The Effects of Exogenous Lactate Administration on the IGF1/Akt/mTOR Pathway in Rat Skeletal Muscle
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Care
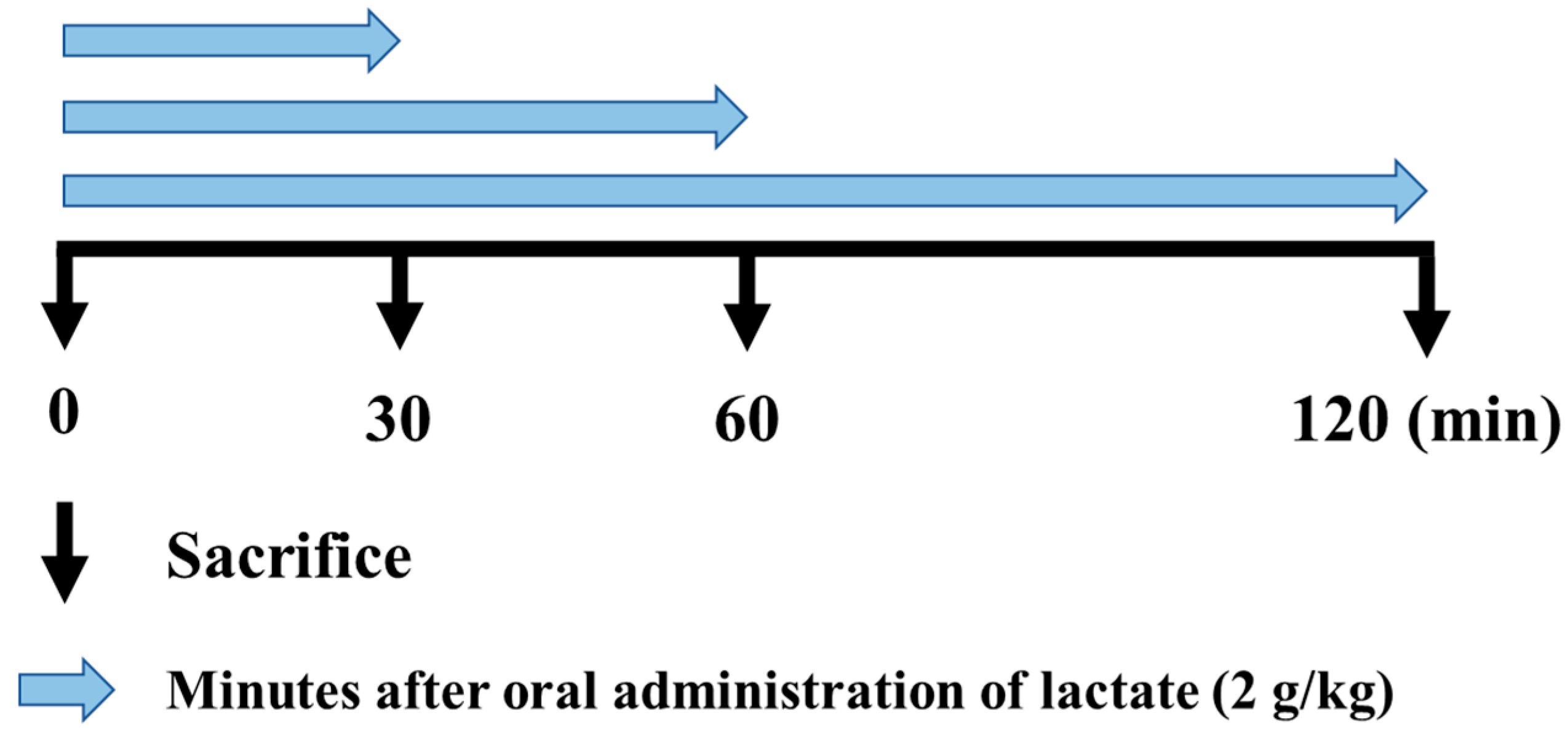
2.2. Animal Protocol
2.3. Blood Parameters
2.4. Reverse Transcription-Polymerase Chain Reaction (RT-PCR) Analysis
2.5. Western Blotting
2.6. Statistical Analysis
3. Results
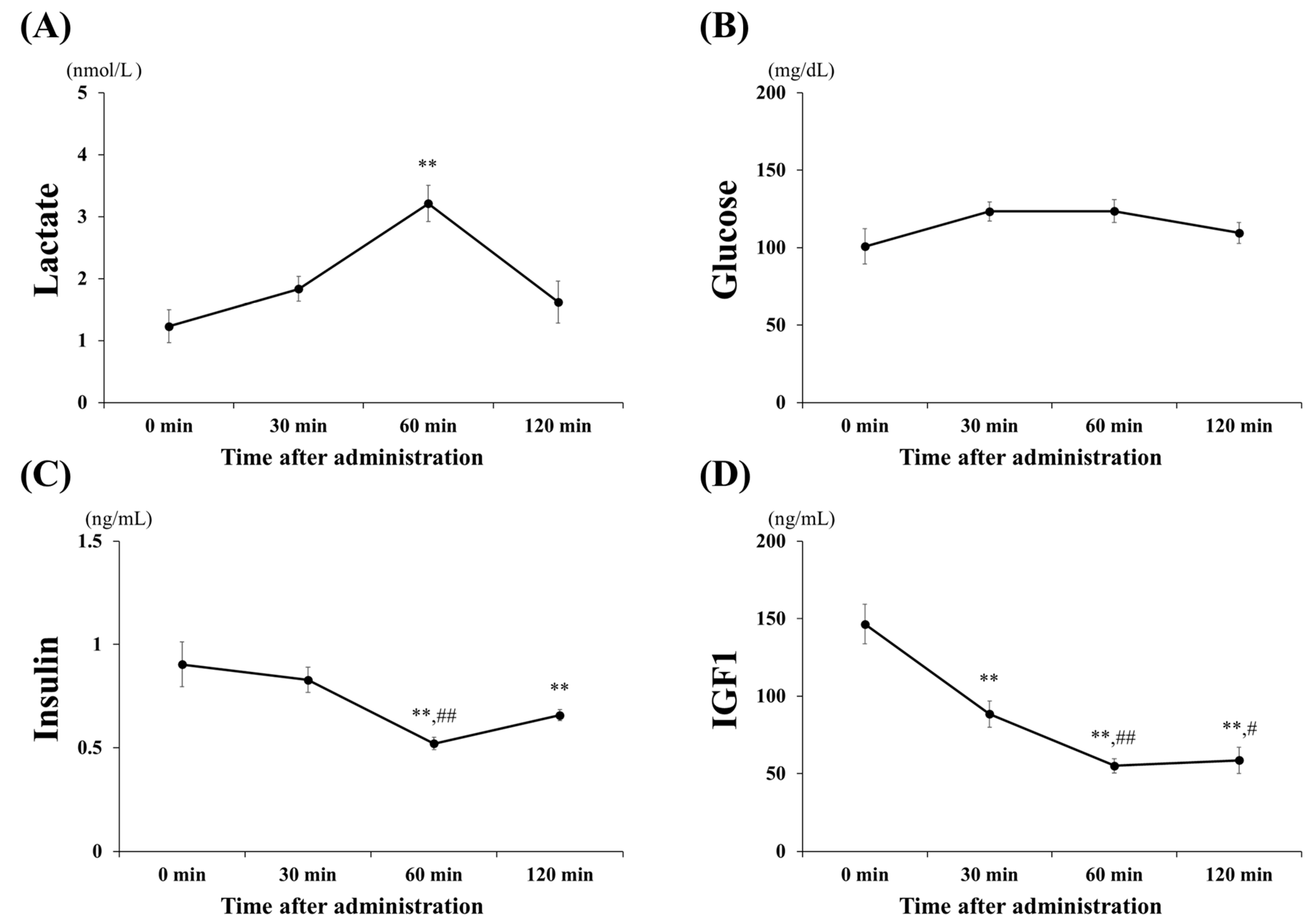
3.1. Blood Concentrations of Lactate, Glucose, Insulin, and IGF1
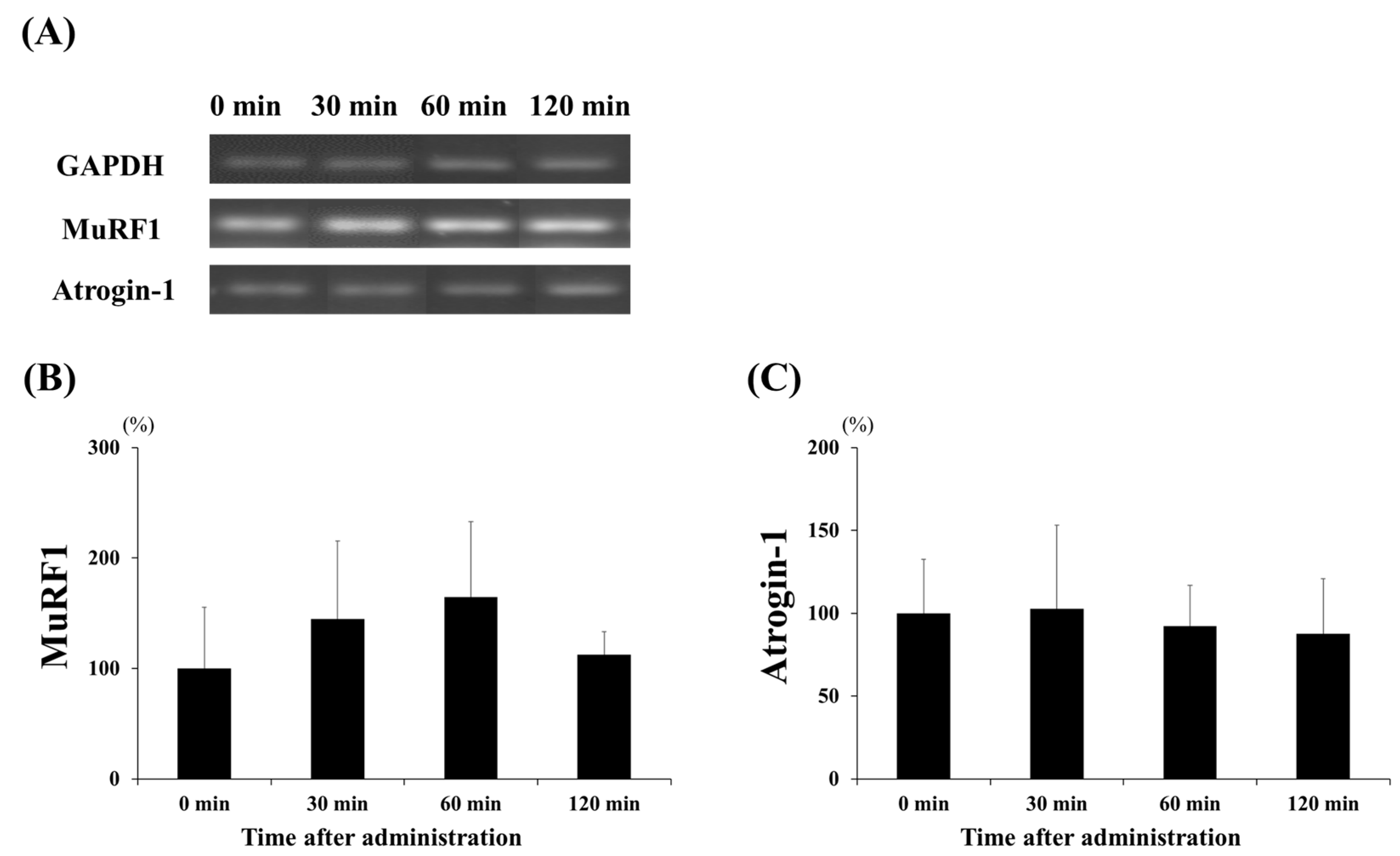
3.2. mRNA Expression of Protein Synthesis and Degradation Factors
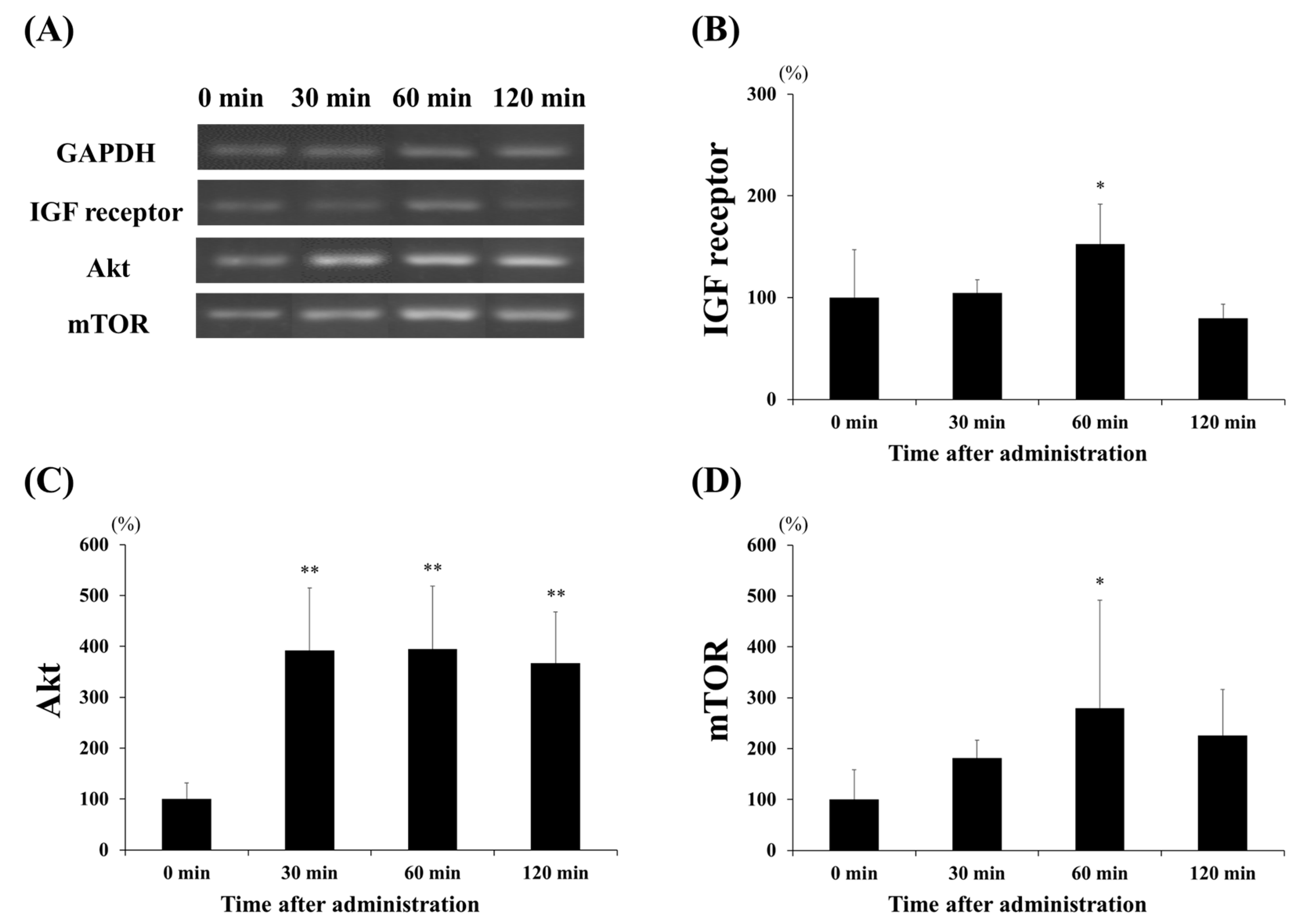
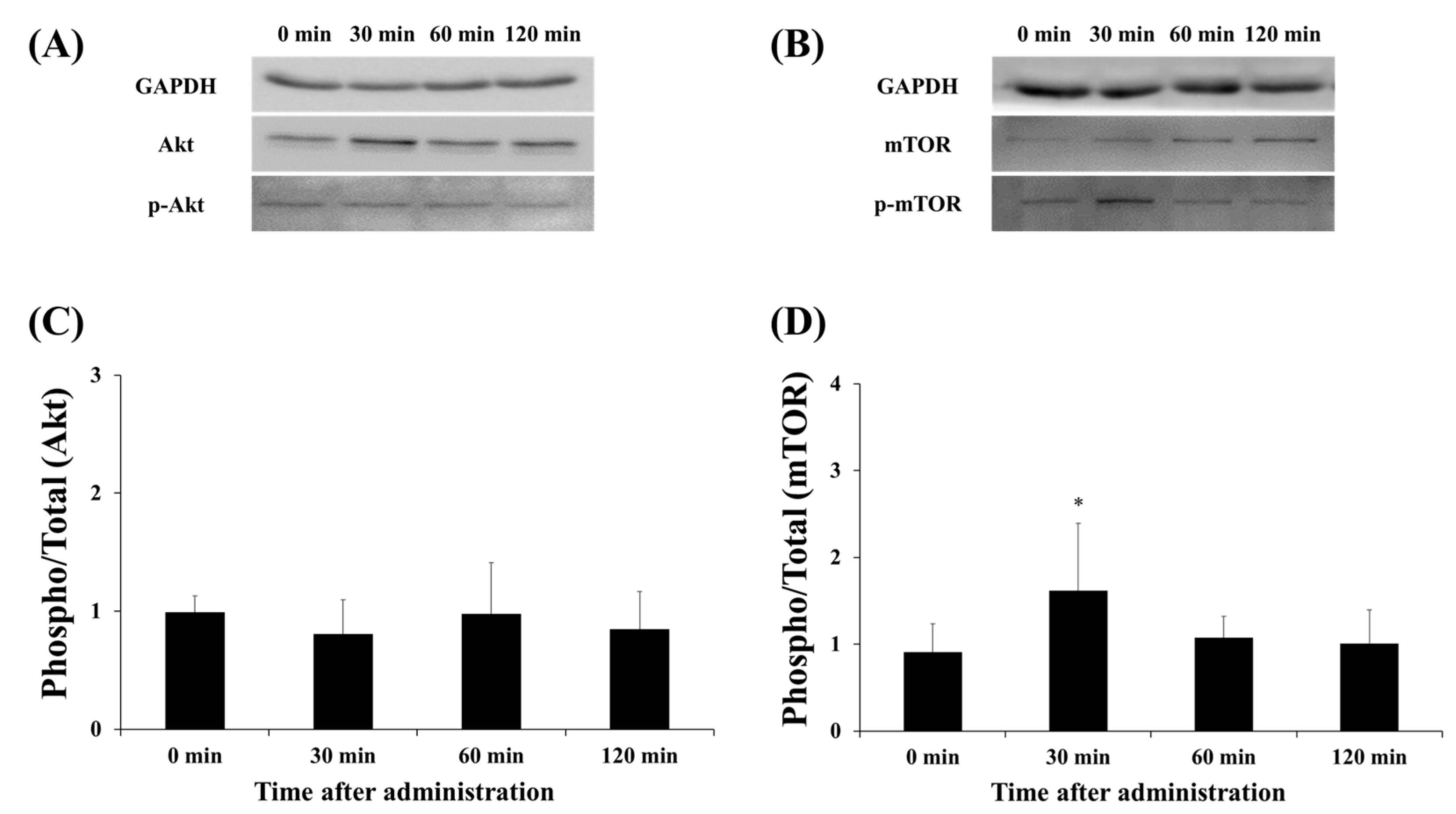
3.3. Protein Expression of Akt and mTOR
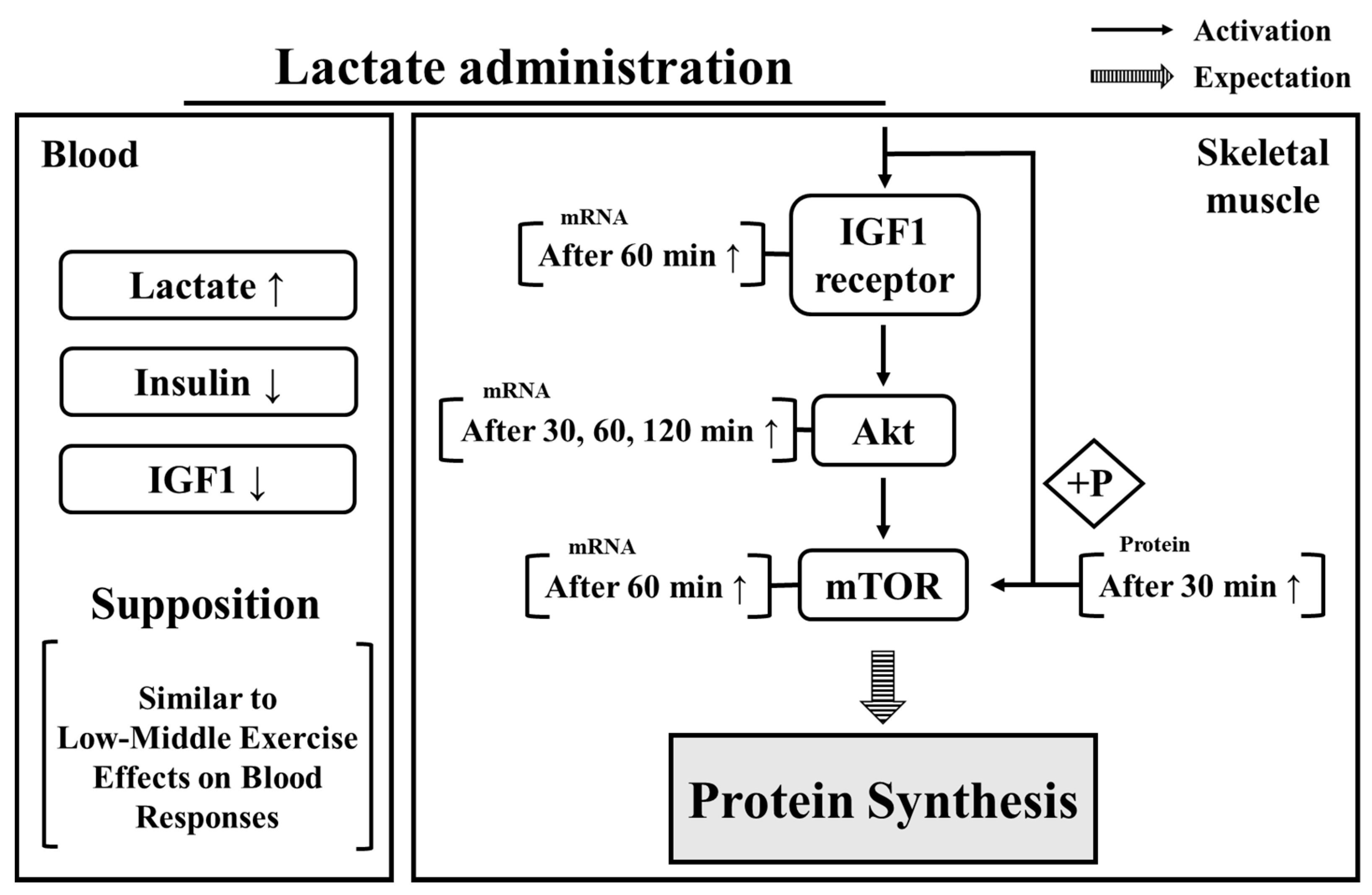
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Tang, J.E.; Phillips, S.M. Maximizing muscle protein anabolism: The role of protein quality. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Mukund, K.; Subramaniam, S. Skeletal muscle: A review of molecular structure and function, in health and disease. WIREs Syst. Biol. Med. 2020, 12, 1–46. [Google Scholar] [CrossRef] [PubMed]
