two misconceptions. Pract. Assess. Res. Eval. 2013, 18, 1–14. [Google Scholar] [CrossRef]
- Cohen, J. The analysis of variance and covariance. Stat. Power Anal. Behav. Sci. 1988, 88, 728. [Google Scholar]
- Koltun, K.J.; Williams, N.I.; Scheid, J.L.; De Souza, M.J. Discriminating hypothalamic oligomenorrhea/amenorrhea from hyperandrogenic oligomenorrhea/amenorrhea in exercising women. Appl. Physiol. Nutr. Metab. 2020, 45, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Arena, B.; Maffulli, N.; Maffulli, F.; Morleo, M.A. Reproductive Hormones and Menstrual Changes with Exercise in Female Athletes. Sports Med. 1995, 19, 278–287. [Google Scholar] [CrossRef] [PubMed]
- Rickenlund, A.; Carlström, K.; Ekblom, B.; Brismar, T.B.; von Schoultz, B.; Hirschberg, A.L. Effects of Oral Contraceptives on Body Composition and Physical Performance in Female Athletes. J. Clin. Endocrinol. Metab. 2004, 89, 4364–4370. [Google Scholar] [CrossRef] [Green Version]
- Rickenlund, A.; Carlström, K.; Ekblom, B.; Brismar, T.B.; Von Schoultz, B.; Hirschberg, A.L. Hyperandrogenicity is an alternative mechanism underlying oligomenorrhea or amenorrhea in female athletes and may improve physical performance. Fertil. Steril. 2003, 79, 947–955. [Google Scholar] [CrossRef]
