Translation and Validation of the Modified A-DIVA Scale to European Portuguese: Difficult Intravenous Access Scale for Adult Patients
Abstract
1. Introduction
2. Materials and Methods
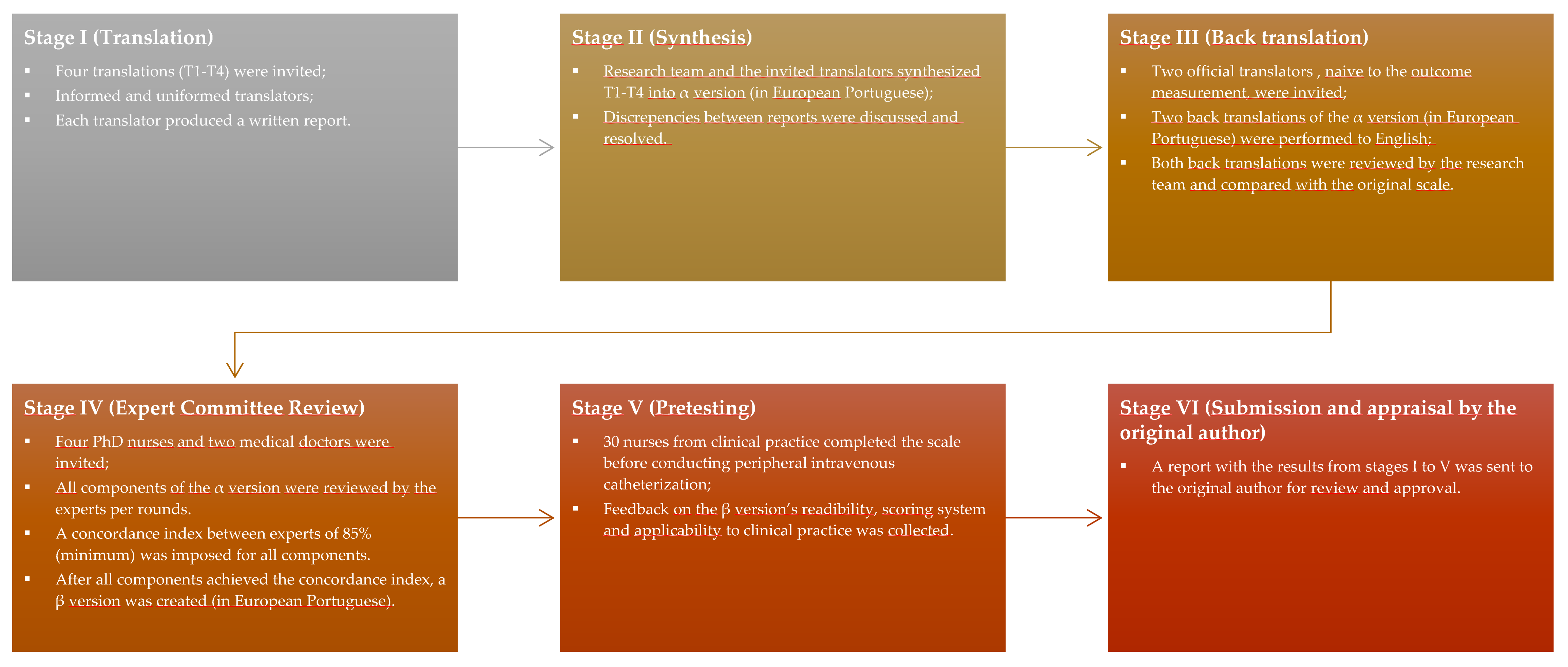
2.1. Phase 1—Translation of the Modified A-DIVA Scale
2.2. Phase 2—Psychometric Validation of the A-DM Scale
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Webster, J.; Morris, H.-L.; Robinson, K.; Sanderson, U. Development and validation of a Vein Assessment Tool (VAT). Aust. J. Adv. Nurs. 2007, 24, 5–7. [Google Scholar] [PubMed]
- Sebbane, M.; Claret, P.-G.; Lefebvre, S.; Mercier, G.; Rubenovitch, J.; Jreige, R.; Eledjam, J.-J.; De La Coussaye, J.-E. Predicting Peripheral Venous Access Difficulty in the Emergency Department Using Body Mass Index and a Clinical Evaluation of Venous Accessibility. J. Emerg. Med. 2013, 44, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Piredda, M.; Biagioli, V.; Barrella, B.; Carpisassi, I.; Ghinelli, R.; Giannarelli, D.; De Marinis, M.G. Factors affecting difficult peripheral intravenous cannulation in adults: A prospective observational study. J. Clin. Nurs. 2017, 26, 1074–1084. [Google Scholar] [CrossRef] [PubMed]
- Alexandrou, E.; Ray-Barruel, G.; Carr, P.J.; Frost, S.A.; Inwood, S.; Higgins, N.; Lin, F.; Alberto, L.; Mermel, L.; Rickard, C.M.; et al. Use of Short Peripheral Intravenous Catheters: Characteristics, Management, and Outcomes Worldwide. J. Hosp. Med. 2018, 13. [Google Scholar] [CrossRef]
- Alexandrou, E.; Ray-Barruel, G.; Carr, P.J.; Frost, S.; Inwood, S.; Higgins, N.; Lin, F.; Alberto, L.; Mermel, L.; Rickard, C.M. International prevalence of the use of peripheral intravenous catheters: Prevalence of the Use of PIVCs. J. Hosp. Med. 2015, 10, 530–533. [Google Scholar] [CrossRef]
- Monteiro, D.; de la Torre-Montero, J.; Nicolussi, A.; Reis, R.; Barbosa, M.; Toffano, S. Prevalence of and factors associated with difficult peripheral venipuncture in adult surgical patients. J. Vasc. Access 2020, 112972982093933. [Google Scholar] [CrossRef]
- Arreguy-Sena, C.; De Melo, L.D.; Braga, L.M.; Krempser, P.; Lemos, R.D.C.P.B.; Lopes, D.D.P. Punção de veias periféricas em adultos hospitalizados: Método misto sequencial aninhado. Enferm. Bras. 2020, 18, 775. [Google Scholar] [CrossRef]
- Horsfield, C. A personal experience of care and the lack of it. J. Infect. Prev. 2014, 15, 82–83. [Google Scholar] [CrossRef]
- Cooke, M.; Ullman, A.J.; Ray-Barruel, G.; Wallis, M.; Corley, A.; Rickard, C.M. Not “just” an intravenous line: Consumer perspectives on peripheral intravenous cannulation (PIVC). An international cross-sectional survey of 25 countries. PLoS ONE 2018, 13, e0193436. [Google Scholar] [CrossRef]
