Does Multiple Sclerosis Differently Impact Physical Activity in Women and Man? A Quantitative Study Based on Wearable Accelerometers
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Data Collection and Processing
- step counts (SC);
- vector magnitude counts (VM), which are composite measures of accelerometric counts considered a proxy for overall physical activity [38]. VM is calculated with the following equation:in which x, y and z are the values of accelerations measured by the device in each of the three orthogonal directions;VM = √(x2+y2+z2)
- levels of PA intensity classified into three categories according to the associated value of metabolic equivalent (MET), namely sedentary behavior (SB, 0–1.5 MET), light intensity PA (LPA, 1.5–3 MET) and moderate-to-vigorous PA (MVPA, > 3 MET). Such a discrimination was carried out based on the cut-points for accelerometric counts per minute (cpm) proposed by Sandroff et al. [39] as reported in Table 2.
2.3. Statistical Analyses
3. Results
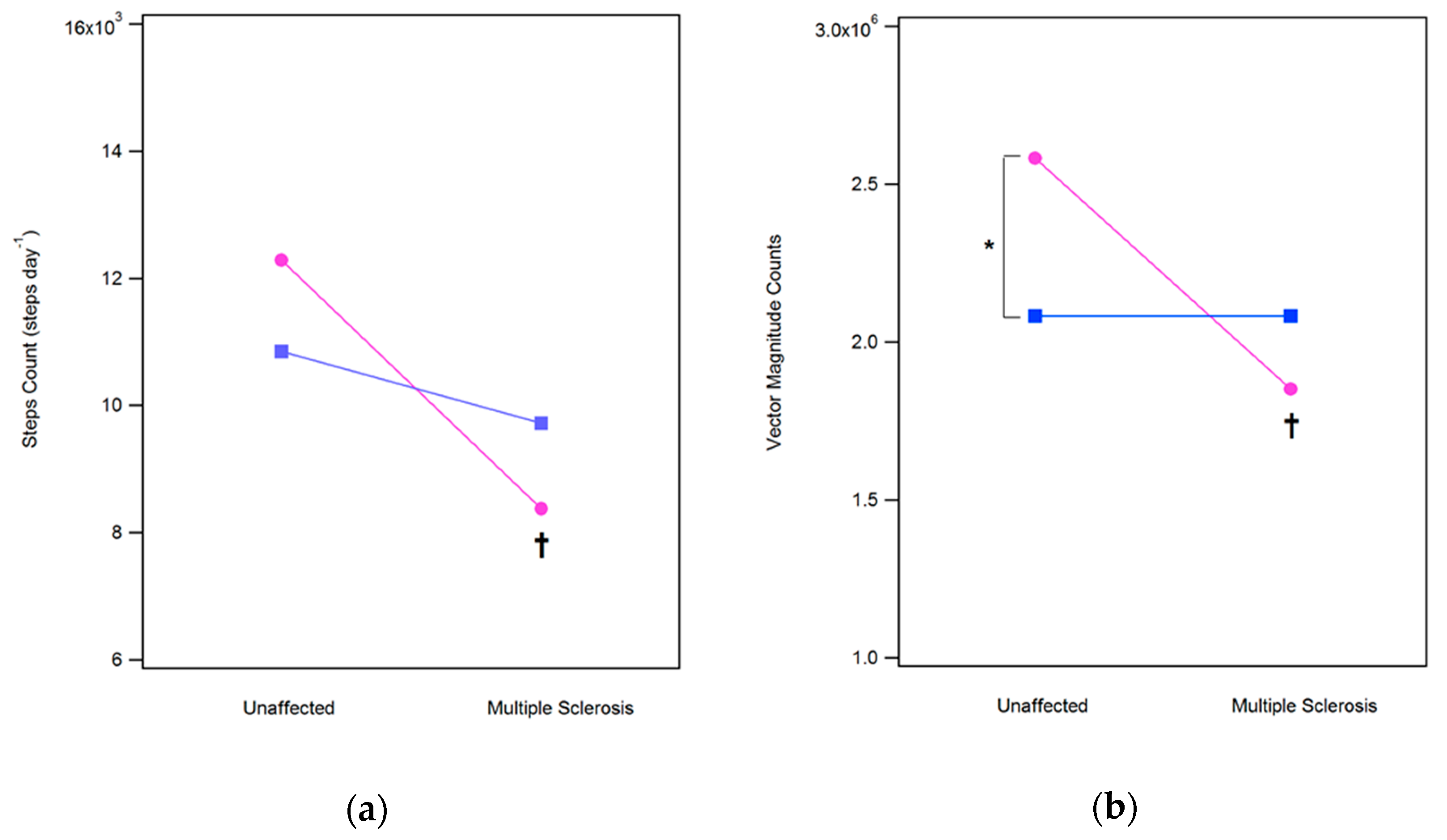
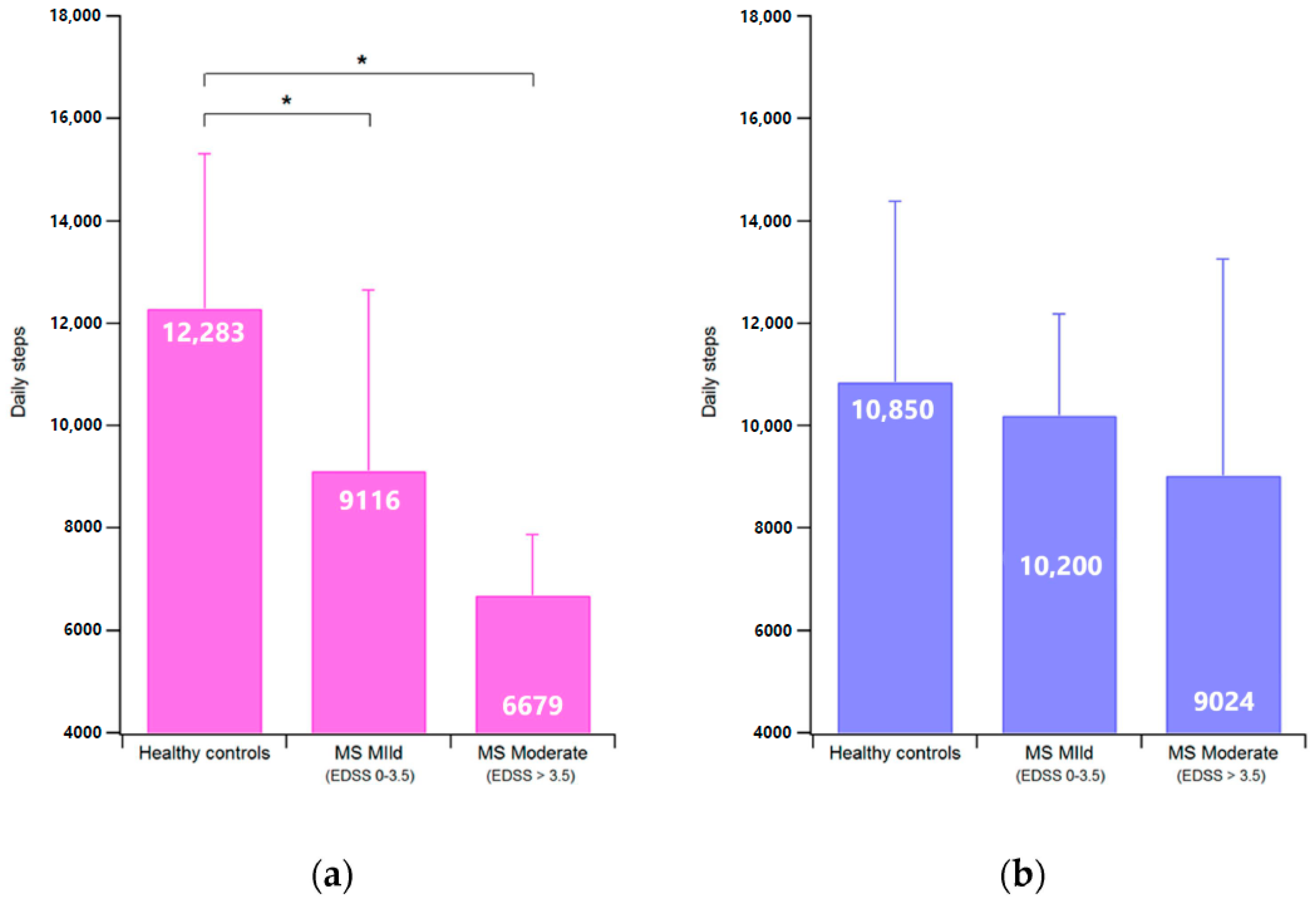
3.1. Steps Count (SC) and Vector Magnitude (VM) Counts
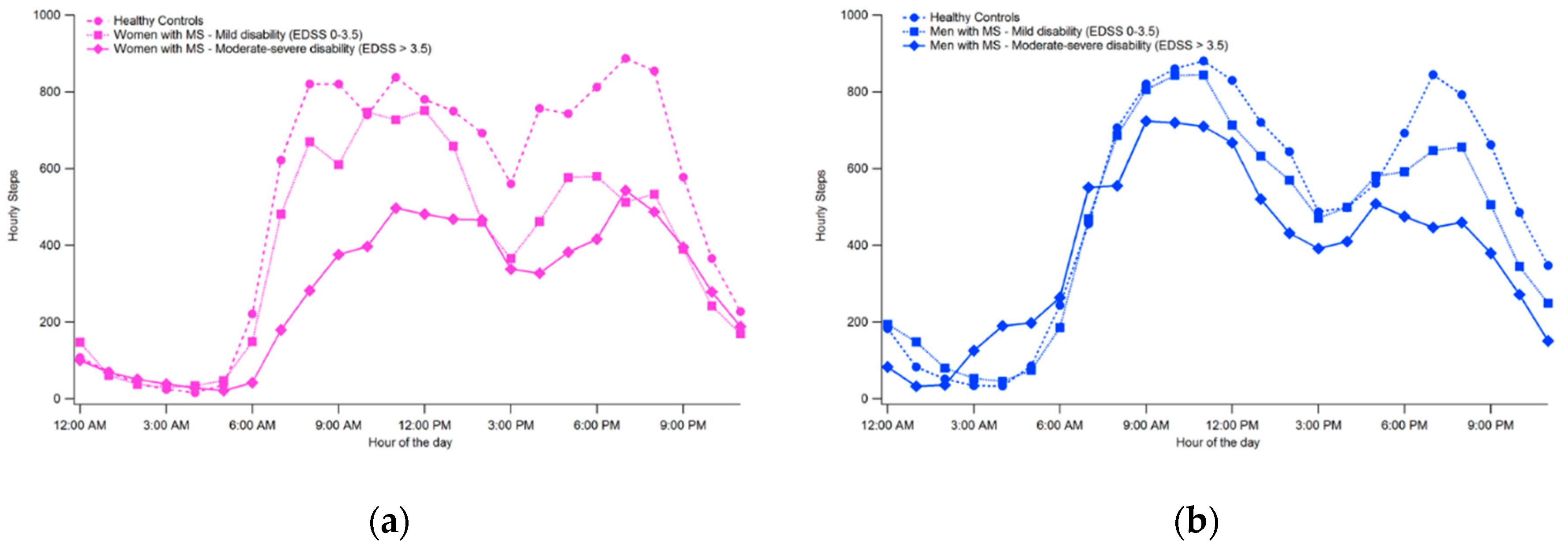
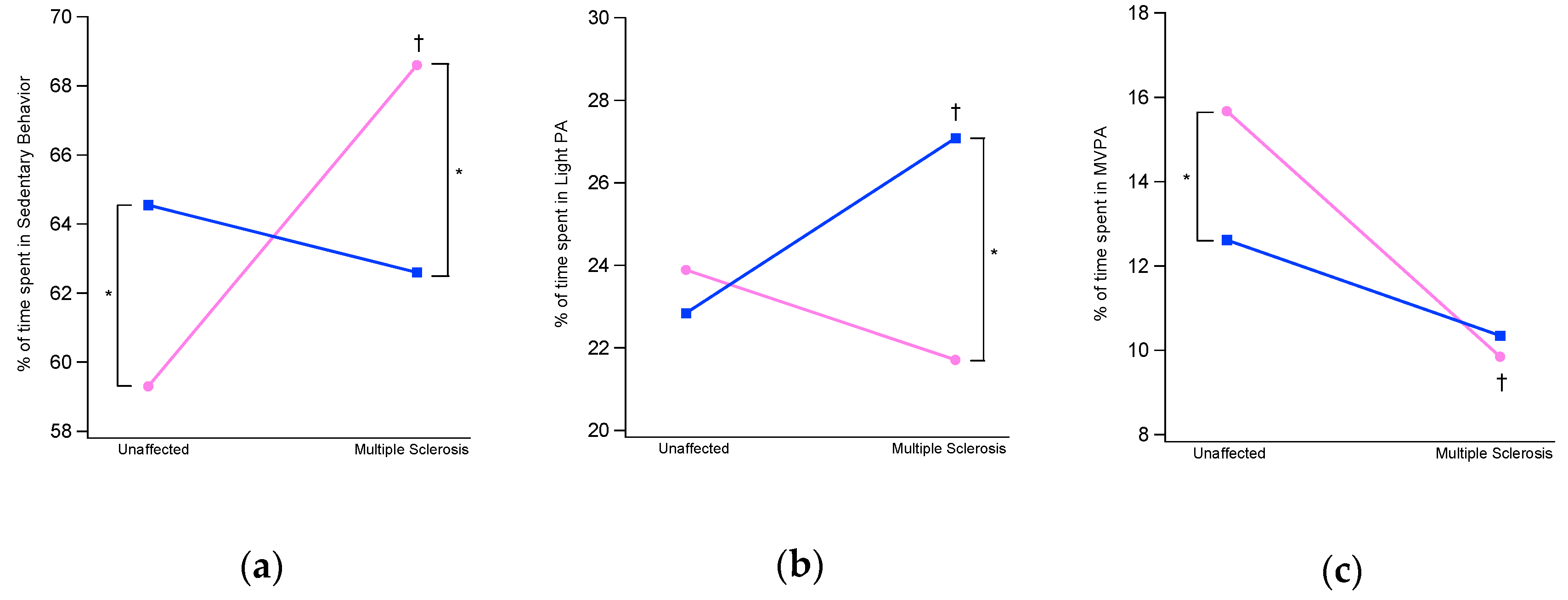
3.2. Physical Activity Intensity Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Compston, A.; Coles, A. Multiple sclerosis. Lancet 2008, 372, 1502–1517. [Google Scholar] [CrossRef]
- Waubant, E.; Lucas, R.; Mowry, E.; Graves, J.; Olsson, T.; Alfredsson, L.; Langer-Gould, A. Environmental and genetic risk factors for MS: An integrated review. Ann. Clin. Transl. Neurol. 2019, 6, 1905–1922. [Google Scholar] [CrossRef] [PubMed]
- Koch-Henriksen, N.; Sørensen, P.S. The changing demographic pattern of multiple sclerosis epidemiology. Lancet Neurol. 2010, 9, 520–532. [Google Scholar] [CrossRef]
- Thompson, A.J.; Baranzini, S.E.; Geurts, J.; Hemmer, B.; Ciccarelli, O. Multiple sclerosis. Lancet 2018, 391, 1622–1636. [Google Scholar] [CrossRef]
- Trojano, M.; Lucchese, G.; Graziano, G.; Taylor, B.V.; Simpson, S.; Lepore, V.; Grand’Maison, F.; Duquette, P.; Izquierdo, G.; Grammond, P.; et al. Geographical variations in sex ratio trends over time in multiple sclerosis. PLoS ONE 2012, 7, e48078. [Google Scholar] [CrossRef]
