Effectiveness of a Multicomponent Intervention in Primary Care That Addresses Patients with Diabetes Mellitus with Two or More Unhealthy Habits, Such as Diet, Physical Activity or Smoking: Multicenter Randomized Cluster Trial (EIRA Study)
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
2.2. Study Area
2.3. Inclusion and Exclusion Criteria
2.4. Intervention (EIRA Study)
2.5. Usual Health Care (the PHCC Control Group)
2.6. Measures and Data Collection
2.7. Sample Size
2.8. Randomization
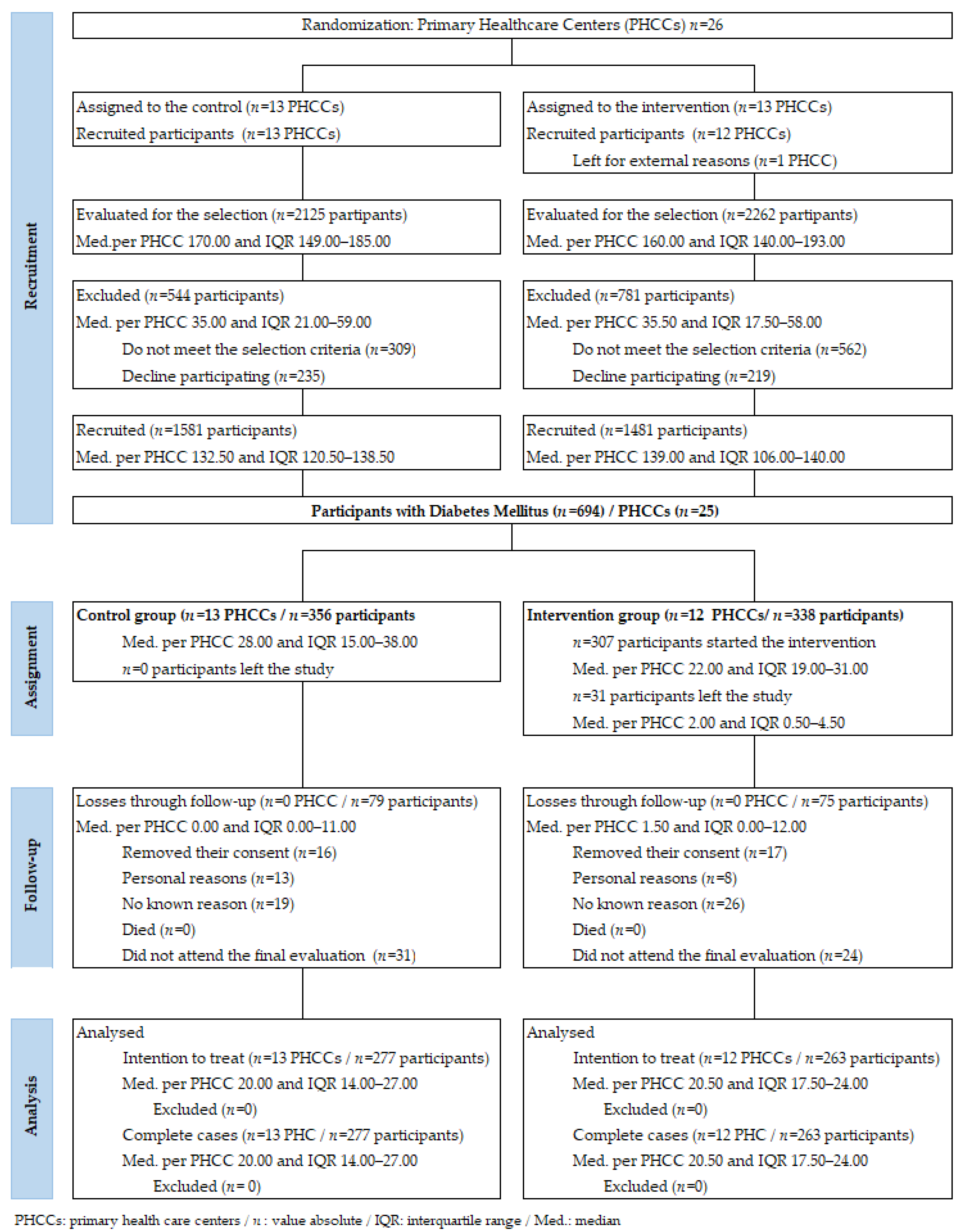
2.9. Recruitment
2.10. Statistical Analysis
3. Results
4. Discussion
4.1. Strengths and Limitations
4.2. Implications for Research and Practice
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Ethical Approval
Nomenclature
| % | Percentage |
| ABI | Ankle-Brachial Index |
| APP | Mobile application |
| BMI | Body Mass Index |
| CAVI | Heart-Ankle Vascular Index |
| CG | Control Group |
| C-HDL | High-Density Lipoprotein Cholesterol |
| CI | Confidence Interval |
| C-LDLCONSORT | Low-Density Lipoprotein CholesterolConsolidated Standards of Reporting Trials |
| COPD | Chronic Obstructive Pulmonary Disease |
| CT | Total Cholesterol |
| CVD | Cerebrovascular Disease |
| DBP | Diastolic Blood Pressure |
| DM | Diabetes Mellitus |
| DQI | Diet Quality |
| DQI-I | International Diet Quality Index |
| GAD-7 | Generalized Anxiety Disorder-7 |
| HbA1c | Glycosylated hemoglobin |
| HTA | Arterial Hypertension |
| IG | Intervention Group |
| IPAQ | International Physical Activity Questionnaire |
| IQR | Interquartile Range |
| ITT | Intention To Treat |
| med. | Median |
| MEDAS | Mediterranean Diet Adherence Questionnaire |
| n | Absolute value |
| VAS | Visual Analogue Scale |
| OR | Odds Ratio |
| P | Statistical significance |
| PC | Primary Care |
| PHCCs | Primary Health Care Centers |
| RCT | Randomized Clinical Trial |
| RedIAPP | Research Network in Preventive and Health Promotion Activities |
| SBP | Systolic blood pressure |
| SMS | Text messages |
References
- American Diabetes Association. Standards of Medical Care in Diabetes 2020. Diabetes Care 2020, 43, S11–S61. [Google Scholar]
- International Diabetes Federation. IDF Diabetes Atlas, 9th ed.; International Diabetes Federation: Brussels, Belgium, 2019. [Google Scholar]
- Stratton, I.M.; Adler, A.I.; Neil, H.A.W.; Matthews, D.R.; Manley, S.E.; Cull, C.A.; Hadden, D.; Turner, R.C.; Holman, R.R. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): Prospective observational study. BMJ 2000, 321, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Norlund, A.; Apelqvist, J.; Bitzen, P.-O.; Nyberg, P.; Scherstén, B. Cost of illness of adult diabetes mellitus underestimated if comorbidity is not considered. J. Intern. Med. 2001, 250, 57–65. [Google Scholar] [CrossRef]
- Coffey, J.T.; Brandle, M.; Zhou, H.; Marriott, D.; Burke, R.; Tabaei, B.P.; Engelgau, M.M.; Kaplan, R.M.; Herman, W.H. Valuing Health-Related Quality of Life in Diabetes. Diabetes Care 2002, 25, 2238–2243. [Google Scholar] [CrossRef] [PubMed]
- Meader, N.; King, K.; Wright, K.; Graham, H.M.; Petticrew, M.; Power, C.; White, M.; Sowden, A.J. Multiple Risk Behavior Interventions: Meta-analyses of RCTs. Am. J. Prev. Med. 2017, 53, e19–e30. [Google Scholar] [CrossRef] [PubMed]
- Galán, I.; Rodríguez-Artalejo, F.; Tobías, A.; Díez-Gañán, L.; Gandarillas, A.; Zorrilla, B. Clustering of behavior-related risk factors and its association with subjective health. Gac Sanit. 2005, 19, 370–378. [Google Scholar] [CrossRef] [PubMed]
- OECD/European Observatory on Health Systems and Policies. Spain: 2019 National Health Profile, STATE of Health in the EU; OECD Publishing, Paris/European Observatory on Health Systems and Policies: Brussels, Belgium, 2019. [Google Scholar]
