Prevalence of Physical Frailty and Its Multidimensional Risk Factors in Korean Community-Dwelling Older Adults: Findings from Korean Frailty and Aging Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Frailty Assessment
2.3. Measurements
2.4. Statistical Analysis
3. Results
3.1. Sociodemographic Characteristics of the Study Population
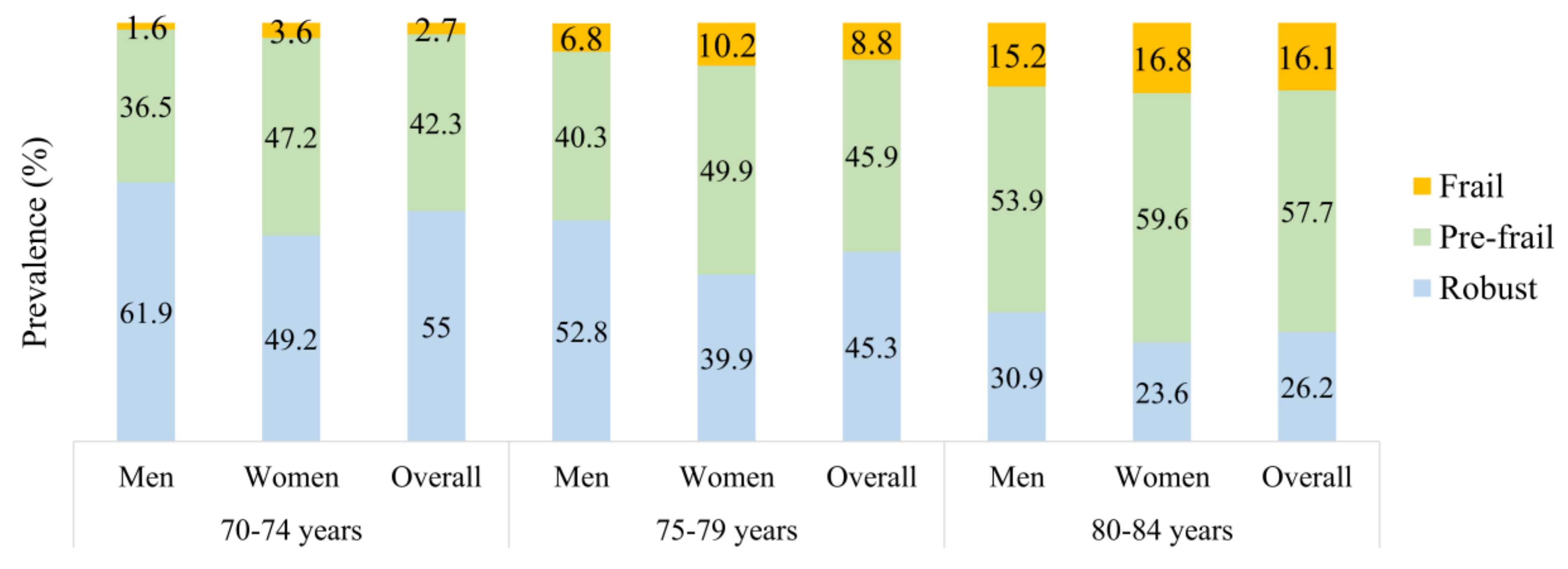
3.2. Prevalence of Frailty
3.3. Characteristics of the Study Population across Frailty Status
3.4. Risk Factors Associated with Physical Frailty
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M157. [Google Scholar] [CrossRef] [PubMed]
- Vermeiren, S.; Vella-Azzopardi, R.; Beckwee, D.; Habbig, A.-K.; Scafoglieri, A.; Jansen, B.; Bautmans, I.; Verté, D.; Beyer, I.; Petrovic, M. Frailty and the prediction of negative health outcomes: A meta-analysis. J. Am. Med. Dir. Assoc. 2016, 17, 1163.e1–1163.e17. [Google Scholar] [CrossRef] [PubMed]
- Collard, R.M.; Boter, H.; Schoevers, R.A.; Oude Voshaar, R.C. Prevalence of frailty in community-dwelling older persons: A systematic review. J. Am. Geriatr. Soc. 2012, 60, 1487–1492. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Gambassi, G.; Abellan van Kan, G.; Vellas, B. The frailty phenotype and the frailty index: Different instruments for different purposes. Age Ageing 2014, 43, 10–12. [Google Scholar] [CrossRef] [PubMed]
- Yaksic, E.; Lecky, V.; Sharnprapai, S.; Tungkhar, T.; Cho, K.; Driver, J.A.; Orkaby, A.R. Defining frailty in research abstracts: A systematic review and recommendations for standardization. J. Frailty Aging 2019, 8, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Prince, M.; Thiyagarajan, J.A.; De Carvalho, I.A.; Bernabei, R.; Chan, P.; Gutierrez-Robledo, L.M.; Michel, J.-P.; Morley, J.E.; Ong, P. Frailty: An emerging public health priority. J. Am. Med. Dir. Assoc. 2016, 17, 188–192. [Google Scholar] [CrossRef]
- Statistics Korea. Population Projections for Korea (2017–2067). Available online: http://kostat.go.kr (accessed on 28 March 2019).