- Beatty, W.W.; Aupperle, R.L. Sex differences in cognitive impairment in multiple sclerosis. Clin. Neuropsychol. 2002, 16, 472–480. [Google Scholar] [CrossRef]
- Bergamaschi, R. Prognostic Factors in Multiple Sclerosis. Int Rev. Neurobiol. 2007, 79, 423–447. [Google Scholar]
- Ribbons, K.A.; McElduff, P.; Boz, C.; Trojano, M.; Izquierdo, G.; Duquette, P.; Girard, M.; Grand’Maison, F.; Hupperts, R.; Grammond, P.; et al. Male Sex Is Independently Associated with Faster Disability Accumulation in Relapse-Onset MS but Not in Primary Progressive MS. PLoS ONE. 2015, 10, e0122686. [Google Scholar] [CrossRef]
- Damasceno, A.; Von Glehn, F.; Brandão, C.O.; Damasceno, B.P.; Cendes, F. Prognostic indicators for long-term disability in multiple sclerosis patients. J. Neurol. Sci. 2013, 324, 29–33. [Google Scholar] [CrossRef]
- Bertoni, R.; Jonsdottir, J.; Feys, P.; Lamers, I.; Cattaneo, D. Modified Functional Walking Categories and participation in people with multiple sclerosis. Mult. Scler. Relat. Disord. 2018, 26, 11–18. [Google Scholar] [CrossRef]
- Théaudin, M.; Romero, K.; Feinstein, A. In multiple sclerosis anxiety, not depression, is related to gender. Mult. Scler. 2016, 22, 239–244. [Google Scholar] [CrossRef] [PubMed]
- Jobin, C.; Larochelle, C.; Parpal, H.; Coyle, P.K.; Duquette, P. Gender issues in multiple sclerosis: An update. Womens Health 2010, 6, 797–820. [Google Scholar] [CrossRef] [PubMed]
- Pau, M.; Corona, F.; Pilloni, G.; Porta, M.; Coghe, G.; Cocco, E. Do gait patterns differ in men and women with multiple sclerosis? Mult. Scler. Relat. Disord. 2017, 18, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Pau, M.; Casu, G.; Porta, M.; Pilloni, G.; Frau, J.; Coghe, G.; Cocco, E. Timed up and go in men and women with multiple sclerosis: Effect of muscular strength. J. Bodyw. Mov. Ther. 2020, 24, 124–130. [Google Scholar] [CrossRef]
- Kister, I.; Bacon, T.E.; Chamot, E.; Salter, A.R.; Cutter, G.R.; Kalina, J.T.; Herbert, J. Natural history of multiple sclerosis symptoms. Int. J. MS Care. 2013, 15, 146–158. [Google Scholar] [CrossRef]
- Motl, R.W.; Learmonth, Y.C.; Pilutti, L.A.; Appmaier, E.; Coote, S. Top 10 research questions related to physical activity and multiple sclerosis. Res. Q. Exerc. Sport. 2015, 86, 117–129. [Google Scholar] [CrossRef]