- Vessel Health and Preservation: The Right Approach for Vascular Access. Available online: https://link.springer.com/book/10.1007/978-3-030-03149-7 (accessed on 14 October 2020).
- Moureau, N.L.; Trick, N.; Nifong, T.; Perry, C.; Kelley, C.; Carrico, R.; Leavitt, M.; Gordon, S.M.; Wallace, J.; Harvill, M.; et al. Vessel Health and Preservation (Part 1): A New Evidence-Based Approach to Vascular Access Selection and Management. J. Vasc. Access 2012, 13, 351–356. [Google Scholar] [CrossRef]
- Carr, P.; Higgins, N.; Cooke, M.; Rippey, J.; Rickard, C. Tools, Clinical Prediction Rules, and Algorithms for the Insertion of Peripheral Intravenous Catheters in Adult Hospitalized Patients: A Systematic Scoping Review of Literature. J. Hosp. Med. 2017, 12, 851–858. [Google Scholar] [CrossRef] [PubMed]
- Carr, P.J.; Rippey, J.C.R.; Cooke, M.L.; Higgins, N.S.; Trevenen, M.L.; Foale, A.; Keijzers, G.; Rickard, C.M. Derivation of a clinical decision-making aid to improve the insertion of clinically indicated peripheral intravenous catheters and promote vessel health preservation. An observational study. PLoS ONE 2019, 14, e0213923. [Google Scholar] [CrossRef]
- Van Loon, F.; van Hooff, L.; de Boer, H.; Koopman, S.; Buise, M.; Korsten, H.; Dierick-van Daele, A.; Bouwman, A. The Modified A-DIVA Scale as a Predictive Tool for Prospective Identification of Adult Patients at Risk of a Difficult Intravenous Access: A Multicenter Validation Study. J. Clin. Med. 2019, 8, 144. [Google Scholar] [CrossRef]
- Van Loon, F.H.J.; Puijn, L.A.P.M.; Houterman, S.; Bouwman, A.R.A. Development of the A-DIVA Scale: A Clinical Predictive Scale to Identify Difficult Intravenous Access in Adult Patients Based on Clinical Observations. Med. 2016, 95, e3428. [Google Scholar] [CrossRef]
- Braga, L. Práticas de enfermagem e a segurança do doente no processo de punção de vasos e na administração da terapêutica endovenosa. Ph.D. Thesis, Universidade de Lisboa, Lisbon, Portugal, 2017. [Google Scholar]
- Parreira, P.; Serambeque, B.; Costa, P.S.; Mónico, L.S.; Oliveira, V.; Sousa, L.B.; Gama, F.; Bernardes, R.A.; Adriano, D.; Marques, I.A.; et al. Impact of an Innovative Securement Dressing and Tourniquet in Peripheral Intravenous Catheter-Related Complications and Contamination: An Interventional Study. Int. J. Environ. Res. Public Health 2019, 16, 3301. [Google Scholar] [CrossRef]
- Oliveira, A.; Costa, P.; Graveto, J.; Costa, F.; Osório, N.; Cosme, A.; Parreira, P. Práticas dos enfermeiros na cateterização intravenosa: Um estudo descritivo. Rev. Enf. Ref. 2019, 111–120. [Google Scholar] [CrossRef]
- Beaton, D.E.; Bombardier, C.; Guillemin, F.; Ferraz, M.B. Guidelines for the Process of Cross-Cultural Adaptation of Self-Report Measures. Spine 2000, 25, 3186–3191. [Google Scholar] [CrossRef]