- Rommel, C.; Bodine, S.C.; Clarke, B.A.; Rossman, R.; Nunez, L.; Stitt, T.N.; Yancopoulos, G.D.; Glass, D.J. Mediation of IGF-1-induced skeletal myotube hypertrophy by PI(3)K/Akt/MTOR and PI(3)K/Akt/GSK3 pathways. Nat. Cell Biol. 2001, 3, 1009–1013. [Google Scholar] [CrossRef] [PubMed]
- Glass, D.J. Signalling pathways that mediate skeletal muscle hypertrophy and atrophy. Nat. Cell Biol. 2003, 5, 87–90. [Google Scholar] [CrossRef]
- Devin, J.L.; Bolam, K.A.; Jenkins, D.G.; Skinner, T.L. The influence of exercise on the insulin-like growth factor axis in oncology: Physiological basis, current, and future perspectives. Cancer Epidemiol. Biol. Prev. Biomark. 2016, 25, 239–249. [Google Scholar] [CrossRef]
- Schiaffino, S.; Mammucari, C. Regulation of skeletal muscle growth by the IGF1-Akt/PKB pathway: Insights from genetic models. Skelet. Muscle 2011, 1, 4. [Google Scholar] [CrossRef]
- Bodine, S.C.; Stitt, T.N.; Gonzalez, M.; Kline, W.O.; Stover, G.L.; Bauerlein, R.; Zlotchenko, E.; Scrimgeour, A.; Lawrence, J.C.; Glass, D.J.; et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy In Vivo. Nat. Cell Biol. 2001, 3, 1014–1019. [Google Scholar] [CrossRef]