- Jung, H.-W.; Kim, S.-W.; Ahn, S.; Lim, J.-Y.; Han, J.-W.; Kim, T.-H.; Kim, K.-W.; Kim, K.-I.; Kim, C.-H. Prevalence and outcomes of frailty in Korean elderly population: Comparisons of a multidimensional frailty index with two phenotype models. PLoS ONE 2014, 9, e87958. [Google Scholar] [CrossRef]
- Jung, H.-W.; Jang, I.-Y.; Lee, Y.S.; Lee, C.K.; Cho, E.-I.; Kang, W.Y.; Chae, J.H.; Lee, E.J.; Kim, D.H. Prevalence of frailty and aging-related health conditions in older Koreans in rural communities: A cross-sectional analysis of the aging study of Pyeongchang rural area. J. Korean Med. Sci. 2016, 31, 345–352. [Google Scholar] [CrossRef]
- Poli, S.; Cella, A.; Puntoni, M.; Musacchio, C.; Pomata, M.; Torriglia, D.; Vello, N.; Molinari, B.; Pandolfini, V.; Torrigiani, C. Frailty is associated with socioeconomic and lifestyle factors in community-dwelling older subjects. Aging Clin. Exp. Res. 2017, 29, 721–728. [Google Scholar] [CrossRef]
- Vaingankar, J.A.; Chong, S.A.; Abdin, E.; Picco, L.; Chua, B.Y.; Shafie, S.; Ong, H.L.; Chang, S.; Seow, E.; Heng, D. Prevalence of frailty and its association with sociodemographic and clinical characteristics, and resource utilization in a population of Singaporean older adults. Geriatr. Gerontol. Int. 2017, 17, 1444–1454. [Google Scholar] [CrossRef]
- Wong, C.H.; Weiss, D.; Sourial, N.; Karunananthan, S.; Quail, J.M.; Wolfson, C.; Bergman, H. Frailty and its association with disability and comorbidity in a community-dwelling sample of seniors in Montreal: A cross-sectional study. Aging Clin. Exp. Res. 2010, 22, 54–62. [Google Scholar] [CrossRef]
- Feng, Z.; Lugtenberg, M.; Franse, C.; Fang, X.; Hu, S.; Jin, C.; Raat, H. Risk factors and protective factors associated with incident or increase of frailty among community-dwelling older adults: A systematic review of longitudinal studies. PLoS ONE 2017, 12, e0178383. [Google Scholar] [CrossRef] [PubMed]
- Won, C.W.; Lee, S.; Kim, J.; Chon, D.; Kim, S.; Kim, C.-O.; Kim, M.K.; Cho, B.; Choi, K.M.; Roh, E. Korean frailty and aging cohort study (KFACS): Cohort profile. BMJ Open 2020, 10, e035573. [Google Scholar] [CrossRef] [PubMed]
- Charlson, M.E.; Pompei, P.; Ales, K.L.; MacKenzie, C.R. A new method of classifying prognostic comorbidity in longitudinal studies: Development and validation. J. Chronic. Dis. 1987, 40, 373–383. [Google Scholar] [CrossRef]
- Chen, L.-K.; Liu, L.-K.; Woo, J.; Assantachai, P.; Auyeung, T.-W.; Bahyah, K.S.; Chou, M.-Y.; Chen, L.-Y.; Hsu, P.-S.; Krairit, O. Sarcopenia in Asia: Consensus report of the Asian Working Group for Sarcopenia. J. Am. Med. Dir. Assoc. 2014, 15, 95–101. [Google Scholar] [CrossRef]
- Kim, S.; Kim, M.; Lee, Y.; Kim, B.; Yoon, T.Y.; Won, C.W. Calf circumference as a simple screening marker for diagnosing sarcopenia in older Korean adults: The Korean frailty and aging cohort study (KFACS). J. Korean Med. Sci. 2018, 33. [Google Scholar] [CrossRef]
- Lean, M.; Han, T.; Morrison, C. Waist circumference as a measure for indicating need for weight management. BMJ 1995, 311, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Cesari, M.; Demougeot, L.; Boccalon, H.; Guyonnet, S.; Van Kan, G.A.; Vellas, B.; Andrieu, S. A self-reported screening tool for detecting community-dwelling older persons with frailty syndrome in the absence of mobility disability: The FiND questionnaire. PLoS ONE 2014, 9, e101745. [Google Scholar] [CrossRef]
- Won, C.W.; Yang, K.Y.; Rho, Y.G.; Kim, S.Y.; Lee, E.J.; Yoon, J.L.; Cho, K.H.; Shin, H.C.; Cho, B.R.; Oh, J.R. The development of Korean activities of daily living (K-ADL) and Korean instrumental activities of daily living (K-IADL) scale. J. Korean Geriatr. Soc. 2002, 6. [Google Scholar] [CrossRef]
- Podsiadlo, D.; Richardson, S. The timed “Up & Go”: A test of basic functional mobility for frail elderly persons. J. Am. Geriatr. Soc. 1991, 39, 142–148. [Google Scholar] [CrossRef]