- de la Torre-Montero, J.-C.; Montealegre-Sanz, M.; Faraldo-Cabana, A.; Oliva-Pellicer, B.; García-Real, I.; Fenwick, M.; Cáceres, E.M.; Rivas-Eguía, B.; Vila-Borrajo, C.; Valles-Andrés, J.; et al. Venous International Assessment, VIA Scale, Validated Classification Procedure for the Peripheral Venous System. J. Vasc. Access 2014, 15, 45–50. [Google Scholar] [CrossRef]
- Landis, J.R.; Koch, G.G. The Measurement of Observer Agreement for Categorical Data. BioM 1977, 33, 159. [Google Scholar] [CrossRef]
- Boateng, G.O.; Neilands, T.B.; Frongillo, E.A.; Melgar-Quiñonez, H.R.; Young, S.L. Best Practices for Developing and Validating Scales for Health, Social, and Behavioral Research: A Primer. Front. Public Health 2018, 6, 149. [Google Scholar] [CrossRef]
- Rodriguez-Calero, M.A.; de Pedro-Gomez, J.E.; Molero-Ballester, L.J.; Fernandez-Fernandez, I.; Matamalas-Massanet, C.; Moreno-Mejias, L.; Blanco-Mavillard, I.; Moya-Suarez, A.B.; Personat-Labrador, C.; Morales-Asencio, J.M. Risk Factors for Difficult Peripheral Intravenous Cannulation. The PIVV2 Multicentre Case-Control Study. J. Clin. Med. 2020, 9, 799. [Google Scholar] [CrossRef]
- Carr, P.J.; Rippey, J.C.R.; Cooke, M.L.; Trevenen, M.L.; Higgins, N.S.; Foale, A.S.; Rickard, C.M. Factors associated with peripheral intravenous cannulation first-time insertion success in the emergency department. A multicentre prospective cohort analysis of patient, clinician and product characteristics. BMJ Open 2019, 9, e022278. [Google Scholar] [CrossRef] [PubMed]
- Fields, J.M.; Piela, N.E.; Au, A.K.; Ku, B.S. Risk factors associated with difficult venous access in adult ED patients. Am. J. Emerg. Med. 2014, 32, 1179–1182. [Google Scholar] [CrossRef]
- Rodríguez-Calero, M.A.; Blanco-Mavillard, I.; Morales-Asencio, J.M.; Fernández-Fernández, I.; Castro-Sánchez, E.; de Pedro-Gómez, J.E. Defining risk factors associated with difficult peripheral venous Cannulation: A systematic review and meta-analysis. Hear. Lung 2020, 49, 273–286. [Google Scholar] [CrossRef] [PubMed]
- Rippey, J.C.; Carr, P.J.; Cooke, M.; Higgins, N.; Rickard, C.M. Predicting and preventing peripheral intravenous cannula insertion failure in the emergency department: Clinician ‘gestalt’ wins again. Emerg. Med. Australas. 2016, 28, 658–665. [Google Scholar] [CrossRef]
- Lapostolle, F.; Catineau, J.; Garrigue, B.; Monmarteau, V.; Houssaye, T.; Vecci, I.; Tréoux, V.; Hospital, B.; Crocheton, N.; Adnet, F. Prospective evaluation of peripheral venous access difficulty in emergency care. Intensiv. Care Med. 2007, 33, 1452–1457. [Google Scholar] [CrossRef] [PubMed]
