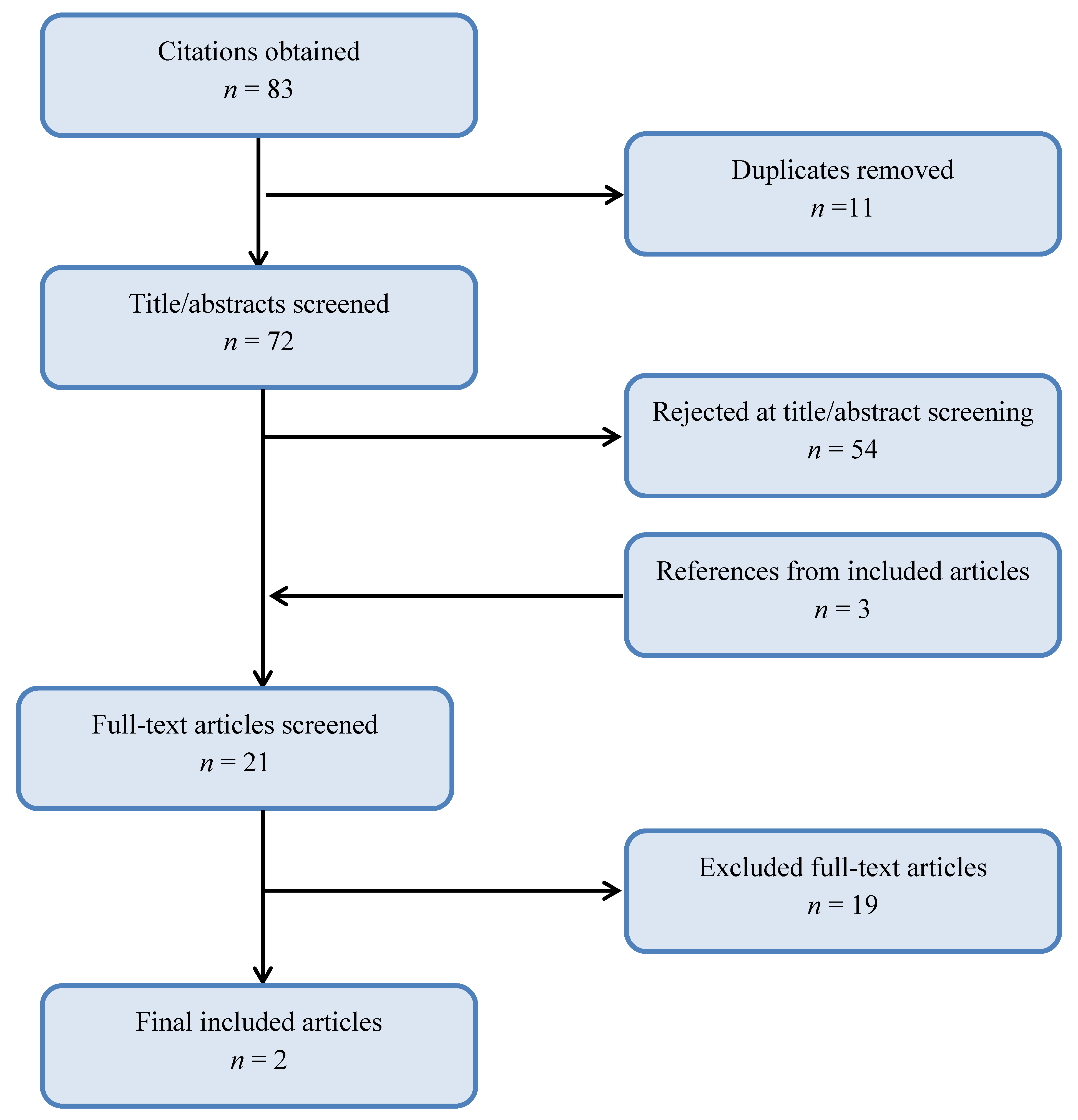
Abdominal Cutaneous Thermography and Perfusion Mapping after Caesarean Section: A Scoping Review
Abstract
1. Background
1.1. Context
1.2. Concept
1.3. Aims and Objectives
2. Methods
2.1. Search Strategy
2.2. Eligibility Criteria
- The participants were postpartum women,
- Infrared thermal imaging of the abdomen was undertaken,
- Abdominal blood flow mapping was reported, and
- The article was published in the English language.