- Stitt, T.N.; Drujan, D.; Clarke, B.A.; Panaro, F.; Timofeyva, Y.; Kline, W.O.; Gonzalez, M.; Yancopoulos, G.D.; Glass, D.J. The IGF-1/PI3K/Akt pathway prevents expression of muscle atrophy-induced ubiquitin ligases by inhibiting FOXO transcription factors. Mol. Cell 2004, 14, 395–403. [Google Scholar] [CrossRef]
- Hollidge-Horvat, M.G.; Parolin, M.L.; Wong, D.; Jones, N.L.; Heigenhauser, G.J.F. Effect of induced metabolic alkalosis on human skeletal muscle metabolism during exercise. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E316–E329. [Google Scholar] [CrossRef]
- Sutton, J.R.; Jones, N.L.; Toews, C.J. Effect of pH on muscle glycolysis during exercise. Clin. Sci. 1981, 61, 331–338. [Google Scholar] [CrossRef]
- Brooks, G.A. The lactate shuttle during exercise and recovery. Med. Sci. Sports Exerc. 1986, 18, 360–380. [Google Scholar] [CrossRef] [PubMed]
- Stevenson, R.W.; Mitchell, D.R.; Hendrick, G.K.; Rainey, R.; Cherrington, A.D.; Frizzell, R.T. Lactate as substrate for glycogen resynthesis after exercise. J. Appl. Physiol. 1987, 62, 2237–2240. [Google Scholar] [CrossRef] [PubMed]
- Nalbandian, M.; Takeda, M. Lactate as a signaling molecule that regulates exercise-induced adaptations. Biology 2016, 5, 38. [Google Scholar] [CrossRef]
