Association between Anemia and Stroke in Females: A Nationwide, Population-Based Cohort Study in Taiwan
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source
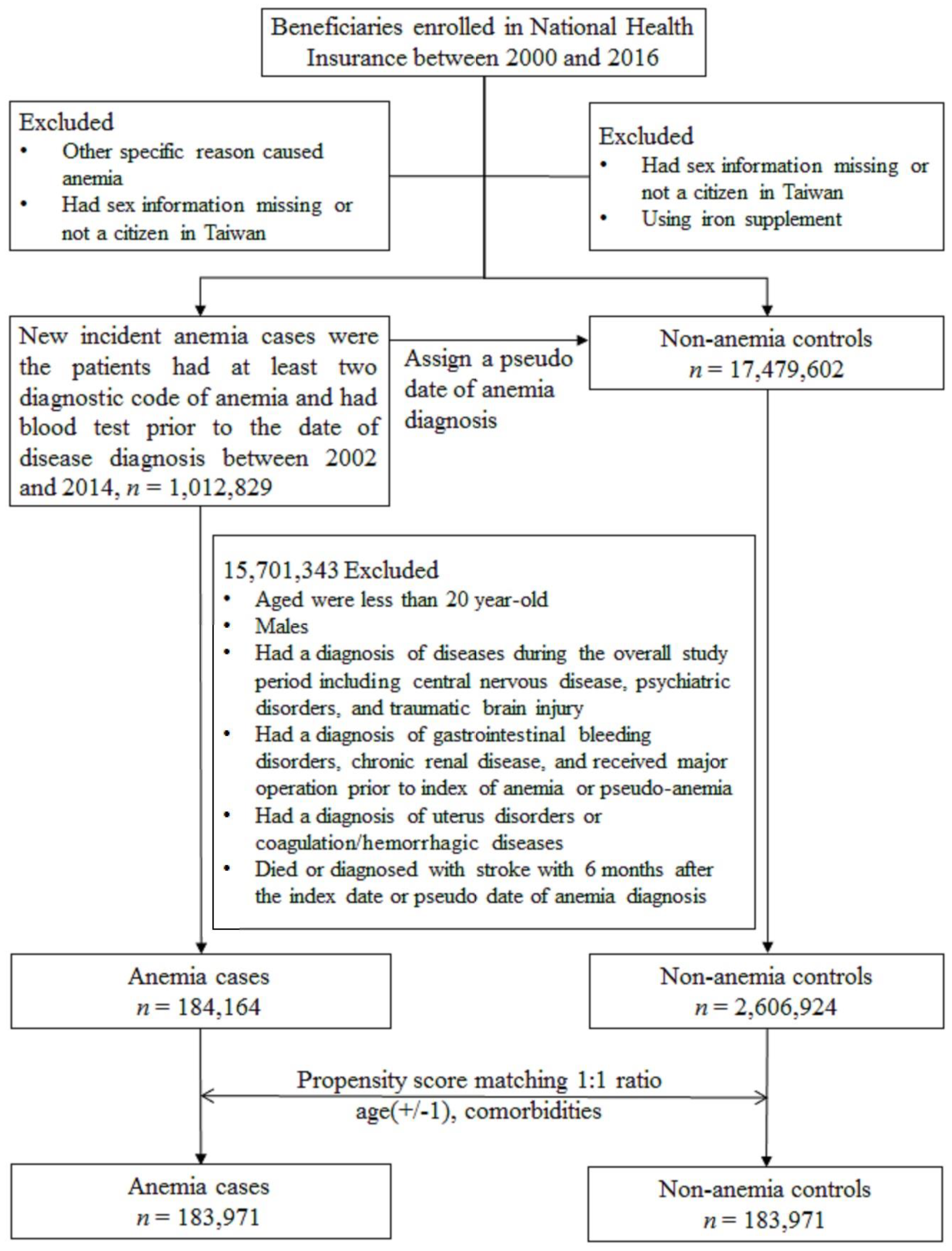
2.2. Study Cohort
2.3. Main Outcome
2.4. Statistical Analysis
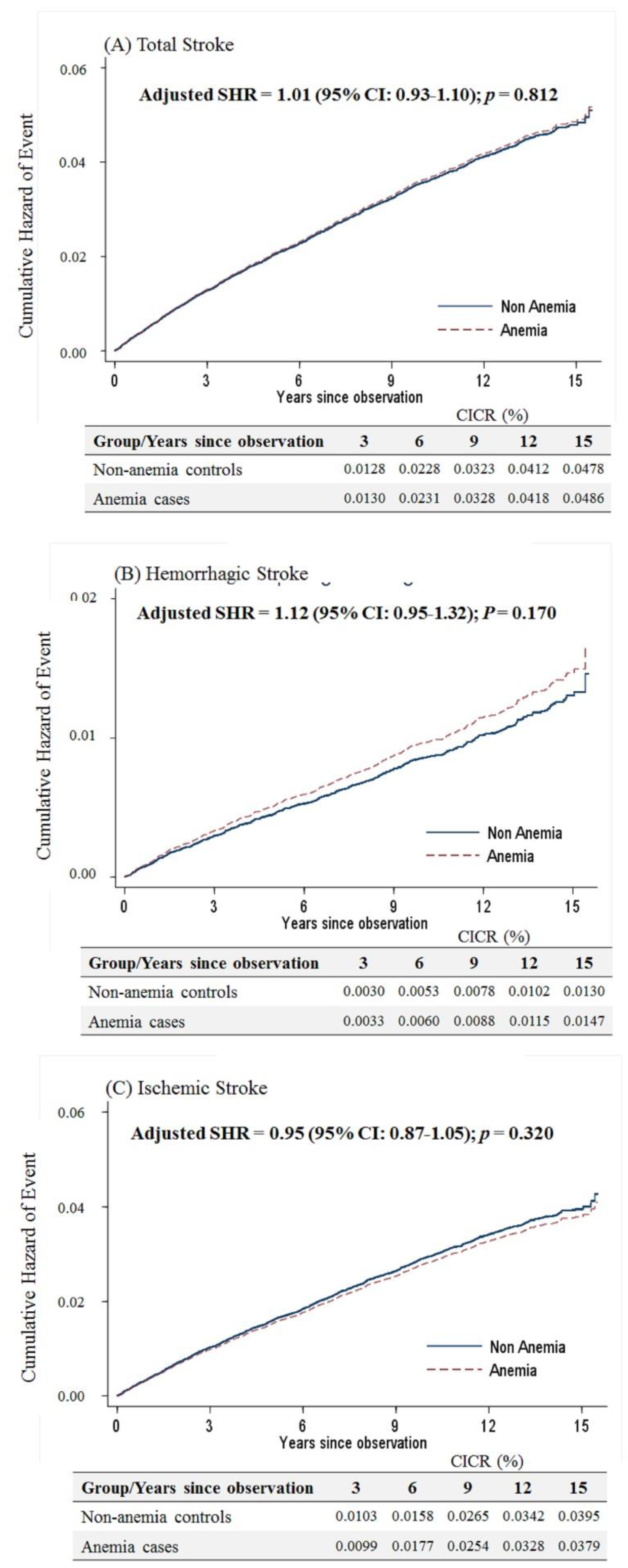
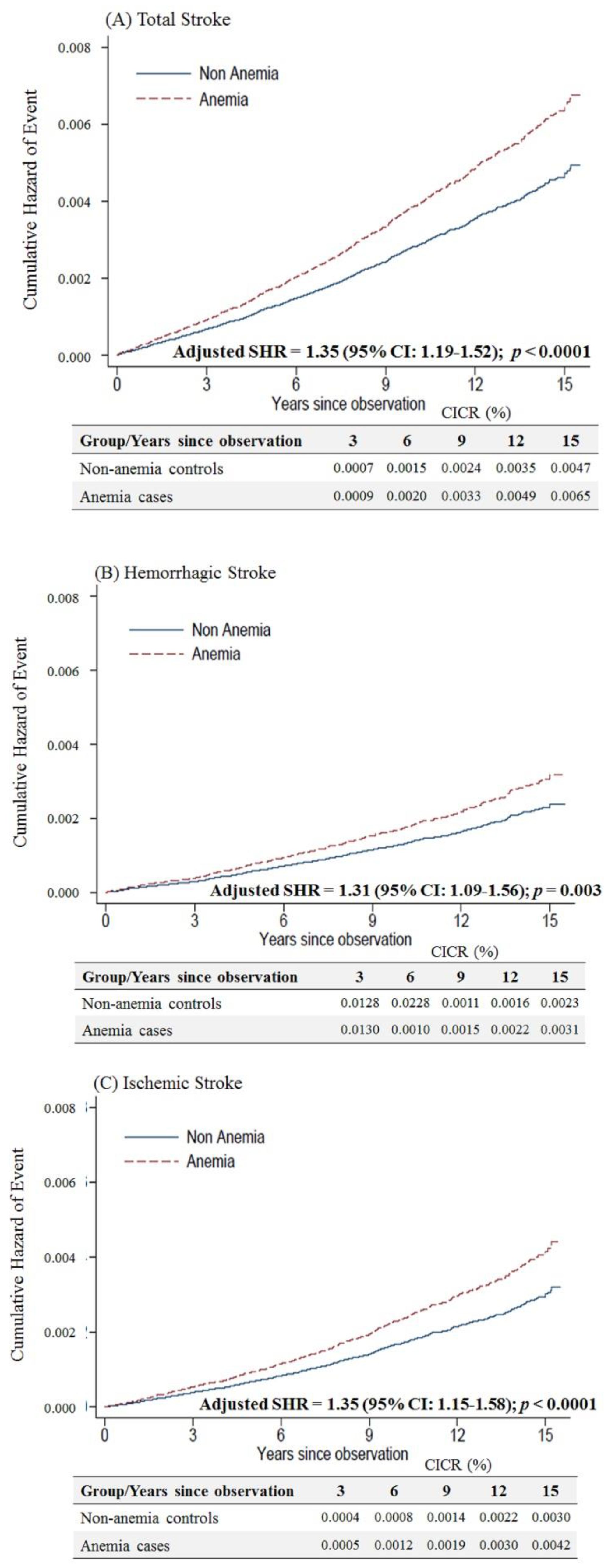
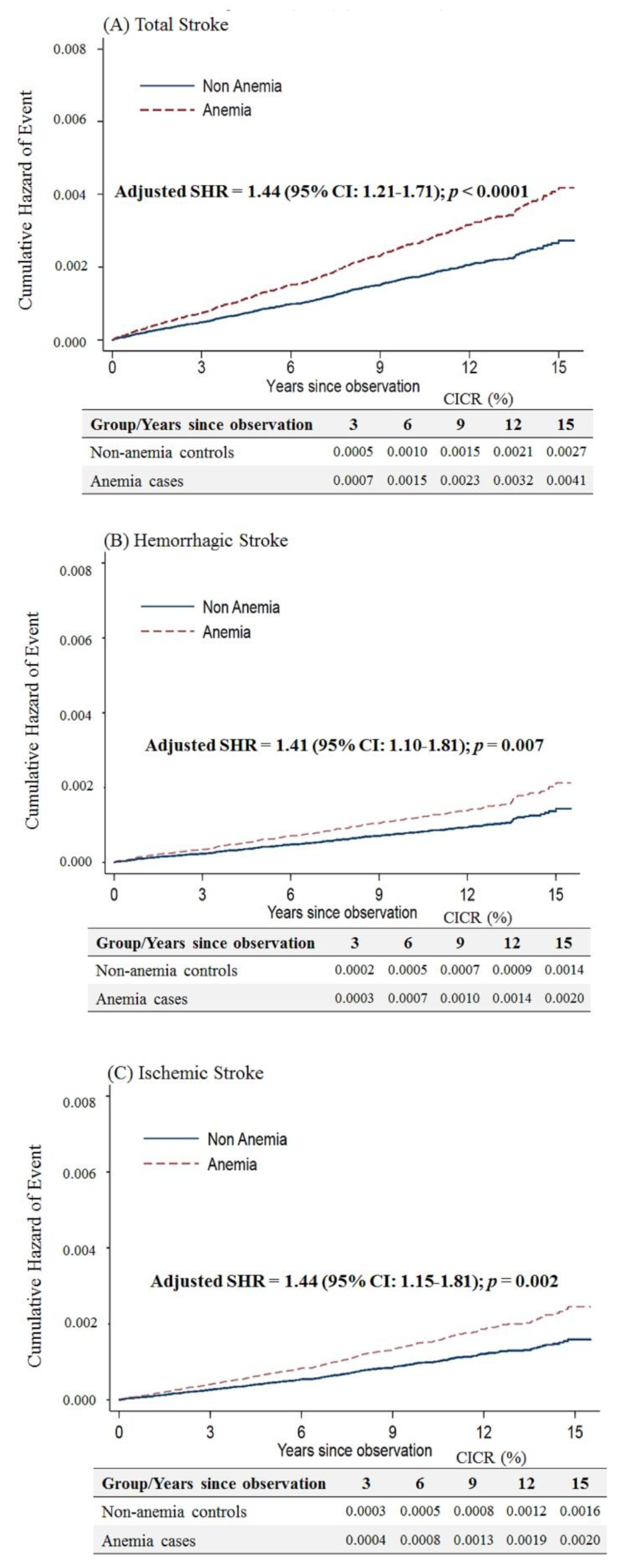
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ASHR | Adjusted sub-distribution hazard ratio |
| ATC | Anatomical Therapeutic Chemical |
| CI | Confidence interval |
| ICD-9-CM | International Classification of Diseases, Ninth Revision, Clinical Modification |
| IDA | Iron-deficiency anemia |
| LDL | Low-density lipoprotein |
| NHI | National Health Insurance |
| NHIRD | National Health Insurance Research Database |
| PSM | Propensity score matching |
| SMD | sSandardized mean difference |
References
- Institute of Health Metrics and Evaluation. Available online: http://www.healthdata.org (accessed on 5 July 2020).
- Guzik, A.; Bushnell, C. Stroke Epidemiology and Risk Factor Management. Contin. Lifelong Learn. Neurol. 2017, 23, 15–39. [Google Scholar] [CrossRef]
- Prabhakaran, S.; Chong, J.Y. Risk Factor Management for Stroke Prevention. Contin. Lifelong Learn. Neurol. 2014, 20, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Andersen, K.K.; Andersen, Z.J.; Olsen, T.S. Age- and Gender-Specific Prevalence of Cardiovascular Risk Factors in 40 102 Patients With First-Ever Ischemic Stroke. Stroke 2010, 41, 2768–2774. [Google Scholar] [CrossRef] [PubMed]
- Demel, S.L.; Kittner, S.; Ley, S.H.; McDermott, M.; Rexrode, K.M. Stroke Risk Factors Unique to Women. Stroke 2018, 49, 518–523. [Google Scholar] [CrossRef] [PubMed]