- Guralnik, J.M.; Simonsick, E.M.; Ferrucci, L.; Glynn, R.J.; Berkman, L.F.; Blazer, D.G.; Scherr, P.A.; Wallace, R.B. A short physical performance battery assessing lower extremity function: Association with self-reported disability and prediction of mortality and nursing home admission. J. Gerontol. 1994, 49, M85–M94. [Google Scholar] [CrossRef] [PubMed]
- Kaiser, M.J.; Bauer, J.M.; Ramsch, C.; Uter, W.; Guigoz, Y.; Cederholm, T.; Thomas, D.R.; Anthony, P.; Charlton, K.E.; Maggio, M. Validation of the Mini Nutritional Assessment Short-Form (MNA®-SF): A practical tool for identification of nutritional status. J. Nutr. Health Aging 2009, 13, 782. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, Y.; Won, C.; Lee, K.; Chon, D. Nutritional status and frailty in community-dwelling older Korean adults: The Korean Frailty and Aging Cohort Study. J. Nutr. 2018, 22, 774–778. [Google Scholar] [CrossRef] [PubMed]
- Gnjidic, D.; Hilmer, S.N.; Blyth, F.M.; Naganathan, V.; Waite, L.; Seibel, M.J.; McLachlan, A.J.; Cumming, R.G.; Handelsman, D.J.; Le Couteur, D.G. Polypharmacy cutoff and outcomes: Five or more medicines were used to identify community-dwelling older men at risk of different adverse outcomes. J. Clin. Epidemiol. 2012, 65, 989–995. [Google Scholar] [CrossRef]
- Yoo, M.; Kim, S.; Kim, B.; Yoo, J.; Lee, S.; Jang, H.; Cho, B.; Son, S.; Lee, J.; Park, Y. Moderate hearing loss is related with social frailty in a community-dwelling older adults: The Korean frailty and aging cohort study (KFACS). Arch. Gerontol. Geriatr. 2019, 83, 126–130. [Google Scholar] [CrossRef]
- Maberley, D.; Hollands, H.; Chuo, J.; Tam, G.; Konkal, J.; Roesch, M.; Veselinovic, A.; Witzigmann, M.; Bassett, K. The prevalence of low vision and blindness in Canada. Eye 2006, 20, 341–346. [Google Scholar] [CrossRef]
- Bae, J.N.; Cho, M.J. Development of the Korean version of the Geriatric Depression Scale and its short form among elderly psychiatric patients. J. Psychoson. Res. 2004, 57, 297–305. [Google Scholar] [CrossRef]
- Lee, D.Y.; Lee, K.U.; Lee, J.H.; Kim, K.W.; Jhoo, J.H.; Kim, S.Y.; Yoon, J.C.; Woo, S.I.; Ha, J.; Woo, J.I. A normative study of the CERAD neuropsychological assessment battery in the Korean elderly. J. Int. Neuropsychol. Soc. 2004, 10, 72–81. [Google Scholar] [CrossRef]
- Won, C.W.; Lee, Y.; Kim, S.; Yoo, J.; Kim, M.; Ng, T.-P.; Kim, H.; Son, S.J. Modified criteria for diagnosing “cognitive frailty”. Psychiatry Investig. 2018, 15, 839. [Google Scholar] [CrossRef]
- Jo, M.-W.; Yun, S.-C.; Lee, S.-I. Estimating quality weights for EQ-5D health states with the time trade-off method in South Korea. Value Health 2008, 11, 1186–1189. [Google Scholar] [CrossRef]
- Kim, S.; Won, C.W.; Kim, B.; Yoo, J.; Byun, S.; Jang, H.C.; Cho, B.; Son, S.; Lee, J.; Park, Y. EuroQol Visual Analogue Scale (EQ-VAS) as a Predicting Tool for Frailty in Older Korean Adults: The Korean Frailty and Aging Cohort Study (KFACS). J. Nutr. 2018, 22, 1275–1280. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Jo, M.W.; Ahn, J.; Ock, M.; Shin, S.; Park, J. Assessment of psychometric properties of the Korean SF-12 v2 in the general population. BMC Public Health 2014, 14, 1086. [Google Scholar] [CrossRef]
- Brazier, J.E.; Roberts, J. The estimation of a preference-based measure of health from the SF-12. Med. Care 2004, 851–859. [Google Scholar] [CrossRef]
- Mitchell, P.H.; Powell, L.; Blumenthal, J.; Norten, J.; Ironson, G.; Pitula, C.R.; Froelicher, E.S.; Czajkowski, S.; Youngblood, M.; Huber, M. A short social support measure for patients recovering from myocardial infarction: The ENRICHD Social Support Inventory. J. Cardiopulm. Rehabil. Prev. 2003, 23, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Shin, N.; Kang, Y. The relationships among health locus of control and resilience, social support and health promoting behavior in patients with newly diagnosed coronary artery diseases. Korean J. Adult Nurs. 2015, 27, 294–303. [Google Scholar] [CrossRef]
