Blood Pressure and Tooth Loss: A Large Cross-Sectional Study with Age Mediation Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Setting
2.2. Participants
2.3. Variables
2.4. Statistical Analysis
3. Results
3.1. Baseline Characteristics
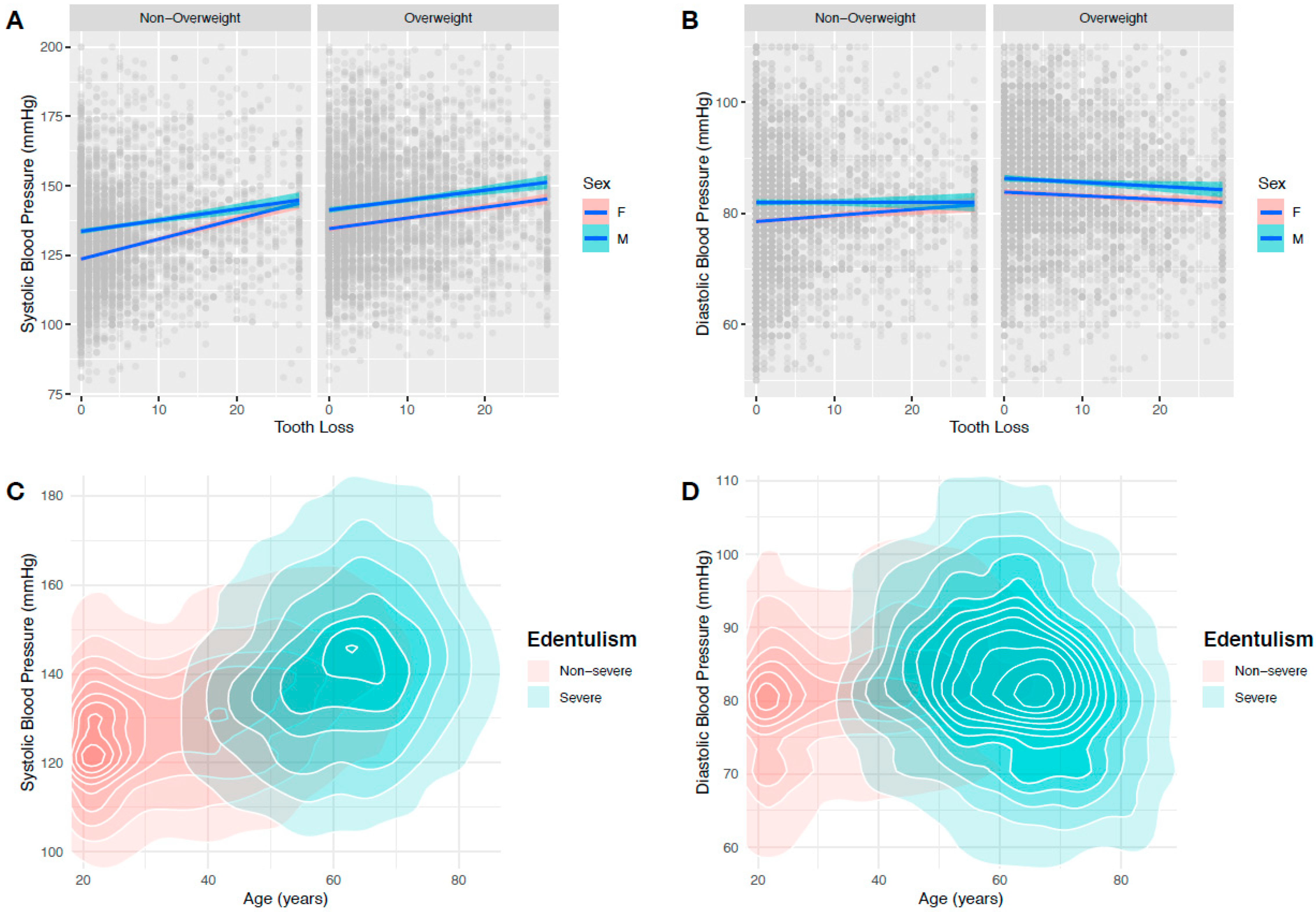
3.2. Relationship between Hypertension and Tooth Loss
3.3. Mediation Analysis of Age
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kearney, P.M.; Whelton, M.; Reynolds, K.; Muntner, P.; Whelton, P.K.; He, J. Global burden of hypertension: Analysis of worldwide data. Lancet 2005, 365, 217–223. [Google Scholar] [CrossRef]
- WHO Hypertension. Available online: https://www.who.int/health-topics/hypertension/#tab=tab_1 (accessed on 17 August 2020).