- Pan, A.; Wang, Y.; Talaei, M.; Hu, F.B. Relation of Smoking with Total Mortality and Cardiovascular Events among Patients With Diabetes Mellitus. Circulation 2015, 132, 1795–1804. [Google Scholar] [CrossRef]
- Liu, M.; Zhang, W.; Yan, Z.; Yuan, X. Smoking increases the risk of diabetic foot amputation: A meta-analysis. Exp. Ther. Med. 2017, 15, 1680–1685. [Google Scholar] [CrossRef] [PubMed]
- Voulgari, C.; Katsilambros, N.; Tentolouris, N. Smoking cessation predicts amelioration of microalbuminuria in newly diagnosed type 2 diabetes mellitus: A 1-year prospective study. Metabolism 2011, 60, 1456–1464. [Google Scholar] [CrossRef] [PubMed]
- Balducci, S.; Zanuso, S.; Cardelli, P.; Salvi, L.; Mazzitelli, G.; Bazuro, A.; Iacobini, C.; Nicolucci, A.; Pugliese, G. Changes in Physical Fitness Predict Improvements in Modifiable Cardiovascular Risk Factors Independently of Body Weight Loss in Subjects With Type 2 Diabetes Participating in the Italian Diabetes and Exercise Study (IDES). Diabetes Care 2012, 35, 1347–1354. [Google Scholar] [CrossRef]
- Boulé, N.G.; Haddad, E.; Kenny, G.P.; Wells, G.A.; Sigal, R.J. Effects of Exercise on Glycemic Control and Body Mass in Type 2 Diabetes Mellitus. JAMA 2001, 286, 1218–1227. [Google Scholar] [CrossRef] [PubMed]
- Owen, N.; Healy, G.N.; Matthews, C.E.; Dunstan, D.W. Too Much Sitting: The Population Health Science of Sedentary Behavior. Exerc. Sport Sci. Rev. 2010, 38, 105–113. [Google Scholar] [CrossRef]
- Dempsey, P.; Larsen, R.; Sethi, P.; Sacre, J.W.; Straznicky, N.E.; Cohen, N.D.; Cerin, E.; Lambert, G.W.; Owen, N.; Kingwell, B.A.; et al. Benefits for Type 2 Diabetes of Interrupting Prolonged Sitting with Brief Bouts of Light Walking or Simple Resistance Activities. Diabetes Care 2016, 39, 964–972. [Google Scholar] [CrossRef] [PubMed]
- Papamichou, D.; Panagiotakos, D.; Itsiopoulos, C. Dietary patterns and management of type 2 diabetes: A systematic review of randomised clinical trials. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 531–543. [Google Scholar] [CrossRef]
- Sotos-Prieto, M.; Bhupathiraju, S.N.; Mattei, J.; Fung, T.T.; Li, Y.; Pan, A.; Willett, W.C.; Rimm, E.B.; Hu, F.B. Changes in Diet Quality Scores and Risk of Cardiovascular Disease Among US Men and Women. Circulation 2015, 132, 2212–2219. [Google Scholar] [CrossRef]
- Sofi, F.; Cesari, F.; Abbate, R.; Gensini, G.F.; Casini, A. Adherence to Mediterranean diet and health status: Meta-analysis. BMJ 2008, 337, a1344. [Google Scholar] [CrossRef]
- Martinez-Lacoba, R.; Pardo-Garcia, I.; Amo-Saus, E.; Escribano-Sotos, F. Mediterranean diet and health outcomes: A systematic meta-review. Eur. J. Public Health Engl. 2018, 18, 1–10. [Google Scholar] [CrossRef] [PubMed]
- King, K.; Meader, N.; Wright, K.; Graham, H.; Power, C.; Petticrew, M.; White, M.; Sowden, A.J. Characteristics of Interventions Targeting Multiple Lifestyle Risk Behaviours in Adult Populations: A Systematic Scoping Review. PLoS ONE 2015, 10, e0117015. [Google Scholar] [CrossRef]
- Lambe, B.; Collins, C. A qualitative study of lifestyle counselling in general practice in Ireland. Fam. Pr. 2009, 27, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health, Social Services and Equality. Comprehensive Council on Lifestyle in Primary Care, linked to Community Resources in the Adult Populatio; Ministry of Health, Social Services and Equality, Publications Center: Madrid, Spain, 2015. [Google Scholar]
- Chrvala, C.A.; Sherr, D.; Lipman, R.D. Diabetes self-management education for adults with type 2 diabetes mellitus: A systematic review of the effect on glycemic control. Patient Educ. Couns. 2016, 99, 926–943. [Google Scholar] [CrossRef]