- Hurn, P.D.; Brass, L.M. Estrogen and Stroke. Stroke 2003, 34, 338–341. [Google Scholar] [CrossRef] [PubMed]
- Green, P.S.; Simpkins, J.W. Neuroprotective effects of estrogens: Potential mechanisms of action. Int. J. Dev. Neurosci. 2000, 18, 347–358. [Google Scholar] [CrossRef]
- Wise, P.M.; Dubal, D.B.; Wilson, M.E.; Rau, S.W.; Böttner, M.; Rosewell, K.L. Estradiol is a protective factor in the adult and aging brain: Understanding of mechanisms derived from in vivo and in vitro studies. Brain Res. Rev. 2001, 37, 313–319. [Google Scholar] [CrossRef]
- Hendrix, S.L.; Wassertheil-Smoller, S.; Johnson, K.C.; Howard, B.V.; Kooperberg, C.; Rossouw, J.E.; Trevisan, M.; Aragaki, A.K.; Baird, A.E.; Bray, P.F.; et al. Effects of Conjugated Equine Estrogen on Stroke in the Women’s Health Initiative. Circulation 2006, 113, 2425–2434. [Google Scholar] [CrossRef]
- Barrett-Connor, E.; Mosca, L.; Collins, P.; Geiger, M.J.; Grady, D.; Kornitzer, M.; McNabb, M.A.; Wenger, N.K. Effects of Raloxifene on Cardiovascular Events and Breast Cancer in Postmenopausal Women. N. Engl. J. Med. 2006, 355, 125–137. [Google Scholar] [CrossRef]
- Chang, Y.-L.; Hung, S.-H.; Ling, W.; Lin, H.-C.; Li, H.-C.; Chung, S.-D. Association between Ischemic Stroke and Iron-Deficiency Anemia: A Population-Based Study. PLoS ONE 2013, 8, 82952. [Google Scholar] [CrossRef]
- Coutinho, J.M.; Zuurbier, S.M.; Gaartman, A.E.; Dikstaal, A.A.; Stam, J.; Middeldorp, S.; Cannegieter, S.C. Association Between Anemia and Cerebral Venous Thrombosis. Stroke 2015, 46, 2735–2740. [Google Scholar] [CrossRef]
