Differences of Gut Microbiota in the Freshwater Blackworm (Lumbriculus variegatus: Oligochaeta) in Two Different Habitat Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Field Sampling
2.2. DNA Extraction
2.3. Pyrosequencing of Bacterial 16S rRNA Genes
2.4. Data Analysis
2.5. Statistical Analysis
3. Results
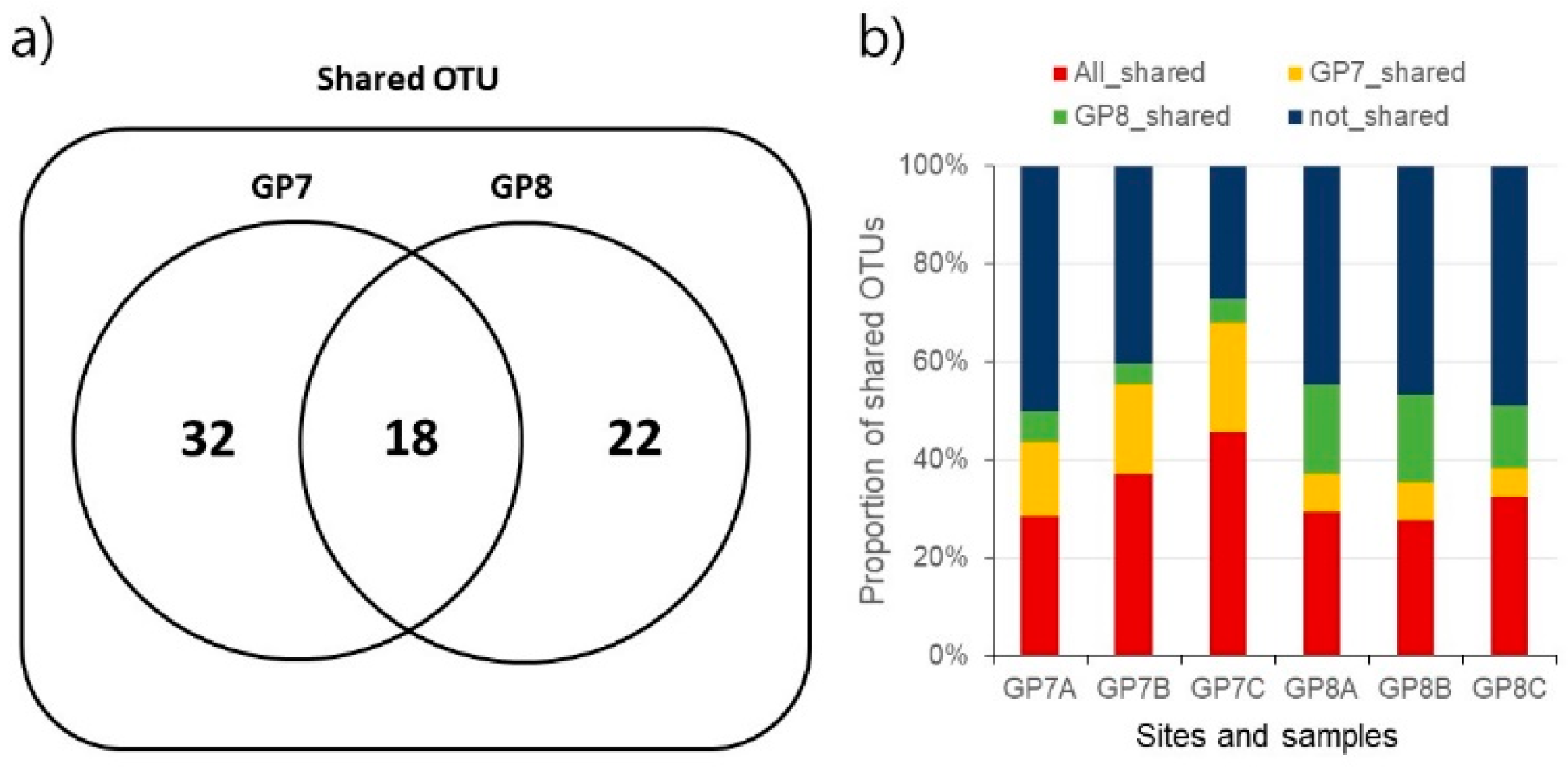
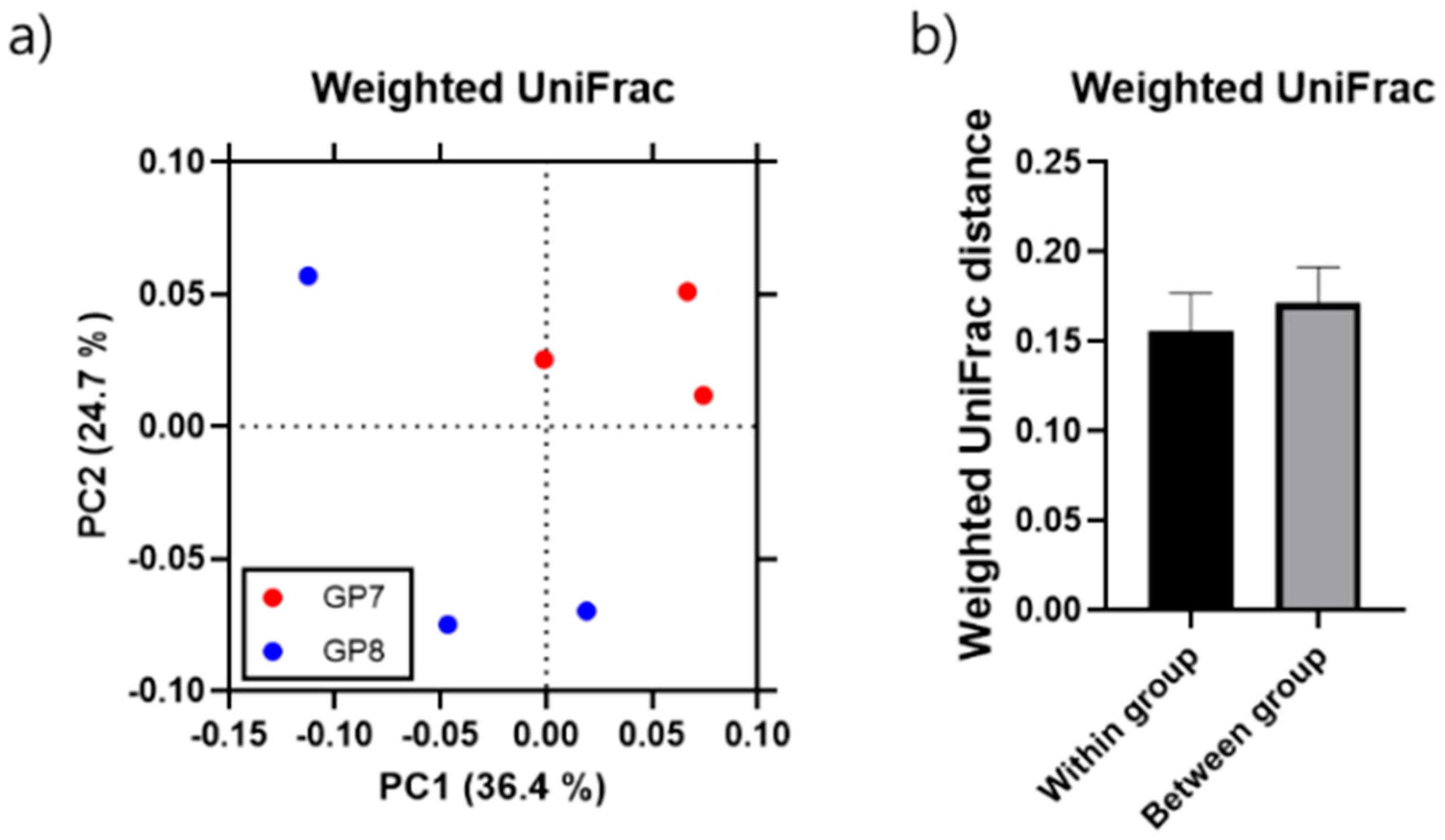
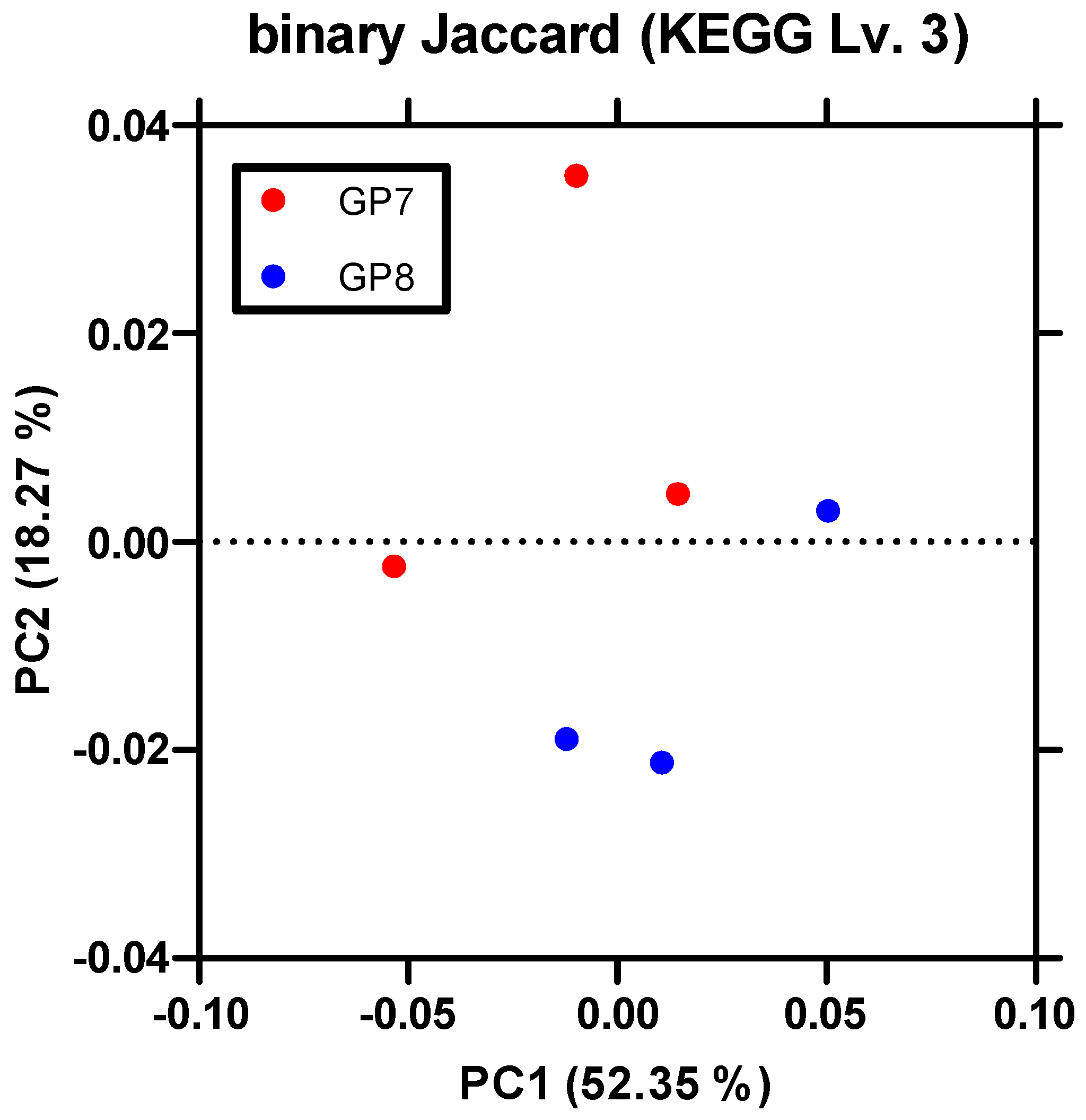
3.1. Gut Microbiome Diversity
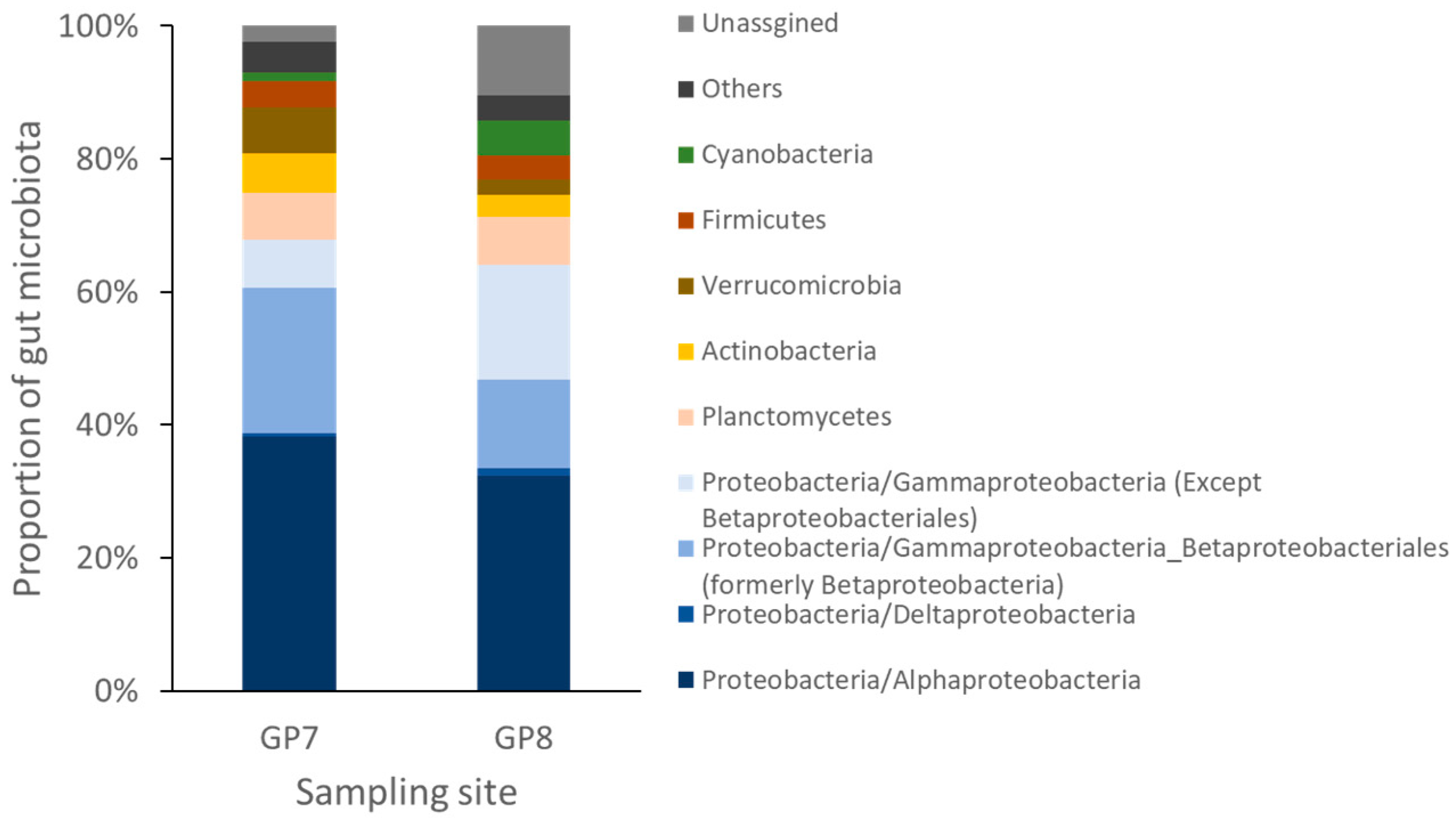
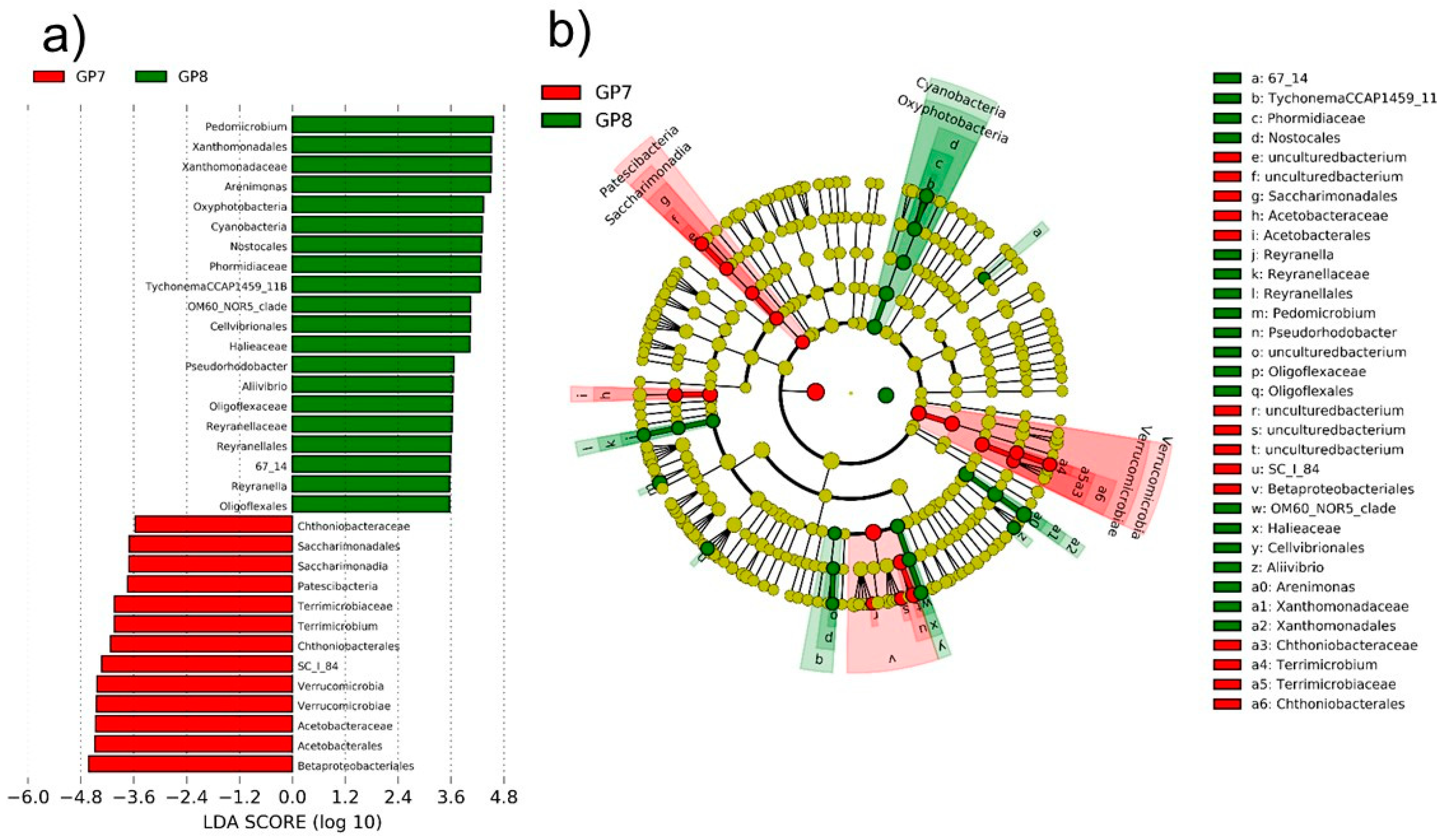
3.2. Dominant Taxa Specificity
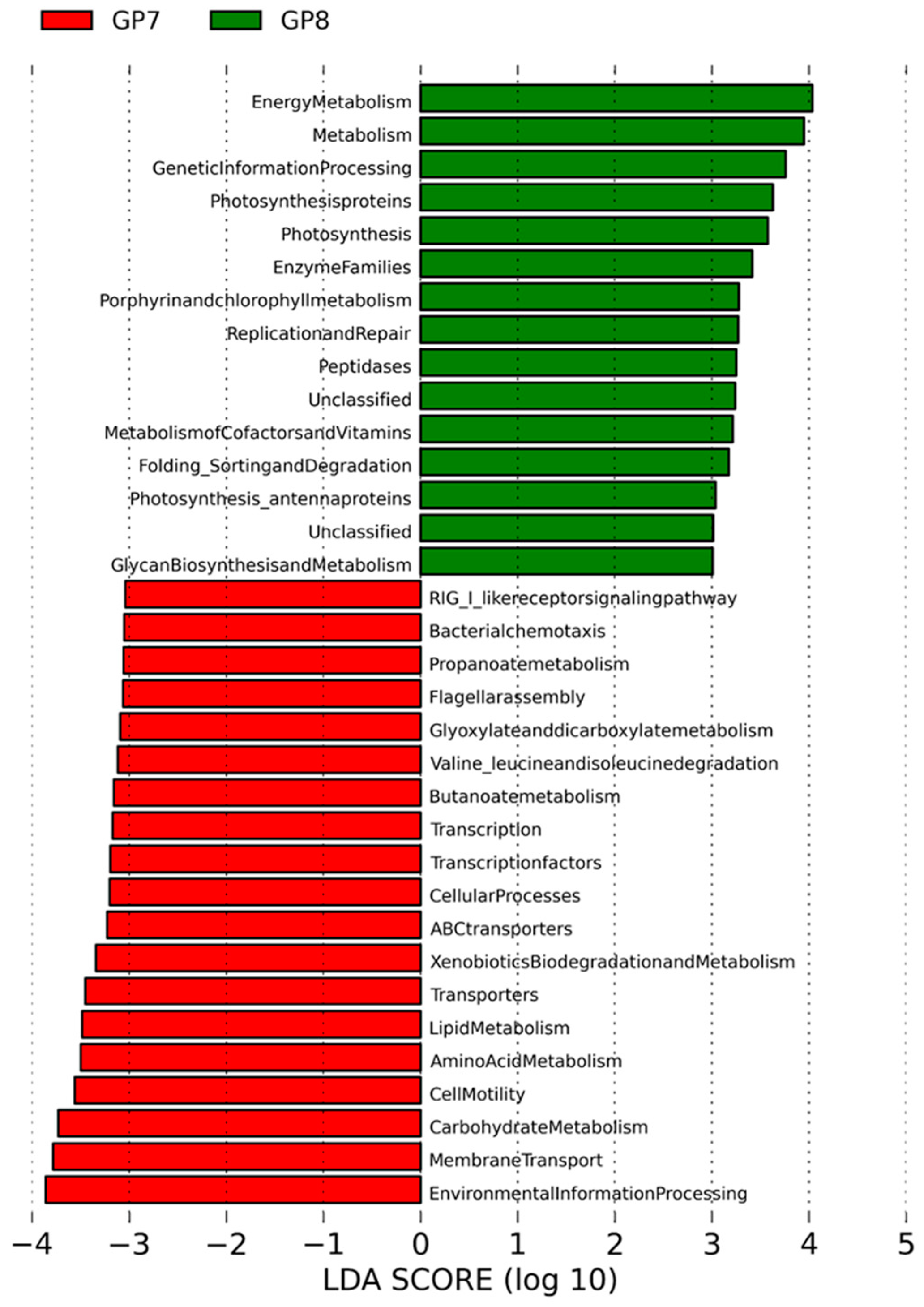
3.3. Functional Significance of Core Microflora
4. Discussion
4.1. Gut Bacterial Community
4.2. Differences in Gut Microbiome in Distinct Habitat Conditions
4.3. Limitation and Further Studies
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Park, Y.-S.; Song, M.-Y.; Oh, K.-H.; Cho, E.; Chon, T.-S. Community patterns of benthic macroinvertebrates collected on the national scale in Korea. Ecol. Model. 2007, 203, 26–33. [Google Scholar] [CrossRef]
- Thursby, E.; Juge, N. Introduction to the human gut microbiota. Biochem. J. 2017, 474, 1823–1836. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, E.; Zilber-Rosenberg, I. Symbiosis and development: The hologenome concept. Birth Defects Res. Part C Embryo Today Rev. 2011, 93, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Aira, M.; Perez-Losada, M.; Domínguez, J. Diversity, structure and sources of bacterial communities in earthworm cocoons. Sci. Rep. 2018, 8, 6632. [Google Scholar] [CrossRef] [PubMed]