- Wenger, G.C.; Tucker, I. Using network variation in practice: Identification of support network type. Health Soc. Care Community 2002, 10, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Siriwardhana, D.D.; Weerasinghe, M.C.; Rait, G.; Falcaro, M.; Scholes, S.; Walters, K.R. Prevalence of frailty in rural community-dwelling older adults in Kegalle district of Sri Lanka: A population-based cross-sectional study. BMJ Open 2019, 9. [Google Scholar] [CrossRef]
- Wu, C.; Smit, E.; Xue, Q.-L.; Odden, M.C. Prevalence and correlates of frailty among community-dwelling Chinese older adults: The China Health and Retirement Longitudinal Study. J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 102–108. [Google Scholar] [CrossRef]
- Zheng, Z.; Guan, S.; Ding, H.; Wang, Z.; Zhang, J.; Zhao, J.; Ma, J.; Chan, P. Prevalence and incidence of frailty in community-dwelling older people: Beijing Longitudinal Study of Aging II. J. Am. Geriatr. Soc. 2016, 64, 1281–1286. [Google Scholar] [CrossRef]
- Lee, Y.; Kim, J.; Han, E.S.; Ryu, M.; Cho, Y.; Chae, S. Frailty and body mass index as predictors of 3-year mortality in older adults living in the community. Gerontology 2014, 60, 475–482. [Google Scholar] [CrossRef]
- Dent, E.; Lien, C.; Lim, W.S.; Wong, W.C.; Wong, C.H.; Ng, T.P.; Woo, J.; Dong, B.; de la Vega, S.; Poi, P.J.H. The Asia-Pacific clinical practice guidelines for the management of frailty. J. Am. Med. Dir. Assoc. 2017, 18, 564–575. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Kiesswetter, E.; Drey, M.; Sieber, C.C. Nutrition, frailty, and sarcopenia. Aging Clin. Exp. Res. 2017, 29, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Samper-Ternent, R.; Reyes-Ortiz, C.; Ottenbacher, K.J.; Cano, C.A. Frailty and sarcopenia in Bogotá: Results from the SABE Bogotá Study. Aging Clin. Exp. Res. 2017, 29, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Clegg, A.; Rogers, L.; Young, J. Diagnostic test accuracy of simple instruments for identifying frailty in community-dwelling older people: A systematic review. Age Ageing 2014, 44, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Da Câmara, S.M.A.; Alvarado, B.E.; Guralnik, J.M.; Guerra, R.O.; Maciel, Á.C.C. Using the Short Physical Performance Battery to screen for frailty in young-old adults with distinct socioeconomic conditions. Geriatr. Gerontol. Int. 2013, 13, 421–428. [Google Scholar] [CrossRef] [PubMed]
- Herr, M.; Robine, J.M.; Pinot, J.; Arvieu, J.J.; Ankri, J. Polypharmacy and frailty: Prevalence, relationship, and impact on mortality in a French sample of 2350 old people. Pharmacoepidemiol. Drug Saf. 2015, 24, 637–646. [Google Scholar] [CrossRef]
- Pérez-Ros, P.; Vila-Candel, R.; López-Hernández, L.; Martínez-Arnau, F.M. Nutritional Status and Risk Factors for Frailty in Community-Dwelling Older People: A Cross-Sectional Study. Nutrients 2020, 12, 1041. [Google Scholar] [CrossRef]
- Walston, J.; Hadley, E.C.; Ferrucci, L.; Guralnik, J.M.; Newman, A.B.; Studenski, S.A.; Ershler, W.B.; Harris, T.; Fried, L.P. Research agenda for frailty in older adults: Toward a better understanding of physiology and etiology: Summary from the American Geriatrics Society/National Institute on Aging Research Conference on Frailty in Older Adults. J. Am. Geriatr. Soc. 2006, 54, 991–1001. [Google Scholar] [CrossRef]
- Dent, E.; Kowal, P.; Hoogendijk, E.O. Frailty measurement in research and clinical practice: A review. Eur. J. Intern. Med. 2016, 31, 3–10. [Google Scholar] [CrossRef]