- Houghton, D.E.; Koh, I.; Ellis, A.; Key, N.S.; Douce, D.R.; Howard, G.; Cushman, M.; Safford, M.; Zakai, N.A. Hemoglobin levels and coronary heart disease risk by age, race, and sex in the reasons for geographic and racial differences in stroke study (REGARDS). Am. J. Hematol. 2019, 95, 258–266. [Google Scholar] [CrossRef]
- Panwar, B.; Judd, S.E.; Warnock, D.G.; McClellan, W.M.; Booth, J.N.; Muntner, P.; Gutiérrez, O.M. Hemoglobin Concentration and Risk of Incident Stroke in Community-Living Adults. Stroke 2016, 47, 2017–2024. [Google Scholar] [CrossRef]
- Hong, C.-T.; Hsieh, Y.-C.; Liu, H.-Y.; Chiou, H.-Y.; Chien, L.-N. Association Between Anemia and Dementia: A Nationwide, Populationbased Cohort Study in Taiwan. Curr. Alzheimer Res. 2020, 17, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Hong, C.T.; Huang, Y.H.; Liu, H.Y.; Chiou, H.-Y.; Chan, L.; Chien, L.-N. Newly Diagnosed Anemia Increases Risk of Parkinson’s disease: A Population-Based Cohort Study. Sci. Rep. 2016, 6, 29651. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Shan, K.S.; O’Sullivan, C.; Nace, T. Iron Deficiency Anemia: An Unexpected Cause of an Acute Occipital Lobe Stroke in an Otherwise Healthy Young Woman. Cureus 2020, 12, 7852. [Google Scholar]
- Gopalratnam, K.; Woodson, K.A.; Rangunwala, J.; Sena, K.; Gupta, M. A Rare Case of Stroke Secondary to Iron Deficiency Anemia in a Young Female Patient. Case Rep. Med. 2017, 2017, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Caglayan, H.Z.B.; Nazliel, B.; Irkeç, C.; Dumlu, A.; Filiz, A.; Ates, M.P. Iron-Deficiency Anemia Leading to Transient Ischemic Attacks due to Intraluminal Carotid Artery Thrombus. Case Rep. Neurol. Med. 2013, 2013, 1–3. [Google Scholar] [CrossRef][Green Version]