- Fiołka, M.J.; Zagaja, M.P.; Piersiak, T.D.; Wróbel, M.; Pawelec, J. Gut bacterium of Dendrobaena veneta (Annelida: Oligochaeta) possesses antimycobacterial activity. J. Invertebr. Pathol. 2010, 105, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Pathma, J.; Sakthivel, N. Microbial diversity of vermicompost bacteria that exhibit useful agricultural traits and waste management potential. SpringerPlus 2012, 1, 26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aira, M.; Domínguez, J. Earthworm Effects without Earthworms: Inoculation of Raw Organic Matter with Worm-Worked Substrates Alters Microbial Community Functioning. PLoS ONE 2011, 6, e16354. [Google Scholar] [CrossRef] [Green Version]
- Koubová, A.; Knapp, B.; Insam, H.; Pižl, V.; Elhottová, D. The effect of passage through the gut of earthworms (Annelida: Lumbricidae) on the diversity of archaea and bacteria. Acta Soc. Zool. Bohem 2010, 74, 69–74. [Google Scholar]
- Breznak, J.A. Intestinal Microbiota of Termites and other Xylophagous Insects. Annu. Rev. Microbiol. 1982, 36, 323. [Google Scholar] [CrossRef]
- Brauman, A.; Dore, J.; Eggleton, P.; Bignell, D.; Breznak, J.; Kane, M. Molecular phylogenetic profiling of prokaryotic communities in guts of termites with different feeding habits. FEMS Microbiol. Ecol. 2001, 35, 27–36. [Google Scholar] [CrossRef]
- Warnecke, F.; Luginbühl, P.; Ivanova, N.; Ghassemian, M.; Richardson, T.H.; Stege, J.T.; Cayouette, M.; McHardy, A.C.; Djordjevic, G.; Aboushadi, N.; et al. Metagenomic and functional analysis of hindgut microbiota of a wood-feeding higher termite. Nature 2007, 450, 560–565. [Google Scholar] [CrossRef]