- Motl, R.W.; Goldman, M. Physical inactivity, neurological disability, and cardiorespiratory fitness in multiple sclerosis. Acta Neurol. Scand. 2011, 123, 98–104. [Google Scholar] [CrossRef]
- Sandroff, B.M.; Klaren, R.E.; Motl, R.W. Relationships among physical inactivity, deconditioning, and walking impairment in persons with multiple sclerosis. J. Neurol Phys. Ther. 2015, 39, 103–110. [Google Scholar] [CrossRef]
- Giesser, B.S. Exercise in the management of persons with multiple sclerosis. Ther. Adv. Neurol. Disord. 2015, 8, 123–130. [Google Scholar] [CrossRef]
- Anens, E.; Emtner, M.; Zetterberg, L.; Hellström, K. Physical activity in subjects with multiple sclerosis with focus on gender differences: A survey. BMC Neurol. 2014, 14, 47. [Google Scholar] [CrossRef]
- Klaren, R.E.; Motl, R.W.; Dlugonski, D.; Sandroff, B.M.; Pilutti, L.A. Objectively quantified physical activity in persons with multiple sclerosis. Arch. Phys. Med. Rehabil. 2013, 94, 2342–2348. [Google Scholar] [CrossRef] [PubMed]
- Dlugonski, D.; Pilutti, L.A.; Sandroff, B.M.; Suh, Y.; Balantrapu, S.; Motl, R.W. Steps per day among persons with multiple sclerosis: Variation by demographic, clinical, and device characteristics. Arch. Phys. Med. Rehabil. 2013, 94, 1534–1539. [Google Scholar] [CrossRef] [PubMed]
- Streber, R.; Peters, S.; Pfeifer, K. Systematic Review of Correlates and Determinants of Physical Activity in Persons with Multiple Sclerosis. Arch. Phys. Med. Rehabil. 2016, 97, 633–645. [Google Scholar] [CrossRef] [PubMed]
- Silfee, V.J.; Haughton, C.F.; Jake-Schoffman, D.E.; Lopez-Cepero, A.; May, C.N.; Sreedhara, M.; Rosal, M.C.; Lemon, S.C. Objective measurement of physical activity outcomes in lifestyle interventions among adults: A systematic review. Prev. Med. Rep. 2018, 11, 74–80. [Google Scholar] [CrossRef]
- Boon, R.M.; Hamlin, M.J.; Steel, G.D. Validation of the New Zealand Physical Activity Questionnaire (NZPAQ-LF) and the International Physical Activity Questionnaire (IPAQ-LF) with accelerometry. Br. J. Sports Med. 2010, 44, 741–746. [Google Scholar] [CrossRef]
- Casey, B.; Coote, S.; Galvin, R.; Donnelly, A. Objective physical activity levels in people with multiple sclerosis: Meta-analysis. Scand. J. Med. Sci. Sports 2018, 28, 1960–1969. [Google Scholar] [CrossRef]
- Sebastião, E.; Learmonth, Y.C.; Motl, R.W. Lower Physical Activity in Persons with Multiple Sclerosis at Increased Fall Risk: A Cross-sectional Study. Am. J. Phys. Med. Rehabil. 2017, 96, 357–361. [Google Scholar] [CrossRef]