- Little, R.J.; Rubin, D.B. Causal Effects in Clinical and Epidemiological Studies Via Potential Outcomes: Concepts and Analytical Approaches. Annu. Rev. Public Health 2000, 21, 121–145. [Google Scholar] [CrossRef]
- Austin, P.C.; Grootendorst, P.; Anderson, G. A comparison of the ability of different propensity score models to balance measured variables between treated and untreated subjects: A Monte Carlo study. Stat. Med. 2007, 26, 734–753. [Google Scholar] [CrossRef]
- Austin, P.C. A critical appraisal of propensity-score matching in the medical literature between 1996 and 2003. Stat. Med. 2008, 27, 2037–2049. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.-Y.; Chen, C.-H.; Li, C.-Y.; Lai, M.-L. Validating the diagnosis of acute ischemic stroke in a National Health Insurance claims database. J. Formos. Med. Assoc. 2015, 114, 254–259. [Google Scholar] [CrossRef] [PubMed]
- Austin, P.C. The use of propensity score methods with survival or time-to-event outcomes: Reporting measures of effect similar to those used in randomized experiments. Stat. Med. 2013, 33, 1242–1258. [Google Scholar] [CrossRef] [PubMed]
- Grammer, T.B.; Kleber, M.E.; Silbernagel, G.; Pilz, S.; Scharnagl, H.; Tomaschitz, A.; König, W.; März, W. Hemoglobin, iron metabolism and angiographic coronary artery disease (The Ludwigshafen Risk and Cardiovascular Health Study). Atherosclerosis 2014, 236, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Kaiafa, G.; Savopoulos, C.; Kanellos, I.; Mylonas, K.S.; Tsikalakis, G.; Tegos, T.; Kakaletsis, N.; Hatzitolios, A.I. Anemia and stroke: Where do we stand? Acta Neurol. Scand. 2016, 135, 596–602. [Google Scholar] [CrossRef]