- Ohkuma, M. Symbioses of flagellates and prokaryotes in the gut of lower termites. Trends Microbiol. 2008, 16, 345–352. [Google Scholar] [CrossRef]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–65. [Google Scholar] [CrossRef] [Green Version]
- Engel, P.; Moran, N.A. The gut microbiota of insects—Diversity in structure and function. FEMS Microbiol. Rev. 2013, 37, 699–735. [Google Scholar] [CrossRef] [PubMed]
- Dillon, R.; Dillon, V. THEGUTBACTERIA OFINSECTS: Nonpathogenic Interactions. Annu. Rev. Èntomol. 2004, 49, 71–92. [Google Scholar] [CrossRef] [PubMed]
- Yun, J.-H.; Roh, S.W.; Whon, T.W.; Jung, M.-J.; Kim, M.-S.; Park, D.-S.; Yoon, C.; Nam, Y.-D.; Kim, Y.-J.; Choi, J.-H.; et al. Insect Gut Bacterial Diversity Determined by Environmental Habitat, Diet, Developmental Stage, and Phylogeny of Host. Appl. Environ. Microbiol. 2014, 80, 5254–5264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, B.; Teh, B.-S.; Sun, C.; Hu, S.; Lu, X.; Boland, W.; Shao, Y. Biodiversity and Activity of the Gut Microbiota across the Life History of the Insect Herbivore Spodoptera littoralis. Sci. Rep. 2016, 6, 29505. [Google Scholar] [CrossRef]
- Kang, H.; Bae, M.-J.; Park, Y.-S. Behavioral Response of Tubifex tubifex to Changes of Water Temperature and Substrate Composition. Korean J. Ecol. Environ. 2017, 50, 355–361. [Google Scholar] [CrossRef]
- Kang, H.-J.; Bae, M.-J.; Park, Y.-S. Ecotoxicological Studies Using Aquatic Oligochaetes: Review. Korean J. Ecol. Environ. 2016, 49, 343–353. [Google Scholar] [CrossRef]