- Mauras, N.; Hayes, V.; Welch, S.; Rini, A.; Helgeson, K.; Dokler, M.; Veldhuis, J.D.; Urban, R.J. Testosterone deficiency in young men: Marked alterations in whole body protein kinetics, strength, and adiposity. J. Clin. Endocrinol. Metab. 1998, 83, 1886–1892. [Google Scholar] [CrossRef]
- Demling, R.H.; Orgill, D.P. The anticatabolic and wound healing effects of the testosterone analog oxandrolone after severe burn injury. J. Crit. Care 2000, 15, 12–17. [Google Scholar] [CrossRef] [Green Version]
- Vingren, J.L.; Kraemer, W.J.; Ratamess, N.A.; Anderson, J.M.; Volek, J.S.; Maresh, C.M. Testosterone physiology in resistance exercise and training: The up-stream regulatory elements. Sports Med. 2010, 40, 1037–1053. [Google Scholar] [CrossRef] [PubMed]
- Berg, U.; Enqvist, J.K.; Mattsson, C.M.; Carlsson-Skwirut, C.; Sundberg, C.J.; Ekblom, B.; Bang, P. Lack of sex differences in the IGF-IGFBP response to ultra endurance exercise. Scand. J. Med. Sci. Sports 2008, 18, 706–714. [Google Scholar] [CrossRef]
- Ginsburg, G.S.; O’Toole, M.; Rimm, E.; Douglas, P.S.; Rifai, N. Gender differences in exercise-induced changes in sex hormone levels and lipid peroxidation in athletes participating in the Hawaii Ironman triathlon: Ginsburg-gender and exercise-induced lipid peroxidation. Clin. Chim. Acta 2001, 305, 131–139. [Google Scholar] [CrossRef]