- Turner, G.; Clegg, A. Best practice guidelines for the management of frailty: A British Geriatrics Society, Age UK and Royal College of General Practitioners report. Age Ageing 2014, 43, 744–747. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, R.E.; O’Mahony, M.S.; Savva, G.M.; Calver, B.L.; Woodhouse, K.W. Inflammation and frailty measures in older people. J. Cell Mol. Med. 2009, 13, 3103–3109. [Google Scholar] [CrossRef]
- Leng, S.; Chaves, P.; Koenig, K.; Walston, J. Serum interleukin-6 and hemoglobin as physiological correlates in the geriatric syndrome of frailty: A pilot study. J. Am. Geriatr. Soc. 2002, 50, 1268–1271. [Google Scholar] [CrossRef] [PubMed]
- Soysal, P.; Stubbs, B.; Lucato, P.; Luchini, C.; Solmi, M.; Peluso, R.; Sergi, G.; Isik, A.T.; Manzato, E.; Maggi, S. Inflammation and frailty in the elderly: A systematic review and meta-analysis. Ageing Res. Rev. 2016, 31, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, R.; Andrew, M.; Fallah, N.; Rockwood, K. Comparison of the prognostic importance of diagnosed diabetes, co-morbidity and frailty in older people. Diabet. Med. 2010, 27, 603–606. [Google Scholar] [CrossRef] [PubMed]
- Ottenbacher, K.J.; Graham, J.E.; Al Snih, S.; Raji, M.; Samper-Ternent, R.; Ostir, G.V.; Markides, K.S. Mexican Americans and frailty: Findings from the Hispanic established populations epidemiologic studies of the elderly. Am. J. Public Health Nations Health 2009, 99, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Soysal, P.; Veronese, N.; Thompson, T.; Kahl, K.G.; Fernandes, B.S.; Prina, A.M.; Solmi, M.; Schofield, P.; Koyanagi, A.; Tseng, P.-T. Relationship between depression and frailty in older adults: A systematic review and meta-analysis. Ageing Res. Rev. 2017, 36, 78–87. [Google Scholar] [CrossRef]
- Gale, C.R.; Westbury, L.; Cooper, C. Social isolation and loneliness as risk factors for the progression of frailty: The English Longitudinal Study of Ageing. Age Ageing 2018, 47, 392–397. [Google Scholar] [CrossRef]
- Ma, L.; Tang, Z.; Zhang, L.; Sun, F.; Li, Y.; Chan, P. Prevalence of frailty and associated factors in the community-dwelling population of China. J. Am. Geriatr. Soc. 2018, 66, 559–564. [Google Scholar] [CrossRef]
- Mello, A.d.C.; Engstrom, E.M.; Alves, L.C. Health-related and socio-demographic factors associated with frailty in the elderly: A systematic literature review. Cad. Saude Publica 2014, 30, 1143–1168. [Google Scholar] [CrossRef]
| Unstandardized Sample, n (%) | Standardized Sample (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | Overall n = 2907 | Men n = 1383 (47.6%) | Women n = 1524 (52.4%) | p-Value | Overall | Men (42.2%) | Women (57.8%) | p-Value | |||
| Age (years) | |||||||||||
| 70–74 | 1154 | (39.7) | 505 | (36.5) | 649 | (42.6) | <0.001 | 41.8 | 45.4 | 39.2 | <0.001 |
| 75–79 | 1080 | (37.2) | 529 | (38.3) | 551 | (36.2) | 36.1 | 35.8 | 36.3 | ||
| 80–84 | 673 | (23.2) | 349 | (25.2) | 324 | (21.3) | 22.1 | 18.8 | 24.5 | ||
| Low education level (<7 years) | 1265 | (43.5) | 361 | (26.1) | 904 | (31.1) | <0.001 | 45.5 | 25.5 | 60.5 | <0.001 |
| Live alone | 659 | (22.7) | 120 | (8.7) | 539 | (35.4) | <0.001 | 24.3 | 8.2 | 36.2 | <0.001 |
| Marital status (without partner) | 948 | (32.6) | 145 | (10.5) | 803 | (52.7) | <0.001 | 35.2 | 9.9 | 53.7 | <0.001 |
| Residence | |||||||||||
| Urban | 822 | (28.4) | 387 | (28.1) | 435 | (28.7) | 0.035 | 28.4 | 29.1 | 27.9 | 0.720 |
| Suburban | 1250 | (43.2) | 569 | (41.4) | 681 | (45.0) | 43.6 | 43.5 | 43.6 | ||
| Rural | 819 | (28.3) | 420 | (30.5) | 399 | (26.3) | 28.0 | 27.3 | 28.4 | ||
| Social security recipient | 204 | (7.0) | 86 | (6.2) | 118 | (7.8) | 0.058 | 7.2 | 6.3 | 7.9 | 0.055 |
| Current worker | 758 | (26.1) | 425 | (30.8) | 333 | (21.9) | <0.001 | 25.8 | 31.1 | 21.9 | <0.001 |
| Overall | Men (42.2%) | Women (57.8%) | |||||
|---|---|---|---|---|---|---|---|
| Variable | % | (95% CI) | % | (95% CI) | % | (95% CI) | p-Value |