- Ratsak, C.H.; Verkuijlen, J. Sludge Reduction by Predatory Activity of Aquatic Oligochaetes in Wastewater Treatment Plants: Science or Fiction? A Review. Hydrobiologia 2006, 564, 197–211. [Google Scholar] [CrossRef]
- Mermillod-Blondin, F.; Nogaro, G.; Datry, T.; Malard, F.; Gibert, J. Do tubificid worms influence the fate of organic matter and pollutants in stormwater sediments? Environ. Pollut. 2005, 134, 57–69. [Google Scholar] [CrossRef]
- Håkanson, L. The relationship between salinity, suspended particulate matter and water clarity in aquatic systems. Ecol. Res. 2005, 21, 75–90. [Google Scholar] [CrossRef]
- Marchand, J. The influence of seasonal salinity and turbidity maximum variations on the nursery function of the Loire estuary (France). Aquat. Ecol. 1993, 27, 427–436. [Google Scholar] [CrossRef]
- Pelegrí, S.; Blackburn, T. Effects of Tubifex tubifex (Oligochaeta: Tubificidae) on N-mineralization in freshwater sediments, measured with 15N isotopes. Aquat. Microb. Ecol. 1995, 9, 289–294. [Google Scholar] [CrossRef]
- Bilotta, G.; Brazier, R. Understanding the influence of suspended solids on water quality and aquatic biota. Water Res. 2008, 42, 2849–2861. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Bae, M.-J.; Lee, D.-S.; Hwang, S.-J.; Moon, J.-S.; Park, Y.-S. Distribution Patterns of the Freshwater Oligochaete Limnodrilus hoffmeisteri Influenced by Environmental Factors in Streams on a Korean Nationwide Scale. Water 2017, 9, 921. [Google Scholar] [CrossRef] [Green Version]