- Hansen, M. Female hormones: Do they influence muscle and tendon protein metabolism? Proc. Nutr. Soc. 2018, 77, 32–41. [Google Scholar] [CrossRef] [Green Version]
- Rechichi, C.; Dawson, B.; Goodman, C. Athletic performance and the oral contraceptive. Int. J. Sports Physiol. Perform. 2009, 4, 151–162. [Google Scholar] [CrossRef]
- Rechichi, C.; Dawson, B.; Goodman, C. Oral Contraceptive Phase Has no Effect on Endurance Test. Int. J. Sports Med. 2008, 29, 277–281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaiksaar, S.; Jürimäe, J.; Mäestu, J.; Purge, P.; Kalytka, S.; Shakhlina, L.; Jürimäe, T. No Effect of Menstrual Cycle Phase and Oral Contraceptive Use on Endurance Performance in Rowers. J. Strength Cond. Res. 2011, 25, 1571–1578. [Google Scholar] [CrossRef] [PubMed]
- Lebrun, C.M.; Joyce, S.M.; Constantini, N.W. Effects of Female Reproductive Hormones on Sports Performance. In Contemporary Endocrinology; Humana Press Inc.: Totowa, NJ, USA, 2020; pp. 267–301. [Google Scholar]
- Keller, M.F.; Harrison, M.L.; Lalande, S. Impact of Menstrual Blood Loss and Oral Contraceptive Use on Oxygen-carrying Capacity. Med. Sci. Sports Exerc. 2020, 52, 1414–1419. [Google Scholar] [CrossRef] [PubMed]
| Baseline Data | Finish Line | 24 h Post-Race | 48 h Post-Race | |
|---|---|---|---|---|
| Estradiol (pg/mL) | 89.06 ± 87.99 | |||