- Kannel, W.B. Hypertension and other risk factors in coronary heart disease. Am. Heart J. 1987, 114, 918–925. [Google Scholar] [CrossRef]
- Peres, M.A.; Tsakos, G.; Barbato, P.R.; Silva, D.A.S.; Peres, K.G. Tooth loss is associated with increased blood pressure in adults—A multidisciplinary population-based study. J. Clin. Periodontol. 2012, 39, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Kiiskinen, U.; Vartiainen, E.; Puska, P.; Aromaa, A. Long-term cost and life-expectancy consequences of hypertension. J. Hypertens. 1998, 16, 1103–1112. [Google Scholar] [CrossRef]
- Zanchetti, A. Factors and consequences of hypertension. J. Hypertens. 2017, 35, 1–2. [Google Scholar] [CrossRef]
- Muñoz Aguilera, E.; Suvan, J.; Buti, J.; Czesnikiewicz-Guzik, M.; Barbosa Ribeiro, A.; Orlandi, M.; Guzik, T.J.; Hingorani, A.D.; Nart, J.; D’Aiuto, F. Periodontitis is associated with hypertension: A systematic review and meta-analysis. Cardiovasc. Res. 2020, 116, 28–39. [Google Scholar] [CrossRef]
- Machado, V.; Aguilera, E.M.; Botelho, J.; Hussain, S.B.; Leira, Y.; Proença, L.; D’Aiuto, F.; Mendes, J.J. Association between Periodontitis and High Blood Pressure: Results from the Study of Periodontal Health in Almada-Seixal (SoPHiAS). J. Clin. Med. 2020, 9, 1585. [Google Scholar] [CrossRef]
- Da, D.; Wang, F.; Zhang, H.; Zeng, X.; Jiang, Y.; Zhao, Q.; Luo, J.; Ding, D.; Zhang, Y.; Wu, B. Association between tooth loss and hypertension among older Chinese adults: A community-based study. BMC Oral Health 2019, 19, 1–7. [Google Scholar] [CrossRef]
- Choe, H.; Kim, Y.H.; Park, J.W.; Kim, S.Y.; Lee, S.Y.; Jee, S.H. Tooth loss, hypertension and risk for stroke in a Korean population. Atherosclerosis 2009, 203, 550–556. [Google Scholar] [CrossRef]
- Del Brutto, O.H.; Mera, R.M.; Del Brutto, V.J.; Zambrano, M.; Montenegro, J.E.; Castillo, P.R. Edentulism associates with poor cardiovascular health. Results from the Atahualpa Project. Int. J. Cardiol. 2014, 176, 1013–1014. [Google Scholar] [CrossRef]
- Del Brutto, O.H.; Mera, R.M.; Recalde, B.Y.; Torpey, A.P.; Hill, J.P.; Generale, L.M.; Peralta, L.D.; Sedler, M.J. Association Between Pulsatile Components of Blood Pressure and Severe Tooth Loss in Rural Ecuador: The Three Villages Study. J. Prim. Care Community Health 2020, 11, 2150132720928670. [Google Scholar]
- Kassebaum, N.J.; Smith, A.G.C.; Bernabé, E.; Fleming, T.D.; Reynolds, A.E.; Vos, T.; Murray, C.J.L.; Marcenes, W. Global, Regional, and National Prevalence, Incidence, and Disability-Adjusted Life Years for Oral Conditions for 195 Countries, 1990–2015: A Systematic Analysis for the Global Burden of Diseases, Injuries, and Risk Factors. J. Dent. Res. 2017, 96, 380–387. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Oral Health. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/oral-health (accessed on 10 December 2020).