- Kuntz, K.L.; Tyler, A.C. Bioturbating invertebrates enhance decomposition and nitrogen cycling in urban stormwater ponds. J. Urban Ecol. 2018, 4. [Google Scholar] [CrossRef]
- Milbrink, G. Evidence for Mutualistic Interactions in Freshwater Oligochaete Communities. Oikos 1993, 68, 317. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; I Gordon, J.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [Green Version]
- Reeder, J.; Knight, R. Rapidly denoising pyrosequencing amplicon reads by exploiting rank-abundance distributions. Nat. Methods 2010, 7, 668–669. [Google Scholar] [CrossRef]
- Edgar, R.C. Search and clustering orders of magnitude faster than BLAST. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2012, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Haas, B.J.; Gevers, D.; Earl, A.M.; Feldgarden, M.; Ward, D.V.; Giannoukos, G.; Ciulla, D.; Tabbaa, D.; Highlander, S.K.; Sodergren, E.; et al. Chimeric 16S rRNA sequence formation and detection in Sanger and 454-pyrosequenced PCR amplicons. Genome Res. 2011, 21, 494–504. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caporaso, J.G.; Bittinger, K.; Bushman, F.; DeSantis, T.Z.; Andersen, G.; Knight, R. PyNAST: A flexible tool for aligning sequences to a template alignment. Bioinformatics 2009, 26, 266–267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Langille, M.G.; Zaneveld, J.; Caporaso, J.G.; McDonald, D.; Knights, D.; Reyes, J.A.; Clemente, J.C.; Burkepile, D.E.; Thurber, R.L.V.; Knight, R.; et al. Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat. Biotechnol. 