- Bhurji, N.; Javer, J.; Gasevic, D.; Khan, N.A. Improving management of type 2 diabetes in South Asian patients: A systematic review of intervention studies. BMJ Open 2016, 6, e008986. [Google Scholar] [CrossRef] [PubMed]
- Guise, J.-M.; Chang, C.; Viswanathan, M.; Glick, S.; Treadwell, J.; Umscheid, C.A.; Whitlock, E.; Fu, R.; Berliner, E.; Paynter, R.; et al. Agency for Healthcare Research and Quality Evidence-based Practice Center methods for systematically reviewing complex multicomponent health care interventions. J. Clin. Epidemiol. 2014, 67, 1181–1191. [Google Scholar] [CrossRef] [PubMed]
- Odgers-Jewell, K.; Ball, L.E.; Kelly, J.; Isenring, E.A.; Reidlinger, D.; Thomas, R. Effectiveness of group-based self-management education for individuals with Type 2 diabetes: A systematic review with meta-analyses and meta-regression. Diabet. Med. 2017, 34, 1027–1039. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, E.J.; Morgan, P.J.; Collins, C.E.; Plotnikoff, R.C.; Callister, R. Efficacy of interventions that include diet, aerobic and resistance training components for type 2 diabetes prevention: A systematic review with meta-analysis. Int. J. Behav. Nutr. Phys. Act. 2014, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- RedIAPP. Primary Care Prevention and Health Promotion Network 2018. Available online: http://www.rediapp.org (accessed on 25 April 2020).
- Zabaleta-del-Olmo, E.; Bolibar, B. Supplement Issue: Health Promotion and Disease Prevention in Primary Health Care: A focus on complex and multi-risk interventions. A focus on complex and multi-risk interventions. Prev. Med. 2015, 76, S1–S104. [Google Scholar] [CrossRef] [PubMed]
- Zabaleta-Del-Olmo, E.; Bolibar, B.; García-Ortiz, L.; García-Campayo, J.; Llobera, J.; Bellón, J.Á.; Ramos, R. Building interventions in primary health care for long-term effectiveness in health promotion and disease prevention. A focus on complex and multi-risk interventions. Prev. Med. 2015, 76, S1–S4. [Google Scholar] [CrossRef] [PubMed]
- Pons-Vigués, M.; Berenguera, A.; Coma-Auli, N.; March, S.; Pombo, H.; Masluk, B.; Pulido-Fuentes, M.; Rodriguez, C.; A Bellón, J.; Pujol-Ribera, E. Health-care users, key community informants and primary health care workers’ views on health, health promotion, health assets and deficits: Qualitative study in seven Spanish regions. Int. J. Equity Health 2017, 16, 99. [Google Scholar] [CrossRef] [PubMed]
- Berenguera, A.; Pons-Vigués, M.; Moreno-Peral, P.; March, S.; Ripoll, J.; Rubio-Valera, M.; Pombo-Ramos, H.; Asensio-Martínez, A.; Bolaños-Gallardo, E.; Martínez-Carazo, C.; et al. Beyond the consultation room: Proposals to approach health promotion in primary care according to health-care users, key community informants and primary care centre workers. Health Expect. 2017, 20, 896–910. [Google Scholar] [CrossRef]
- Zabaleta-Del-Olmo, E.; Pombo, H.; Pons-Vigués, M.; Casajuana-Closas, M.; Pujol-Ribera, E.; López-Jiménez, T.; Cabezas-Peña, C.; Martín-Borràs, C.; Serrano-Blanco, A.; Rubio-Valera, M.; et al. Complex multiple risk intervention to promote healthy behaviours in people between 45 to 75 years attended in primary health care (EIRA study): Study protocol for a hybrid trial. BMC Public Health 2018, 18, 874. [Google Scholar] [CrossRef]
- Boutron, I.; Altman, D.G.; Moher, D.; Schulz, K.F.; Ravaud, P. CONSORT NPT Group. CONSORT Statement for Randomized Trials of Nonpharmacologic Treatments: A 2017 Update and a CONSORT Extension for Nonpharmacologic Trial Abstracts. Ann. Intern Med. 2017, 167, 40–47. [Google Scholar] [CrossRef] [PubMed]
- University of Manitoba-Concept Dictionary and Glossary for Population Based Research [Internet]. Manitoba: Centre for Health Policy; 2020 [Consulted 2020 Mar 31]. Available online: http://www.mdanderson.org/departments/CIMER/ (accessed on 20 March 2021).