- Craddock, H.L. Consequences of tooth loss: 1. The patient perspective--aesthetic and functional implications. Dent. Update 2009, 36, 616–619. [Google Scholar] [CrossRef] [PubMed]
- Chauncey, H.H.; Muench, M.E.; Kapur, K.K.; Wayler, A.H. The effect of the loss of teeth on diet and nutrition. Int. Dent. J. 1984, 34, 98–104. [Google Scholar] [PubMed]
- McCance, R.A. The Nutritional Revolution. BMJ 1952, 1, 145–146. [Google Scholar] [CrossRef][Green Version]
- Haag, D.G.; Peres, K.G.; Brennan, D.S. Tooth loss and general quality of life in dentate adults from Southern Brazil. Qual. Life Res. 2017, 26, 2647–2657. [Google Scholar] [CrossRef]
- Darnaud, C.; Thomas, F.; Pannier, B.; Danchin, N.; Bouchard, P. Oral health and blood pressure: The IPC cohort. Am. J. Hypertens. 2015, 28, 1257–1261. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: Guidelines for reporting observational studies. Int. J. Surg. 2014, 12, 1495–1499. [Google Scholar] [CrossRef]
- Machado, V.; Mesquita, M.F.; Bernardo, M.A.; Casal, E.; Proença, L.; Mendes, J.J. IL-6 and TNF-α salivary levels according to the periodontal status in Portuguese pregnant women. PeerJ 2018, 6, e4710. [Google Scholar] [CrossRef]
- Kintiraki, E.; Papakatsika, S.; Kotronis, G.; Goulis, D.; Kotsis, V. Pregnancy-Induced hypertension. Hormones 2015, 10, 211–223. [Google Scholar] [CrossRef]
- Vallée, A.; Yannoutsos, A.; Zhang, Y.; Henry-Bonniot, G.; Protogerou, A.; Topouchian, J.; Safar, M.E.; Blacher, J. Determinants of pulse pressure amplification in hypertensive and diabetic patients. Hypertens. Res. 2019, 42, 374–384. [Google Scholar] [CrossRef] [PubMed]
- Muntner, P.; Shimbo, D.; Carey, R.M.; Charleston, J.B.; Gaillard, T.; Misra, S.; Myers, M.G.; Ogedegbe, G.; Schwartz, J.E.; Townsend, R.R.; et al. Measurement of Blood Pressure in Humans: A Scientific Statement from the American Heart Association. Hypertension 2019, 73, E35–E66. [Google Scholar] [CrossRef] [PubMed]
- Mancia, G.; De Backer, G.; Dominiczak, A.; Cifkova, R.; Fagard, R.; Germano, G.; Grassi, G.; Heagerty, A.M.; Kjeldsen, S.E.; Laurent, S.; et al. 2007 Guidelines for the Management of Arterial Hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). J. Hypertens. 2007, 25, 1105–1187. [Google Scholar] [CrossRef] [PubMed]
- Mancia, G.; Fagard, R.; Narkiewicz, K.; Redon, J.; Zanchetti, A.; Böhm, M.; Christiaens, T.; Cifkova, R.; De Backer, G.; Dominiczak, A.; et al. 2013 ESH/ESC guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur. Heart J. 2013, 34, 2159–2219. [Google Scholar] [PubMed]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef] [PubMed]
- Del Brutto, O.H.; Mera, R.M.; Zambrano, M.; Del Brutto, V.J. Severe edentulism is a major risk factor influencing stroke incidence in rural Ecuador (The Atahualpa Project). Int. J. Stroke 2017, 12, 201–204. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Use of Glycated Haemoglobin (HbA1c) in the Diagnosis of Diabetes Mellitus. Available online: https://www.who.int/diabetes/publications/report-hba1c_2011.pdf?ua=1 (accessed on 10 December 2020).
- Hussain, M.M.; Solomon, I.E.; Pazdernik, V.; Bandarpalle, S.; Navarro, I.; Shneyder, T.; Lebeau, L. Relationship between elevated automatic blood pressure readings and manual blood pressure readings in adult patients with normal and high body mass index. J. Am. Osteopath. Assoc. 2019, 119, 364–370. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, D.; Pang, Z.; Jiang, W.; Wang, S.; Li, S.; Von Bornemann Hjelmborg, J.; Tan, Q. Multivariate modeling of body mass index, pulse pressure, systolic and diastolic blood pressure in Chinese twins. Twin Res. Hum. Genet. 2015, 18, 73–78. [Google Scholar] [CrossRef]