2.3. Data-Charting Process
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| #Set | Query | Limiters/Expanders |
|---|---|---|
| S1 | maternal OR mother * OR postnatal OR postpartum OR after childbirth | Limiters—human; Expanders—apply equivalent subjects; Search modes—Boolean/phrase. |
| S2 | abdominal OR skin OR cutaneous OR subcutaneous | |
| S3 | blood flow OR perforator* OR skin perforation | |
| S4 | (S1 AND S2 AND S3) NOT Fet* NOT newborn NOT infant NOT placenta | |
| S5 | abdom* AND (thermal imaging OR thermograph*) NOT fet* NOT newborn NOT infant NOT placenta NOT mast* NOT breast | |
| S7 | S5 OR S6 |
Appendix B. Charting of Key Review Items
| 1. | Author |
| 2. | Year of publication |
| 3. | Title |
| 4. | Publication: Journal/Book |
| 5. | Source origin/country |
| 6. | Aims/Objectives/Purpose |
| 7. | Study population |
| 8. | Sample size |
| 9. | CONCEPT: Phenomenon of interest relating to the measurement of abdominal temperature and cutaneous blood perfusion |
| 10. | Additional references found from citation list |
| 11. | Methods used |
| 12. | Equipment and cameras |
| 13. | Calibration |
| 14. | Camera resolution |
| 15. | Duration of use |
| 16. | Any comparator? |
| 17. | CONTEXT: medical specialty, geography, clinical, experimental |
| 18. | Whether meets aim of review—caesarean section Yes/No |
| 19. | Outcomes and details of the results (e.g., how measured) |
| 20. | Explanation |
| 21. | Quantitative mapping YES NO |
| 22. | Qualitative mapping YES NO |
| 23. | Temperature values (°C) if any. How documented? |
| 24. | Key findings that relate to scoping review questions |
| 25. | Actions |
References
- Weiser, T.G.; Regenbogen, S.E.; Thompson, K.D.; Haynes, A.B.; Lipsitz, S.R.; Berry, W.R.; Gawande, A.A. An estimation of the global volume of surgery: A modelling strategy based on available data. Lancet 2008, 372, 139–144. [Google Scholar] [CrossRef]
- Visser, G.H.A.; Ayres-De-Campos, D.; Barnea, E.R.; De Bernis, L.; Di Renzo, G.C.; Vidarte, M.F.E.; Lloyd, I.; Nassar, A.H.; Nicholson, W.; Shah, P.K.; et al. FIGO position paper: How to stop the caesarean section epidemic. Lancet 2018, 392, 1286–1287. [Google Scholar] [CrossRef]
- Gibbons, L.; Belizán, J.; Lauer, J.; Betrán, A.; Merialdi, M.; Althabe, F. The global numbers and costs of additionally needed and unnecessary caesarean sections performed per year: Overuse as a barrier to universal coverage health systems financing. World Health Rep. 2010, 2010, 30. [Google Scholar]
- Soltani, H.; Sandall, J. Organisation of maternity care and choices of mode of birth: A worldwide view. Midwifery 2012, 28, 146–149. [Google Scholar] [CrossRef]
- EUROSTAT: Surgical Operations and Procedures Statistics. 2019. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php/Surgical_operations_and_procedures_statistics#Number_of_surgical_operations_and_procedures (accessed on 22 November 2020).
- Risk Factors for Wound Infection Following Cesarean Deliveries. Available online: https://obgyn.onlinelibrary.wiley.com/doi/abs/10.1016/j.ijgo.2005.03.020 (accessed on 22 November 2020).
- Anderson, V.; Chaboyer, W.; Gillespie, B. The relationship between obesity and surgical site infections in women undergoing caesarean sections: An integrative review. Midwifery 2013, 29, 1331–1338. [Google Scholar] [CrossRef]
- Brenes-Monge, A.; Saavedra-Avendaño, B.; Alcalde-Rabanal, J.; Darney, B.G. Are overweight and obesity associated with increased risk of cesarean delivery in Mexico? A cross-sectional study from the National Survey of Health and Nutrition. BMC Pregnancy Childbirth 2019, 19, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ward, M.C.; Agarwal, A.; Bish, M.; James, R.; Faulks, F.; Pitson, J.; Yuen, N.; Mnatzaganian, G. Trends in obesity and impact on obstetric outcomes in a regional hospital in Victoria, Australia. Aust. N. Z. J. Obstet. Gynaecol. 2020, 60, 204–211. [Google Scholar] [CrossRef]
- Machado, L.S. Cesarean section in morbidly obese parturients: Practical implications and complications. N. Am. J. Med. Sci. 2012, 4, 13–18. [Google Scholar] [CrossRef]
- Wloch, C.; Wilson, J.; Lamagni, T.; Harrington, P.; Charlett, A.; Sheridan, E. Risk factors for surgical site infection following caesarean section in England: Results from a multicentre cohort study. BJOG Int. J. Obstet. Gynaecol. 2012, 119, 1324–1333. [Google Scholar] [CrossRef]
- Tierney, N.M.; Lukacz, E.; Hebert, S.; Kelly, T.F.; Lacoursiere, D.Y. Incidence of wound complications after cesarean delivery. Obstet. Gynecol. 2015, 125, 5S. [Google Scholar] [CrossRef]
- Monahan, M.; Jowett, S.; Pinkney, T.; Brocklehurst, P.; Morton, D.G.; Abdali, Z.; Roberts, T.E. Surgical site infection and costs in low- and middle-income countries: A systematic review of the economic burden. PLoS ONE 2020, 15, e0232960. [Google Scholar] [CrossRef] [PubMed]
- Wloch, C.; Van Hoek, A.J.; Green, N.; Conneely, J.; Harrington, P.; Sheridan, E.; Wilson, J.; Lamagni, T. Cost-benefit analysis of surveillance for surgical site infection following caesarean section. BMJ Open 2020, 10, e036919. [Google Scholar] [CrossRef] [PubMed]
- Horan, T.C.; Gaynes, R.P.; Martone, W.J.; Jarvis, W.R.; Emori, T.G. CDC definitions of nosocomial surgical site infections, 1992: A modification of CDC definitions of surgical wound infections. Infect. Control. Hosp. Epidemiol. 1992, 13, 606–608. [Google Scholar] [CrossRef] [PubMed]