| Frailty status | |||||||
| Robust | 45.2 | (43.3–47.0) | 52.8 | (50.1–55.5) | 39.6 | (37.1–42.0) | <0.001 |
| Prefrail | 47.0 | (45.1–48.8) | 41.1 | (38.5–43.8) | 51.3 | (48.7–53.8) | |
| Frail | 7.9 | (6.8–8.9) | 6.0 | (4.9–7.2) | 9.2 | (7.7–10.7) | |
| Frailty component | |||||||
| Exhaustion | 32.5 | (30.7–34.2) | 21.1 | (18.9–23.2) | 40.8 | (38.3–43.3) | <0.001 |
| Low physical activity | 10.2 | (8.7–11.8) | 10.7 | (9.1–12.3) | 10.5 | (9.4–11.6) | 0.695 |
| Slowness | 20.1 | (18.6–21.6) | 18.8 | (16.8–20.8) | 21.1 | (19.0–23.2) | 0.131 |
| Weakness | 19.7 | (18.3–21.2) | 18.0 | (16.0–20.0) | 21.0 | (18.9–23.1) | 0.042 |
| Unintentional weight loss | 4.9 | (4.1–5.7) | 5.2 | (4.0–6.3) | 4.6 | (3.5–5.7) | 0.527 |
| Frailty score | |||||||
| 0 | 45.2 | (43.3–47.0) | 52.8 | (50.1–55.5) | 39.6 | (37.1–42.0) | <0.001 |
| 1 | 32.1 | (30.3–33.8) | 28.8 | (26.3–31.2) | 34.5 | (32.1–36.9) | |
| 2 | 15.0 | (13.6–16.3) | 12.4 | (10.7–14.1) | 16.8 | (14.9–18.7) | |
| 3 | 5.8 | (4.9–6.7) | 4.5 | (3.4–5.5) | 6.8 | (5.5–8.1) | |
| 4 | 1.9 | (1.4–2.4) | 1.5 | (1.0–2.1) | 2.2 | (1.4–2.9) | |
| 5 | 0.1 | (0.0–0.3) | 0.1 | (0.0–0.2) | 0.2 | (0.0–0.4) | |
| Variable | Overall | Robust (45.2%) | Prefrail (47.0%) | Frail (7.9%) | p-Value | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sociodemographic | |||||||||||||
| Age (years) | 75.8 | ± | 0.07 | 74.9 | ± | 0.09 | 76.3 | ± | 0.10 | 78.5 | ± | 0.23 | <0.001 |
| 70–74 | 41.8 | 51.0 | 37.7 | 14.5 | <0.001 | ||||||||
| 75–79 | 36.1 | 36.2 | 35.2 | 40.4 | |||||||||
| 80–84 | 22.1 | 12.8 | 27.1 | 45.2 | |||||||||
| Female sex | 57.8 | 50.6 | 63.1 | 67.5 | <0.001 | ||||||||
| Low education level (<7 years) | 45.5 | 33.7 | 52.5 | 71.8 | <0.001 | ||||||||
| Live alone | 24.3 | 19.4 | 27.5 | 33.6 | <0.001 | ||||||||
| Marital status (without partner) | 35.3 | 29.3 | 39.2 | 46.2 | <0.001 | ||||||||
| Residence | |||||||||||||
| Urban | 28.4 | 34.2 | 25.5 | 13.2 | <0.001 | ||||||||
| Suburban | 43.6 | 43.9 | 43.6 | 41.7 | |||||||||
| Rural | 28.0 | 21.9 | 30.9 | 45.2 | |||||||||
| Social security recipient | 7.2 | 5.8 | 8.0 | 11.1 | 0.017 | ||||||||
| Current worker | 25.8 | 26.2 | 26.6 | 17.9 | 0.010 | ||||||||
| Physical | |||||||||||||
| Underweight (BMI < 18.5 kg/m2) | 1.6 | 1.1 | 1.7 | 4.0 | 0.030 | ||||||||
| Low appendicular skeletal muscle (lower 20%) | 22.3 | 16.3 | 25.9 | 35.6 | <0.001 | ||||||||
| Low calf circumference (<32 cm) | 27.6 | 19.5 | 31.9 | 48.1 | <0.001 | ||||||||
| High waist circumference (M ≥ 102 cm; F ≥ 88 cm) | 51.4 | 48.1 | 54.1 | 53.6 | 0.007 | ||||||||
| Sarcopenia (AWGS-defined) | 10.1 | 1.1 | 14.6 | 34.5 | <0.001 | ||||||||
| ADL disability (>1 point) | 2.2 | 0.8 | 2.1 | 10.2 | <0.001 | ||||||||
| IADL disability (>2 points) | 6.3 | 3.9 | 6.8 | 17.3 | <0.001 | ||||||||
| Falls in the past year | 20.6 | 16.0 | 22.3 | 36.6 | <0.001 | ||||||||
| Physical function | |||||||||||||
| Severe mobility limitation | 10.1 | 1.1 | 14.6 | 34.5 | <0.001 | ||||||||
| Timed Up and Go (seconds) | 10.5 | ± | 0.05 | 9.4 | ± | 0.04 | 10.8 | ± | 0.1 | 14.9 | ± | 0.3 | <0.001 |
| Short Physical Performance Battery (score) | 10.8 | ± | 0.03 | 11.4 | ± | 0.02 | 10.6 | ± | 0.04 | 8.6 | ± | 0.15 | <0.001 |
| Gait speed (m/s) | 1.10 | ± | 0.00 | 1.22 | ± | 0.01 | 1.04 | ± | 0.00 | 0.76 | ± | 0.01 | <0.001 |
| Grip strength (kg) | 25.7 | ± | 0.1 | 28.7 | ± | 0.2 | 24.0 | ± | 0.2 | 18.9 | ± | 0.4 | <0.001 |
| Biological | |||||||||||||
| Albumin (g/dL) | 4.4 | ± | 0.00 | 4.4 | ± | 0.01 | 4.3 | ± | 0.01 | 4.3 | ± | 0.02 | <0.001 |
| Serum creatinine (mg/dL) | 0.84 | ± | 0.01 | 0.83 | ± | 0.01 | 0.84 | ± | 0.01 | 0.87 | ± | 0.02 | 0.271 |
| HbA1c (%) | 6.0 | ± | 0.02 | 6.0 | ± | 0.02 | 6.0 | ± | 0.02 | 6.2 | ± | 0.08 | <0.001 |
| WBC (X1000/uL) | 5.9 | ± | 0.03 | 5.7 | ± | 0.04 | 6.0 | ± | 0.05 | 6.2 | ± | 0.12 | <0.001 |