- Wu, O.; Leng, J.H.; Yang, F.F.; Yang, H.M.; Zhang, H.; Li, Z.F.; Zhang, X.Y.; Yuan, C.D.; Li, J.J.; Pan, Q.; et al. A comparative research on obesity hypertension by the comparisons and associations between waist circumference, body mass index with systolic and diastolic blood pressure, and the clinical laboratory data between four special Chinese adult groups. Clin. Exp. Hypertens. 2018, 40, 16–21. [Google Scholar] [CrossRef]
- Pavlović, M.; Milković-Kraus, S.; Jovanović, V.; Hercigonja-Szekeres, M. Ageing, arterial blood pressure, body mass index, and diet. Arh. Hig. Rada Toksikol. 2012, 63, 3–9. [Google Scholar] [CrossRef]
- Chapple, I.L.C.; Bouchard, P.; Cagetti, M.G.; Campus, G.; Carra, M.C.; Cocco, F.; Nibali, L.; Hujoel, P.; Laine, M.L.; Lingstrom, P.; et al. Interaction of lifestyle, behaviour or systemic diseases with dental caries and periodontal diseases: Consensus report of group 2 of the joint EFP/ORCA workshop on the boundaries between caries and periodontal diseases. J. Clin. Periodontol. 2017, 44, S39–S51. [Google Scholar] [CrossRef] [PubMed]
- Darveau, R.P. Periodontitis: A polymicrobial disruption of host homeostasis. Nat. Rev. Microbiol. 2010, 8, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Mikolajczyk, T.P.; Nosalski, R.; Szczepaniak, P.; Budzyn, K.; Osmenda, G.; Skiba, D.; Sagan, A.; Wu, J.; Vinh, A.; Marvar, P.J.; et al. Role of chemokine RANTES in the regulation of perivascular inflammation, T-cell accumulation, and vascular dysfunction in hypertension. FASEB J. 2016, 30, 1987–1999. [Google Scholar] [CrossRef] [PubMed]
- Czesnikiewicz-Guzik, M.; Nosalski, R.; Mikolajczyk, T.P.; Vidler, F.; Dohnal, T.; Dembowska, E.; Graham, D.; Harrison, D.G.; Guzik, T.J. Th1-type immune responses to Porphyromonas gingivalis antigens exacerbate angiotensin II-dependent hypertension and vascular dysfunction. Br. J. Pharmacol. 2019, 176, 1922–1931. [Google Scholar] [CrossRef]
- Dar-Odeh, N.; Borzangy, S.; Babkair, H.; Farghal, L.; Shahin, G.; Fadhlalmawla, S.; Alhazmi, W.; Taher, S.; Abu-Hammad, O. Association of dental caries, retained roots, and missing teeth with physical status, diabetes mellitus and hypertension in women of the reproductive age. Int. J. Environ. Res. Public Health 2019, 16, 2565. [Google Scholar] [CrossRef]
- Zhu, Y.; Hollis, J.H. Tooth loss and its association with dietary intake and diet quality in American adults. J. Dent. 2014, 42, 1428–1435. [Google Scholar] [CrossRef]
- Rapsomaniki, E.; Timmis, A.; George, J.; Pujades-Rodriguez, M.; Shah, A.D.; Denaxas, S.; White, I.R.; Caulfield, M.J.; Deanfield, J.E.; Smeeth, L.; et al. Blood pressure and incidence of twelve cardiovascular diseases: Lifetime risks, healthy life-years lost, and age-specific associations in 1·25 million people. Lancet 2014, 383, 1899–1911. [Google Scholar] [CrossRef]
- Haag, D.G.; Peres, K.G.; Balasubramanian, M.; Brennan, D.S. Oral Conditions and Health-Related Quality of Life: A Systematic Review. J. Dent. Res. 2017, 96, 864–874. [Google Scholar] [CrossRef]