- Prochaska, J.O.; Redding, C.A.; Evers, K.E. The Transtheoretical model and stages of change. In Health Behavior and Health Education. Theory, Research, and Practice, 4th ed.; Glanz, K., Rimer, B.K., Viswanath, K., Eds.; John Wiley & Sons, Inc.: San Francisco, CA, USA, 2008; pp. 97–122. [Google Scholar]
- Boveda Fontan, J.; Perula de Torres, L.A.; Campinez Navarro, M.; Bosch Fontcuberta, J.M.; Barragan Brun, N.; Prados Castillejo, J.A. Current evidence on the motivational interview in the approach to health care problems in primary care. Atención Primaria 2013, 45, 486–495. [Google Scholar] [CrossRef][Green Version]
- Bickerdike, L.; Booth, A.; Wilson, P.M.; Farley, K.; Wright, K. Social prescribing: Less rhetoric and more reality. A systematic review of the evidence. BMJ Open 2017, 7, e013384. [Google Scholar] [CrossRef] [PubMed]
- Botello, B.; Palacio, S.; García, M.; Margolles, M.; Fernández, F.; Hernán, M.; Nieto, J.; Cofiño, R. Methodology for health assets mapping in a community. Gac Sanit. 2013, 27, 180–183. [Google Scholar] [CrossRef] [PubMed]
- García, R.C.; Guillem, F.C.; Seco, E.M.; Puente, J.M.G.; Manent, J.I.R.; Arango, J.S.J.; Cantera, C.M.; Muñoz, M.J.; Santiago, A.L. Grupo de Educación Sanitaria y Promoción de la Salud del PAPPS. Recomendaciones sobre el estilo de vida. Actualizacón PAPPS 2018. Aten Primaria. 2018, 50 (Suppl. 1), S29–S40. [Google Scholar] [CrossRef]
- Cuixart, C.B.; Sánchez, J.J.A.; Banegas, J.R.B.; León, C.F.; Lobos-Bejarano, J.M.; Rioboó, E.M.; Pérez, J.N.; Orozco-Beltrán, D.; Álvarez, F.V. Recomendaciones preventivas cardiovasculares. Actualización PAPPS 2018. Atención Primaria 2018, 50, 4–28. [Google Scholar] [CrossRef]
- Schröder, H.; Fitó, M.; Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Lamuela-Raventós, R.; Ros, E.; Salaverría, I.; Fiol, M.; et al. A short screener is valid for assessing Mediterranean diet adherence among older Spanish men and women. J. Nutr. 2011, 141, 1140–1145. [Google Scholar] [CrossRef]
- Kim, S.; Haines, P.S.; Siega-Riz, A.M.; Popkin, B.M. The Diet Quality Index-International (DQI-I) Provides an Effective Tool for Cross-National Comparison of Diet Quality as Illustrated by China and the United States. J. Nutr. 2003, 133, 3476–3484. [Google Scholar] [CrossRef] [PubMed]
- Roman-Viñas, B.; Serra-Majem, L.; Hagströmer, M.; Ribas-Barba, L.; Sjöström, M.; Segura-Cardona, R. International Physical Activity Questionnaire: Reliability and validity in a Spanish population. Eur. J. Sport Sci. 2010, 10, 297–304. [Google Scholar] [CrossRef]
- Crespo-Salgado, J.J.; Delgado-Martín, J.L.; Blanco-Iglesias, O.; Aldecoa-Landesa, S. Basic guidelines for detecting sedentarism and recommendations for physical activity in primary care. Atención Primaria 2015, 47, 175–183. [Google Scholar] [CrossRef]
- Badia, X.; Roset, M.; Montserrat, S. La versión española del EuroQol: Descripción y aplicaciones. Med. Clin. 1999, 112 (Suppl. 1), S79–S86. [Google Scholar]
- Adams, G.; Gulliford, M.C.; Ukoumunne, O.C.; Eldridge, S.; Chinn, S.; Campbell, M.J. Patterns of intra-cluster correlation from primary care research to inform study design and analysis. J. Clin. Epidemiol. 2004, 57, 785–794. [Google Scholar] [CrossRef]
- Parra-Sánchez, J.; Moreno-Jiménez, M.; Nicola, C.M.; Nocua-Rodríguez, I.I.; Amegló-Parejo, M.R.; del Carmen-Peña, M.; Cordero-Prieto, C.; Gajardo-Barrena, M.J. Evaluation of a supervised physical exercise program in sedentary patients over 65 years with type 2 diabetes mellitus. Atención Primaria 2015, 47, 555–562. [Google Scholar] [CrossRef][Green Version]
- Chudyk, A.; Petrella, R.J. Effects of exercise on cardiovascular risk factors in type 2 diabetes: A meta-analysis. Diabetes Care 2011, 34, 1228–1237. [Google Scholar] [CrossRef] [PubMed]
- Umpierre, D.; Ribeiro, P.A.B.; Kramer, C.K.; Leitao, C.; Zucatti, A.T.N.; Azevedo, M.J.; Gross, J.L.; Ribeiro, J.P.; Schaan, B.D. Physical Activity Advice Only or Structured exercise training and association With HbA 1c Levels in Type 2 Diabetes. JAMA J. Am. Med. Assoc. 2011, 305, 1790–1799. [Google Scholar] [CrossRef]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Van Buuren, S. Flexible Imputation of Missing Data, 2nd ed.; Chapman & Hall/CRC Interdisciplinary Statistics: Boca Ratón, FL, USA, 2018. [Google Scholar]
- R Core Team. R. A language and Environment for Statistical Computing; Foundation for Statistical Computing: Vienna, Austria, 2015; Available online: http://www.r-project.org (accessed on 20 March 2021).