| RBC (Mil/uL) | 4.4 | ± | 0.01 | 4.4 | ± | 0.01 | 4.3 | ± | 0.01 | 4.2 | ± | 0.03 | <0.001 |
| Cortisol (ug/dL) at 8 a.m. | 10.1 | ± | 0.08 | 10.2 | ± | 0.10 | 10.1 | ± | 0.11 | 10.2 | ± | 0.30 | 0.740 |
| hs-CRP (mg/L) | 1.34 | ± | 0.04 | 1.29 | ± | 0.05 | 1.31 | ± | 0.05 | 1.85 | ± | 0.19 | <0.001 |
| Vitamin B12 (pg/mL) | 610.1 | ± | 5.34 | 614.8 | ± | 7.77 | 608.3 | ± | 7.77 | 593.4 | ± | 22.02 | 0.551 |
| TSH (ulU/mL) | 2.8 | ± | 0.10 | 2.9 | ± | 0.20 | 2.6 | ± | 0.07 | 2.7 | ± | 0.22 | 0.306 |
| Insulin (uU/mL) | 8.0 | ± | 0.18 | 7.3 | ± | 0.20 | 8.3 | ± | 0.21 | 9.9 | ± | 1.41 | <0.001 |
| Triglyceride (mg/dL) | 122.6 | ± | 1.15 | 120.3 | ± | 1.69 | 122.5 | ± | 1.60 | 137.1 | ± | 5.15 | 0.001 |
| Total cholesterol (mg/dL) | 174.6 | ± | 0.68 | 176.5 | ± | 1.01 | 173.1 | ± | 1.01 | 172.5 | ± | 2.26 | 0.037 |
| HDL-cholesterol (mg/dL) | 52.5 | ± | 0.26 | 53.4 | ± | 0.39 | 52.0 | ± | 0.38 | 50.5 | ± | 0.93 | 0.003 |
| LDL-cholesterol (mg/dL) | 108.2 | ± | 0.63 | 109.7 | ± | 0.95 | 107.1 | ± | 0.92 | 106.0 | ± | 2.10 | 0.069 |
| 25-hydroxy vitamin D (mg/mL) | 23.2 | ± | 0.19 | 23.6 | ± | 0.28 | 23.2 | ± | 0.27 | 21.0 | ± | 0.56 | 0.001 |
| eGFR (mL/min/1.73 m2) a | 77.5 | ± | 0.27 | 78.9 | ± | 0.35 | 76.9 | ± | 0.41 | 73.3 | ± | 1.14 | <0.001 |
| Lifestyle | |||||||||||||
| Current smoker | 5.2 | 4.7 | 5.3 | 7.7 | 0.238 | ||||||||
| Alcohol intake (≥2–3 time/week) | 16.3 | 19.1 | 14.1 | 13.4 | 0.001 | ||||||||
| Sleep latency (>1 h) | 4.3 | 3.1 | 4.9 | 7.0 | 0.019 | ||||||||
| Long night-time sleep (>8 h) | 5.9 | 4.8 | 6.5 | 9.2 | <0.001 | ||||||||
| Dairy products (not every day) | 60.9 | 56.3 | 62.8 | 74.0 | <0.001 | ||||||||
| Legumes and eggs intake (<2 times/week) | 19.7 | 13.8 | 22.8 | 33.5 | <0.001 | ||||||||
| Meat, fish, and poultry intake (not every day) | 80.8 | 78.0 | 83.2 | 82.0 | 0.045 | ||||||||
| Risk of malnutrition (MNA score ≤ 11) | 8.0 | 3.7 | 10.0 | 20.0 | <0.001 | ||||||||
| Health condition | |||||||||||||
| Number of drugs | 4.4 | ± | 0.06 | 3.9 | ± | 0.08 | 4.7 | ± | 0.09 | 5.7 | ± | 0.23 | <0.001 |
| Comorbidity (≥2 diseases) | 55.6 | 48.3 | 61.3 | 63.2 | <0.001 | ||||||||
| Polypharmacy (≥5 medications) | 32.2 | 24.4 | 36.0 | 55.0 | <0.001 | ||||||||
| Hospitalization in the past year | 12.9 | 8.6 | 15.8 | 20.5 | <0.001 | ||||||||
| Hearing impairment | 15.3 | 13.9 | 15.7 | 21.2 | 0.033 | ||||||||
| Visual impairment | 2.6 | 1.4 | 3.4 | 4.4 | 0.002 | ||||||||
| Low chewing ability | 46.7 | 39.1 | 48.8 | 64.0 | <0.001 | ||||||||
| Low pronouncing ability | 25.0 | 18.9 | 27.4 | 45.5 | <0.001 | ||||||||
| Medical condition | |||||||||||||
| Hypertension | 58.4 | 54.0 | 61.4 | 66.2 | <0.001 | ||||||||
| Diabetes | 21.9 | 18.3 | 23.9 | 31.3 | <0.001 | ||||||||
| Urinary incontinence | 4.1 | 2.2 | 5.0 | 10.4 | <0.001 | ||||||||
| Cardiovascular disease b | 13.3 | 11.2 | 14.4 | 18.6 | 0.004 | ||||||||
| Dyslipidemia | 33.6 | 34.2 | 33.7 | 29.1 | 0.326 | ||||||||
| Osteoarthritis | 26.7 | 20.1 | 31.6 | 34.8 | <0.001 | ||||||||
| Osteoporosis | 17.5 | 13.2 | 20.6 | 23.4 | <0.001 | ||||||||
| Rheumatoid arthritis | 2.2 | 1.0 | 3.0 | 4.2 | <0.001 | ||||||||
| Digestive system ulceration | 6.3 | 4.9 | 7.5 | 7.2 | 0.019 | ||||||||
| Chronic obstructive pulmonary disease | 0.9 | 0.9 | 0.8 | 1.3 | 0.703 | ||||||||
| Allergic rhinitis | 4.1 | 4.8 | 3.6 | 3.3 | 0.241 | ||||||||
| Bronchitis | 1.5 | 1.0 | 1.9 | 2.1 | 0.137 | ||||||||
| Asthma | 3.6 | 3.1 | 3.7 | 5.2 | 0.369 | ||||||||
| Thyroid disease | 4.7 | 4.9 | 4.5 | 4.4 | 0.889 | ||||||||
| Kidney disease | 1.5 | 1.0 | 1.8 | 2.8 | 0.087 | ||||||||
| Prostate disease | 14.7 | 16.8 | 13.0 | 12.6 | 0.011 | ||||||||
| Depressive disorder | 3.0 | 1.9 | 2.8 | 10.7 | <0.001 | ||||||||
| Psychological | |||||||||||||
| EQ-5D index | 0.88 | ± | 0.00 | 0.92 | ± | 0.00 | 0.86 | ± | 0.00 | 0.73 | ± | 0.01 | <0.001 |