- Teh, C.H.; Chan, Y.Y.; Lim, K.H.; Kee, C.C.; Lim, K.K.; Yeo, P.S.; Azahadi, O.; Fadhli, Y.; Tahir, A.; Lee, H.L.; et al. Association of physical activity with blood pressure and blood glucose among Malaysian adults: A population-based study Chronic Disease epidemiology. BMC Public Health 2015, 15, 1–7. [Google Scholar] [CrossRef]
- Rippe, J.M.; Angelopoulos, T.J. Sugars, obesity, and cardiovascular disease: Results from recent randomized control trials. Eur. J. Nutr. 2016, 55, 45–53. [Google Scholar] [CrossRef]
- Hoare, E.; Varsamis, P.; Owen, N.; Dunstan, D.W.; Jennings, G.L.; Kingwell, B.A. Sugar-and intense-sweetened drinks in Australia: A systematic review on cardiometabolic risk. Nutrients 2017, 9, 1075. [Google Scholar] [CrossRef] [PubMed]
- Mansoori, S.; Kushner, N.; Suminski, R.R.; Farquhar, W.B.; Chai, S.C. Added sugar intake is associated with blood pressure in older females. Nutrients 2019, 11, 2060. [Google Scholar] [CrossRef] [PubMed]
- Ferrannini, E.; Cushman, W.C. Diabetes and hypertension: The bad companions. Lancet 2012, 380, 601–610. [Google Scholar] [CrossRef]
- Virdis, A.; Giannarelli, C.; Fritsch Neves, M.; Taddei, S.; Ghiadoni, L. Cigarette Smoking and Hypertension. Curr. Pharm. Des. 2010, 16, 2518–2525. [Google Scholar] [CrossRef] [PubMed]
- Carson, S.J.; Burns, J. Impact of smoking on tooth loss in adults. Evid. Based. Dent. 2016, 17, 73–74. [Google Scholar] [CrossRef] [PubMed]
- Preshaw, P.M.; Alba, A.L.; Herrera, D.; Jepsen, S.; Konstantinidis, A.; Makrilakis, K.; Taylor, R. Periodontitis and diabetes: A two-way relationship Matrix metalloproteinase NHANES National Health and Nutrition Examination Survey. Diabetologia 2012, 55, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Chow, C.K.; Teo, K.K.; Rangarajan, S.; Islam, S.; Gupta, R.; Avezum, A.; Bahonar, A.; Chifamba, J.; Dagenais, G.; Diaz, R.; et al. Prevalence, awareness, treatment, and control of hypertension in rural and urban communities in high-, middle-, and low-income countries. JAMA—J. Am. Med. Assoc. 2013, 310, 959–968. [Google Scholar] [CrossRef]
- Chau, K.; Girerd, N.; Zannad, F.; Rossignol, P.; Boivin, J.M. Health-related determinants of undiagnosed arterial hypertension: A population-based study. Fam. Pract. 2018, 36, 276–283. [Google Scholar] [CrossRef]
- Melgarejo, J.D.; Maestre, G.E.; Thijs, L.; Asayama, K.; Boggia, J.; Casiglia, E.; Hansen, T.W.; Imai, Y.; Jacobs, L.; Jeppesen, J.; et al. Prevalence, Treatment, and Control Rates of Conventional and Ambulatory Hypertension Across 10 Populations in 3 Continents. Hypertension 2017, 70, 50–58. [Google Scholar] [CrossRef]
- Scholes, S.; Conolly, A.; Mindell, J.S. Income-based inequalities in hypertension and in undiagnosed hypertension: Analysis of Health Survey for England data. J. Hypertens. 2020, 38, 912–924. [Google Scholar] [CrossRef]
- Benjamin, E.J.; Virani, S.S.; Callaway, C.W.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Chiuve, S.E.; Cushman, M.; Delling, F.N.; Deo, R.; et al. Heart Disease and Stroke Statistics—2018 Update: A Report from the American Heart Association. Circulation 2018, 137, E67–E492. [Google Scholar] [CrossRef] [PubMed]
- Wall, H.K.; Hannan, J.A.; Wright, J.S. Patients with undiagnosed hypertension hiding in plain sight. JAMA—J. Am. Med. Assoc. 2014, 312, 1973–1974. [Google Scholar] [CrossRef] [PubMed]