- Zejnullahu, V.; Isjanovska, R.; Sejfija, Z.; Zejnullahu, V. Surgical site infections after cesarean sections at the University Clinical Center of Kosovo: Rates, microbiological profile and risk factors. BMC Infect. Dis. 2019, 19, 752–759. [Google Scholar] [CrossRef]
- Sway, A.; Nthumba, P.; Solomkin, J.; Tarchini, G.; Gibbs, R.; Ren, Y.; Wanyoro, A. Burden of surgical site infection following cesarean section in sub-Saharan Africa: A narrative review. Int. J. Women’s Heal. 2019, 11, 309–318. [Google Scholar] [CrossRef]
- Saeed, K.B.M.; Greene, R.A.; Corcoran, P.; O’Neill, S.M. Incidence of surgical site infection following caesarean section: A systematic review and meta-analysis protocol. BMJ Open 2017, 7, e013037. [Google Scholar] [CrossRef]
- Alfouzan, W.; Al Fadhli, M.; Abdo, N.; Alali, W.; Dhar, R. Surgical site infection following cesarean section in a general hospital in Kuwait: Trends and risk factors. Epidemiol. Infect. 2019, 147, e287. [Google Scholar] [CrossRef]
- Jasim, H.; Sulaiman, S.; Khan, A.; Dawood, O.T.; Abdulameer, A.; Rajah, U. Incidence and risk factors of surgical site infection among patients undergoing cesarean section. Clin. Med. Insights Ther. 2017, 9. [Google Scholar] [CrossRef]
- Yeeles, H.; Trinick, S.; Childs, C.; Soltani, H.; Farrell, T. Postpartum infection in morbidly obese women after caesarean section: Does early prophylactic oral antibiotic use make a difference? Open J. Obstet. Gynecol. 2014, 4, 547–549. [Google Scholar] [CrossRef][Green Version]
- Soper, D.E.; Bump, R.C.; Hurt, W. Wound infection after abdominal hysterectomy: Effect of the depth of subcutaneous tissue. Am. J. Obstet. Gynecol. 1995, 173, 465–471. [Google Scholar] [CrossRef]
- Vermillion, S.T.; Lamoutte, C.; Soper, D.E.; Verdeja, A. Wound infection after cesarean: Effect of subcutaneous tissue thickness. Obstet. Gynecol. 2000, 95, 923–926. [Google Scholar] [CrossRef] [PubMed]
- Chelmow, D.; Rodriguez, E.J.; Sabatini, M.M. Suture closure of subcutaneous fat and wound disruption after cesarean delivery: A meta-analysis. Obstet. Gynecol. 2004, 103, 974–980. [Google Scholar] [CrossRef] [PubMed]
- Childs, C.; Wright, N.; Willmott, J.; Davies, M.; Kilner, K.; Ousey, K.; Soltani, H.; Madhuvrata, P.; Stephenson, J. The surgical wound in infrared: Thermographic profiles and early stage test-accuracy to predict surgical site infection in obese women during the first 30 days after caesarean section. Antimicrob. Resist. Infect. Control. 2019, 8, 7. [Google Scholar] [CrossRef]
- Barnes, R.B. Thermography of the human body. Science 1963, 140, 870. [Google Scholar] [CrossRef] [PubMed]
- De Weerd, L.; Miland, Å.O.; Mercer, J.B. Perfusion dynamics of free DIEP and SIEA flaps during the first postoperative week monitored with dynamic infrared thermography. Ann. Plast. Surg. 2009, 62, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Cracowski, J.-L.; Roustit, M. Human skin microcirculation. Compr. Physiol. 2020, 10, 1105–1154. [Google Scholar] [CrossRef]
- Bell, J.; Bell, S.; Vahratian, A.; Awonuga, A.O. Abdominal surgical incisions and perioperative morbidity among morbidly obese women undergoing cesarean delivery. Eur. J. Obstet. Gynecol. Reprod. Biol. 2011, 154, 16–19. [Google Scholar] [CrossRef]
- Ellis, H. Anatomy of the abdominal wall. Surgery 2008, 26, e9–e16. [Google Scholar]
- Taylor, G.I.; Pan, W.-R. The angiosome concept and tissue transfer. In Plastic Surgery; Part 1. Principles, Techniques and Basic Science. The Blood Supply of the Skin and Skin Flaps; QPM/CRC Press: St. Louis, MO, USA, 2014. [Google Scholar]
- Jonsson, K.; Jensen, J.A.; Goodson, W.H.; Scheuenstuhl, H.; West, J.; Hopf, H.W.; Hunt, T.K. Tissue oxygenation, anemia, and perfusion in relation to wound healing in surgical patients. Ann. Surg. 1991, 214, 605–613. [Google Scholar] [CrossRef]
- Levy, B.I.; Schiffrin, E.L.; Mourad, J.J.; Agostini, D.; Vicaut, E.; Safar, M.E.; Struijker-Boudier, H.A. Impaired tissue perfusion: A pathology common to hypertension, obesity, and diabetes mellitus. Circulation 2008, 118, 968–976. [Google Scholar] [CrossRef]
- Sen, C.K. Wound healing essentials: Let there be oxygen. Wound Repair Regen. 2009, 17, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Anhalt, K.; Machin, G. Thermodynamic temperature by primary radiometry. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2016, 374, 20150041. [Google Scholar] [CrossRef] [PubMed]
- Siah, C.J.; Childs, C. Thermographic mapping of the abdomen in healthy subjects and patients after enterostoma. J. Wound Care 2016, 24, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Betrán, A.P.; Ye, J.; Moller, A.-B.; Zhang, J.; Gülmezoglu, A.M.; Torloni, M.R. The increasing trend in caesarean section rates: Global, regional and national estimates: 1990–2014. PLoS ONE 2016, 11, e0148343. [Google Scholar] [CrossRef]
- Badia, J.M.; Casey, A.L.; Petrosillo, N.; Hudson, P.; Mitchell, S.; Crosby, C. Impact of surgical site infection on healthcare costs and patient outcomes: A systematic review in six European countries. J. Hosp. Infect. 2017, 96, 1–15. [Google Scholar] [CrossRef]
- Smollock, W.; Montenegro, P.; Czenis, A.; He, Y. Hypoperfusion and wound healing. Adv. Ski. Wound Care 2018, 31, 72–77. [Google Scholar] [CrossRef]
- Arksey, H.; O’Malley, L. Scoping studies: Towards a methodological framework. Intl. J. Soc. Res. Methodol. 2005, 8, 19–32. [Google Scholar] [CrossRef]