2013, 31, 814–821. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, T.Z.; Hugenholtz, P.; Larsen, N.; Rojas, M.; Brodie, E.; Keller, K.; Huber, T.; Dalevi, D.; Hu, P.; Andersen, G.L. Greengenes, a Chimera-Checked 16S rRNA Gene Database and Workbench Compatible with ARB. Appl. Environ. Microbiol. 2006, 72, 5069–5072. [Google Scholar] [CrossRef] [Green Version]
- Ogata, H.; Goto, S.; Sato, K.; Fujibuchi, W.; Bono, H.; Kanehisa, M. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 1999, 27, 29–34. [Google Scholar] [CrossRef] [Green Version]
- Segata, N.; Izard, J.; Waldron, L.; Gevers, D.; Miropolsky, L.; Garrett, W.S.; Huttenhower, C. Metagenomic biomarker discovery and explanation. Genome Biol. 2011, 12, R60. [Google Scholar] [CrossRef] [Green Version]
- Baquiran, J.-P.; Thater, B.; Sedky, S.; De Ley, P.; Crowley, D.; Orwin, P.M. Culture-Independent Investigation of the Microbiome Associated with the Nematode Acrobeloides maximus. PLoS ONE 2013, 8, e67425. [Google Scholar] [CrossRef]
- Dubilier, N.; Giere, O.; Distel, D.; Cavanaugh, C.M. Characterization of chemoautotrophic bacterial symbionts in a gutless marine worm Oligochaeta, Annelida) by phylogenetic 16S rRNA sequence analysis and in situ hybridization. Appl. Environ. Microbiol. 1995, 61, 2346–2350. [Google Scholar] [CrossRef] [Green Version]
- Shin, N.-R.; Whon, T.W.; Bae, J.-W. Proteobacteria: Microbial signature of dysbiosis in gut microbiota. Trends Biotechnol. 2015, 33, 496–503. [Google Scholar] [CrossRef]