- Sandroff, B.M.; Dlugonski, D.; Pilutti, L.A.; Pula, J.H.; Benedict, R.H.; Motl, R.W. Physical activity is associated with cognitive processing speed in persons with multiple sclerosis. Mult. Scler. Relat. Disord. 2014, 3, 123–128. [Google Scholar] [CrossRef]
- Motl, R.W.; Snook, E.M.; McAuley, E.; Gliottoni, R.C. Symptoms, self-efficacy, and physical activity among individuals with multiple sclerosis. Res. Nurs Health 2006, 29, 597–606. [Google Scholar] [CrossRef]
- Motl, R.W.; McAuley, E. Pathways between physical activity and quality of life in adults with multiple sclerosis. Health Psychol. 2009, 28, 682–689. [Google Scholar] [CrossRef]
- Motl, R.W.; McAuley, E.; Snookm, E.M.; Gliottoni, R.C. Physical activity and quality of life in multiple sclerosis: Intermediary roles of disability, fatigue, mood, pain, self-efficacy and social support. Psychol. Health Med. 2009, 14, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Polman, C.H.; Reingold, S.C.; Edan, G.; Filippi, M.; Hartung, H.; Kappos, L.; Lublin, F.D.; Metz, L.M.; McFarland, H.F.; O’Connor, P.W.; et al. Diagnostic criteria for multiple sclerosis: 2005 revisions to the McDonald criteria. Ann. Neurol. 2005, 58, 840–846. [Google Scholar] [CrossRef] [PubMed]
- Polman, C.H.; Reingold, S.C.; Banwell, B.; Clanet, M.; Cohen, J.A.; Filippi, M.; Fujihara, K.; Havrdova, E.; Hutchinson, M.; Kappos, L.; et al. Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann. Neurol. 2011, 69, 292–302. [Google Scholar] [CrossRef] [PubMed]
- Kurtze, J.F. Rating neurologic impairment in multiple sclerosis: An expanded disability status scale (EDSS). Neurology 1983, 33, 1444–1452. [Google Scholar] [CrossRef] [PubMed]
- Sandroff, B.M.; Motl, R.W.; Suh, Y. Accelerometer output and its association with energy expenditure in persons with multiple sclerosis. J. Rehabil Res. Dev. 2012, 49, 467–475. [Google Scholar] [CrossRef]
- Kos, D.; Nagels, G.; D’Hooghe, M.B.; Ilsbrouks, S.; Delbeke, S.; Kerckhofs, E. Measuring activity patterns using actigraphy in multiple sclerosis. Chronobiol. Int. 2007, 24, 345–356. [Google Scholar] [CrossRef]