| Testosterone (ng/dL) | 19.34 ± 9.34 | |||
| T/E ratio | 1.25 ± 1.99 | |||
| CK (ui/L) | 137.59 ± 54.73 | 5075.76 ± 3871.18 * | 2036.61 ± 1389.83 * # | 905.30 ± 534.95 * # |
| LDH (ui/L) | 185.88 ± 25.29 | 380.59 ± 112.80 * | 320.93 ± 93.97 * # | 303.38 ± 86.67 * # |
| MM (%) | 39.20 ± 3.45 | 38.00 ± 4.86 * | ||
| HG (kg) | 31.33 ± 3.69 | 28.67 ± 4.10 * | ||
| SJ (cm) | 20.76 ± 2.72 | 18.47 ± 2.38 * |
| Estradiol Correlation r Value/p Value | Testosterone Correlation r Value/p Value | T/E Ratio Correlation r Value/p Value | |
|---|---|---|---|
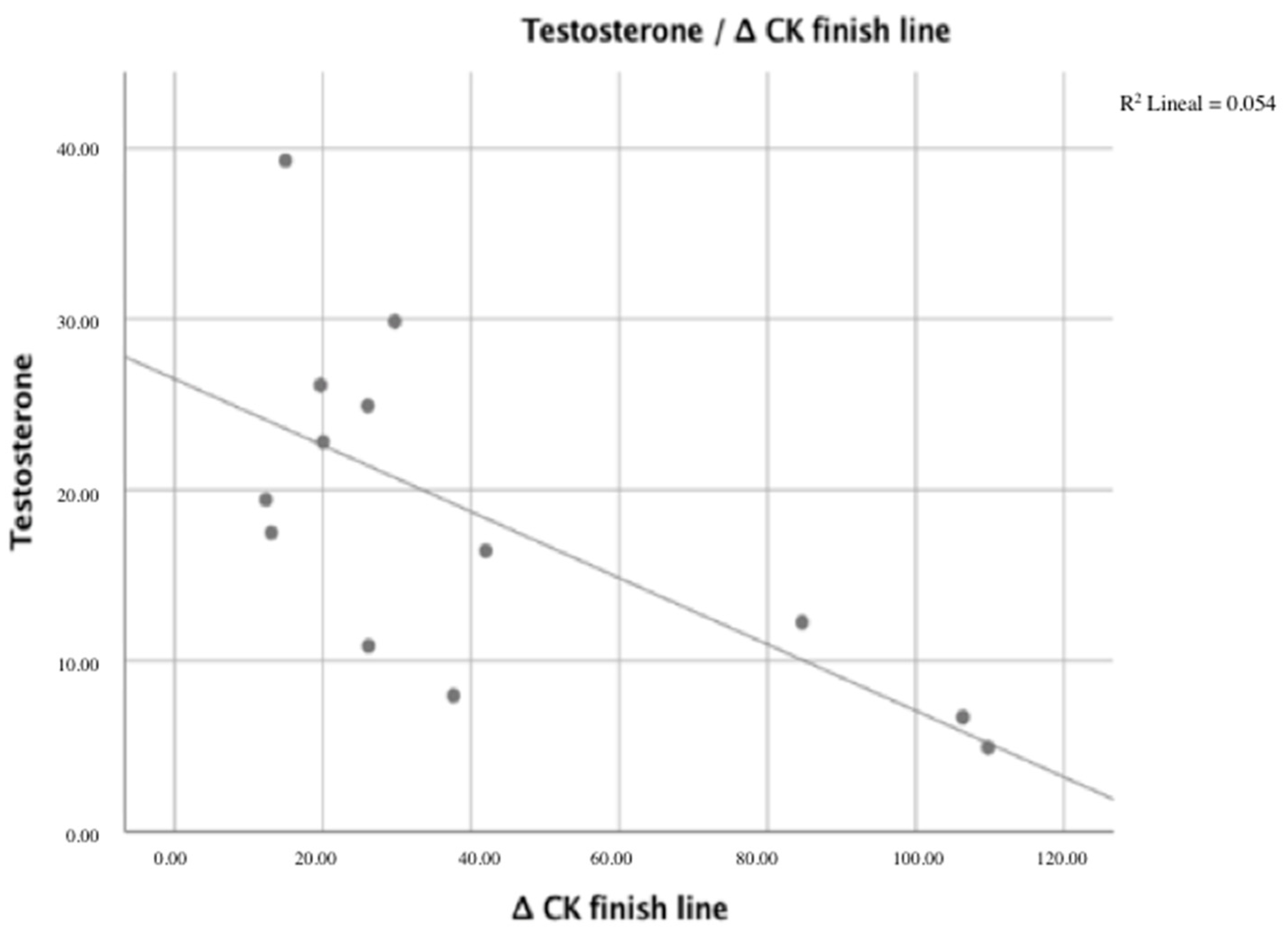
| Δ CK finish line | −0.199/0.514 | −0.674 */0.012 | 0.435/0.137 |
| Δ CK 24 h | −0.324/0.280 | −0.652 */0.016 | 0.319/0.288 |
| Δ CK 48 h | −0.428/0.145 | −0.619 */0.024 | 0.134/0.662 |
| Δ LDH finish line | −0.065/0.832 | −0.541/0.056 | 0.581 */0.037 |
| Δ LDH 24 h | −0.114/0.712 | −0.585 */0.036 | 0.595 */0.032 |
| Δ LDH 48 h | −0.147/0.631 | −0.615 */0.025 | 0.620 */0.024 |
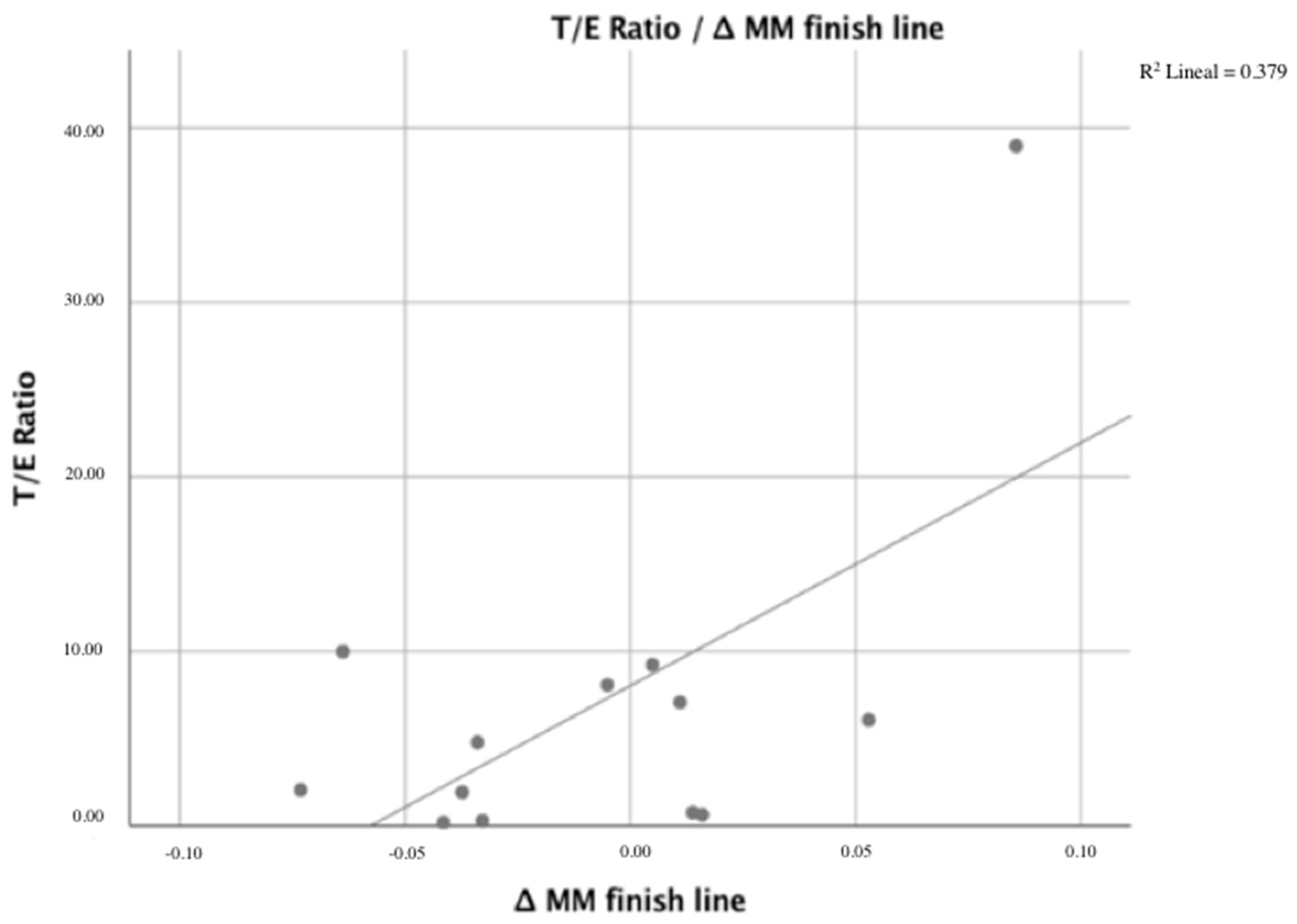
| Δ MM finish line | 0.202/0.508 | −0.466/0.109 | 0.625 */0.02 |
| Δ SJ finish line | −0.206/0.520 | 0.132/0.683 | −0.065/0.841 |
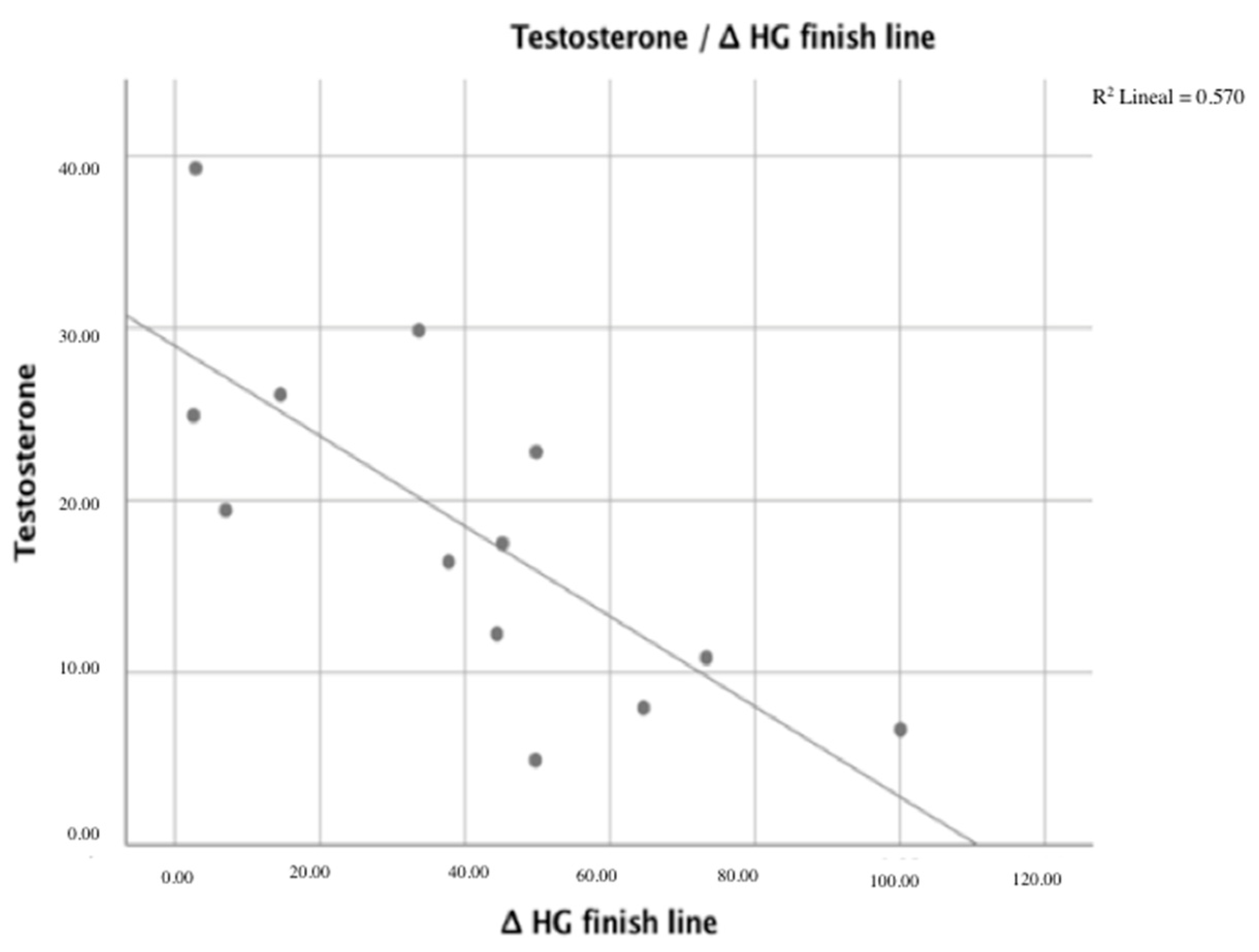
| Δ HG finish line | −0.472/0.104 | −0.755 */0.003 | −0.034/0.912 |
| No Hormonal Contraceptive Methods | Hormonal Contraceptive Methods | p-Value/d Cohen | |