- Brune, A.; Emerson, D.; Breznak, J.A. The Termite Gut Microflora as an Oxygen Sink: Microelectrode Determination of Oxygen and pH Gradients in Guts of Lower and Higher Termites. Appl. Environ. Microbiol. 1995, 61, 2681–2687. [Google Scholar] [CrossRef] [Green Version]
- Balykin, A.V. On the relationship between the microflora and the habitat of the Tubificidae. In Proceedings of the Aquatic Oligochaeta—Proceedings of The Fourth All-Union Symposim, Tbilisi, Georgia, 5–7 October 1983; pp. 17–22. [Google Scholar]
- Wavre, M.; Brinkhurst, R.O. Interactions Between Some Tubificid Oligochaetes and Bacteria Found in the Sediments of Toronto Harbour, Ontario. J. Fish. Res. Board Can. 1971, 28, 335–341. [Google Scholar] [CrossRef]
- Whitley, L.S.; Seng, T.N. Studies on the bacterial flora of tubificid worms. Hydrobiologia 1976, 48, 79–83. [Google Scholar] [CrossRef]
- Brinkhurst, R.O.; Chua, K.E. Preliminary Investigation of the Exploitation of Some Potential Nutritional Resources by Three Sympatric Tubificid Oligochaetes. J. Fish. Res. Board Can. 1969, 26, 2659–2668. [Google Scholar] [CrossRef]
- Rodríguez, P.; Martínez-Madrid, M.; Arrate, J.A.; Navarro, E. Selective feeding by the aquatic oligochaete Tubifex tubifex (Tubificidae, Clitellata). Hydrobiologia 2001, 463, 133–140. [Google Scholar] [CrossRef]

| Environmental Variable | Sampling Site | |
|---|---|---|
| GP7 | GP8 | |
| Location (latitude, longitude) | 37°52′27″ N, 127°31′34″ E | 37°50′03″ N, 127°30′57″ E |
| Water velocity (m/s) | 0.3 ± 0.1 | 0.2 ± 0.0 |
| Water depth (cm) | 46.9 ± 9.3 | 48.2 ± 10.9 |
| Water width (m) | 47.8 ± 2.5 | 77.5 ± 19.3 |