- Buchan, D.S.; McSeveney, F.; McLellan, G. A comparison of physical activity from Actigraph GT3X+ accelerometers worn on the dominant and non-dominant wrist. Clin. Physiol. Funct. Imaging 2019, 39, 51–56. [Google Scholar] [CrossRef]
- Doherty, A.; Jackson, D.; Hammerla, N.; Plotz, T.; Olivier, P.; Granat, M.H.; van Hees, V.T.; Trenell, M.I.; Owen, C.G. Large-Scale Population Assessment of Physical Activity Using Wrist-Worn Accelerometers: The UK Biobank Study. PLoS ONE 2017, 12, e0169649. [Google Scholar] [CrossRef]
- Sandroff, B.M.; Riskin, B.J.; Agiovlasitis, S.; Motl, R.W. Accelerometer cut-points derived during over-ground walking in persons with mild, moderate, and severe multiple sclerosis. J. Neurol. Sci. 2014, 340, 50–57. [Google Scholar] [CrossRef]
- The Lancet Public Health. Time to tackle the physical activity gender gap. Lancet Pub. Health 2019, 4, e360. [Google Scholar] [CrossRef]
- Azevedo, M.R.; Araújo, C.L.; Reichert, F.F.; Wernek, G.L.; Griep, R.H.; Ponce de Leon, A.C.; Faerestin, E. Gender differences in leisure-time physical activity. Int. J. Pub. Health 2007, 52, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Meyer, K.; Niemann, S.; Abel, T. Gender differences in physical activity and fitness—Association with self-reported health and health-relevant attitudes in a middle-aged Swiss urban population. J. Pub. Health 2004, 12, 283–290. [Google Scholar] [CrossRef][Green Version]
- McMunn, A.; Bird, L.; Webb, E.; Saker, A. Gender Divisions of Paid and Unpaid Work in Contemporary UK Couples. Work Employ. Soc. 2020, 34, 155–173. [Google Scholar] [CrossRef]
- Wennman, H.; Pietilä, A.; Rissanen, H.; Valkeinen, H.; Partonen, T.; Maki-Opas, T.; Borodulin, B. Gender, age and socioeconomic variation in 24-hour physical activity by wrist-worn accelerometers: The Fin Health 2017 Survey. Sci. Rep. 2019, 9, 6534. [Google Scholar] [CrossRef]
- Shammas, L.; Zentek, T.; von Haaren, B.; Schlesinger, S.; Hey, S.; Rashid, A. Home-based system for physical activity monitoring in patients with multiple sclerosis (Pilot study). Biomed. Eng. Online 2014, 13, 10. [Google Scholar] [CrossRef][Green Version]
- Romberg, A.; Ruutianen, J.; Daumer, M. Physical Activity in Finnish Persons with Multiple Sclerosis. J. Nov. Physiother. 2013, 3, 3. [Google Scholar] [CrossRef]
- Norris, M.; Anderson, R.; Motl, R.W.; Heyes, S.; Coote, S. Minimum number of days required for a reliable estimate of daily step count and energy expenditure, in people with MS who walk unaided. Gait Posture 2017, 53, 201–206. [Google Scholar] [CrossRef]