- Tricco, A.C.; Lillie, E.; Zarin, W.; O’Brien, K.K.; Colquhoun, H.; Levac, D.; Moher, D.; Peters, M.D.J.; Horsley, T.; Weeks, L.; et al. PRISMA extension for scoping reviews (PRISMA-ScR): Checklist and explanation. Ann. Intern. Med. 2018, 169, 467–473. [Google Scholar] [CrossRef]
- JBI. Appendix 11.1 JBI template source of evidence details, characteristics and results extraction instrument. In JBI Reviewer Handbook; Joanna Briggs Institute: Adelaide, Australia, 2020. [Google Scholar]
- Childs, C.; Siraj, M.; Fair, F.; Selvan, A.; Soltani, H.; Wilmott, J.; Farrell, T. Thermal territories of the abdomen after caesarean section birth: Infrared thermography and analysis. J. Wound Care 2016, 25, 499–512. [Google Scholar] [CrossRef]
- Falzon, O.; Ciantar, A.; Sammut, L.; Schembri, M.; Baron, Y.M.; Calleja-Agius, J.; Demicoli, P.; Camilleri, K.P. Principal component analysis of dynamic thermography data from pregnant and non-pregnant women. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 17–21 July 2018; Volume 2018, pp. 5664–5667. [Google Scholar]
- Savastano, D.M.; Gorbach, A.M.; Eden, H.S.; Brady, S.M.; Reynolds, J.C.; Yanovski, J.A. Adiposity and human regional body temperature. Am. J. Clin. Nutr. 2009, 90, 1124–1131. [Google Scholar] [CrossRef]
- Chandra, S.; Persad, V.; Young, D.; Baskett, T. A Preliminary study of cutaneous blood flow associated with postpartum use of oral misoprostol. J. Obstet. Gynaecol. Can. 2004, 26, 1073–1076. [Google Scholar] [CrossRef]
- Ciantar, A.; Falzon, O.; Sammut, L.; Schembri, M.; Baron, Y.M.; Calleja-Agius, J.; Demicoli, P.P.; Camilleri, K.P. Registration of dynamic thermography data of the abdomen of pregnant and non-pregnant women. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 17–21 July 2018; Volume 2018, pp. 5668–5671. [Google Scholar]
- Chudecka, M.; Lubkowska, A.; Kempińska-Podhorodecka, A. Body surface temperature distribution in relation to body composition in obese women. J. Therm. Biol. 2014, 43, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Thermal Imaging of Body Surface Temperature Distribution in Women with Anorexia Nervosa. Available online: https://www.semanticscholar.org/paper/Thermal-Imaging-of-Body-Surface-Temperature-in-with-Chudecka-Lubkowska/cee9f97b17c0246fe55416d2a719f1a345025ac4 (accessed on 22 November 2020).
- King, H.H.; Cayce, C.T.; Herrin, J. Thermography examination of abdominal area skin temperatures in individuals with and without focal-onset epilepsy. Explore 2017, 13, 46–52. [Google Scholar] [CrossRef] [PubMed]
- McFarlin, B.; Venable, A.; Williams, R.; Jackson, A. Comparison of techniques for the measurement of skin temperature during exercise in a hot, humid environment. Biol. Sport 2014, 32, 11–14. [Google Scholar] [CrossRef]
- Dutta, E.H.; Burns, R.N.; Pacheco, L.D.; Marrs, C.C.; Koutrouvelis, A.; Koutrouvelis, G.L.O. Lower extremity blood flow velocity in obese versus nonobese pregnant women. Am. J. Perinatol. 2019, 37, 384–389. [Google Scholar] [CrossRef]
- Goodlin, R.C.; Brooks, P.G. Abdominal wall hot spots in pregnant women. J. Reprod Med. 1987, 32, 177–180. [Google Scholar]
- Jo, J.; Kim, H. Comparison of abdominal skin temperature between fertile and infertile women by infrared thermography: A diagnostic approach. J. Therm. Biol. 2016, 61, 133–139. [Google Scholar] [CrossRef]
- Hu, H.F.; Hsiu, H.; Sung, C.J.; Lee, C.H. Combining laser-Doppler flowmetry measurements with spectral analysis to study different microcirculatory effects in human prediabetic and diabetic subjects. Lasers Med. Sci. 2017, 32, 327–334. [Google Scholar] [CrossRef]
- Bruins, A.A.; Kistemaker, K.R.J.; Boom, A.; Klaessens, J.H.G.M.; Verdaasdonk, R.M.; Boer, C. Thermographic skin temperature measurement compared with cold sensation in predicting the efficacy and distribution of epidural anesthesia. J. Clin. Monit. 2017, 32, 335–341. [Google Scholar] [CrossRef]
- Key, D.J. Preliminary demonstration using localized skin temperature elevation as observed with thermal imaging as an indicator of fat-specific absorption during focused-field radiofrequency therapy. J. Drugs Dermatol. JDD 2014, 13, 864–866. [Google Scholar]
- Willman, M.K. Pitfalls of abdominal thermography. J. Am. Osteopat. Assoc. 1973, 72, 913–920. [Google Scholar]
- Kliot, D.A.; Birnbaum, S.J. Thermographic studies of wound healing. Am. J. Obstet. Gynecol. 1965, 93, 515–521. [Google Scholar] [CrossRef]
- Gershon-Cohen, J.; Haberman-Brueschke, J.D.; Brueschke, E.E. Obstetric and gynecologic thermography. Obstet. Gynecol. 1965, 26, 842–847. [Google Scholar] [PubMed]
- Simoes, R.; Vardasca, R.; Nogueira-Silva, C. Thermal skin reference values in healthy late pregnancy. J. Therm. Biol. 2012, 37, 608–614. [Google Scholar] [CrossRef]
- Selvan, A.N.; Childs, C. Infrared thermal mapping, analysis and interpretation in biomedicine. In Application of Infrared to Biomedical Sciences; Ng, E., Etehadtavakol, M., Eds.; Springer: Singapore, 2017; pp. 377–394. [Google Scholar]
- Siah, C.-J.R.; Childs, C.; Chia, C.K.; Cheng, K.K.F. An observational study of temperature and thermal images of surgical wounds for detecting delayed wound healing within four days after surgery. J. Clin. Nurs. 2019, 28, 2285–2295. [Google Scholar] [CrossRef]
- Manchot, C. The Cutaneous Arteries of the Human Body; Springer Science and Business Media LLC: Berlin/Heidelberg, Germany, 1983. [Google Scholar]
- Itoh, Y.; Arai, K. Use of recovery-enhanced thermography to localize cutaneous perforators. Ann. Plast. Surg. 1995, 34, 507–511. [Google Scholar] [CrossRef]