- Basak, R.; Chowdhury, A.; Fatmi, L.; Saha, N.; Mollah, A.; Yasmin, S. Stroke in the young: Relationship with iron deficiency anemia and thrombocytosis. Mymensingh Med. J. 2008, 17, 74–77. [Google Scholar]
- Cappellini, M.D.; Motta, I.; Musallam, K.M.; Taher, A.T. Redefining thalassemia as a hypercoagulable state. Ann. N. Y. Acad. Sci. 2010, 1202, 231–236. [Google Scholar] [CrossRef]
- Taher, A.T.; Cappellini, M.D.; Bou-Fakhredin, R.; Coriu, D.; Musallam, K.M. Hypercoagulability and Vascular Disease. Hematol. Clin. N. Am. 2018, 32, 237–245. [Google Scholar] [CrossRef]
- Gotoh, S.; Hata, J.; Ninomiya, T.; Hirakawa, Y.; Nagata, M.; Mukai, N.; Fukuhara, M.; Ikeda, F.; Ago, T.; Kitazono, T.; et al. Hematocrit and the risk of cardiovascular disease in a Japanese community: The Hisayama Study. Atherosclerosis 2015, 242, 199–204. [Google Scholar] [CrossRef]
- Evstatiev, R. Iron deficiency, thrombocytosis and thromboembolism. Wien Med. Wochenschr. 2016, 166, 437–446. [Google Scholar] [CrossRef]
- Tang, X.; Fang, M.; Cheng, R.; Zhang, Z.; Wang, Y.; Shen, C.; Han, Y.; Lu, Q.; Du, Y.; Liu, Y.; et al. Iron-Deficiency and Estrogen Are Associated With Ischemic Stroke by Up-Regulating Transferrin to Induce Hypercoagulability. Circ. Res. 2020, 127, 651–663. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Zhang, Z.; Fang, M.; Han, Y.; Wang, G.; Wang, S.; Xue, M.; Li, Y.; Zhang, L.; Wu, J.; et al. Transferrin plays a central role in coagulation balance by interacting with clotting factors. Cell Res. 2019, 30, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Abe, H.; Semba, H.; Takeda, N. The Roles of Hypoxia Signaling in the Pathogenesis of Cardiovascular Diseases. J. Atheroscler. Thromb. 2017, 24, 884–894. [Google Scholar] [CrossRef] [PubMed]