- Ohno, Y.; Ando, K.; Ito, T.; Suda, Y.; Matsui, Y.; Oyama, A.; Kaneko, H.; Yokoyama, S.; Egawa, T.; Goto, K. Lactate stimulates a potential for hypertrophy and regeneration of mouse skeletal muscle. Nutrients 2019, 11, 869. [Google Scholar] [CrossRef]
- Oishi, Y.; Tsukamoto, H.; Yokokawa, T.; Hirotsu, K.; Shimazu, M.; Uchida, K.; Tomi, H.; Higashida, K.; Iwanaka, N.; Hashimoto, T. Mixed lactate and caffeine compound increases satellite cell activity and anabolic signals for muscle hypertrophy. J. Appl. Physiol. 2015, 118, 742–749. [Google Scholar] [CrossRef] [PubMed]
- Cerda-Kohler, H.; Henríquez-Olguín, C.; Casas, M.; Jensen, T.E.; Llanos, P.; Jaimovich, E. Lactate administration activates the ERK1/2, MTORC1, and AMPK pathways differentially according to skeletal muscle type in mouse. Physiol. Rep. 2018, 6, e13800. [Google Scholar] [CrossRef]
- Oh, S.S.J.; Kim, S.; Moon, S.; Park, D.H.; Kang, J.H. Lactate overload inhibits myogenic activity in C2C12 myotubes. Open Life Sci. 2019, 14, 29–37. [Google Scholar] [CrossRef]