- Kabon, M.B.; Nagele, R.A.; Reddy, M.D.; Eagon, M.C.; Fleshman, M.J.W.; Sessler, M.D.I.; Kurz, M.A. Obesity decreases perioperative tissue oxygenation. Anesthesiology 2004, 100, 274–280. [Google Scholar] [CrossRef]
- El-Mrakby, H.H.; Milner, R.H. The vascular anatomy of the lower anterior abdominal wall: A microdissection study on the deep inferior epigastric vessels and the perforator branches. Plast. Reconstr. Surg. 2002, 109, 539–543. [Google Scholar] [CrossRef]
- Charkoudian, N. Skin blood flow in adult human thermoregulation: How it works, when it does not, and why. In Mayo Clinic Proceedings; Elsevier BV: Amsterdam, The Netherlands, 2003; Volume 78, pp. 603–612. [Google Scholar]
- Hallock, G.G.; Brown, C.L.; DeSantis, M.J. Preoperative identification of perforating vessels to the skin by color duplex imaging. J. Ultrasound Med. 1995, 14, 263–266. [Google Scholar] [CrossRef]
- Ahn, C.; Narayanan, S. In vivo anatomic study of cutaneous perforarors in free flaps using magnetic resonance imaging. J. Reocstr. Microsurg. 1994, 10, 57. [Google Scholar]
- Pestana, I.A.; Zenn, M.R. Correlation between abdominal perforator vessels identified with preoperative CT angiography and intraoperative fluorescent angiography in the microsurgical breast reconstruction patient. Ann. Plast. Surg. 2014, 72, S144–S149. [Google Scholar] [CrossRef] [PubMed]
- De Weerd, L.; Mercer, J.B.; Weum, S. Dynamic infrared thermography. Clin. Plast. Surg. 2011, 38, 277–292. [Google Scholar] [CrossRef] [PubMed]
- Tenorio, X.; Mahajan, A.L.; Elias, B.; Van Riempst, J.S.; Wettstein, R.; Harder, Y.; Pittet, B. Locating perforator vessels by dynamic infrared imaging and flow doppler with no thermal cold challenge. Ann. Plast. Surg. 2011, 67, 143–146. [Google Scholar] [CrossRef] [PubMed]
- Hennessy, O.; McLoughlin, R.; McInerney, N.; Hussey, A.; Potter, S. Smartphone thermal imaging for preoperative perforator mapping in DIEP flap breast reconstruction. Eur. J. Plast. Surg. 2020, 43, 743–750. [Google Scholar] [CrossRef]
- Cifuentes, I.J.; Dagnino, B.L.; Salisbury, M.C.; Perez, M.E.; Ortega, C.; Maldonado, D. Augmented reality and dynamic infrared thermography for perforator mapping in the anterolateral thigh. Arch. Plast. Surg. 2018, 45, 284–288. [Google Scholar] [CrossRef]
- Badiul, P.O.; Sliesarenko, S.V. Multidetector-row computed tomographic angiography in the planning of the local perforator flaps. Plast. Reconstr. Surg. Glob. Open 2015, 3, e516. [Google Scholar] [CrossRef]
- Hennessy, O.; Potter, S.M. Use of infrared thermography for the assessment of free flap perforators in autologous breast reconstruction: A systematic review. JPRAS Open 2020, 23, 60–70. [Google Scholar] [CrossRef]
- Mercer, J.B.; De Weerd, L.; Weum, S. Thermography in Plastic Surgery; European Association of Thermography (EAT): Wien, Austria, 2012. [Google Scholar]
- John, H.E.; Niumsawatt, V.; Rozen, W.M.; Whitaker, I.S. Clinical applications of dynamic infrared thermography in plastic surgery: A systematic review. Gland Surg. 2016, 5, 122–132. [Google Scholar]
- Ring, E.F.J.; Ammer, K. Infrared thermal imaging in medicine. Physiol. Meas. 2012, 33, R33–R46. [Google Scholar] [CrossRef]
- Cañedo-Dorantes, L.; Cañedo-Ayala, M. Skin acute wound healing: A comprehensive review. Int. J. Inflamm. 2019, 2019, 3706315. [Google Scholar] [CrossRef]
| Author | Title | Design, Setting, Methods | Sample Size/Participants | Findings |
|---|---|---|---|---|
| Childs et al., 2016 [43] | Thermal territories of the abdomen after caesarean section birth: infrared thermography and analysis | Design: prospective feasibility and exploratory study Setting: postnatal wards, thermography on day 2 postoperative Methods: Body Temperature: tympanic membrane Ambient Conditions: air temperature, relative humidity, air velocity (postnatal ward). Thermal Imaging Equipment: LWIR using FLIR™ 450sc uncooled camera in static mode. Pixel resolution 320 × 240, NETD <30 mK, accuracy ±2 °C, spatial resolution 1.36 mRAD (milliradians) Calibration: Certification via blackbody source (FLIR™ Systems) Statistical analyses: SPSS V22 (IBM) Qualitative: Patient narratives from participants on perspectives of acceptability of thermal imaging at the bedside | Convenience sample 20 women by BMI group Women undergoing elective CS birth All BMI categories; normal weight: 18.5–24.9 kg/m2, overweight: 25.0–29.9 kg/m2, obese: >30 kg/m2 | Recruitment: women aged 20–39 (median 33) years; obese n = 8, overweight n-7, normal weight n = 5. All participants apyrexial at time of imaging. Temperature (°C) profiling: of abdomen (healthy reference site and wound). HCS pixel clustering of the incision and healthy adjacent region revealed differences in the thermal territory profiles (scar and adjacent region) Maximum temperature difference between healthy reference abdominal skin and wound site exceeds 2 °C in those who were suspected of developing SSI. For the majority (n = 16) small differences in wound site and reference site temperature were observed Outcome: this study shows that temperature differences between wound and reference skin territories could provide a thermal ‘signature’ of possible infection risk with ‘cold spots’ proposed as a putative marker of an ‘avascular’ region along the incision site Women reported positive feedback. LWIR is an acceptable technology as a future wound surveillance technology. The study meets some but not all inclusion criteria, e.g., postpartum women (with incisional wounds following caesarean section) with abdominal thermal mapping but does not include any measures of cutaneous blood flow in relation to abdominal temperature |