|---|---|---|---|
| Baseline ferritin (ng/mL) | 24.11 ± 11.37 | 24.72 ± 8.75 | 0.912 |
| Red blood cells (ng/mL) | 4.649 ± 0.25 | 4.90 ± 0.29 | 0.084 |
| Hemoglobin (g/dL) | 14.24 ± 1.31 | 14.41 ± 1.23 | 0.792 |
| Baseline red blood cells (%) | 43.05 ± 3.14 | 43.78 ± 2.91 | 0.645 |
| Estradiol (pg/mL) | 129.20 ± 93.85 | 33.10 ± 32.83 | 0.012 */2.16 |
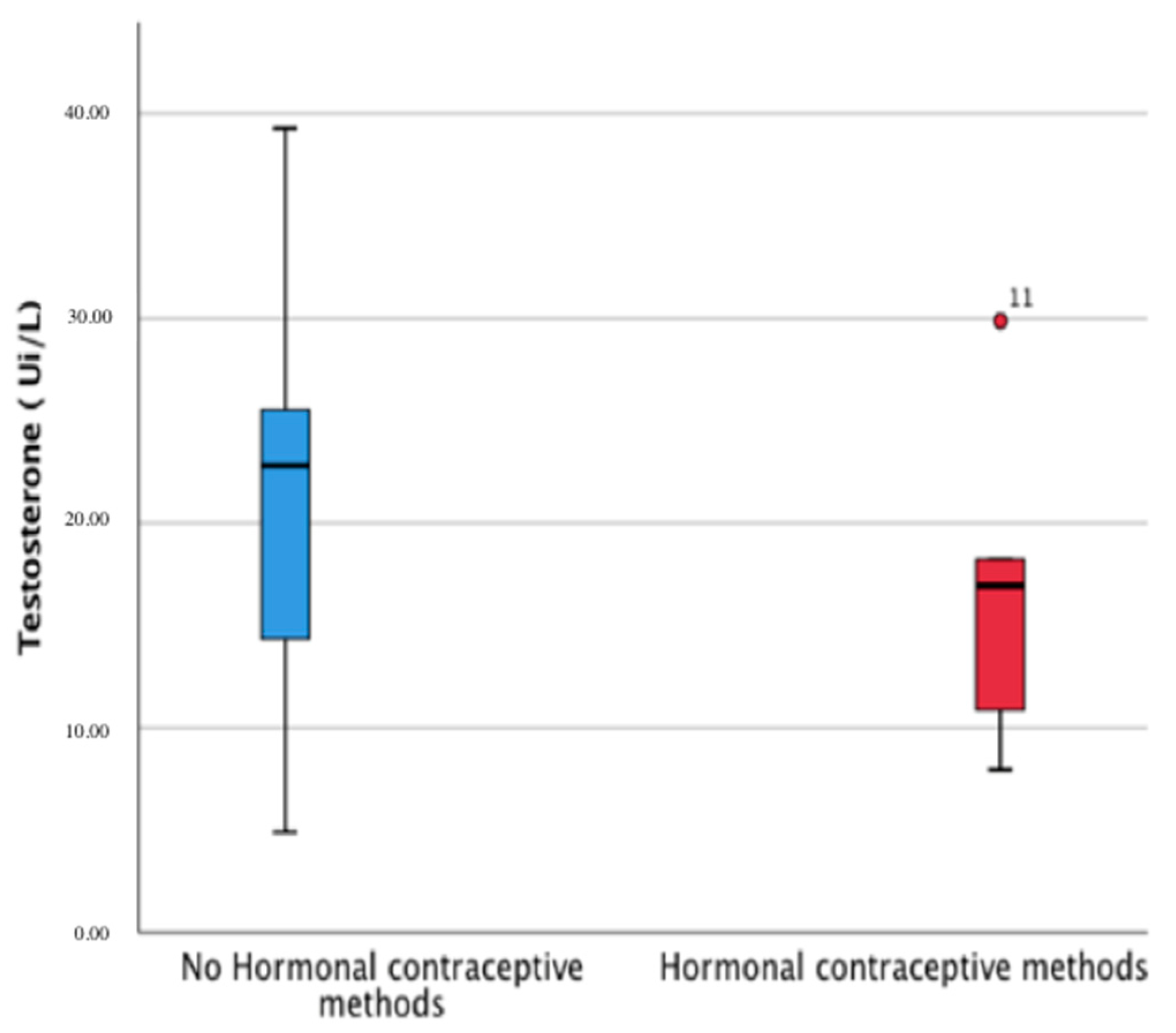
| Testosterone (ng/dL) | 21.58 ± 10.37 | 16.79 ± 7.57 | 0.313 |
| T/E ratio | 0.86 ± 1.78 | 1.97 ± 2.33 | 0.290 |
| CK (IU/L) baseline | 147.80 ± 66.65 | 126.83 ± 31.012 | 0.485 |
| CK (IU/L) finish line | 5640.55 ± 4572.41 | 3072.91 ± 940.02 | 0.303 |
| CK (IU/L) 24 h | 2202.62 ± 1613.55 | 1392.00 ± 415.22 | 0.356 |
| CK (IU/L) 48 h | 921.25 ± 563.60 | 646.50 ± 197.30 | 0.246 |
| LDH (IU/L) baseline | 187.50 ± 31.28 | 181.00 ± 14.71 | 0.643 |
| LDH (IU/L) finish line | 393.36 ± 132.56 | 327 ± 41.45 | 0.366 |
| LDH (IU/L) 24 h | 331.37 ± 117.06 | 290.75 ± 37.50 | 0.522 |
| LDH (IU/L) 48 h | 309.62 ± 110.85 | 286.50 ± 28.06 | 0.696 |
| MM (%) baseline | 43.85 ± 3.65 | 45.26 ± 3.16 | 0.439 |
| MM (%) finish line | 43.36 ± 5.12 | 44.27 ± 4.86 | 0.439 |
| Δ SJ finish line | −0.16 ± 0.07 | −0.06 ± 0.14 | 0.112 |
| Δ HG finish line | −0.10 ± 0,12 | −0.01 ± /0.10 | 0.771 |
| a. Linear regression models. | ||||