- Motl, R.W.; McAuley, E.; Snook, E.M.; Scott, J.A. Validity of physical activity measures in ambulatory individuals with multiple sclerosis. Disabil. Rehabil. 2006, 28, 1151–1156. [Google Scholar] [CrossRef]
- Sandroff, B.M.; Dlugonski, D.; Weikert, M.; Suh, S.; Motl, R.W. Physical activity and multiple sclerosis: New insights regarding inactivity. Acta Neurol. Scand. 2012, 126, 256–262. [Google Scholar] [CrossRef]
- Fjeldstad, C.; Fjeldstad, A.S.; Pardo, G. Use of Accelerometers to Measure Real-Life Physical Activity in Ambulatory Individuals with Multiple Sclerosis: A Pilot Study. Int. J. MS Care 2015, 17, 215–220. [Google Scholar] [CrossRef]
- Engelhard, M.M.; Patek, S.D.; Lach, J.C.; Goldman, M.D. Real-world walking in multiple sclerosis: Separating capacity from behavior. Gait Posture 2018, 59, 211–216. [Google Scholar] [CrossRef] [PubMed]
- Bassett, D.R.; Wyatt, H.R.; Thompson, H.; Peters, J.C.; Hill, J.O. Pedometer-Measured Physical Activity and Health Behaviors in U.S. Adults. Med. Sci Sports Exerc. 2010, 42, 1819–1825. [Google Scholar] [CrossRef] [PubMed]
- Althoff, T.; Sosi, R.; Hicks, J.L.; King, A.C.; Delp, S.L.; Leskovec, J. Large-scale physical activity data reveal worldwide activity inequality. Nature 2017, 547, 336–339. [Google Scholar] [CrossRef] [PubMed]
- Kosecki, D. Fitbit’s Healthiest Cities, Countries, and Days for 2017. 2017. Available online: https://blog.fitbit.com/fitbit-year-in-review/ (accessed on 24 June 2020).
- Neal, W.N.; Cederberg, K.L.; Jeng, B.; Sasaki, J.E.; Motl, R.W. Is Symptomatic Fatigue Associated with Physical Activity and Sedentary Behaviors Among Persons with Multiple Sclerosis? Neurorehabil. Neural. Repair 2020, 34, 505–511. [Google Scholar] [CrossRef]
- Hildebrand, M.; Van Hees, V.T.; Hansen, B.H.; Ekelund, U. Age group comparability of raw accelerometer output from wrist- and hip-worn monitors. Med. Sci Sports Exerc. 2014, 46, 1816–1824. [Google Scholar] [CrossRef]
- Stuifbergen, A.K.; Roberts, G.J. Health promotion practices of women with multiple sclerosis. Arch. Phys. Med. Rehabil. 1997, 78, S3–S9. [Google Scholar] [CrossRef]