- Tsukamoto, S.; Shibasaki, A.; Naka, A.; Saito, H.; Iida, K. Lactate promotes myoblast differentiation and myotube hypertrophy via a pathway involving MyoD in vitro and enhances muscle regeneration in vivo. Int. J. Mol. Sci. 2018, 19, 3649. [Google Scholar] [CrossRef]
- Kim, J.; Lee, K.P.; Beak, S.; Kang, H.R.; Kim, Y.K.; Lim, K. Effect of black chokeberry on skeletal muscle damage and neuronal cell death. J. Exerc. Nutr. Biochem. 2019, 23, 26–31. [Google Scholar] [CrossRef]
- Kim, N.; Kim, J.; Lim, K.; Park, J. Role of dihydrotestosterone in whole-body energy utilization during acute running exercise in mice. J. Exerc. Nutr. Biochem. 2018, 22, 7–11. [Google Scholar] [CrossRef]
- Park, Y.; Jang, I.; Park, H.Y.; Kim, J.; Lim, K. Hypoxic exposure can improve blood glycemic control on high-fat diet-induced obese mice. Phys. Act. Nutr. 2020, 24, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Lezi, E.; Lu, J.; Selfridge, J.E.; Burns, J.M.; Swerdlow, R.H. Lactate administration reproduces specific brain and liver exercise-related changes. J. Neurochem. 2013, 127, 91–100. [Google Scholar] [CrossRef]
- Kyun, S.; Yoo, C.; Hashimoto, T.; Tomi, H.; Teramoto, N.; Kim, J.; Lim, K. Effects of exogenous lactate administration on fat metabolism and glycogen synthesis factors in rats. Phys. Act. Nutr. 2020, 24, 1. [Google Scholar] [CrossRef] [PubMed]