- Ataklte, F.; Erqou, S.; Kaptoge, S.; Taye, B.; Echouffo-Tcheugui, J.B.; Kengne, A.P. Burden of undiagnosed hypertension in sub-saharan africa: A systematic review and meta-analysis. Hypertension 2015, 65, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Sogunuru, G.P.; Mishra, S. Asian management of hypertension: Current status, home blood pressure, and specific concerns in India. J. Clin. Hypertens. 2020, 22, 479–482. [Google Scholar] [CrossRef]
- Bovet, P.; Chiolero, A. Prevalence and control of hypertension. Lancet 2018, 392, 1305–1306. [Google Scholar] [CrossRef]
- Olsen, M.H.; Angell, S.Y.; Asma, S.; Boutouyrie, P.; Burger, D.; Chirinos, J.A.; Damasceno, A.; Delles, C.; Gimenez-Roqueplo, A.P.; Hering, D.; et al. A call to action and a lifecourse strategy to address the global burden of raised blood pressure on current and future generations: The Lancet Commission on hypertension. Lancet 2016, 388, 2665–2712. [Google Scholar] [CrossRef]
- Rodrigues, A.P.; Gaio, V.; Kislaya, I.; Graff-Iversen, S.; Cordeiro, E.; Silva, A.C.; Namorado, S.; Barreto, M.; Gil, A.P.; Antunes, L.; et al. Sociodemographic disparities in hypertension prevalence: Results from the first Portuguese National Health Examination Survey. Rev. Port. Cardiol. 2019, 38, 547–555. [Google Scholar] [CrossRef]
- Landry, R.G.; Jean, M. Periodontal Screening and Recording (PSR) Index: Precursors, utility and limitations in a clinical setting. Int. Dent. J. 2002, 52, 35–40. [Google Scholar] [CrossRef]
- Machado, V.; Botelho, J.; Amaral, A.; Proença, L.; Alves, R.; Rua, J.; Cavacas, M.A.; Delgado, A.S.; Mendes, J.J. Prevalence and extent of chronic periodontitis and its risk factors in a Portuguese subpopulation: A retrospective cross-sectional study and analysis of Clinical Attachment Loss. PeerJ 2018, 6, e5258. [Google Scholar] [CrossRef]
- Botelho, J.; Machado, V.; Proença, L.; Alves, R.; Cavacas, M.A.; Amaro, L.; Mendes, J.J. Study of Periodontal Health in Almada-Seixal (SoPHiAS): A cross-sectional study in the Lisbon Metropolitan Area. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Botelho, J.; Machado, V.; Proença, L.; Oliveira, M.J.; Cavacas, M.A.; Amaro, L.; Águas, A.; Mendes, J.J. Perceived xerostomia, stress and periodontal status impact on elderly oral health-related quality of life: Findings from a cross-sectional survey. BMC Oral Health 2020, 20, 199. [Google Scholar] [CrossRef] [PubMed]
- Machado, V.; Botelho, J.; Proença, L.; Alves, R.; Oliveira, M.J.; Amaro, L.; Águas, A.; Mendes, J.J. Periodontal status, perceived stress, diabetes mellitus and oral hygiene care on quality of life: A structural equation modelling analysis. BMC Oral Health 2020, 20, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Machado, V.; Botelho, J.; Ramos, C.; Proença, L.; Alves, R.; Cavacas, M.A.; Mendes, J.J. Psychometric properties of the Brief Illness Perception Questionnaire (Brief-IPQ) in periodontal diseases. J. Clin. Periodontol. 2019, 46, 1183–1191. [Google Scholar] [CrossRef] [PubMed]
- Machado, V.; Botelho, J.; Proença, L.; Mendes, J.J. Self-reported illness perception and oral health related quality of life predict adherence to initial periodontal treatment. J. Clin. Periodontol. 2020, in press. [Google Scholar] [CrossRef]
- Santos, J.; Antunes, L.; Namorado, S.; Kislaya, I.; João Santos, A.; Rodrigues, A.P.; Braz, P.; Gaio, V.; Barreto, M.; Lyshol, H.; et al. Oral hygiene habits in Portugal: Results from the first Health Examination Survey (INSEF 2015). Acta Odontol. Scand. 2019, 77, 334–339. [Google Scholar] [CrossRef]
| Variable | Global (n = 10,576) | ≥10 Teeth Lost (n = 2465) | <10 Teeth Lost (n = 8111) | p-Value * |