- International Business Machines Corporation. IBM SPSS Statistics 25.0.; IBM: Armonk, NY, USA, 2017. [Google Scholar]
- Wulff, J.N.; Jeppesen, L.E. Multiple imputation by chained equations in praxis: Guidelines and review. J. Res. Natl. Inst. Stand. Technol. 2017, 15, 41–56. [Google Scholar]
- White, I.R.; Royston, P.; Wood, A.M. Multiple imputation using chained equations: Issues and guidance for practice. Stat Med. 2011, 30, 377–399. [Google Scholar] [CrossRef] [PubMed]
- Pocock, S.J.; Assmann, S.E.; Enos, L.E.; Kasten, L.E. Subgroup analysis, covariate adjustment and baseline comparisons in clinical trial reporting: Current practice and problems. Stat. Med. 2002, 21, 2917–2930. [Google Scholar] [CrossRef]
- Represas-Carrera, F.J.; Méndez-López, F.; Couso-Viana, S.; Masluk, B.; Magallón-Botaya, R.; Clavería, A. Baseline characteristics and quality of life in patients with Diabetes Mellitus included in the EIRA randomized clinical trial. Rev. Esp. Salud. Publica. 2021, 95, e202103034. [Google Scholar] [PubMed]
- Alonso-Domínguez, R.; García-Ortiz, L.; Patino-Alonso, M.C.; Sánchez-Aguadero, N.; Gómez-Marcos, M.A.; Recio-Rodríguez, J.I. Effectiveness of a multifactorial intervention in increasing adherence to the mediterranean diet among patients with diabetes mellitus type 2: A controlled and randomized study (EMID study). Nutrients 2019, 11, 162. [Google Scholar] [CrossRef]
- Salas-Salvadó, J.; Guasch-Ferré, M.; Lee, C.H.; Estruch, R.; Clish, C.B.; Ros, E. Protective Effects of the Mediterranean Diet on Type 2 Diabetes and Metabolic Syndrome. J. Nutr. 2015, 146, 920S–927S. [Google Scholar] [CrossRef]
- Olivares, D.; Flores, J. The effect of an educational intervention on the metabolic control of diabetic patients. Rev. Med. Chil. 2019, 147, 1024–1028. [Google Scholar] [CrossRef]
- Pan, B.; Wu, Y.; Yang, Q.; Ge, L.; Gao, C.; Xun, Y.; Tian, J.; Ding, G. The impact of major dietary patterns on glycemic control, cardiovascular risk factors, and weight loss in patients with type 2 diabetes: A network meta-analysis. J. Evid. Based Med. 2019, 12, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Khunti, K.; Gray, L.J.; Skinner, T.; Carey, M.E.; Realf, K.; Dallosso, H.; Fisher, H.; Campbell, M.; Heller, S.; Davies, M.J. Effectiveness of a diabetes education and self management programme (DESMOND) for people with newly diagnosed type 2 diabetes mellitus: Three year follow-up of a cluster randomised controlled trial in primary care. BMJ 2012, 344, e2333. [Google Scholar] [CrossRef]
- Ebrahim, S.; Taylor, F.; Ward, K.; Beswick, A.; Burke, M.; Smith, G.D. Multiple risk factor interventions for primary prevention of coronary heart disease. Cochrane Database Syst. Rev. 2011. [Google Scholar] [CrossRef]
- Zhang, P.; Hire, D.; Espeland, M.A.; Knowler, W.C.; Thomas, S.; Tsai, A.G.; Glick, H.A.; Look AHEAD Research Group. Impact of intensive lifestyle intervention on preference-based quality of life in type 2 diabetes: Results from the Look AHEAD trial. Obesity (Silver Spring) 2016, 24, 856–864. [Google Scholar] [CrossRef]
- Casillas, A.; Iglesias, K.; Flatz, A.; Burnand, B.; Peytremann-Bridevaux, I. No consistent association between processes-of-care and health-related quality of life among patients with diabetes: A missing link? BMJ Open Diabetes Res. Care 2015, 3, e000042. [Google Scholar] [CrossRef]
- Hamilton, M.T.; Hamilton, D.G.; Zderic, T.W. Sedentary behavior as a mediator of type 2 diabetes. Med. Sport Sci. 2014, 60, 11–26. [Google Scholar] [CrossRef]
- Nguyen, B.; Bauman, A.; Ding, D. Incident Type 2 Diabetes in a Large Australian Cohort Study: Does Physical Activity or Sitting Time Alter the Risk Associated With Body Mass Index? J. Phys. Act. Health 2017, 14, 13–19. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Global Strategy on Diet, Physical Activity and Health; World Health Organization: Geneva, Switzerland, 2004; Available online: https://www.who.int/publications/list/9241592222/es/ (accessed on 20 March 2021).