- Lévy, P.; Kohler, M.; McNicholas, W.T.; Barbé, F.; McEvoy, R.D.; Somers, V.K.; Lavie, L.; Pépin, J.-L. Correction: Obstructive sleep apnoea syndrome. Nat. Rev. Dis. Prim. 2015, 1, 15024. [Google Scholar] [CrossRef]
- Stefanska, A.; Bergmann, K.; Sypniewska, G. Metabolic Syndrome and Menopause: Pathophysiology, Clinical and Diagnostic Significance. Adv. Clin. Chem. 2015, 72, 1–75. [Google Scholar]
- Shen, T.-Y.; Strong, C.; Yu, T. Age at menopause and mortality in Taiwan: A cohort analysis. Maturitas 2020, 136, 42–48. [Google Scholar] [CrossRef]
- Dimitrova, K.R.; DeGroot, K.; Myers, A.K.; Kim, Y.D. Estrogen and homocysteine. Cardiovasc. Res. 2002, 53, 577–588. [Google Scholar] [CrossRef]
- Li, W.; Sun, Q.; Duan, X.; Yi, F.; Zhou, Y.; Hu, Y.; Yao, L.; Xu, H.; Zhou, L. Etiologies and risk factors for young people with intracerebral hemorrhage. Zhong Nan Da Xue Xue Bao Yi Xue Ban 2018, 43, 1246–1250. [Google Scholar]
- Tabuchi, S. Relationship between Postmenopausal Estrogen Deficiency and Aneurysmal Subarachnoid Hemorrhage. Behav. Neurol. 2015, 2015, 1–6. [Google Scholar] [CrossRef]
- Shalev, H.; Kapelushnik, J.; Moser, A.; Knobler, H.; Tamary, H. Hypocholesterolemia in chronic anemias with increased erythropoietic activity. Am. J. Hematol. 2007, 82, 199–202. [Google Scholar] [CrossRef]
- Valappil, A.V.; Chaudhary, N.V.; Praveenkumar, R.; Gopalakrishnan, B.; Girija, A.S. Low cholesterol as a risk factor for primary intracerebral hemorrhage: A case–control study. Ann. Indian Acad. Neurol. 2012, 15, 19–22. [Google Scholar] [CrossRef]