|---|---|---|---|---|
| Age, mean (SD) (years) | 44.9 (17.9) | 61.4 (12.4) | 39.9 (16.3) | <0.001 |
| Female gender, % (n) | 59.7 (6312) | 59.6 (1470) | 59.7 (4842) | 0.996 |
| BMI, mean (SD) (kg/m2) | 25.5 (4.7) | 27.1 (4.7) | 25.0 (4.5) | <0.001 |
| Current smoker, % (n) | 25.7 (2722) | 27.0 (667) | 25.3 (2055) | <0.001 |
| SBP, mean (SD) (mmHg) | 135.3 (20.1) | 142.1 (21.0) | 133.2 (19.3) | <0.001 |
| DBP, mean (SD) (mmHg) | 83.0 (12.3) | 83.5 (12.7) | 82.8 (12.2) | 0.008 |
| PP, mean (SD) (mmHg) | 76.7 (2.8) | 76.4 (2.9) | 76.8 (2.7) | <0.001 |
| Missing teeth, mean (SD) | 5.8 (7.2) | 17.1 (5.7) | 2.4 (2.7) | <0.001 |
| Medical conditions, mean (SD) | 1 (1) | 2 (1) | 1 (1) | <0.001 |
| Self-reported hypertension, % (n) | 18.1 (1919) | 22.2 (1800) | 4.8 (119) | <0.001 |
| Use of antihypertensive drugs, % (n) | 21.1 (2230) | 21.7 (1757) | 19.2 (473) | 0.008 |
| Hypertension, % (n) | 43.2 (5470) | 56.1 (1384) | 39.3 (3186) | <0.001 |
| Blood Pressure, % (n) | ||||
| Optimal | 16.5 (1747) | 10.7 (264) | 18.3 (1483) | <0.001 |
| Normal | 18.9 (1996) | 12.8 (316) | 20.7 (1680) | |
| High Normal | 21.4 (2263) | 20.3 (501) | 21.7 (1762) | |
| Grade 1 | 27.8 (2943) | 34.0 (838) | 26.0 (2105) | |
| Grade 2 | 10.8 (1139) | 15.6 (384) | 9.3 (755) | |
| Grade 3 | 4.6 (488) | 6.6 (162) | 4.0 (326) | |
| Hypertension (excluding self-reported hypertension and taking antihypertensive drugs) (n = 8069), % (n) | 43.4 (3504) | 55.8 (1086) | 39.5 (2418) | <0.001 |
| Variable | Hypertension | Hypertension Grade 3 |
|---|---|---|
| All participants | ||
| Model 1 | 1.05 (1.05–1.06) *** | 1.04 (1.03–1.05) *** |
| Model 2 | 1.05 (1.05–1.06) *** | 1.04 (1.03–1.05) *** |
| Model 3 | 1.05 (1.05–1.06) *** | 1.04 (1.03–1.05) *** |
| Model 4 | 1.04 (1.03–1.05) *** | 1.03 (1.02–1.05) *** |
| Model 5 | 1.00 (1.00–1.01) | 1.01 (1.00–1.03) |
| Participants not taking antihypertensive medication (n = 8346) | ||
| Model 1 | 1.06 (1.05–1.07) *** | 1.05 (1.03–1.06) *** |
| Model 2 | 1.06 (1.05–1.08) *** | 1.03 (1.01–1.06) * |
| Model 3 | 1.06 (1.05–1.08) *** | 1.03 (1.01–1.06) * |
| Model 4 | 1.05 (1.03–1.06) *** | 1.02 (1.00–1.05) |
| Model 5 | 1.00 (0.99–1.02) | 1.00 (0.97–1.03) |
| Variable | Overall | No Antihypertensive Use (n = 8346) | Antihypertensive Use (n = 2230) | |||
|---|---|---|---|---|---|---|
| β Coefficient (SE) | p-Value | β Coefficient (SE) | p-Value | β Coefficient (SE) | p-Value | |
| SBP (mmHg) | ||||||
| Age (years) | 0.28 (0.01) | <0.001 | 0.27 (0.02) | <0.001 | 0.30 (0.03) | <0.001 |
| BMI (kg/m2) | 0.91 (0.04) | <0.001 | 0.93 (0.05) | <0.001 | 0.54 (0.06) | <0.001 |
| Missing teeth (n) | 0.06 (0.03) | 0.076 | 0.07 (0.04) | 0.049 | 0.00 (0.07) | 0.955 |
| DBP (mmHg) | ||||||
| Age (years) | 0.01 (0.01) | 0.135 | 0.01 (0.01) | 0.511 | 0.04 (0.06) | 0.042 |
| BMI (kg/m2) | 0.59 (0.03) | <0.001 | 0.61 (0.03) | <0.001 | 0.54 (0.06) | <0.001 |
| Missing teeth (n) | −0.03 (0.02) | 0.115 | −0.02 (0.02) | 0.415 | −0.09 (0.05) | 0.059 |
| Variable | Females (Overall) (n = 6312) | Females not Taking Antihypertensive Use (n = 4937) | Females Taking Antihypertensive Use (n = 1375) | Males (Overall) (n = 4264) | Males not Taking Antihypertensive Use (n = 3409) | Males Taking Antihypertensive Use (n = 855) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| β Coefficient (SE) | p-Value | β Coefficient (SE) | p-Value | β Coefficient (SE) | p-Value | β Coefficient (SE) | p-Value | β Coefficient (SE) | p-Value | β Coefficient (SE) | p-Value | |