- González, M.; Perpinyà, D.; Mir, S.; Casellas, P.; Melció, D.; García, M.T. Efectividad de un programa de educación grupal estructurada en personas con diabetes mellitus tipo 2. Enferm Clin. 2005, 15, 141–146. [Google Scholar] [CrossRef]
- Copado, C.A.; Palomar, V.G.; Ureña, A.M.; Mengual, F.A.; Martínez, M.S.; Serralta, J.R.L. Mejora en el control de los diabéticos tipo 2 tras una intervención conjunta: Educación diabetológica y ejercicio físico. Aten Primaria. 2011, 43, 398–406. [Google Scholar] [CrossRef]
- Nongnut, O.; McCaffrey, R.; Choonhapran, P.; Chutug, P.; Rueangram, S. Development of a community participation program for diabetes mellitus prevention in a primary care unit, Thailand. Nurs. Health Sci. 2011, 13, 352–359. [Google Scholar] [CrossRef]
- Gladys, A.; Sánchez, G.; Padilla, G.; Fernández, M.; Roselló, M.; Guzmán, S. Intervención educativa comunitaria sobre la diabetes en el ámbito de la atención primaria. Rev. Panam. Salud. Publica. 2001, 9, 145–153. [Google Scholar] [CrossRef][Green Version]
- Cleland, C.; Ferguson, S.; Ellis, G.; Hunter, R.F. Validity of the International Physical Activity Questionnaire (IPAQ) for assessing moderate-to-vigorous physical activity and sedentary behaviour of older adults in the United Kingdom. BMC Med. Res. Methodol. 2018, 18, 176. [Google Scholar] [CrossRef] [PubMed]
- Limb, E.S.; Ahmad, S.; Cook, D.G.; Kerry, S.M.; Ekelund, U.; Whincup, P.H.; Victor, C.R.; Iliffe, S.; Ussher, M.; Fox-Rushby, J. Measuring change in trials of physical activity interventions: A comparison of self-report questionnaire and accelerometry within the PACE-UP trial. Int. J. Behav. Nutr. Phys Act. 2019, 16, 10. [Google Scholar] [CrossRef] [PubMed]
- Heltberg, A.; Andersen, J.S.; Kragstrup, J.; Siersma, V.; Sandholdt, H.; Ellervik, C. Social disparities in diabetes care: A general population study in Denmark. Scand. J. Prim. Health Care 2017, 35, 54–63. [Google Scholar] [CrossRef]
- Alghafri, T.S.; Alharthi, S.M.; Al-Farsi, Y.; Alrawahi, A.H.; Bannerman, E.; Craigie, A.M.; Anderson, A.S. ‘MOVEdiabetes’: A cluster randomized controlled trial to increase physical activity in adults with type 2 diabetes in primary health in Oman. BMJ Open Diabetes Res. Care 2018, 6, e000605. [Google Scholar] [CrossRef]
- Lobelo, F.; Young, D.R.; Sallis, R.; Garber, M.D.; Billinger, S.A.; Duperly, J.; Hutber, A.; Pate, R.R.; Thomas, R.J.; Widlansky, M.E.; et al. Routine Assessment and Promotion of Physical Activity in Healthcare Settings: A Scientific Statement from the American Heart Association. Circulation 2018, 137, e495–e522. [Google Scholar] [CrossRef]
- Cassetti, V.; Paredes-Carbonell, J.J. Theory of change: A tool for participatory planning and evaluation in community health. Gac Sanit. 2020, 34, 305–307. [Google Scholar] [CrossRef]
| Variables | Control (n = 356) | Intervention (n = 338) | p | ||
|---|---|---|---|---|---|
| n | med. (IQR)/% (95% CI) * | n | med. (IQR)/% (95% CI) *) | ||
| Age | 356 | 60.00 (54.00–66.00) | 338 | 60.00 (54.00–67.00) | 0.901 |
| Gender* | 356 | 338 | 1.000 | ||
| Male | 222 | 62.36 (57.24–67.28) | 210 | 62.13 (56.87–67.18) | |
| Female | 134 | 37.64 (32.72–42.76) | 128 | 37.87 (32.82–43.13) | |
| Civil status* | 354 | 335 | 0.296 | ||
| Single | 48 | 13.56 (10.29–17.42) | 31 | 9.25 (6.50–12.71) | |
| Married/Live with a partner | 244 | 68.93 (63.97–73.58) | 238 | 71.04 (66.02–75.71) | |
| Separated and/or divorced | 36 | 10.17 (7.35–13.64) | 43 | 12.84 (9.58–16.73) | |
| Widow/Widower | 25 | 7.06 (4.74–10.08) | 23 | 6.87 (4.52–9.95) | |
| Other | 1 | 0.28 (0.03–1.31) | 0 | 0.00 (0.00–0.00) | |
| Country born in* | 355 | 335 | 0.420 | ||