| Childs et al., 2019 [25] | The surgical wound in infrared: thermographic profiles and early stage test accuracy to predict surgical site infection during the first 30 days after caesarean section | Design: Prospective early stage test-accuracy study Setting: Postnatal wards and 30 day community follow-up Methods: Body Temperature: tympanic membrane Ambient conditions: air temperature, relative humidity, air velocity of postnatal ward and patient’s own room at home Wound Assessment: Centre for Disease Control (CDC) SSI criteria Antibiotic Prophylaxis: type/dose/duration documented (in patient) General Practitioner (GP) outpatient prescribing: type/dose/duration Thermal Imaging Equipment: LWIR using FLIR™ 450sc uncooled camera in static mode. Pixel resolution 320 × 240, NETD < 30 mK, accuracy ±2 °C, spatial resolution 1.36 mRAD (milliradians) Calibration: across temperature range 30–45 °C against blackbody source (P80P Amtek Land Instruments, Dronfield UK) Measurement Reliability: LWIR temperature measurements compared against independent certified (UKAS, UK) platinum resistance thermometer (type 100 Ω, platinum resistance thermometer (PRT) 100 Isotech, Skelmersdale UK) Imaging Protocol: LWIR thermography performed before hospital discharge (24–48 h postoperative) and on day 7, 15 and 30 after surgery Wound Visual Assessment: digital photographs taken alongside LWIR thermal image Follow Up: confirmed at 30 days postoperative via GP surgery | Obese women (BMI ≥ 30 kg/m2 at booking in) giving birth by elective or emergency CS Sample size: With an estimated SSI incidence (obese population) of 20%, test-accuracy for early stage identification of SSI to correctly identify women with (sensitivity) and without (specificity) infection, sample size: n = 50 women Abdominal skin regions of interest (ROIs) selected for each woman: ROI 1: Abdomen (umbilicus centrally) ROI 2: Wound site | Recruitment: 53 afebrile women aged 21–44 (median 32) years with BMI 30.1–43.9 median 34.2 kg/m2) recruited. Fifty women entered the study. First thermal image taken in hospital between day 1–3 (median 2) days postoperative. Full sets of thermal images taken on 4 occasions (one in hospital and three at home) were achived in 78% of sample Visual assessment: of the wound from digital photographs showed poor agreement with the eventual clinical wound outcome, indicating no improvement of wound assessment over chance. Agreement for the rater’s wound assessment for likely SSI indicated no consensus of opinion by clinicians At the 30 day postoperative follow-up time point, 16 women (32%) returned to the GP (6–24: median 10) days after surgery with suspected wound infection; one woman returning on two occasions. Fourteen women in this obese sample were given a clinical diagnosis of SSI (28%) Cutaneous temperature measurement: ROI 1 (abdomen) and ROI 2 (wound). Wound site (ROI 2) was consistently at a higher temperature than ROI 1 (abdomen). ROI 1 was at a significantly lower temperature in women who subsequently developed SSI. A unit (1 °C) reduction in abdominal temperature led to a 3-fold raised odds of infection. A 1 °C widening of temperature between ROI 2-ROI 1 (wound minus abdomen) was associated with an odds ratio for SSI of 2.25 (at day 2) and 2.5 (at day 7) Correct prediction for wound outcome using logistic regression models ranged from 70–79%. Thermal mapping: Although the wound was observed on the thermal image as ‘hotter’ than the abdomen, ‘cold spots’ were observed along the scar from day 2 in many women who later developed SSI. The origin of wound’ cold spots’ was proposed as attributed to a reduction in cutaneous blood flow. No direct measures of cutaneous blood flow were taken This study meets some but not all inclusion criteria. Infrared thermography was performed in obese women at high risk of postoperative wound infection (SSI). No confirmatory evidence was obtained to validate the link between low cutaneous temperature and tissue perfusion |
| Author | Title | Include | Reason |
|---|---|---|---|
| Falzon et al., 2018 [44] | Principal component analysis of dynamic thermography data from pregnant and non-pregnant women | NO | Although studies were undertaken in women (pregnant and non-pregnant) using infrared thermography of the abdomen as the region of interest (ROI) the study did not include measures of related cutaneous perfusion. |
| Savastano et al., 2009 [45] | Adiposity and human regional temperature | NO | Does not address all aspects of the inclusion criteria but does include infrared thermography mapping of abdomen but in the context of adiposity and body composition providing some background relevance to the research aim. |
| Chandra et al., 2004 [46] | A preliminary study of cutaneous blood flow associated with postpartum use of oral misoprostol | NO | Study aim was a randomized controlled trial to compare effects of oral misoprostol (a uterotonic drug with shivering side effects) with intravenous oxytocin in women at risk of postpartum haemorrhage. No abdominal thermography was performed but Doppler flux was used to assess changes in tissue perfusion (of triceps) postpartum temperature. |