- Higashi, Y.; Sukhanov, S.; Anwar, A.; Shai, S.Y.; Delafontaine, P. Aging, atherosclerosis, and IGF-1. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 626–639. [Google Scholar] [CrossRef]
- Duan, C.; Ren, H.; Gao, S. Insulin-like growth factors (IGFs), IGF receptors, and IGF-binding proteins: Roles in skeletal muscle growth and differentiation. Gen. Comp. Endocrinol. 2010, 167, 344–351. [Google Scholar] [CrossRef]
- Machida, S.; Booth, F.W. Insulin-like growth factor 1 and muscle growth: Implication for satellite cell proliferation. Proc. Nutr. Soc. 2004, 63, 337–340. [Google Scholar] [CrossRef]
- Morville, T.; Sahl, R.E.; Trammell, S.A.J.; Svenningsen, J.S.; Gillum, M.P.; Helge, J.W.; Clemmensen, C. Divergent effects of resistance and endurance exercise on plasma bile acids, FGF19, and FGF21 in humans. JCI Insight 2018, 3, e122737. [Google Scholar] [CrossRef]
- Boisseau, N.; Delamarche, P.; Rannou, F.; Bentue-Ferrer, D.; Gratas-Delamarche, A. Effects of glucose ingestion at the onset of moderate-intensity, prolonged exercise in women as compared to men. Eur. J. Appl. Physiol. 2000, 81, 93–99. [Google Scholar] [CrossRef]