| Spain | 326 | 91.83 (88.64–94.34) | 318 | 94.93 (92.18–96.90) | |
| Rest of Europe | 8 | 2.25 (1.07–4.21) | 5 | 1.49 (0.57–3.24) | |
| America | 17 | 4.79 (2.92–7.39) | 9 | 2.69 (1.34–4.85) | |
| Asia | 0 | 0.00 (0.00–0.00) | 0 | 0.00 (0.00–0.00) | |
| Africa | 4 | 1.13 (0.38–2.66) | 3 | 0.90 (0.25–2.37) | |
| Spanish Autonomous Community* | 356 | 338 | 0.000 | ||
| Andalusia | 52 | 14.61 (11.23–18.56) | 48 | 14.20 (10.79–18.22) | |
| Aragón | 29 | 8.15 (5.64–11.33) | 56 | 16.57 (12.90–20.81) | |
| Balearics | 79 | 22.19 (18.11–26.72) | 72 | 21.30 (17.19–25.90) | |
| Castilla y León | 33 | 9.27 (6.59–12.61) | 49 | 14.50 (11.05–18.55) | |
| Catalonia | 49 | 13.76 (10.48–17.63) | 43 | 12.72 (9.49–16.59) | |
| Galicia | 59 | 16.57 (12.99–20.70) | 46 | 13.61 (10.27–17.57) | |
| Basque Country | 55 | 15.45 (11.98–19.48) | 24 | 7.10 (4.71–10.21) | |
| Level of education* | 353 | 335 | 0.882 | ||
| Higher Education | 42 | 11.90 (8.83–15.58) | 39 | 11.64 (8.54–15.40) | |
| Secondary Education | 123 | 34.84 (30.01–39.92) | 118 | 35.22 (30.25–40.45) | |
| Primary Education | 157 | 44.48 (39.35–49.69) | 154 | 45.97 (40.69–51.32) | |
| No studies | 31 | 8.78 (6.16–12.07) | 24 | 7.16 (4.77–10.30) | |
| Occupation* | 355 | 335 | 0.746 | ||
| Student | 0 | 0.00 (0.00–0.00) | 1 | 0.30 (0.03–1.39) | |
| Employee | 91 | 25.63 (21.30–30.36) | 83 | 24.78 (20.38–29.60) | |
| Self-employed | 37 | 10.42 (7.57–13.92) | 34 | 10.15 (7.26–13.72) | |
| Leave from work >3 months | 8 | 2.25 (1.07–4.21) | 11 | 3.28 (1.76–5.61) | |
| Unemployed and paid | 24 | 6.76 (4.49–9.73) | 14 | 4.18 (2.41–6.73) | |
| Unemployed not paid | 14 | 3.94 (2.28–6.35) | 14 | 4.18 (2.41–6.73) | |
| Household tasks | 37 | 10.42 (7.57–13.92) | 41 | 12.24 (9.06–16.07) | |
| Permanent disability | 17 | 4.79 (2.92–7.39) | 12 | 3.58 (1.97–5.99) | |
| Retired | 127 | 35.77 (30.92–40.86) | 125 | 37.31 (32.26–42.58) | |
| Glycemic control | |||||
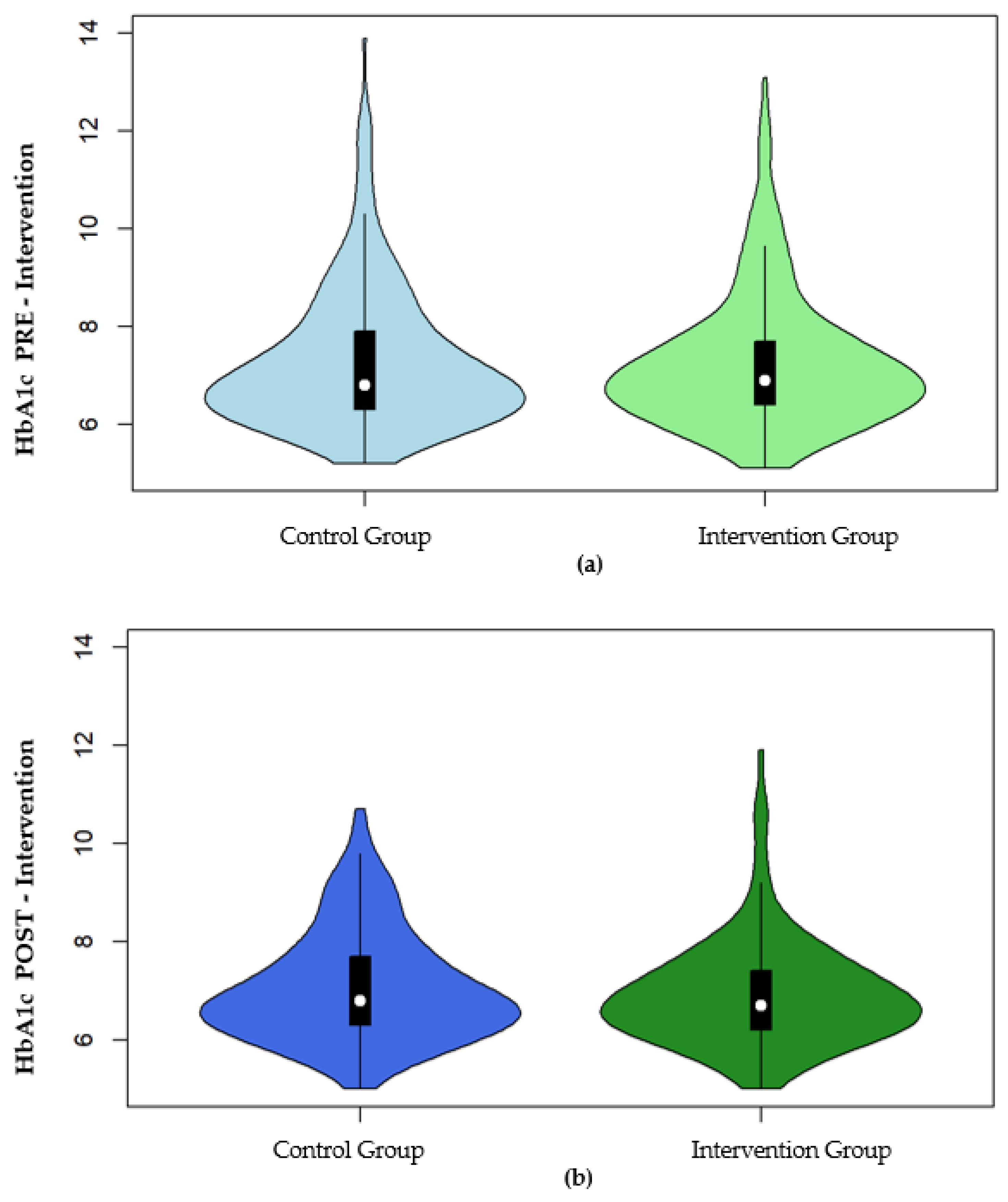
| HbA1c (%) | 279 | 6.80 (6.30–7.90) | 253 | 6.90 (6.40–7.70) | 0.884 |
| Regular/good control (HbA1c <8%) (Yes) * | 215 | 77.06 (71.87–81.70) | 205 | 81.03 (75.86–85.49) | 0.322 |
| Mediterranean diet | |||||
| Adhering to Mediterranean diet (MEDAS) | 354 | 7.00 (5.00–8.00) | 338 | 7.00 (5.00–8.00) | 0.828 |
| Good adherence (MEDAS ≥9) (Yes) * | 73 | 20.62 (16.66–25.06) | 55 | 16.27 (12.63–20.49) | 0.238 |
| Diet quality (DQI-I) | 354 | 38.00 (36.00–41.00) | 335 | 38.00 (36.00–40.00) | 0.576 |
| Physical activity | |||||
| Little physical activity (IPAQ) | 353 | 328 | 0.818 | ||
| Moderate/Intensive * | 168 | 47.59 (42.42–52.80) | 153 | 46.65 (41.30–52.05) | |
| Low (<600 MET.min/week) * | 185 | 52.41 (47.20–57.58) | 175 | 53.35 (47.95–58.70) | |
| Sedentary lifestyle (≥6 h/week sat down) (Yes) * | 111 | 42.05 (36.20–48.06) | 98 | 38.13 (32.35–44.18) | 0.373 |
| Smoking habit | |||||
| ≥1 cigarette/day (Yes) * | 145 | 40.73 (35.72–45.89) | 127 | 37.57 (32.53–42.83) | 0.437 |
| Quality of life | |||||
| VAS (EuroQol-5D5L) | 348 | 70.00 (50.00–80.00) | 335 | 70.00 (50.00–80.00) | 0.676 |
| Variables | Control (n = 356) | Intervention (n = 338) | ||
|---|---|---|---|---|
| n | med. (IQR)/% (95% CI) * | n | med. (IQR)/% (95% CI) * | |
| Glycemic control | ||||
| HbA1c (%) | 188 | 6.80 (6.30–7.70) | 169 | 6.70 (6.20–7.40) |
| Regular/good control (HbA1c <8%) (Yes) * | 153 | 81.38 (75.36–86.45) | 152 | 89.94 (84.73–93.80) |
| Mediterranean diet | ||||
| Adhering to Mediterranean diet (MEDAS) | 268 | 8.00 (6.00–9.00) | 262 | 8.00 (7.00–9.00) |
| Good adherence (MEDAS ≥9) (Yes) * | 88 | 32.84 (27.42–38.62) | 106 | 40.46 (34.65–46.48) |