| Ciantar et al., 2018 [47] | Registration of dynamic thermography data of the abdomen of pregnant and non-pregnant women | NO | This methodological paper, in an obstetric population, set out to find reliable methods to obtain accurate thermography images using dynamic mode whereas typically, this is undertaken in ‘static’ thermography mode. Changes in the ROI can be affected by movement over time, but dynamic mode eliminates sources of error in spatial alignment. Pregnant and non-pregnant (but not postpartum) women were included. |
| Chudecka et al., 2014 [48] | Body surface temperature distribution in relation to body composition in obese women | NO | This study used infrared thermography to compare profiles (total body and regional) between obese and normal weight women to identify body regions within which heat transfer is impeded. This work adds further supportive evidence that mean body surface temperature of the abdomen decreases as the percentage of body fat rises. No measures of cutaneous blood flow linked to abdominal infrared thermography. |
| Chudecka and Lubkowska 2016 [49] | Thermal imaging of body surface temperature distribution in women with anorexia nervosa | NO | The aim of this study was to assess the relationship between subcutaneous (and visceral) fat and skin temperature in 20 young women with Anorexia Nervosa (AN) compared to a reference group of healthy women. Infrared thermography was used to measure skin temperature in 12 body regions. Mean abdominal temperature in patients with AN was higher than the reference group. Higher skin (and abdominal) temperatures were attributed to significantly reduced subcutaneous fat content. No measurement or indication of cutaneous blood flow was undertaken in association with infrared thermography. |
| King et al., 2017 [50] | Thermography examination of abdominal area skin temperatures in individuals with and without focal epilepsy | NO | This study revisited an osteopathic theory that the abdomen of people with epilepsy manifest ‘cold spots’ due to inflammatory lesions originating with injuries to viscera and or the musculoskeletal system. Using infrared thermography to map abdominal regions, adults with focal-onset epilepsy had colder abdominal areas than controls. No measures reported of cutaneous blood flow in target abdominal regions. |
| McFarlin et al., 2015 [51] | Comparison of techniques for the measurement of skin temperature during exercise in a hot humid environment | NO | Thermal imaging was used as one of three methods for skin (abdomen and biceps) temperature measurement using wired skin electrodes and iButtons mounted on skin using adhesive tape, thermal imaging for temperature values. Limits of agreement between measures were performed. Differences between skin electrode, iButton and thermography were comparable for biceps but not abdomen. Underlying subcutaneous tissue was proposed as an explanation for large measurement bias with thermography skin temperature measurement. Not postpartum women. |
| Dutta et al., 2020 [52] | Lower extremity blood flow velocity in obese versus non-obese pregnant women | NO | The objective was to assess risk factors for venous thrombo-embolism using Duplex ultrasound in pregnant women during the third trimester, with and without obesity. No thermal imaging was undertaken and the method of blood flow estimation was lower limbs not abdomen. Blood flow in lower extremities was higher in obese compared with non-obese women. |
| Goodlin and Brooks 1987 [53] | Abdominal wall hot spots in pregnant women | NO | In a group of 140 women (non-pregnant and pregnant) an infrared temperature probe was used to ‘map’ seven regions of the abdominal wall. Hot spots were defined as temperatures ≥0.3 °C than the rest of abdomen. Left and right inguinal hot spots were most common (72%) followed by inguinal areas (58%) in normal pregnancy. Authors noted that whilst obese, non-pregnant, women had inguinal hot spots this was thought to be due to heat trapping by the abdominal pannus contributing to heat retention in this region. No postpartum or abdominal skin blood flow mapping was undertaken. |
| Jo and Kim 2016 [54] | Comparison of abdominal skin temperature between fertile and infertile women by infrared thermography: A diagnostic approach | NO | Abdominal skin thermographic profiles of fertile and non-fertile women (using an infrared camera) were compared, showing a higher temperature in fertile women. The authors comment that temperature difference between the groups could be due to blood perfusion differences, but no abdominal skin blood flow mapping was undertaken. The study population did not include postnatal women. |
| Hu et al., 2017 [55] | Combining laser Doppler flowmetry measurements with spectral analysis to study different microcirculatory effects in human pre diabetic and diabetic subjects | NO | Laser Doppler flowmetry (LDF) and spectral analysis was undertaken in adult males and females to establish microcirculatory regulatory mechanisms at the ankle skin in diabetic, pre-diabetic and ‘normal’ subjects. Results showed that endothelial, neurogenic and myogenic activities of blood vessels were all smaller in pre-diabetic or diabetic patients compared to ‘normal’. No measures of thermography nor cutaneous blood flow were performed at the abdomen and no postpartum women were studied. |