| EQ-VAS | 74.2 | ± | 0.33 | 79.6 | ± | 0.40 | 71.3 | ± | 0.49 | 60.1 | ± | 1.49 | <0.001 |
| SF-12 | ± | ± | ± | ± | |||||||||
| Physical health | 43.3 | ± | 0.21 | 48.1 | ± | 0.23 | 40.9 | ± | 0.30 | 30.7 | ± | 0.72 | <0.001 |
| Mental health | 52.7 | ± | 0.20 | 55.6 | ± | 0.22 | 51.3 | ± | 0.32 | 44.7 | ± | 0.87 | <0.001 |
| Poor self-perceived health | 31.0 | 17.2 | 37.9 | 68.9 | <0.001 | ||||||||
| Depressive symptoms (GDS score ≥ 6) | 22.7 | 9.4 | 29.8 | 57.0 | <0.001 | ||||||||
| Cognitive dysfunction (MMSE score < 24) | 22.3 | 12.4 | 27.3 | 49.6 | <0.001 | ||||||||
| Cognitive impairment | 24.4 | 17.9 | 27.7 | 41.7 | <0.001 | ||||||||
| Social | |||||||||||||
| Social support | 5.5 | ± | 0.02 | 5.5 | ± | 0.03 | 5.4 | ± | 0.04 | 5.3 | ± | 0.10 | 0.026 |
| Poor social capital | 6.4 | 5.2 | 6.3 | 13.7 | 0.002 | ||||||||
| Social network | |||||||||||||
| Low interaction with family | 39.5 | 37.1 | 40.6 | 46.4 | 0.019 | ||||||||
| Low interaction with friends | 23.1 | 17.1 | 25.8 | 41.9 | <0.001 | ||||||||
| Low interaction with neighbor | 28.7 | 30.2 | 27.6 | 26.9 | 0.294 | ||||||||
| Religious activities (none) | 41.7 | 41.5 | 40.6 | 49.8 | 0.044 | ||||||||
| Social activities (none) | 21.6 | 20.6 | 22.3 | 23.3 | 0.491 | ||||||||
| Variable | B | S.E. | p-Value | OR | 95% CI | |
|---|---|---|---|---|---|---|
| Lower | Upper | |||||
| At risk of malnutrition a | 0.922 | 0.240 | <0.001 | 2.51 | 1.57 | 4.03 |
| Sarcopenia b | 0.872 | 0.202 | <0.001 | 2.39 | 1.61 | 3.56 |
| Severe mobility limitation c | 0.758 | 0.199 | <0.001 | 2.13 | 1.45 | 3.15 |
| Poor social capital d | 0.687 | 0.287 | 0.017 | 1.99 | 1.13 | 3.49 |
| Residence | ||||||
| Urban | Ref. | |||||
| Suburban | 0.571 | 0.259 | 0.027 | 1.77 | 1.07 | 2.94 |
| Rural | 0.673 | 0.265 | 0.016 | 1.89 | 1.13 | 3.18 |
| Depressive e | 0.634 | 0.194 | 0.001 | 1.89 | 1.29 | 2.76 |
| Poor self-perceived health | 0.502 | 0.199 | 0.012 | 1.65 | 1.12 | 2.44 |
| Polypharmacy f | 0.478 | 0.181 | 0.008 | 1.61 | 1.13 | 2.30 |
| hs-CRP, mg/L | 0.252 | 0.095 | 0.008 | 1.29 | 1.07 | 1.55 |
| HbA1c, % | 0.243 | 0.103 | 0.018 | 1.28 | 1.04 | 1.56 |
| Timed Up and Go, seconds | 0.236 | 0.039 | <0.001 | 1.27 | 1.17 | 1.37 |
| Age, years | 0.081 | 0.025 | 0.001 | 1.08 | 1.03 | 1.14 |
| 25-hydroxyvitamin D, ng/mL | −0.023 | 0.010 | 0.024 | 0.98 | 0.96 | 1.00 |
| Short Physical Performance Battery | −0.144 | 0.064 | 0.025 | 0.87 | 0.76 | 0.98 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, H.; Kim, M.; Lee, Y.; Won, C.W. Prevalence of Physical Frailty and Its Multidimensional Risk Factors in Korean Community-Dwelling Older Adults: Findings from Korean Frailty and Aging Cohort Study. Int. J. Environ. Res. Public Health 2020, 17, 7883. https://doi.org/10.3390/ijerph17217883
Jung H, Kim M, Lee Y, Won CW. Prevalence of Physical Frailty and Its Multidimensional Risk Factors in Korean Community-Dwelling Older Adults: Findings from Korean Frailty and Aging Cohort Study. International Journal of Environmental Research and Public Health. 2020; 17(21):7883. https://doi.org/10.3390/ijerph17217883
Chicago/Turabian StyleJung, Heeeun, Miji Kim, Yunhwan Lee, and Chang Won Won. 2020. "Prevalence of Physical Frailty and Its Multidimensional Risk Factors in Korean Community-Dwelling Older Adults: Findings from Korean Frailty and Aging Cohort Study" International Journal of Environmental Research and Public Health 17, no. 21: 7883. https://doi.org/10.3390/ijerph17217883
APA StyleJung, H., Kim, M., Lee, Y., & Won, C. W. (2020). Prevalence of Physical Frailty and Its Multidimensional Risk Factors in Korean Community-Dwelling Older Adults: Findings from Korean Frailty and Aging Cohort Study. International Journal of Environmental Research and Public Health, 17(21), 7883. https://doi.org/10.3390/ijerph17217883