- Carr, P.J.; Rippey, J.C.R.; Budgeon, C.A.; Cooke, M.L.; Higgins, N.; Rickard, C.M. Insertion of Peripheral Intravenous Cannulae in the Emergency Department: Factors Associated with First-time Insertion Success. J. Vasc. Access 2016, 17, 182–190. [Google Scholar] [CrossRef]
- Piredda, M.; Fiorini, J.; Facchinetti, G.; Biagioli, V.; Marchetti, A.; Conti, F.; Iacorossi, L.; Giannarelli, D.; Matarese, M.; De Marinis, M.G. Risk factors for a difficult intravenous access: A multicentre study comparing nurses’ beliefs to evidence. J. Clin. Nurs. 2019, 28, 3492–3504. [Google Scholar] [CrossRef]
- Costa, P.; Sousa, L.; Marques, I.; Oliveira, A.; Parreira, P.; Vieira, M.; Graveto, J. Estudos realizados em Portugal no âmbito do cateterismo venoso periférico: Protocolo de scoping review. Rev. Enf. Ref. 2020, 5, e20004. [Google Scholar] [CrossRef]
| Original Version (In English) | European Portuguese Version |
|---|---|
| Factor | Fator |
| Score | Pontuação |
| Is there a known history of a difficult intravenous access? | A pessoa apresenta história prévia de acesso endovenoso periférico considerado difícil? |
| Do you expect a failed first attempt or a difficult intravenous access? | É esperada uma primeira tentativa de punção sem sucesso ou que o acesso endovenoso periférico seja difícil? |
| Is there an inability to identify a dilated vein by palpating the upper extremity? | É incapaz de identificar uma veia dilatada após observação de um membro superior? |
| Is there an inability to identify a dilated vein by visualizing the upper extremity? | É incapaz de identificar uma veia dilatada após palpação de um membro superior? |
| Has the largest dilated vein a diameter less than 3 millimeters? | A veia dilatada de maior calibre apresenta um diâmetro menor que 3 milímetros? |
| Demographic and Clinical Variables | Patients’ Characteristics (n = 100) |
|---|---|
| Age | 63.4 years (28–92; SD ± 14.2) |
| Sex | |
| Male | 8% |
| Female | 92% |
| Comorbidities | |
| Type 2 Diabetes Mellitus | 16% |
| Arterial Hypertension | 44% |
| Dyslipidemia | 16% |
| Smoker | 8% |
| Previous cancer treatment | |
| Chemotherapy | 24% |
| Radiotherapy | 2% |
| Hormone Therapy | 2% |
| None | 74% |
| Body Mass Index (kg/m2) | |
| Below 18.5 | 5% |
| 18.5–24.9 | 26% |
| 25.0–29.9 | 49% |
| 30.0 and above | 17% |
| Missing | 3% |
| Item | Cohen’s Kappa | p-Value |
|---|---|---|
| A pessoa apresenta história prévia de acesso endovenoso periférico considerado difícil? | 0.917 | <0.001 |
| É esperada uma primeira tentativa de punção sem sucesso ou que o acesso endovenoso periférico seja difícil? | 0.847 | <0.001 |
| É incapaz de identificar uma veia dilatada após palpação de um membro superior? | 0.864 | <0.001 |
| É incapaz de identificar uma veia dilatada após observação de um membro superior? | 0.847 | <0.001 |