| Bruins et al. 2018 [56] | Thermographic skin temperature measurement compared with cold sensation in predicting efficacy and distribution of epidural anaesthesia | NO | Thermography was used as an alternative to ‘gold-standard’ cold sensation test of epidural anaesthesia. Thermographic imaging was performed in patients undergoing abdominal, thoracic and orthopoedic surgery. The decrease in skin temperature due to body heat distribution and vasodilation induced by epidural block was detected using thermography indicating that thermal imaging could provide an additional and objective assessment method to the cold-sensation test. No cutaneous blood flow measurements. |
| Siah and Childs 2016 [36] | Thermographic mapping of the abdomen in healthy subjects and patients after enterostoma | NO | Male and female adults were recruited in this study. Thermal patterns in nine abdominal regions and at the surgical wound (enterostoma) revealed some differences between healing and infected wounds. Healing wounds showed a trend in the thermal ‘map’; an increase in temperature on the first postoperative day, and ‘warming’ over the subsequent five days. ‘Cold spots’ emerged on the thermogram of the surgical wounds which subsequently became infected. Subjects with higher BMI had significantly lower mean abdominal temperatures in 4 of 9 regions suggesting that body composition influences skin temperature. Infected surgical wounds appear ‘colder’ than healing wounds. Postpartum women were not included in this study. No cutaneous blood flow mapping was undertaken by independent method such as Doppler. |
| Key 2014 [57] | Preliminary demonstration using localized skin temperature elevation as observed with thermal imaging as an indicator of fat-specific absorption during focused-field radiofrequency therapy | NO | This study investigated the safe duration of focused-field radiofrequency therapy skin heating to temperatures in excess of 40 °C. Five subjects only studied. Sample did not include pregnant or postpartum women. Peak heating of skin (hottest spots) were abdomen and flank where areas of subcutaneous fat were greatest and where visible fat was ‘pinchable’. |
| Willman 1973 [58] | Pitfalls of Abdominal Thermography | NO | A qualitative review of body heat patterns using a ‘thermograph’ system. Images were produced on polaroid film. Four inflammatory conditions in the abdominal region were investigated to determine similarities of the thermograph. Temperature change and underlying disease was observed, particularly of the upper right quadrant. Participants were male and female but none were postpartum and no measures of cutaneous blood flow were performed at sites of interest. |
| Kliot and Birnbaum 1964 [59] | Thermographic studies of wound healing | NO | Pre- and postoperative abdominal thermography was used in different populations of patients undergoing gynaecological, pelvic and abdominal surgery to follow phases of uncomplicated wound healing. The study provides insights of classical wound healing stages in relation to what is seen on thermography, i.e., scar looks ‘cold’ along the incision. The scar ‘cold’ spot gradually disappears, ‘blending in’ to surrounding abdominal skin. No direct measures of cutaneous blood flow. |
| Gershon-Cohen et al., 1965 [60] | Obstetric & Gynecological thermography | NO | Skin temperature thermal imaging was proposed as an improvement to conventional body (oral) temperature methods and as a means to portray the temperature of the entire body surface. Conditions examined include breast, placental localisation and pregnancy with the objective of identifying thermographic ‘hot spots’ commensurate with pathological lesions. No measures of the relationship between hot spots on thermography and concomitant changes in cutaneous blood flow were made. |
| Simoes et al., 2012 [61] | Thermal skin reference values in healthy late pregnancy | NO | Healthy pregnant women during the third trimester. Postpartum women were not included. Objective: assessment of distribution pattern of thermal symmetry using infrared thermography with regions of interest over the abdomen. By contrast to current literature in healthy adults, temperature of lower abdominal ROIs were lowest in healthy pregnant women of normal weight and highest in those in obese categories. No explanation is provided. No measures of cutaneous blood flow were undertaken. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Childs, C.; Soltani, H. Abdominal Cutaneous Thermography and Perfusion Mapping after Caesarean Section: A Scoping Review. Int. J. Environ. Res. Public Health 2020, 17, 8693. https://doi.org/10.3390/ijerph17228693
Childs C, Soltani H. Abdominal Cutaneous Thermography and Perfusion Mapping after Caesarean Section: A Scoping Review. International Journal of Environmental Research and Public Health. 2020; 17(22):8693. https://doi.org/10.3390/ijerph17228693
Chicago/Turabian StyleChilds, Charmaine, and Hora Soltani. 2020. "Abdominal Cutaneous Thermography and Perfusion Mapping after Caesarean Section: A Scoping Review" International Journal of Environmental Research and Public Health 17, no. 22: 8693. https://doi.org/10.3390/ijerph17228693
APA StyleChilds, C., & Soltani, H. (2020). Abdominal Cutaneous Thermography and Perfusion Mapping after Caesarean Section: A Scoping Review. International Journal of Environmental Research and Public Health, 17(22), 8693. https://doi.org/10.3390/ijerph17228693