| SBP (mmHg) | ||||||||||||
| Age (years) | 0.29 (0.02) | <0.001 | 0.28 (0.02) | <0.001 | 0.32 (0.04) | <0.001 | 0.24 (0.02) | <0.001 | 0.24 (0.02) | <0.001 | 0.24 (0.05) | <0.001 |
| BMI (kg/m2) | 0.90 (0.05) | <0.001 | 0.914 (0.05) | <0.001 | 0.82 (0.11) | <0.001 | 0.74 (0.07) | <0.001 | 0.76 (0.08) | <0.001 | 0.70 (0.15) | <0.001 |
| Missing teeth (n) | 0.07 (0.04) | 0.065 | 0.08 (0.04) | 0.065 | 0.04 (0.09) | 0.690 | 0.08 (0.05) | 0.114 | 0.10 (0.06) | 0.087 | 0.02 (0.11) | 0.896 |
| DBP (mmHg) | ||||||||||||
| Age (years) | 0.01 (0.01) | 0.402 | 0.00 (0.01) | 0.929 | 0.04 (0.02) | 0.090 | 0.01 (0.01) | 0.626 | 0.00 (0.02) | 0.905 | 0.03 (0.03) | 0.397 |
| BMI (kg/m2) | 0.55 (0.03) | <0.001 | 0.56 (0.04) | <0.001 | 0.52 (0.10) | <0.001 | 0.59 (0.05) | <0.001 | 0.611 (0.05) | <0.001 | 0.52 (0.10) | <0.001 |
| Missing teeth (n) | −0.02 (0.03) | 0.559 | 0.01 (0.03) | 0.853 | −0.08 (0.06) | 0.103 | −0.03 (0.03) | 0.445 | −0.02 (0.04) | 0.617 | −0.05 (0.08) | 0.480 |
| Exposure: Missing Teeth/Outcome: Systolic Blood Pressure/Mediator: Age | ||||
| Effect | Estimate | SE | p-Value | 95% CI |
| Exposure → Mediator | 1.25 | 0.18 | <0.001 | 1.21–1.28 |
| Mediator → Exposure | 0.36 | 0.12 | <0.001 | 0.34–0.39 |
| Total Effect | 0.50 | 0.02 | <0.001 | 0.45–0.55 |
| Direct Effect | 0.05 | 0.03 | 0.073 | −0.00–0.10 |
| Mediated Effect | 0.45 | 0.02 | - | 0.42–0.49 |
| AB/C (Age percentage mediated) = 80% | ||||
| Exposure: Missing Teeth/Outcome: Diastolic Blood Pressure/Mediator: Age | ||||
| Effect | Estimate | SE | p-Value | 95% CI |
| Exposure → Mediator | 1.25 | 0.02 | <0.001 | 1.21–1.28 |
| Mediator → Exposure | 0.06 | 0.01 | <0.001 | 0.04–0.07 |
| Total Effect | 0.08 | 0.01 | <0.001 | 0.05–0.11 |
| Direct Effect | 0.01 | 0.02 | 0.726 | −0.03–0.04 |
| Mediated Effect | 0.07 | 0.01 | - | 0.05–0.09 |
| AB/C (Age percentage mediated) = 87.5% | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendes, J.J.; Viana, J.; Cruz, F.; Pereira, D.; Ferreira, S.; Pereira, P.; Proença, L.; Machado, V.; Botelho, J.; Rua, J.; et al. Blood Pressure and Tooth Loss: A Large Cross-Sectional Study with Age Mediation Analysis. Int. J. Environ. Res. Public Health 2021, 18, 285. https://doi.org/10.3390/ijerph18010285
Mendes JJ, Viana J, Cruz F, Pereira D, Ferreira S, Pereira P, Proença L, Machado V, Botelho J, Rua J, et al. Blood Pressure and Tooth Loss: A Large Cross-Sectional Study with Age Mediation Analysis. International Journal of Environmental Research and Public Health. 2021; 18(1):285. https://doi.org/10.3390/ijerph18010285
Chicago/Turabian StyleMendes, José João, João Viana, Filipe Cruz, Dinis Pereira, Sílvia Ferreira, Paula Pereira, Luís Proença, Vanessa Machado, João Botelho, João Rua, and et al. 2021. "Blood Pressure and Tooth Loss: A Large Cross-Sectional Study with Age Mediation Analysis" International Journal of Environmental Research and Public Health 18, no. 1: 285. https://doi.org/10.3390/ijerph18010285
APA StyleMendes, J. J., Viana, J., Cruz, F., Pereira, D., Ferreira, S., Pereira, P., Proença, L., Machado, V., Botelho, J., Rua, J., & Delgado, A. S. (2021). Blood Pressure and Tooth Loss: A Large Cross-Sectional Study with Age Mediation Analysis. International Journal of Environmental Research and Public Health, 18(1), 285. https://doi.org/10.3390/ijerph18010285