- Stuifbergen, A.K.; Becker, H.; Blozis, S.; Timmerman, G.; Kullberg, V. A randomized clinical trial of a wellness intervention for women with multiple sclerosis. Arch. Phys. Med. Rehabil. 2003, 84, 467–476. [Google Scholar] [CrossRef]
- Ploughman, M. Breaking down the barriers to physical activity among people with multiple sclerosis—A narrative review. Phys. Ther. Rev. 2017, 22, 124–132. [Google Scholar] [CrossRef]
- Kalron, A.; Menascu, S.; Frid, L.; Aloni, R.; Achiron, A. Physical activity in mild multiple sclerosis: Contribution of perceived fatigue, energy cost, and speed of walking. Disabil. Rehabil. 2020, 42, 1240–1246. [Google Scholar] [CrossRef]
- Mezini, S.; Soundy, A. A Thematic Synthesis Considering the Factors which Influence Multiple Sclerosis Related Fatigue during Physical Activity. Behav. Sci. 2019, 1, 70. [Google Scholar] [CrossRef]
- Salter, A.; Fox, R.J.; Tyry, T.; Cutter, G.; Marrie, R.A. The association of fatigue and social participation in multiple sclerosis as assessed using two different instruments. Mult. Scler. Relat. Disord. 2019, 31, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Dyck, I. Hidden geographies: The changing lifeworlds of women with multiple sclerosis. Soc. Sci. Med. 1995, 40, 307–320. [Google Scholar] [CrossRef]
- John, D.; Morton, A.; Arguello, D.; Lyden, K.; Bassett, D. “What is a step?” differences in how a step is detected among three popular activity monitors that have impacted physical activity research. Sensors 2018, 18, 1206. [Google Scholar] [CrossRef] [PubMed]
- Santos-Lozano, A.; Torres-Luque, G.; Garatachea, N. Inter-trial variability of GT3X accelerometer. Sci. Sports 2014, 29, e7. [Google Scholar] [CrossRef]
| Healthy Controls | Multiple Sclerosis | |||
|---|---|---|---|---|
| Women | Men | Women | Men | |
| Participants | 21 | 20 | 23 | 22 |
| Age (years) | 46.7 ± 14.6 | 49.6 ± 14.4 | 49.4 ± 9.0 | 51.2 ± 11.8 |
| Height (cm) | 163.2 ± 6.4 | 171.0 ± 6.8 | 159.2 ± 6.5 | 172.8 ± 6.5 |
| Body Mass (kg) | 61.5 ± 10.4 | 73.9 ± 11.7 | 61.5 ± 10.4 | 71.8 ± 9.8 |
| EDSS score | NA | 3.6 ± 1.8 | 3.6 ± 1.8 | |
| Type of MS | NA | 16 RR/4 PP/3 SP | 14 RR/4 PP/4 SP | |
| Disease duration (years) | NA | 17.6 ± 10.2 | 18.4 ± 13.4 | |
| Level of Physical Activity Intensity | Metabolic Equivalent (MET) | Accelerometric Counts Per Minute (cpm) | |
|---|---|---|---|
| Healthy Controls and People with MS with EDSS Score ≤ 3.5 | People with MS with EDSS Score > 3.5 | ||