| Turbidity (NTU) | 1.3 ± 0.4 | 1.6 ± 0.3 |
| Electric conductivity (μS/cm) | 69.8 ± 3.2 | 79.7 ± 4.5 |
| pH | 7.5 ± 0.1 | 7.7 ± 0.2 |
| Dissolved oxygen (mg/L) | 12.6 ± 0.5 | 12.4 ± 0.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, P.S.; Lee, Y.-R.; Kwon, Y.-S.; Bae, J.-W.; Lee, S.-J.; Park, Y.-S. Differences of Gut Microbiota in the Freshwater Blackworm (Lumbriculus variegatus: Oligochaeta) in Two Different Habitat Conditions. Int. J. Environ. Res. Public Health 2021, 18, 10298. https://doi.org/10.3390/ijerph181910298
Kim PS, Lee Y-R, Kwon Y-S, Bae J-W, Lee S-J, Park Y-S. Differences of Gut Microbiota in the Freshwater Blackworm (Lumbriculus variegatus: Oligochaeta) in Two Different Habitat Conditions. International Journal of Environmental Research and Public Health. 2021; 18(19):10298. https://doi.org/10.3390/ijerph181910298
Chicago/Turabian StyleKim, Pil Soo, Yeo-Rang Lee, Yong-Su Kwon, Jin-Woo Bae, Sung-Jae Lee, and Young-Seuk Park. 2021. "Differences of Gut Microbiota in the Freshwater Blackworm (Lumbriculus variegatus: Oligochaeta) in Two Different Habitat Conditions" International Journal of Environmental Research and Public Health 18, no. 19: 10298. https://doi.org/10.3390/ijerph181910298
APA StyleKim, P. S., Lee, Y.-R., Kwon, Y.-S., Bae, J.-W., Lee, S.-J., & Park, Y.-S. (2021). Differences of Gut Microbiota in the Freshwater Blackworm (Lumbriculus variegatus: Oligochaeta) in Two Different Habitat Conditions. International Journal of Environmental Research and Public Health, 18(19), 10298. https://doi.org/10.3390/ijerph181910298