| Model | R2 Adjusted | Standardized Coefficients Beta | Standard Error | F (p) |
| Dependent variable: Δ CK finish line Covariables: Testosterone. | 0.405 | –0.674 | 2.69663 | 9.151 (0.012) |
| Dependent variable: Δ LDH finish line Covariables: T/E ratio. | 0.479 | –0.579 | 0.36986 | 6.507 (0.015) |
| Dependent variable: Δ HG finish line Covariables: Testosterone | 0.531 | –0.755 | 0.19891 | 14.567 (0.003) |
| Dependent variable: Δ MM finish line Covariables: T/E ratio. | 0.335 | 0.625 | 3.96370 | 7.056 (0.022) |
| b. Linear regression models for women not receiving hormonal contraception (n = 11; 66.7%). | ||||
| Model | R2 Adjusted | Standardized Coefficients Beta | Standard Error | F (p) |
| Dependent Variable: Δ CK finish line Covariables: Testosterone. | 0.689 | –3.222 | 0.744 | 18.756 (0.003) |
| Dependent Variable: Δ LDH finish line Covariables: Testosterone. | 0.702 | –0.046 | 0.31096 | 19.819 (0.003) |
| Dependent Variable: Δ HG finish line Covariables: Testosterone | 0.556 | –0.022 | 0.19891 | 8.777 (0.021) |
| Dependent Variable: Δ MM finish line Covariables: T/E Ratio. | 0.658 | 0.370 | 2.9994 | 16.371 (0.005) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Collado-Boira, E.; Baliño, P.; Boldo-Roda, A.; Martínez-Navarro, I.; Hernando, B.; Recacha-Ponce, P.; Hernando, C.; Muriach, M. Influence of Female Sex Hormones on Ultra-Running Performance and Post-Race Recovery: Role of Testosterone. Int. J. Environ. Res. Public Health 2021, 18, 10403. https://doi.org/10.3390/ijerph181910403