| Sedentary Behavior (SB) | <1.5 | 0–319 | 0–87 |
| Light Physical Activity (LPA) | 1.5–3 | 320–1980 | 88–1185 |
| Moderate-to-Vigorous Physical Activity (MVPA) | >3 | >1980 | >1185 |
| Healthy Controls | Multiple Sclerosis | ||||
|---|---|---|---|---|---|
| Women | Men | Women | Men | ||
| SB | % | 59.3 ± 7.8 | 66.1 ± 8.0 * | 68.6 ± 8.2 † | 62.6 ± 9.8 * |
| min | 804.1 ± 132.0 | 899.9 ± 112.2 * | 859.5 ± 149.1 † | 810.1 ± 108.0 * | |
| LPA | % | 23.9 ± 3.9 | 23.0 ± 2.0 | 20.9 ± 6.5 | 27.1 ± 7.3 * † |
| min | 311.9 ± 49.2 | 313.3 ± 70.4 | 266.0 ± 77.7 | 347.0 ± 95.4 † | |
| MVPA | % | 16.8 ± 5.3 | 10.8 ± 4.6 * | 10.5 ± 5.5 † | 10.3 ± 5.5 |
| min | 209.1 ± 69.8 | 147.7 ± 64.0 * | 123.0 ± 74.2 † | 134.9 ± 73.6 | |
| SC (steps/day) | 12726 ± 2771 | 10850 ± 3532 | 8375 ± 3199 † | 9719 ± 3071 | |
| VMC (106 counts/day) | 2.68 ± 0.54 | 2.08 ± 0.63 * | 1.85 ± 0.55 † | 2.08 ± 0.66 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pau, M.; Porta, M.; Coghe, G.; Frau, J.; Lorefice, L.; Cocco, E. Does Multiple Sclerosis Differently Impact Physical Activity in Women and Man? A Quantitative Study Based on Wearable Accelerometers. Int. J. Environ. Res. Public Health 2020, 17, 8848. https://doi.org/10.3390/ijerph17238848
Pau M, Porta M, Coghe G, Frau J, Lorefice L, Cocco E. Does Multiple Sclerosis Differently Impact Physical Activity in Women and Man? A Quantitative Study Based on Wearable Accelerometers. International Journal of Environmental Research and Public Health. 2020; 17(23):8848. https://doi.org/10.3390/ijerph17238848
Chicago/Turabian StylePau, Massimiliano, Micaela Porta, Giancarlo Coghe, Jessica Frau, Lorena Lorefice, and Eleonora Cocco. 2020. "Does Multiple Sclerosis Differently Impact Physical Activity in Women and Man? A Quantitative Study Based on Wearable Accelerometers" International Journal of Environmental Research and Public Health 17, no. 23: 8848. https://doi.org/10.3390/ijerph17238848
APA StylePau, M., Porta, M., Coghe, G., Frau, J., Lorefice, L., & Cocco, E. (2020). Does Multiple Sclerosis Differently Impact Physical Activity in Women and Man? A Quantitative Study Based on Wearable Accelerometers. International Journal of Environmental Research and Public Health, 17(23), 8848. https://doi.org/10.3390/ijerph17238848