- Lee, G.; Choi, S.; Kim, K.; Yun, J.; Son, J.S.; Jeong, S.; Kim, S.M.; Park, S.M. Association of Hemoglobin Concentration and Its Change With Cardiovascular and All-Cause Mortality. J. Am. Heart Assoc. 2018, 7, 007723. [Google Scholar] [CrossRef]
- Napolitano, M.; Dolce, A.; Celenza, G.; Grandone, E.; Perilli, M.G.; Siragusa, S.; Carta, G.; Orecchioni, A.; Mariani, G. Iron-dependent erythropoiesis in women with excessive menstrual blood losses and women with normal menses. Ann. Hematol. 2013, 93, 557–563. [Google Scholar] [CrossRef]
| Before Matching | After Matching | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control n | (%) | Case n | (%) | SMD | Control n | (%) | Case n | (%) | SMD | |
| Sample size | 2,606,924 | 100% | 184,164 | 100.0% | 183,971 | 100% | 183,971 | 100.0% | ||
| Age, mean (SD) | 38.9 (13.4) | 38.5 (13.5) | 38.4 (13.4) | 38.4 (13.4) | ||||||
| 20–39 | 1,444,920 | 55.4% | 111,237 | 60.4% | 0.101 | 109,562 | 59.6% | 111,195 | 60.4% | 0.018 |
| 40–49 | 595,065 | 22.8% | 43,179 | 23.4% | 0.015 | 43,292 | 23.5% | 43,163 | 23.5% | 0.002 |
| 50–59 | 366,931 | 14.1% | 15,789 | 8.6% | 0.174 | 16,884 | 9.2% | 15,769 | 8.6% | 0.021 |
| 60–69 | 140,054 | 5.4% | 7046 | 3.8% | 0.074 | 7147 | 3.9% | 7028 | 3.8% | 0.003 |
| 70+ | 59,954 | 2.3% | 6913 | 3.8% | 0.085 | 7086 | 3.9% | 6816 | 3.7% | 0.008 |
| Comorbidity, yes | ||||||||||
| Hypertension | 143,174 | 5.5% | 10,288 | 5.6% | 0.004 | 10,172 | 5.5% | 10,169 | 5.5% | <0.0001 |
| Diabetes | 56,471 | 2.2% | 5511 | 3.0% | 0.052 | 5400 | 2.9% | 5407 | 2.9% | <0.0001 |
| Hyperlipidemia | 61,961 | 2.4% | 4499 | 2.4% | 0.004 | 4446 | 2.4% | 4448 | 2.4% | <0.0001 |
| Coronary artery disease | 20,318 | 0.8% | 2001 | 1.1% | 0.032 | 1919 | 1.0% | 1936 | 1.1% | 0.001 |
| Heart failure | 3136 | 0.1% | 593 | 0.3% | 0.043 | 491 | 0.3% | 533 | 0.3% | 0.004 |