- Kido, K.; Ato, S.; Yokokawa, T.; Makanae, Y.; Sato, K.; Fujita, S. Acute resistance exercise-induced IGF1 expression and subsequent GLUT4 translocation. Physiol. Rep. 2016, 4, e12907. [Google Scholar] [CrossRef]
- Gumucio, J.P.; Mendias, C.L. Atrogin-1, MuRF-1, and sarcopenia. Endocrine 2013, 43, 12–21. [Google Scholar] [CrossRef]
- Hashimoto, T.; Yokokawa, T.; Narusawa, R.; Okada, Y.; Kawaguchi, R.; Higashida, K. A lactate-based compound containing caffeine in addition to voluntary running exercise decreases subcutaneous fat mass and improves glucose metabolism in obese rats. J. Funct. Foods 2019, 56, 84–91. [Google Scholar] [CrossRef]
| Gene | Sequences |
|---|---|
| IGF receptor | F–5′ GAG AAC AAT GAG TGC TGC CA 3′ |
| R–5′ ACC CTT GGA GCA TCT GGG CA 3′ | |
| Akt | F–5′ TGC TGG AGG ACA ACG ACT AT 3′ |
| R–5′ TGT CAT CTT GAT CAG GCG GT 3′ | |
| mTOR | F–5′ TTG AGG TTG CTA TGA CCA GAG AGA A 3′ |
| R–5′ TTA CCA GAA AGG ACA CCA GCC AAT G 3′ | |
| MuRF1 | F–5′ ACA TCT TCC AGG CTG CCA AT 3′ |
| R–5′ GTT CTC CAC CAG CAG GTT CC 3′ | |
| Atrogin-1 | F–5′ GAC TGG ACT TCT CGA CTG CC 3′ |