Collado-Boira E, Baliño P, Boldo-Roda A, Martínez-Navarro I, Hernando B, Recacha-Ponce P, Hernando C, Muriach M. Influence of Female Sex Hormones on Ultra-Running Performance and Post-Race Recovery: Role of Testosterone. International Journal of Environmental Research and Public Health. 2021; 18(19):10403. https://doi.org/10.3390/ijerph181910403
Chicago/Turabian StyleCollado-Boira, Eladio, Pablo Baliño, Ana Boldo-Roda, Ignacio Martínez-Navarro, Bárbara Hernando, Paula Recacha-Ponce, Carlos Hernando, and María Muriach. 2021. "Influence of Female Sex Hormones on Ultra-Running Performance and Post-Race Recovery: Role of Testosterone" International Journal of Environmental Research and Public Health 18, no. 19: 10403. https://doi.org/10.3390/ijerph181910403
APA StyleCollado-Boira, E., Baliño, P., Boldo-Roda, A., Martínez-Navarro, I., Hernando, B., Recacha-Ponce, P., Hernando, C., & Muriach, M. (2021). Influence of Female Sex Hormones on Ultra-Running Performance and Post-Race Recovery: Role of Testosterone. International Journal of Environmental Research and Public Health, 18(19), 10403. https://doi.org/10.3390/ijerph181910403