| Atrial fibrillation | 1441 | 0.1% | 215 | 0.1% | 0.021 | 149 | 0.1% | 194 | 0.1% | 0.008 |
| Peripheral artery disease | 1747 | 0.1% | 208 | 0.1% | 0.015 | 174 | 0.1% | 186 | 0.1% | 0.002 |
| Malignant neoplasm | 11,071 | 0.4% | 2867 | 1.6% | 0.114 | 2795 | 1.5% | 2766 | 1.5% | 0.001 |
| Rheumatic disease | 40,173 | 1.5% | 3893 | 2.1% | 0.043 | 3827 | 2.1% | 3829 | 2.1% | <0.0001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sui, Y.; Hong, C.-T.; Chien, L.-N.; Liu, H.-Y.; Chiou, H.-Y.; Hsieh, Y.-C. Association between Anemia and Stroke in Females: A Nationwide, Population-Based Cohort Study in Taiwan. Int. J. Environ. Res. Public Health 2020, 17, 7440. https://doi.org/10.3390/ijerph17207440
Sui Y, Hong C-T, Chien L-N, Liu H-Y, Chiou H-Y, Hsieh Y-C. Association between Anemia and Stroke in Females: A Nationwide, Population-Based Cohort Study in Taiwan. International Journal of Environmental Research and Public Health. 2020; 17(20):7440. https://doi.org/10.3390/ijerph17207440
Chicago/Turabian StyleSui, Yuan, Chien-Tai Hong, Li-Nien Chien, Hung-Yi Liu, Hung-Yi Chiou, and Yi-Chen Hsieh. 2020. "Association between Anemia and Stroke in Females: A Nationwide, Population-Based Cohort Study in Taiwan" International Journal of Environmental Research and Public Health 17, no. 20: 7440. https://doi.org/10.3390/ijerph17207440
APA StyleSui, Y., Hong, C.-T., Chien, L.-N., Liu, H.-Y., Chiou, H.-Y., & Hsieh, Y.-C. (2020). Association between Anemia and Stroke in Females: A Nationwide, Population-Based Cohort Study in Taiwan. International Journal of Environmental Research and Public Health, 17(20), 7440. https://doi.org/10.3390/ijerph17207440