| R–5′ GAC TTG CCG ACT CTC TGG AC 3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kyun, S.; Yoo, C.; Park, H.-Y.; Kim, J.; Lim, K. The Effects of Exogenous Lactate Administration on the IGF1/Akt/mTOR Pathway in Rat Skeletal Muscle. Int. J. Environ. Res. Public Health 2020, 17, 7805. https://doi.org/10.3390/ijerph17217805
Kyun S, Yoo C, Park H-Y, Kim J, Lim K. The Effects of Exogenous Lactate Administration on the IGF1/Akt/mTOR Pathway in Rat Skeletal Muscle. International Journal of Environmental Research and Public Health. 2020; 17(21):7805. https://doi.org/10.3390/ijerph17217805
Chicago/Turabian StyleKyun, Sunghwan, Choongsung Yoo, Hun-Young Park, Jisu Kim, and Kiwon Lim. 2020. "The Effects of Exogenous Lactate Administration on the IGF1/Akt/mTOR Pathway in Rat Skeletal Muscle" International Journal of Environmental Research and Public Health 17, no. 21: 7805. https://doi.org/10.3390/ijerph17217805
APA StyleKyun, S., Yoo, C., Park, H.-Y., Kim, J., & Lim, K. (2020). The Effects of Exogenous Lactate Administration on the IGF1/Akt/mTOR Pathway in Rat Skeletal Muscle. International Journal of Environmental Research and Public Health, 17(21), 7805. https://doi.org/10.3390/ijerph17217805