| A veia dilatada de maior calibre apresenta um diâmetro menor que 3 milímetros? | 0.970 | <0.001 |
| Total | 0.910 | <0.001 |
| Variables | r/rpb | p-Value | |
|---|---|---|---|
| Patient-related variables | Age | 0.154 | 0.030 |
| Gender | 0.069 | 0.330 | |
| Body Mass Index | 0.151 | 0.033 | |
| Visible vein diameter (in millimeters) | −0.523 | <0.001 | |
| Previous antineoplastic treatment | 0.157 | 0.027 | |
| Comorbidities: Arterial Hypertension | 0.072 | 0.309 | |
| Comorbidities: Dyslipidemia | 0.011 | 0.879 | |
| Comorbidities: Type-2 Diabetes | 0.226 | 0.001 | |
| Procedural variables | Caliber (Gauge) | 0.517 | <0.001 |
| Insertion site: hand | −0.063 | 0.375 | |
| Insertion site: forearm | 0.027 | 0.708 | |
| Insertion site: antecubital fossa | 0.072 | 0.311 | |
| First-attempt success | 0.512 | <0.001 | |
| Number of puncture attempts | 0.414 | <0.001 | |
| Time for PIVC insertion (in minutes) | −0.016 | 0.825 | |
| Easiness of puncture | 0.620 | <0.001 | |
| PIVC outcomes | Premature removal | 0.336 | <0.001 |
| Complication: infiltration | 0.086 | 0.226 | |
| Complication: phlebitis | 0.122 | 0.085 | |
| Complications (overall) | 0.157 | 0.027 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santos-Costa, P.; Sousa, L.B.; van Loon, F.H.J.; Salgueiro-Oliveira, A.; Parreira, P.; Vieira, M.; Graveto, J. Translation and Validation of the Modified A-DIVA Scale to European Portuguese: Difficult Intravenous Access Scale for Adult Patients. Int. J. Environ. Res. Public Health 2020, 17, 7552. https://doi.org/10.3390/ijerph17207552
Santos-Costa P, Sousa LB, van Loon FHJ, Salgueiro-Oliveira A, Parreira P, Vieira M, Graveto J. Translation and Validation of the Modified A-DIVA Scale to European Portuguese: Difficult Intravenous Access Scale for Adult Patients. International Journal of Environmental Research and Public Health. 2020; 17(20):7552. https://doi.org/10.3390/ijerph17207552
Chicago/Turabian StyleSantos-Costa, Paulo, Liliana B. Sousa, Fredericus H.J. van Loon, Anabela Salgueiro-Oliveira, Pedro Parreira, Margarida Vieira, and João Graveto. 2020. "Translation and Validation of the Modified A-DIVA Scale to European Portuguese: Difficult Intravenous Access Scale for Adult Patients" International Journal of Environmental Research and Public Health 17, no. 20: 7552. https://doi.org/10.3390/ijerph17207552
APA StyleSantos-Costa, P., Sousa, L. B., van Loon, F. H. J., Salgueiro-Oliveira, A., Parreira, P., Vieira, M., & Graveto, J. (2020). Translation and Validation of the Modified A-DIVA Scale to European Portuguese: Difficult Intravenous Access Scale for Adult Patients. International Journal of Environmental Research and Public Health, 17(20), 7552. https://doi.org/10.3390/ijerph17207552