| Diet quality (DQI-I) | 259 | 39.00 (37.00–41.00) | 246 | 39.00 (37.00–41.00) |
| Physical activity | ||||
| Little physical activity (IPAQ) | 256 | 250 | ||
| Moderate/Intensive * | 138 | 52.08 (46.07–58.04) | 137 | 52.29 (46.24–58.29) |
| Low (<600 MET.min/week) * | 127 | 47.92 (41.96–53.93) | 175 | 47.71 (46.71–53.76) |
| Sedentary lifestyle (≥6 h/week sat down) (Yes) * | 149 | 43.57 (38.38–48.86) | 138 | 42.86 (37.54–48.31) |
| Smoking habit | ||||
| ≥1 cigarette/day (Yes) * | 103 | 38.85 (32.89–44.51) | 85 | 32.57 (27.10–38.42) |
| Quality of life | ||||
| VAS (EuroQol-5D5L) | 261 | 70.00 (60.00–80.00) | 260 | 70.00 (60.00–80.00) |
| Variables | Adjusted Mean Difference | OR | 95% CI | p | |
|---|---|---|---|---|---|
| HbA1c (%) | −0.09 | −0.29–0.10 | 0.327 | NS | |
| Regular/good glycemia control (Yes) | 0.57 | 0.25–1.31 | 0.170 | NS | |
| Diet quality (DQI-I) | 0.25 | −0.32–0.82 | 0.392 | NS | |
| Adhering to Mediterranean diet (MEDAS) | 0.45 | 0.01–0.89 | 0.043 | * | |
| Good adherence to Mediterranean diet (Yes) | 1.62 | 1.03–2.54 | 0.036 | * | |
| Moderate/intensive physical activity (Yes) | 1.09 | 0.64–1.86 | 0.740 | NS | |
| Sedentary lifestyle (No) | 0.97 | 0.55–1.73 | 0.922 | NS | |
| Smoke ≥1 cigarette/day (No) | 0.61 | 0.54–1.06 | 0.079 | NS | |
| VAS (EuroQol-5D5L) | −1.26 | −4.98–2.45 | 0.504 | NS | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Represas-Carrera, F.; Couso-Viana, S.; Méndez-López, F.; Masluk, B.; Magallón-Botaya, R.; Recio-Rodríguez, J.I.; Pombo, H.; Leiva-Rus, A.; Gil-Girbau, M.; Motrico, E.; et al. Effectiveness of a Multicomponent Intervention in Primary Care That Addresses Patients with Diabetes Mellitus with Two or More Unhealthy Habits, Such as Diet, Physical Activity or Smoking: Multicenter Randomized Cluster Trial (EIRA Study). Int. J. Environ. Res. Public Health 2021, 18, 5788. https://doi.org/10.3390/ijerph18115788
Represas-Carrera F, Couso-Viana S, Méndez-López F, Masluk B, Magallón-Botaya R, Recio-Rodríguez JI, Pombo H, Leiva-Rus A, Gil-Girbau M, Motrico E, et al. Effectiveness of a Multicomponent Intervention in Primary Care That Addresses Patients with Diabetes Mellitus with Two or More Unhealthy Habits, Such as Diet, Physical Activity or Smoking: Multicenter Randomized Cluster Trial (EIRA Study). International Journal of Environmental Research and Public Health. 2021; 18(11):5788. https://doi.org/10.3390/ijerph18115788
Chicago/Turabian StyleRepresas-Carrera, Francisco, Sabela Couso-Viana, Fátima Méndez-López, Bárbara Masluk, Rosa Magallón-Botaya, Jose I. Recio-Rodríguez, Haizea Pombo, Alfonso Leiva-Rus, Montserrat Gil-Girbau, Emma Motrico, and et al. 2021. "Effectiveness of a Multicomponent Intervention in Primary Care That Addresses Patients with Diabetes Mellitus with Two or More Unhealthy Habits, Such as Diet, Physical Activity or Smoking: Multicenter Randomized Cluster Trial (EIRA Study)" International Journal of Environmental Research and Public Health 18, no. 11: 5788. https://doi.org/10.3390/ijerph18115788
APA StyleRepresas-Carrera, F., Couso-Viana, S., Méndez-López, F., Masluk, B., Magallón-Botaya, R., Recio-Rodríguez, J. I., Pombo, H., Leiva-Rus, A., Gil-Girbau, M., Motrico, E., Martí-Lluch, R., Gude, F., & Clavería, A. (2021). Effectiveness of a Multicomponent Intervention in Primary Care That Addresses Patients with Diabetes Mellitus with Two or More Unhealthy Habits, Such as Diet, Physical Activity or Smoking: Multicenter Randomized Cluster Trial (EIRA Study). International Journal of Environmental Research and Public Health, 18(11), 5788. https://doi.org/10.3390/ijerph18115788








